CHE108 - States of Matter and Analytical Chemistry
1/170
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
171 Terms
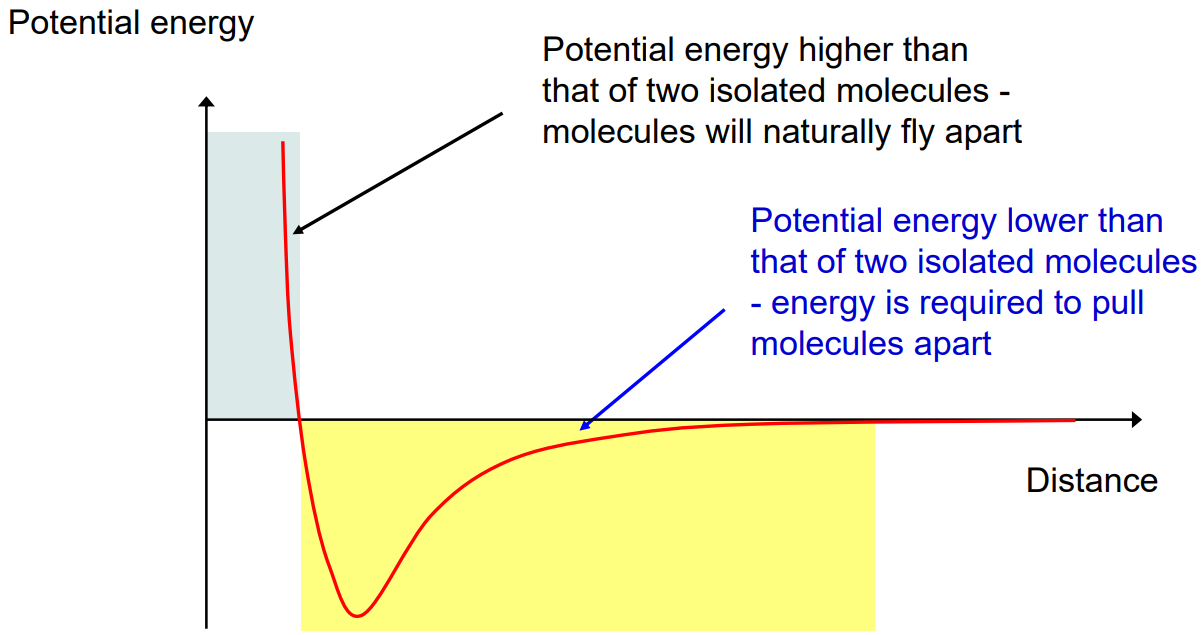
In this potential energy curve, what does the minimum point represent?
The most stable separation between two molecules, where attractive and repulsive forces balance.
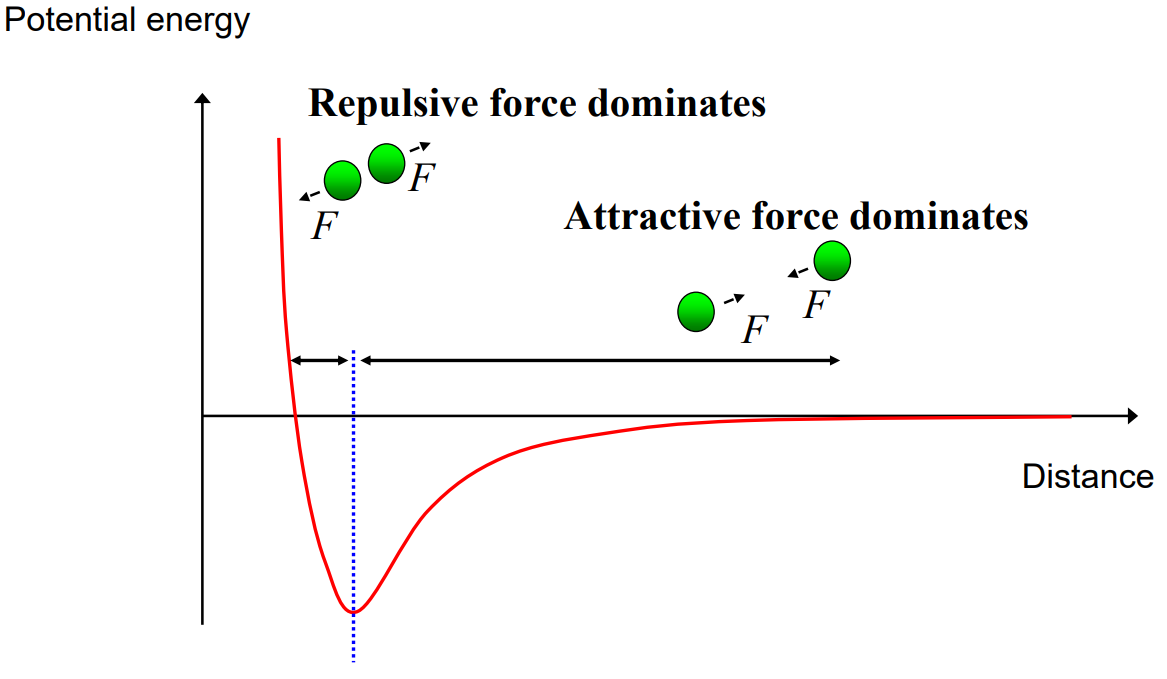
According to this diagram, when does repulsion dominate and when does attraction dominate?
