Inorganic Year 1
1/17
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
18 Terms
What are the Trends in Atomic Radius, Reactivity, Ionisation Energy, Boiling Point and Electronegativity down group 7?
Atomic Radius - The atomic radius increases down the group due to additional electron shells.
Reactivity - The reactivity decreases down the group due to additional electron shielding weakening the attraction from the nucleus to accept an electron.
Ionisation Energy - The ionisation energy decreases down the group due to greater atomic radius and increased electron shielding.
Boiling Point - The boiling point increases down the group due to increasing van der Waals forces holding the molecule together.
Electronegativity - The electronegativity decreases down the group due to increased atomic radius and electron shielding.
What is Oxidising Power and the trend down group 7?
Oxidising Power - The ability of a oxidising agent to accept electrons in a reaction and be reduced.
Oxidising Power/Ability decreases down the group due to their ability to attract electrons decreasing from electron shielding and atomic radius. This means that a halide will displace and halogen below it in the periodic table in a displacement reaction: Cl2(aq) + 2NaBr(aq) → Br2(aq) + 2NaCl(aq)
What is Reducing Power and the trend for halide ions down group 7?
Reducing Power - The ability of a reducing agent to donate electrons in a reaction and be oxidised.
Reducing Power/Ability increases down the group for halide ions. This is due it the ability to lose electrons increasing from electron shielding and atomic radius. This trend can be seen in the reactions of solid sodium halides with concentrated sulfuric acid.
Describe the reactions of Halide Ions with Concentrated Sulfuric Acid?
Fluoride and Chloride Ions:
NaF + H2SO4 → NaHSO4 + HF
NaCl + H2SO4 → NaHSO4 + HCl
Bromide Ions:
NaBr + H2SO4 → NaHSO4 + HBr
2HBr + H2SO4 → Br2 + SO2 + 2H2O
Iodide Ions:
NaI + H2SO4 → NaHSO4 + HI
2HI + H2SO4 → I2 + SO2 + 2H2O
6HI + SO2 → H2S + 3I2 + 2H2O
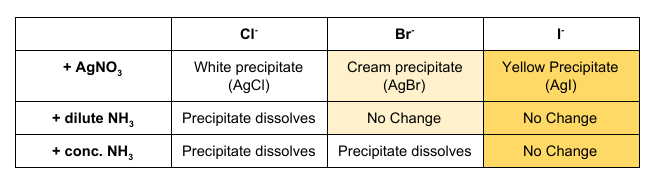
How can Halide Ions be distinguished using Silver Nitrate and Ammonia?
Acidified Silver Nitrate is used to test for halide ions as they react to form coloured precipitates. As the precipitate may not be clear to distinguish, different concentrations of ammonia can be added. Silver Nitrate is acidified to remove any ions that may interfere with the reaction.
See Table for Results.
How does Chlorine react with water and why is it used?
Chlorine reacts with water in a disproportionation reaction as chlorine is both oxidised and reduced: Cl2 + H2O → ClO- + Cl- + 2H+
Chlorine is used in small quantities to kill bacteria and sterilise water. Even though chlorine can be toxic, the benefits still outweigh the risks of using chlorine.
In the presence of UV light, chlorine decomposes with water, where chlorine is reduced in this reaction: 2Cl2 + 2H2O → 4HCl + O2
How can Chlorine be used in Bleach Production?
Chlorine can be mixed with cold, aqueous sodium hydroxide to produce sodium hypochlorite, a key ingredient in bleach: 2NaOH + Cl2 → NaClO + NaCl + H2O
This reaction is a disproportionation reaction.
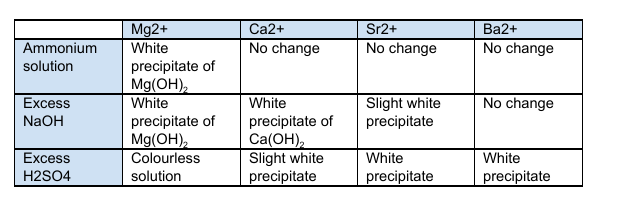
Required Practical 4 - Carrying out simple test-tube reactions to identify cations and anions?
