Unit 3 - Equilibrium, Acids and Bases
1/29
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
30 Terms
![<p>Concentration [ ] vs Time will plot a straight line for a reaction of the</p><p></p>](https://knowt-user-attachments.s3.amazonaws.com/bdfeb798-2d37-4ffb-a081-7c30d2e80360.jpg)
Concentration [ ] vs Time will plot a straight line for a reaction of the
Zero Order
![<p>ln[ ] vs Time will plot a straight line for a reaction of the</p>](https://knowt-user-attachments.s3.amazonaws.com/06554f1e-fa8b-4ce6-a4e9-b49c29d7d1a4.jpg)
ln[ ] vs Time will plot a straight line for a reaction of the
First Order
![<p>1/[ ] vs Time will plot a straight line for a reaction of the </p>](https://knowt-user-attachments.s3.amazonaws.com/11374eab-df1e-4a3f-9f1b-6202702d17e3.jpg)
1/[ ] vs Time will plot a straight line for a reaction of the
Second Order
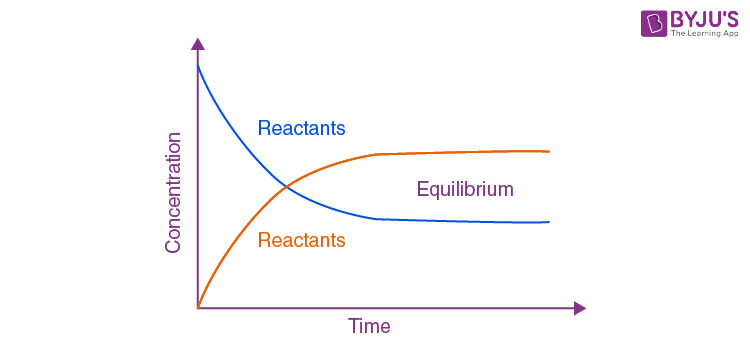
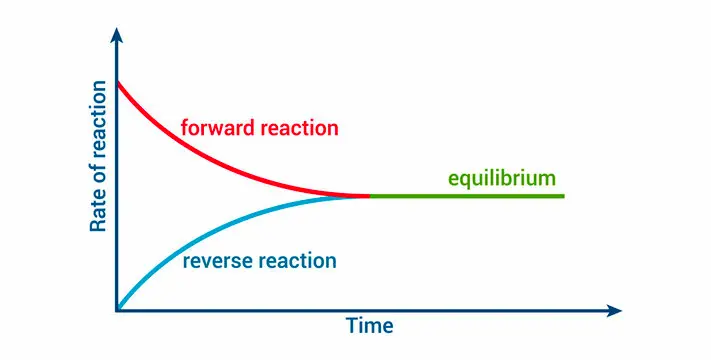
What is an equilibrium reaction, and what happens to products and reactants when reached?
A reaction that is reversible, and the forward ad reverse rates become equal at equilibrium.
Are the concentrations of reactants and products at equilibrium always equal? Why or why not?
No, because equilibrium calls for equal RATES, not concentrations. If reactants and products react at the same rate, they are balanced; even if equilibrium concentrations differ.
What is the equilibrium expression?
K={\frac{{[C]^c[D]^d}}{[A]^{a}[B]^{b}}}
where:
C and D = product concentrations
A and B = reactants concentrations
c, d, a, b = stoich coefficients
When can simplifying assumption be used?
if inital [ ] / k > 1000
How to check is x is valid in ICE table after using simplifying assumption
x / initial [ ] *100 < 5%
What is the Haber Process
Start w/ N2 from air and H2 natural gasses
Reacted in a reactor at 400 - 450ºC, 200 atm with iron catalyst
Products and reactants cooled, resulting in liquid ammonia- unreacted N2 and H2 gas recycled to be used again
The rxn 2 reactants → 1 product has a specific Kc. Find:
Kc for 4 reactants → 2 products
Kc for 1 product → 2 reactants
Kc for 2 product → 4 reactants
(Kc)² → [prdts]² / [rxts']4 = ([prdts] / [rxts']2)²
Kc-1
(Kc-1)² = Kc-2
What is the difference between Q and K?