Very short distance: Repulsion dominates (electron cloud overlap)
Longer distance: Attraction dominates (intermolecular forces pulling molecules together)
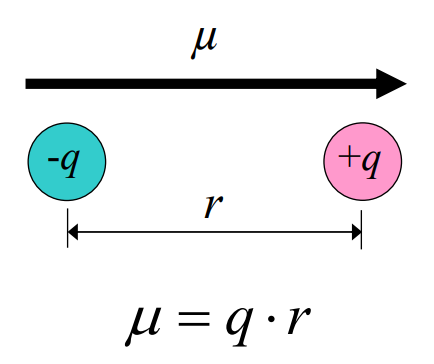
In the dipole diagram, what does the arrow for μ represent?
The dipole moment vector, pointing from negative charge (−q) to positive charge (+q), with magnitude μ = q × r.
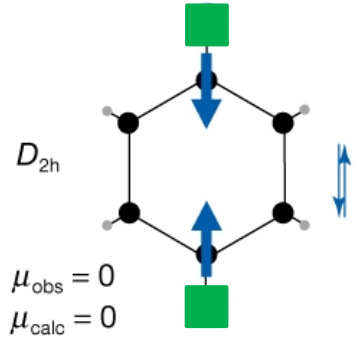
Why does the molecule labeled D₂h have μ = 0 in this diagram?
Because the bond dipoles cancel due to symmetry, giving no net dipole moment.
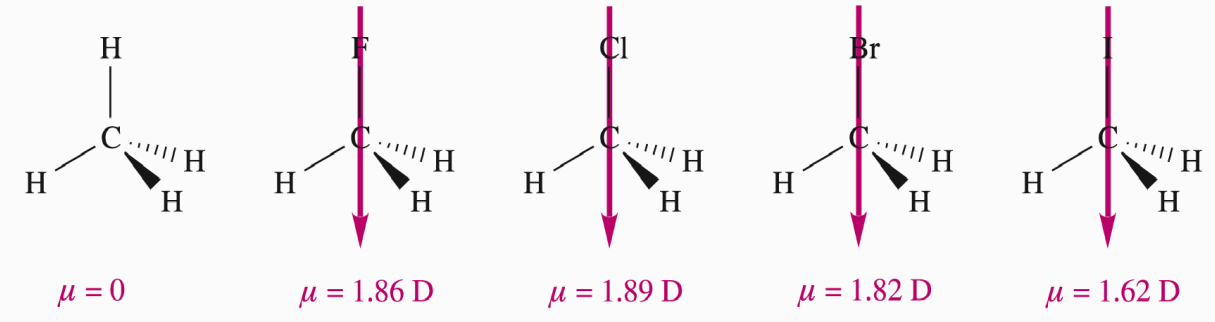
The diagram shows C–F, C–Cl, C–Br, C–I dipoles. Why doesn’t dipole moment strictly follow electronegativity?
Because bond length also affects μ — dipole moment depends on both charge difference and distance between charges.
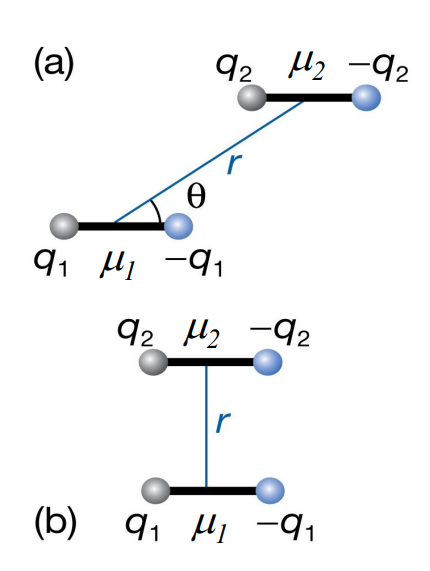
In this dipole–dipole diagram, what determines whether the interaction is attractive or repulsive?
The relative orientation (angle θ) and distance r between dipoles.
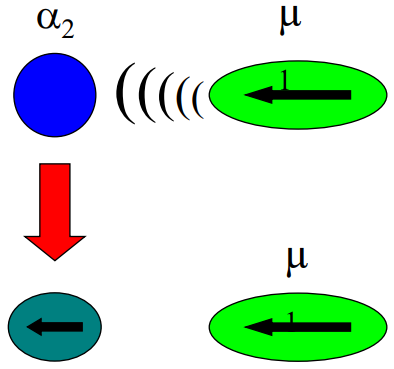
What process is shown when the blue nonpolar molecule distorts near a green polar molecule?