Cations (Group 2 metals or Ammonia):
Group 2 Metals
Add 10 drops of your unknown solution, and 10 drops of dilute sodium hydroxide into a test tube
Stir the test tube and record any observations
Continue to add sodium hydroxide dropwise and continue to record observations
Once completed, dispose of any contents in a bowl of water and repeat
The dilute sodium hydroxide could be substituted with dilute sulphuric acid
The results are in the table attached
Ammonium Ions
Place 10 drops of your unknown solution in a test tube with 10 drops of sodium hydroxide and shake
Heat the mixture in a water bath gently and test the fumes produced
It should turn damp red litmus paper to blue
Anions (Halides, Sulfates, Hydroxides, Carbonates)
Hydroxides
Test a 1cm depth of your solution with damp red litmus paper
It should turn the litmus paper blue if it is a hydroxide
Carbonates
Add an equal volume of dilute HCl to your unknown solution and transfer the gas produced to a tube of lime water
The limewater will go cloudy if it is a carbonate
Sulfates
Add an equal volume of dilute HCl and barium chloride to your unknown solution
Once reacted add a bit more HCl to test if the precipitate dissolves
If the precipitate remains, sulphate or hydrogen sulphate ions are present
Halides
Silver Nitrate Test
What are the trends in Ionisation Energy, Atomic Radius, Reactivity and Melting Points down group 2?
Ionisation Energy - The first ionisation energy decreases down group 2 due to a greater atomic radius and increased electron shielding.
Atomic Radius - The atomic radius increased down group 2 due to additional electron shells.
Reactivity - Reactivity increases down group 2 due to increased electron shielding making the outer electrons easier to lose.
Melting Point - Melting Point decreases down group 2 due to the ions getting larger meaning the attractive forces are weaker as they have to travel a further distance.
What are the Reactions of Group 2 Elements with Water?
The group 2 metals increasingly react down the group with water in a redox reaction to produce a metal hydroxide and hydrogen. The metal hydroxides form as an alkaline solution.
Example Reaction: M(s) + 2H2O(l) → M(OH)2(aq) + H2(g), Where M is any group 2 metal
Magnesium reacts very slowly with water, but rapidly with steam as it provides the reaction with extra energy. It also causes magnesium to burn with a bright white flame to form magnesium oxide, a white powder: Mg(s) + H2O(g) → MgO(s) + H2(g)
What is the Solubility of Group 2 Hydroxides?
The solubility of group 2 hydroxides increases down the group. The hydroxides are all white solids. Magnesium hydroxide is sparingly soluble and Barium hydroxide is the most soluble.
As magnesium hydroxide is sparingly soluble, it is used in medicine (milk of magnesia) as an antacid as it is alkaline, and can neutralise acids.
Calcium hydroxide is used in agriculture to treat acidic soil and neutralise it due to its alkalinity.
What is the Solubility of Group 2 Sulfates?
The solubility of group 2 sulfates decreases down the group. Magnesium Sulfate is the most soluble and Barium Sulfate is insoluble.
As Barium Sulfate is insoluble, it is used in medicine as barium meals which allow internal tissue and organs to be imaged. It is toxic if it enters the blood steam but as it is insoluble it cannot be absorbed into the blood.
What are some other Uses of group 2 compounds?