Q compares the ratio of products to reactants at any given time, while K compares this ratio at equilibrium.
Can compare Q and K to find which direction system will favor
Kp and Kc equation
Kp = Kc(RT)Δn
R is 8.314
Kp in kPa
Le Chatelier stresses
Adding/removing species
Changing temp
Changing pressure
Adding catalyst
Response if volume of rxn container increases
Favors side with more mols:
↑V, ↓P, all partial Ps decrease
Side with more mols is slowed more relative to equilib due to exponents making more affected
Rxn favors side w more mols to increase rate
Response if volume of rxn container decreases
Favors side w less mols:
↓V, ↑P, all partial Ps increase
Side with less mols rate relatively slower, bc side w more mols rate increases much more due to exponents
To catch up to equilib, rxn favors side w less mols
Response if inert gas is added at constant volume
No response.
Inert gas in same volume ↑P, but all partial pressures remain same
Therefore no affect on Q and stays at equilib
Response if inert gas added at constant pressure
Favors side w more mols:
Volume changes to maintain pressure with added inert gas
↑V, all partial pressures decrease
Side w more mols rate affected more (slower)
Rxn favors side w more mols
According to Lewis theory, define acid and base
Lewis acid: Electron pair ACCEPTOR
Lewis base: Electron pair DONOR
What is an oxyacid?
Acid where acidic H is bonded to O, e.g. H3PO4

How do bond strength and polarity affect acid strength?
Bond strength: More strong bonds with central atom = WEAKER acid, harder to peel H+ off
Smaller central atoms increase bond strength (more e- overlap)
Bond polarity: More polar H-X bond (x is electronegative element) = STRONGER acid, water molecules more attracted to more positive H+
Bonded atom and number of non-protonated oxygens pull e- cloud away from H+
Percent ionization formula
[H3O+]/[HA] * 100
How does dilution affect percent ionization
Increases percent ionization
Diluting decreases all concentrations, and product rate is more affected
LCP, system favours products (ions)
↑[A-], ↑% ionization
Is the reverse or forward reaction favoured + why?
CH3COOH+OH−⇌CH3COO−+H2O
Ka(CH₃COOH) = 1.8×10−5
Ka(H₂O) = 1.0×10−14
Forward, bc Ka(CH₃COOH) > Ka(H₂O) → products of forward rxn more favoured
Proof for autoionization of water
for any conjugate pair:
Kw = Ka * Kb
=\left(\frac{\left\lbrack H3O+\right\rbrack\left\lbrack A-\right\rbrack}{\left\lbrack HA\right\rbrack}\right)\left(\frac{\left\lbrack HA\right\rbrack\left\lbrack OH-\right\rbrack}{\left\lbrack A-\right\rbrack}\right)
= [H3O+][OH-] = Kw
Is this salt acidic or basic?
NH₄Cl
Break down into ions:
NH₄+ is weak acid, Cl- is extremely weak base
NH₄+ can react with water, makes salt sol’n ACIDIC
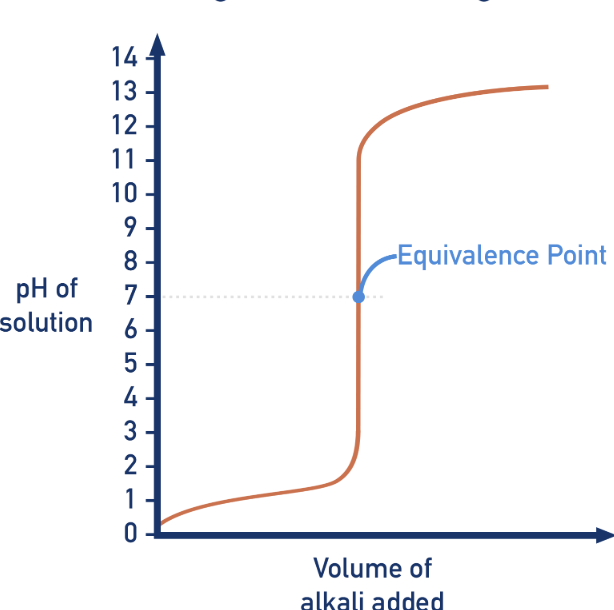
What type of titration?