Dipole - Induced Dipole
A permanent dipole induces a temporary dipole in a nonpolar molecule. This attraction depends on polarizability (α).
How do dispersion forces arise?
Temporary electron fluctuations create a dipole in molecule A, which induces a dipole in molecule B, leading to attraction.
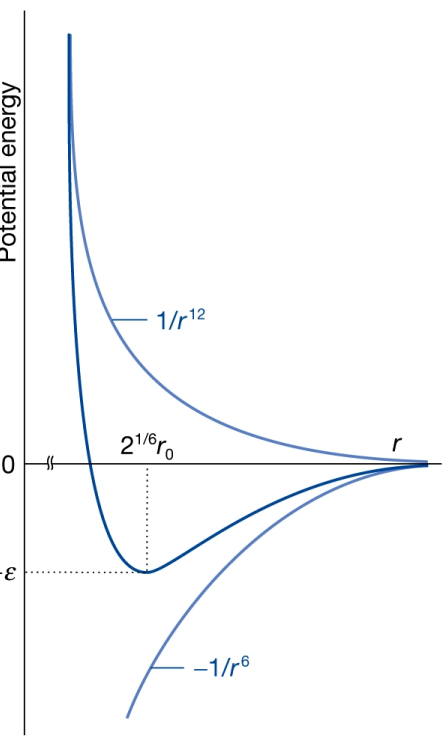
In this Lennard-Jones graph, what do the two curves represent?
−1/r⁶: Attractive van der Waals forces
+1/r¹²: Strong short-range repulsion
The minimum shows the equilibrium intermolecular distance.
Hydrogen bonding typically occurs in molecules containing which bonds?
N–H, O–H, or F–H bonds, where hydrogen is attached to a highly electronegative atom.
What are the three main types of van der Waals attractive interactions?
Dipole–dipole
Dipole-induced dipole
Dispersion
What type of van der Waals attractive force is strongest in small molecules?

How are the strength van der Waals attractive forces different in large molecules?
The “contact area” increases and dispersion forces become
very significant.
What four variables define the state of a pure gas?
P = Pressure
V = Volume
T = Temperature
n = Amount of gas (moles)
Where does gas pressure come from?
From molecular collisions with container walls.
What assumptions define an ideal gas?
Particles move in random motion
No intermolecular forces
Particles have negligible volume
What does Boyle’s Law state? (constant n, T)
The relationship between presume and volume is inversely proportional.
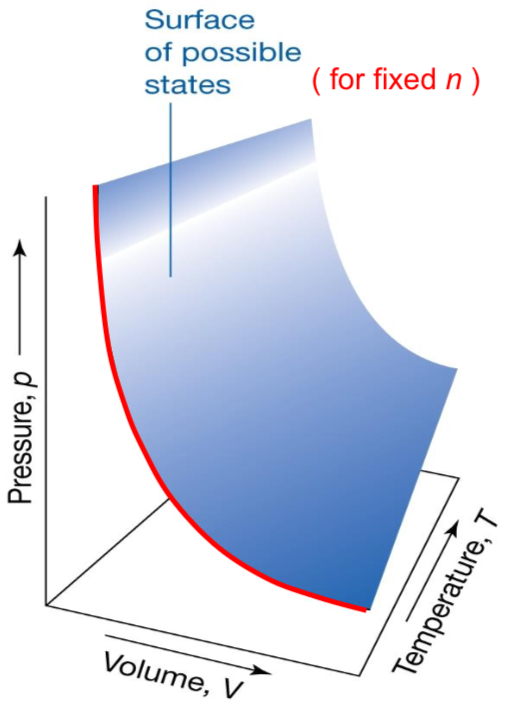
What does Charles’ Law state? (constant n, P)
Volume and temperature are proportional?
