Hormonal control of growth and puberty
1/65
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
66 Terms
Growth requires
net synthesis of proteins
lengthening of the long bones
increase in number and size of cells comprising soft tissue
Growth in humans
continuous process
begins before birth
growth rates in children not steady
spurts of growth and development
requires cooperation of several endocrine organs
growth rate
not continuous
factors responsible for promoting growth are not the same throughout growth period
fetal growth
promotes largely by hormones from placenta
GH plays no role in fetal development
postnatal growth
displayed during first two years of life
pubertal growth
occurs during adolescence
normal growth rate depends on
hormones
Children do not grow without adequate GH
Thyroid hormone, insulin and sex hormones at puberty play direct and permissive roles
Deficiency leads to abnormal growth and development
genetic
potential adult size genetically determined at conception
absence of stress
cortisol released from adrenals during stress is catabolic and inhibits growth
adequate diet
require adequate protein, calorie intake, vitamins and minerals
obtained from food or manufactured in body
growth hormone
produced by somatotrophs of anterior pituitary
release GH periodically throughout the day
peak secretion during sleep
is anabolic hormones
major targets are bone and skeletal muscle
stimulation of the epiphyseal plate leads to long bone growth
stimulation of skeletal muscles promotes increased muscle mass
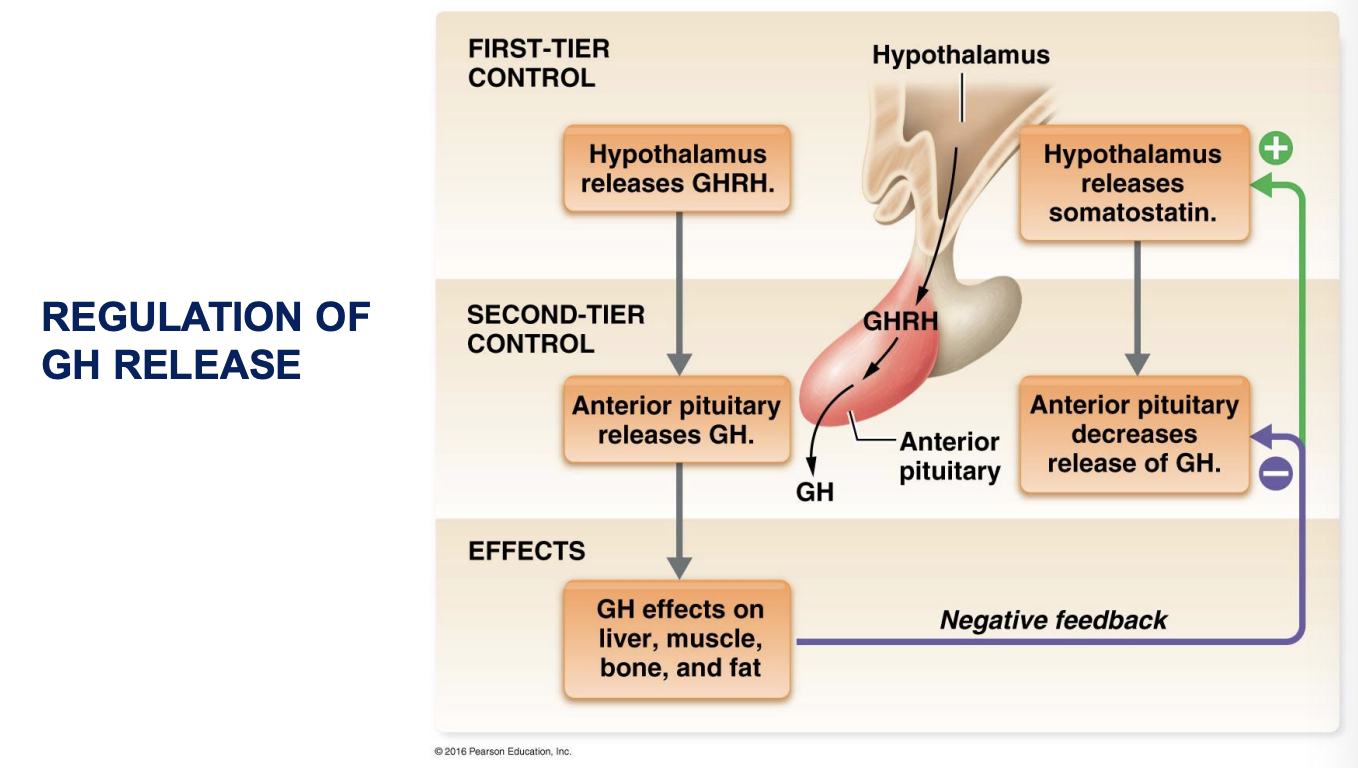
regulated by two hypothalamic hormones with antagonistic effects
growth hormone releasing hormone stimulated GH release (GHRH)
growth hormone inhibiting hormone inhibits GH release (GHIH)
Regulation of GH release