Strong acid-strong base
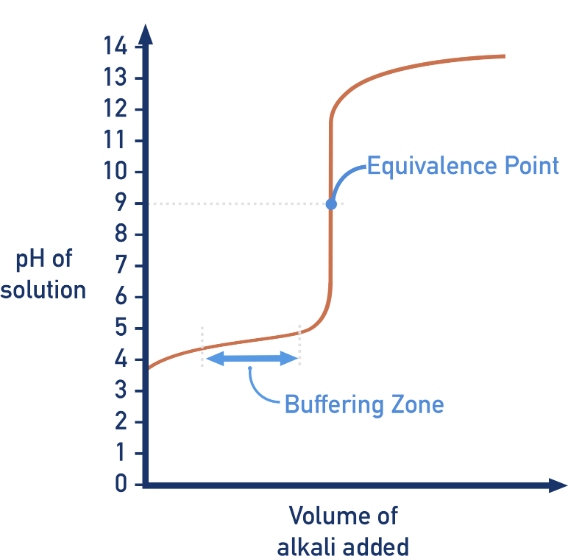
What type of titration?
Weak acid-strong base
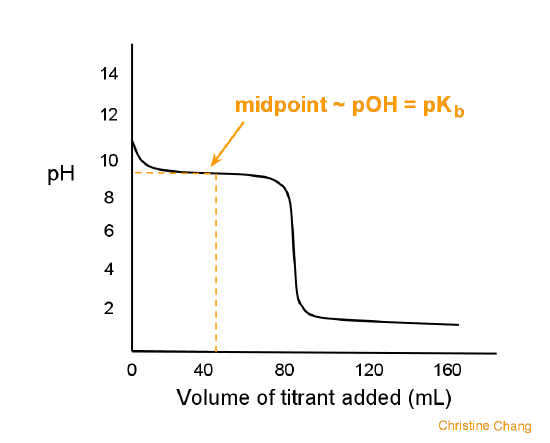
What type of titration?
Weak base-strong acid
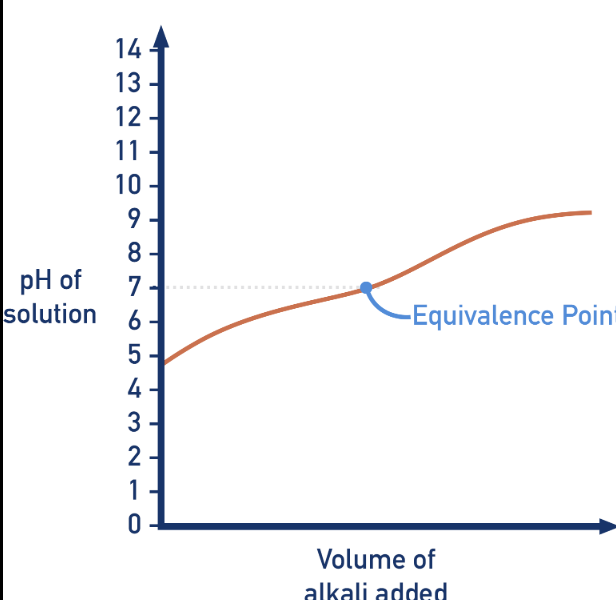
What type of titration?
weak acid-weak base