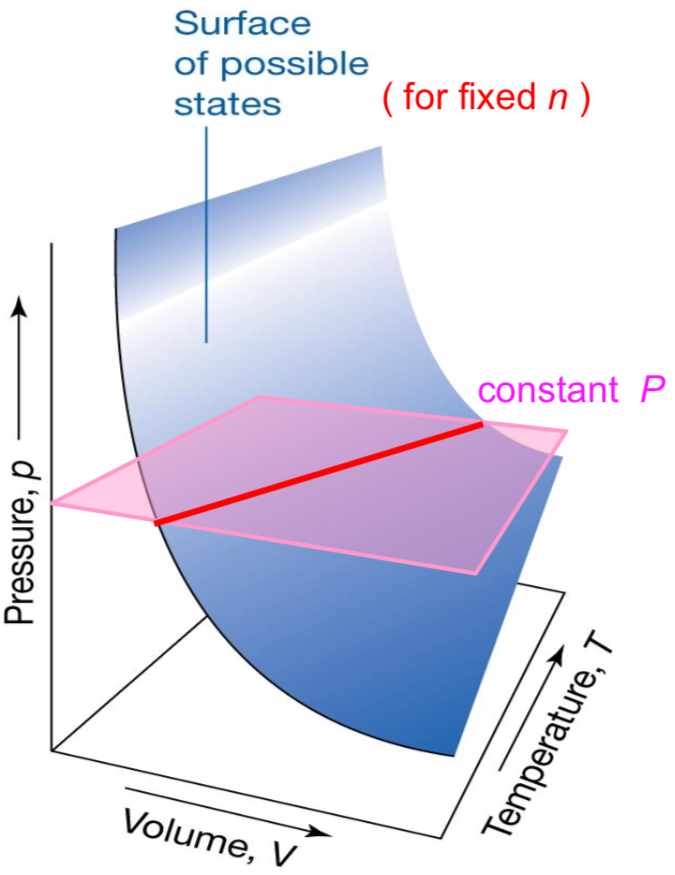
What does Gay-Lussac’s Law state? (constant n, V)
Pressure and Temperature are proportional
Why do real gases deviate from ideal behavior?
Because:
Molecules have finite size
Molecules experience intermolecular forces
What does the van der Waals equation correct for?
a → accounts for attractive forces (reduces pressure)
b → accounts for finite molecular volume (reduces free volume)
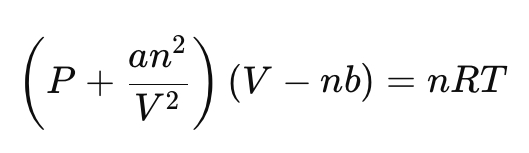
What do real gas isotherms show compared to ideal gases?
Deviations at low volume / high pressure, and possible phase transition regions (e.g., CO₂).
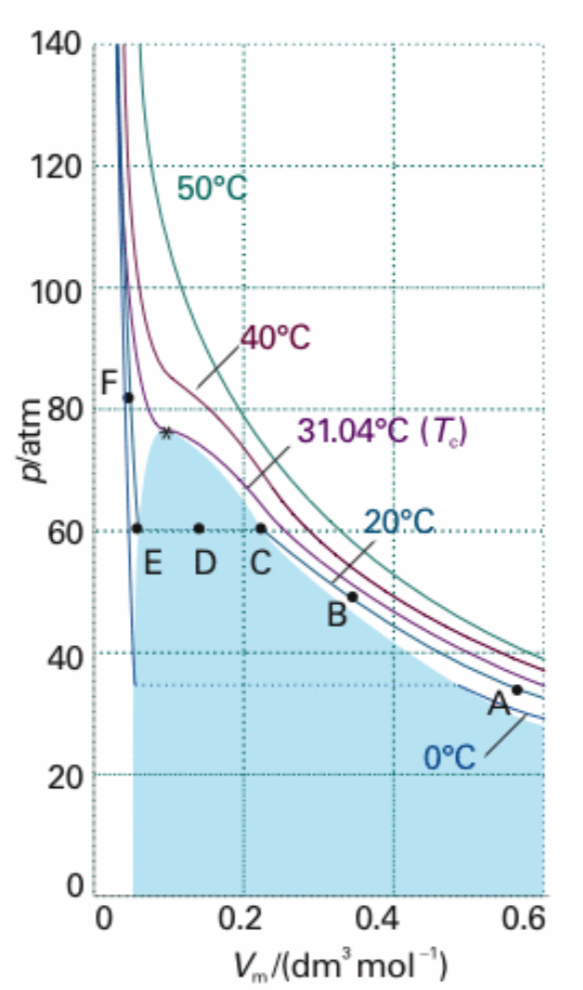
What is the average translational kinetic energy of a gas molecule?
Depends only on temperature.
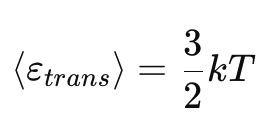
How does temperature affect the Maxwell–Boltzmann distribution?