Metal Extraction - Magnesium is used to extract titanium from titanium chloride via a displacement reaction: TiCl4 + 2Mg → 2MgCl2 + Ti
Flue Gas Removal - Calcium Oxide reacts with sulfur dioxide to remove it from factory pollutants and prevent it from reaching the atmosphere. It forms calcium sulfite and water: CaO + 2H2O + SO2 → CaSO3 + 2H2O
Testing Sulfate Ions - Acidified Barium Chloride solution is used as a test for sulfate ions as it reacts to form barium sulfate, a white precipitate when sulfate ions are present. It is acidified to remove interfering ions that cause a false positive. Ba2+ + SO42- → BaSO4(s)
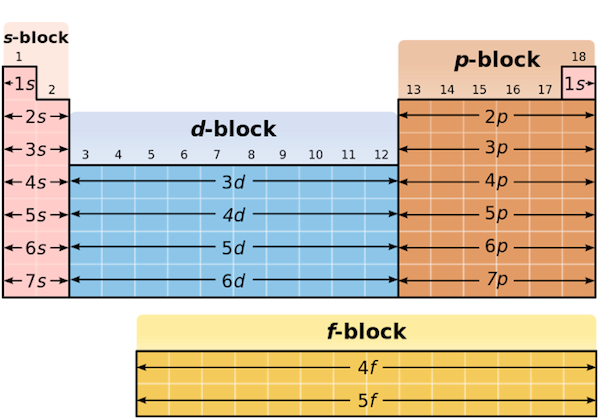
How are Elements classifies in the Periodic Table?
They are arranged due to proton number. Elements in a period have the same number of electron shells. Elements in a group have the same number of outer electrons. Elements are also classified into s-, p-, d- and f- blocks to show electron configuration.
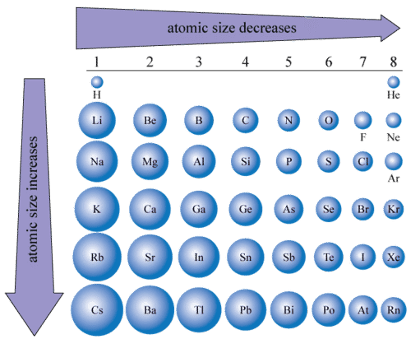
Explain the trend in Atomic Radius across a Period and down a Group?
Across a Period:
Atomic Radius decreases. This is due to an increased nuclear charge of the elements with the same number of electron shells. This means that there is a greater attraction from the nucleus so the outer electrons are pulled in closer, reducing the atomic radius.
Down a Group:
Atomic Radius Increases. This is due to an increasing amount of electron shells. This increases the distance between the outer electrons and the nucleus, reducing the attraction. More shells also increase nuclear shielding by blocking the attractive forces, reducing the nuclear attraction further, increasing the atomic radius.
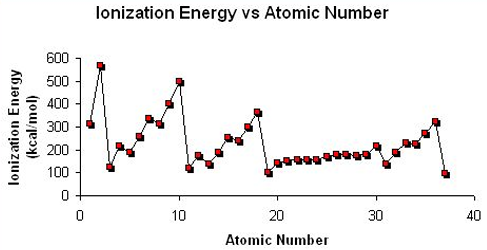
Explain the trend in Ionisation Energy across a Period and down a Group?
Across a Period:
Ionisation Energy generally increases. This is due to the decreasing atomic radius and increasing nuclear charge, causing electrons to be attracted more to the nucleus, so more energy is required to overcome the attraction and remove the electron.
Down a Group:
Ionisation Energy decreases. This is due to the nuclear attraction between the nucleus and outer electrons reducing and the increasing amounts of shielding means that less energy is required to remove an outer electron.