Higher T →
Broader curve
Peak shifts to higher speeds
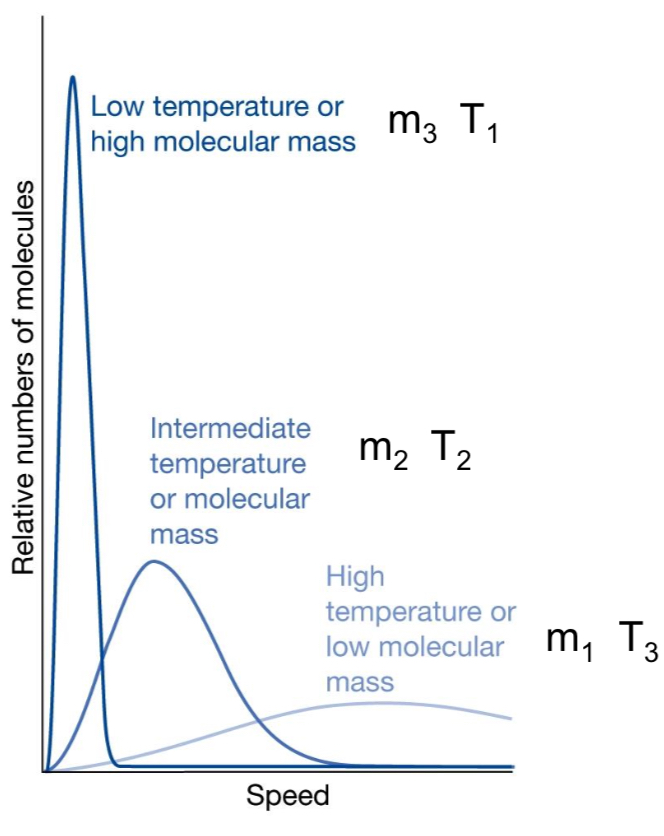
How does molecular mass affect the speed distribution?
Lighter molecules → higher speeds
Heavier molecules → lower speeds
Why do gases behave more ideally at low pressure?
Molecules are far apart
Few intermolecular interactions
Large mean free path
Why do gases deviate from ideal behavior at high pressure?
Molecules are closer together
More collisions and interactions
Finite molecular size becomes important
Do gases always mix? Why?
Yes. Gases spontaneously interdiffuse due to random molecular motion, regardless of chemical type.
State Dalton’s Law of Partial Pressures.
Ptotal=PA+PB+PC+…
What is partial pressure?
The pressure a gas would exert if it alone occupied the container.
How is the liquid state described structurally?
Liquids are relatively disordered
Much denser than gases
Molecules:
Constantly interact via intermolecular forces
Are in motion but less free than in gases
Is there any order in liquids?
Yes — short-range order, but no long-range order.
Molecules form fluctuating shells around a central molecule.
Radial distribution function g(r):
First peak → nearest neighbours
Oscillations decay → no long-range structure
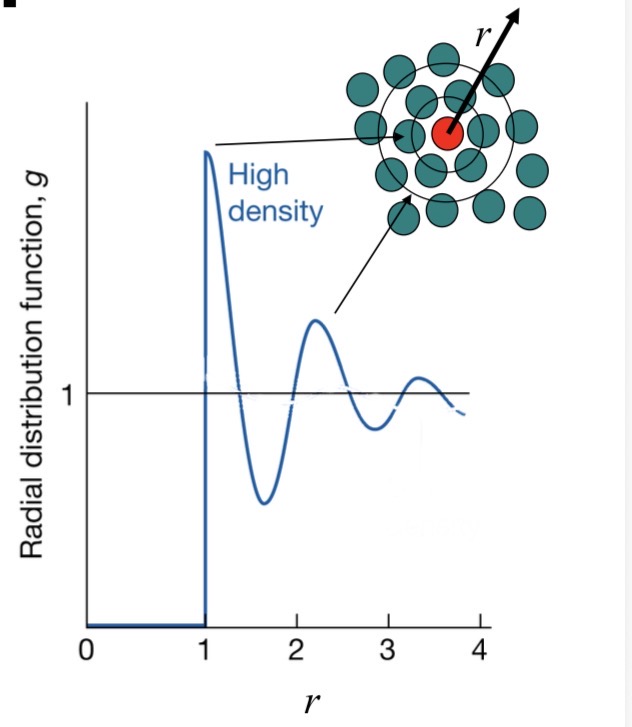
What molecular motions are suppressed in liquids?
Translation: almost completely suppressed
(molecules migrate slowly, randomly)Rotation: restricted
(small stepwise rotations ~5–60°)Vibrations: largely unaffected
Why is potential energy important in liquids?
The intermolecular interaction potential energy is a major component of the liquid’s internal energy.
Liquids have significant intermolecular forces → unlike gases.
What is vapour pressure?
Vapour pressure = pressure of gas in equilibrium with its liquid
Why does vapour pressure increase with temperature?
Higher temperature → more molecules have enough energy to enter gas phase.
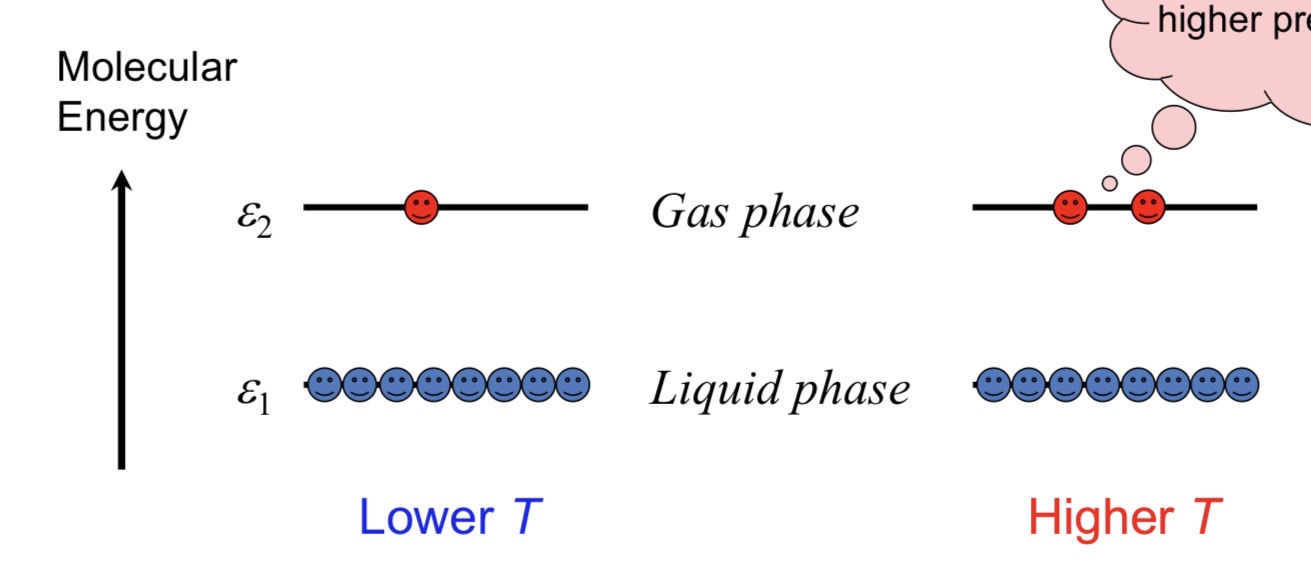
When does boiling occur?
Boiling occurs when:
Vapour pressure=Atmospheric pressureVapour pressure=Atmospheric
What are miscible and immiscible liquids?
Miscible → form a mixture (e.g., ethanol + water)
Immiscible → do not mix (e.g., oil + water)
May be partially miscible
Miscibility depends on temperature
Why are some liquids immiscible?
Entropy
Mixing always increases disorder → favours mixing
Enthalpy (intermolecular forces)
Mixing favoured if:
VA−B<VA−A,VB−BVA−B<VA−A,VB−B
Mixing NOT favoured if:
VA−B>VA−A,VB−B
What is a colloid?
A colloid = dispersion of small particles
(typically d < 500 nm) in another material.
What is an emulsion?
Milk
Cosmetics
Food products
Are colloids thermodynamically stable?
Thermodynamically unstable
Kinetically stable (non-labile)
They want to separate into phases, but separation can be slow.
How are solid-state materials broadly classified based on atomic arrangement?
As ordered (crystalline) or disordered (amorphous/glassy) materials.

What determines the bulk properties and function of a solid?
The inter-atomic arrangement and degree of ordering.
Why is atomic ordering important in solids?
It governs bulk properties, phase relations, and material functionality.
What defines a crystalline solid?
A periodic atomic structure with long-range order (LRO) and a homogeneous structure.
What defines an amorphous solid?
A non-periodic structure with no long-range order (LRO) and a heterogeneous structure.
What is short-range order (SRO)?
Local atomic ordering over a few nearest-neighbour distances.
Can amorphous materials have any order?
Yes, they may exhibit short-range order (SRO) but lack long-range order (LRO).
What is a Bravais lattice?
A distinct 3D periodic arrangement of points that describes all possible crystal lattices.
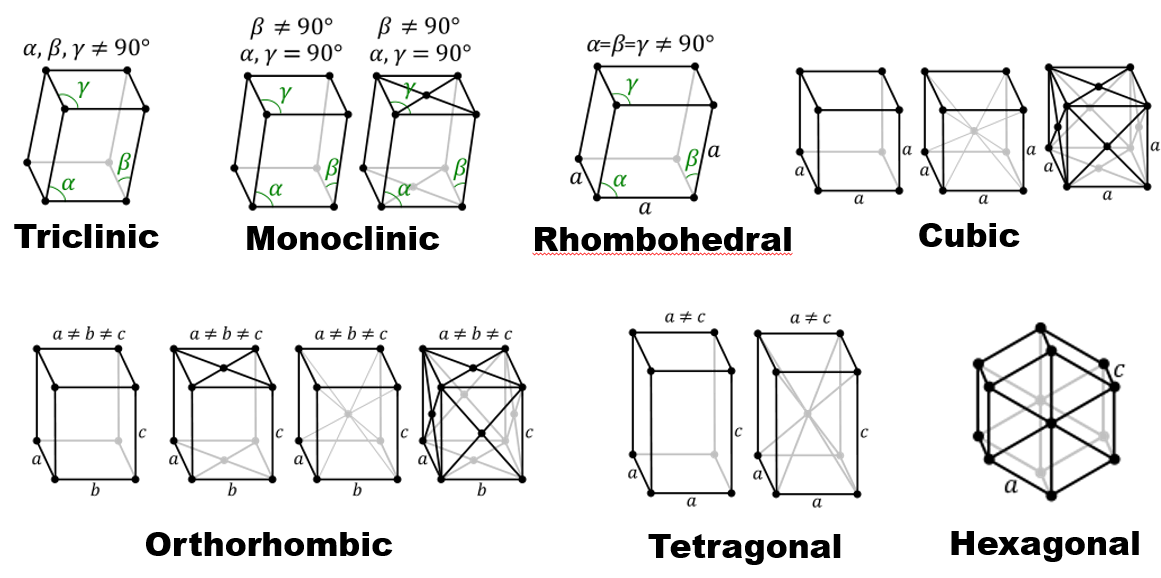
How many lattice systems and Bravais lattices exist?