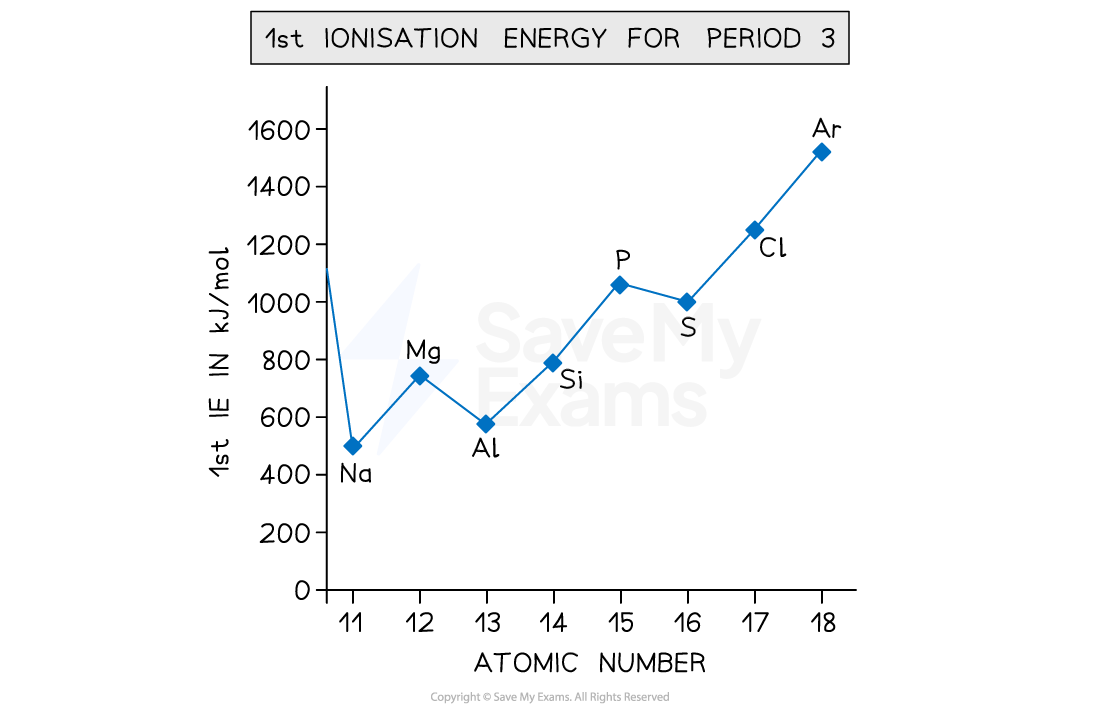
Explain the trend in Ionisation Energy across Period 3?
Ionisation Energy generally increases along Period 3. This is due to the decreasing atomic radius and increasing nuclear charge, causing electrons to be attracted more to the nucleus, so more energy is required to overcome the attraction and remove the electron.
There is a drop in First Ionisation Energy between groups 2 and 3 as it is now removing a p-block electron which is in a higher energy level compared to the s-electrons so less energy is required to remove it.
There is a drop in First Ionisation Energy between groups 5 and 6 as electrons are now being added to half-filled orbitals. This means that due to two electrons being in the same orbital, there will be electron-electron repulsion form the two negative charges, so it is easier to lose.
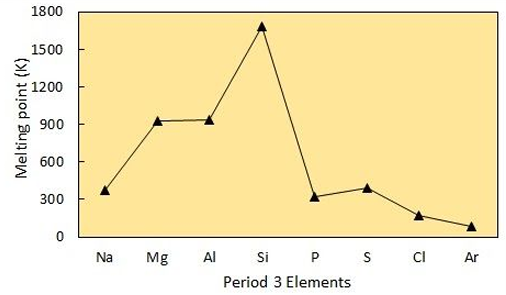
Explain the trend in Melting Point across Period 3?
Sodium - Aluminium:
These 3 elements all have metallic bonding. This means that their melting point increases based on charge. As your go along the period, the charge on the ion increases. This means that there are more electrons released so join the sea of delocalised electrons. As a result, the electrostatic forces increase so the metallic bond becomes stronger.
Silicon:
Silicon has a macromolecular structure. This means that it contains very strong covalent bonds which require lots of energy to overcome giving it the highest melting point.
Phosphorus - Chlorine:
These 3 elements are all simple covalent molecules (P4, S8, Cl2) held together by weak van der Waals forces. As these don’t require much energy to overcome their melting points are lower. The molecules with more electrons have more van der Waals forces so it has a higher melting point, making the order S8 > P4 > Cl2.
Argon:
Argon is a noble gas that exists as an individual atom due to its full outer shell. This makes it very stable but with very weak van der Waals forces. As a result, it has the lowest melting point causing it to exist as a gas at room temperature.