7 lattice systems and 14 Bravais lattices.
Name the seven lattice systems.
Triclinic, Monoclinic, Orthorhombic, Tetragonal, Cubic, Rhombohedral, Hexagonal.
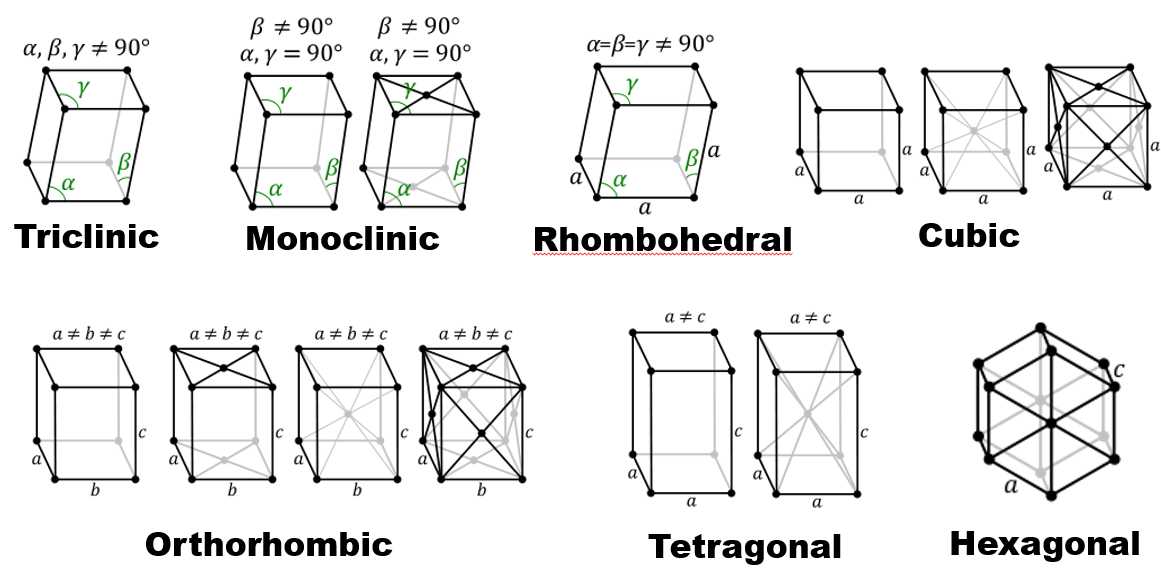
What are the four common cubic Bravais lattices?
Simple cubic, body-centred cubic (BCC), face-centred cubic (FCC), base-centred cubic.
What is a unit cell?
The smallest repeating structural unit in a crystalline solid.
How does atomic packing differ in crystalline and amorphous solids?
Crystalline solids have regular packing and uniform voids, while amorphous solids have random packing and polydisperse voids.
What structural model describes amorphous solids?
A random continuous network (RCN).
What drives changes in bulk properties of solids?
Order–disorder transitions.
Quartz vs Silica
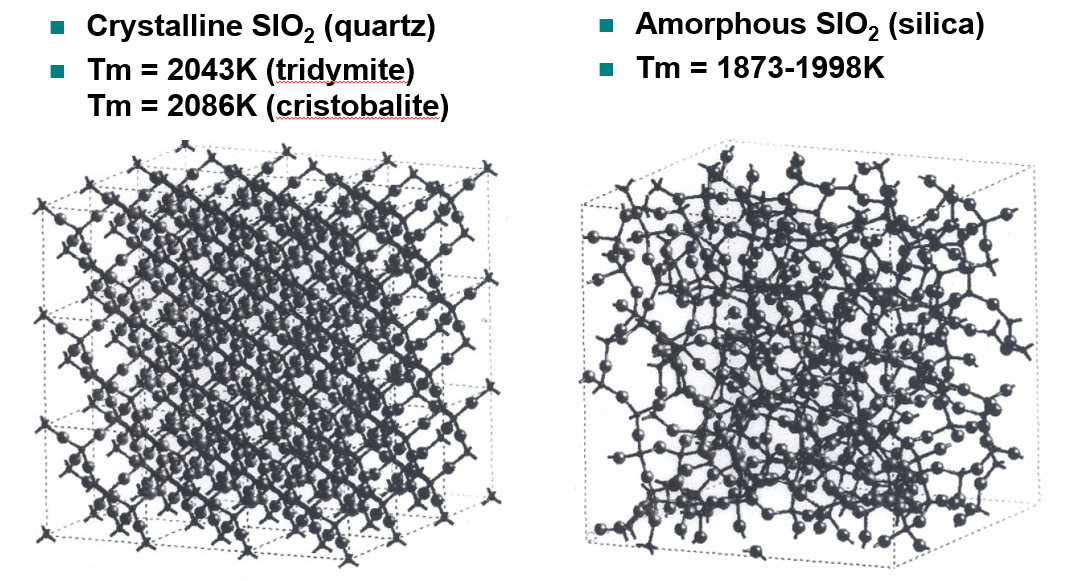
Why does amorphous SiO₂ have a lower melting temperature than crystalline forms?
Due to structural disorder and weaker collective bonding.
What is a network modifier in glass science?
A lower-valency cation added to a glass network that breaks bridging bonds.
How do network modifiers affect glass structure?
They introduce non-bridging oxygens, disrupting the network.
What is the effect of network modifiers on glass transition temperature (Tg)?
They lower Tg.
Why are network modifiers important industrially?
They reduce processing temperatures and improve glass workability.
What are the two main types of chemical analysis?
Qualitative
Quantative
What is qualitative analysis?
Identifies what substances are present.
What is quantitative analysis?
Determines how much of a substance is present.
What is a representative sample?
A sample that accurately reflects the composition of the entire material being analysed.
Why is sampling especially important for heterogeneous materials?
Because the composition varies throughout the material, so incorrect sampling can produce inaccurate results.
What is the purpose of the coning and quartering method?
To reduce sample size while maintaining a representative composition.
Why are samples dried before analysis?
To remove moisture content, which could affect mass measurements.
Give the methods used to separate analytes during sample pre-treatment.
Distillation
Filtration
Centrifugation
Solvent extraction
Why might analytes be concentrated before analysis?
Because chemical reactions and measurements are easier and more accurate in solution.
What is aqua regia?
A mixture of:
3 parts HCl
1 part HNO₃
Used to dissolve gold and other metals.
Why is calibration required in analytical chemistry?
To determine the relationship between instrument response and analyte concentration.
What is the matrix effect?
When other substances in the sample alter the detector signal for the analyte.
How can matrix effects be minimised?
Matrix matching
Standard addition method
What is volumetric analysis?
A method where the volume of a reagent needed to react with an analyte is measured.
What is a titrant?
The standard solution added during a titration.
What are the four common types of titration reactions?
Acid–base
Redox
Complexometric
Precipitation
What is the equivalence point in a titration?
The point where stoichiometrically equivalent amounts of titrant and analyte have reacted.
What is the end point in a titration?
The observable change indicating the titration is complete.
Examples:
Colour change
Voltage change
Absorbance change.
What causes titration error?
The difference between the equivalence point and the end point.
What is back titration?
A method where excess reagent is added to the analyte and the remaining excess is titrated.
Why is back titration sometimes used?
The direct titration endpoint is unclear
The reaction is slow
Excess reagent ensures complete reaction.
What is a primary standard?
A highly pure, stable substance used to prepare standard solutions.
What properties must a primary standard have?
High purity (≈99.9%)
Stable in air and solution
Known composition
Easily weighed.
What is a secondary standard?
A solution whose concentration is determined by standardisation with a primary standard.
What is measurement uncertainty?
The range within which the true value of a measurement is expected to lie.
How is instrument uncertainty determined?
Uncertainty = half the smallest scale division.
What is percentage error?

How can percentage error be reduced?
Use more precise equipment
Measure larger volumes
Repeat measurements.
What is a systematic error?
An error caused by consistent faults in equipment or method, producing biased results.
Give examples of systematic errors.
Faulty instrument calibration
Method error
Observer error.
What is a random error?
An error caused by uncontrolled variations in measurement, producing unpredictable results.
What is accuracy?
How close a measurement is to the true value.
What is precision?
How close repeated measurements are to each other.
Which type of error mainly affects precision?
Random errors.
How is total percentage uncertainty calculated?
Square each percentage error, sum them, then take the square root.
What characterizes a solid?
A solid is defined by its rigid shape and fixed volume, with particles that are tightly packed, allowing it to maintain its form independently of any container.