mechanism of GH action
indirect actions
via insulin-like growth factors
acts on wide variety of cell types
direct actions
more selective
involved in regulation of blood glucose and amino acid concentrations
cell division and differentiation
Insulin-like growth factor-1
Produced by liver in response to GH stimulation
Mediates most of GH’s actions
Synthesis affected by
Age
Increases at puberty – corresponds to increase in GH
Nutrition
Inadequate nutrition reduces production despite GH levels increasing
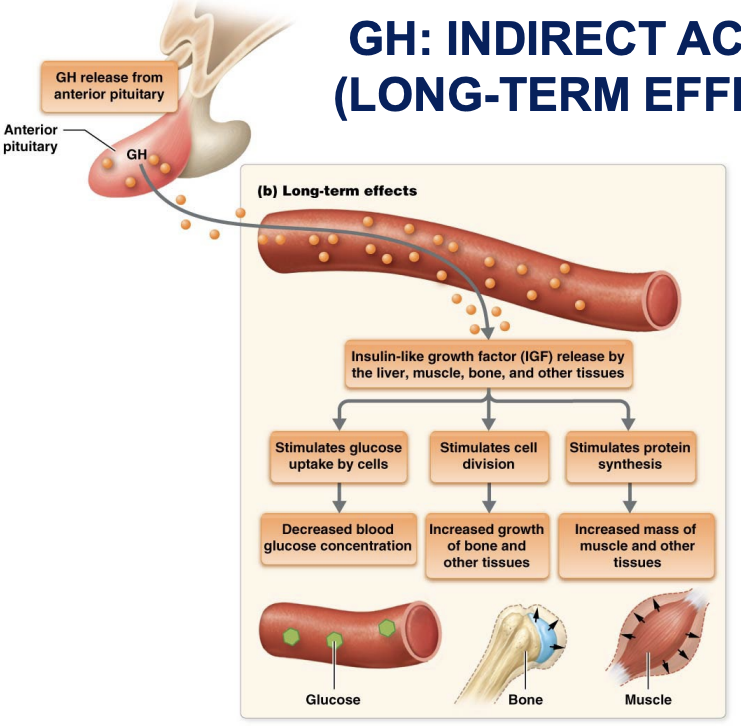
GH: indirect action long term effects
Promotes growth indirectly
stimulates liver’s production of somatomedins
Primary somatomedin is insulin-like growth factor (IGF-1) which
Acts directly on bone and soft tissues to bring about most growth promoting actions
Stimulates protein synthesis, cell division, and lengthening and thickening of bones
Stimulates uptake of sulphur into cartilage matrix
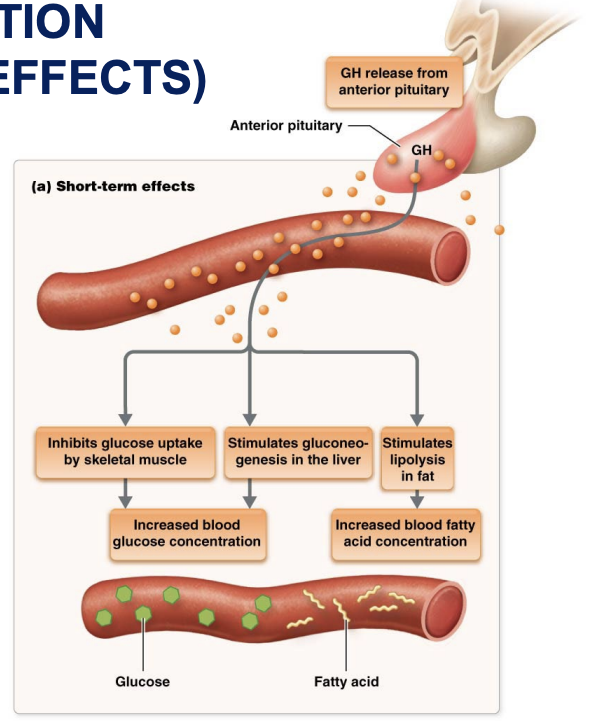
GH: direct action short term effects
Increases fatty acid levels in blood
enhances breakdown of triglyceride fat stored in adipose tissue
Increases blood glucose levels
decreases glucose uptake by muscles
Stimulates stem cell division and differentiation in epithelial and connective tissue
Effects of GH on soft tissue
Hyperplasia: Increases cell number
Stimulates cell division
Prevents apoptosis (cell death)
Hypertrophy: Increases cell size
Promotes protein synthesis
Inhibits protein degradation
Increases uptake of amino acids by cells
Stimulates the cellular mechanisms responsible for protein synthesis
Effects of GH on bone
stimulates osteoblast activity (formation of bone tissue)
promotes proliferation of epiphyseal cartilage
promotes lengthening of bone if epiphyseal plate remains “open” ie cartilaginous
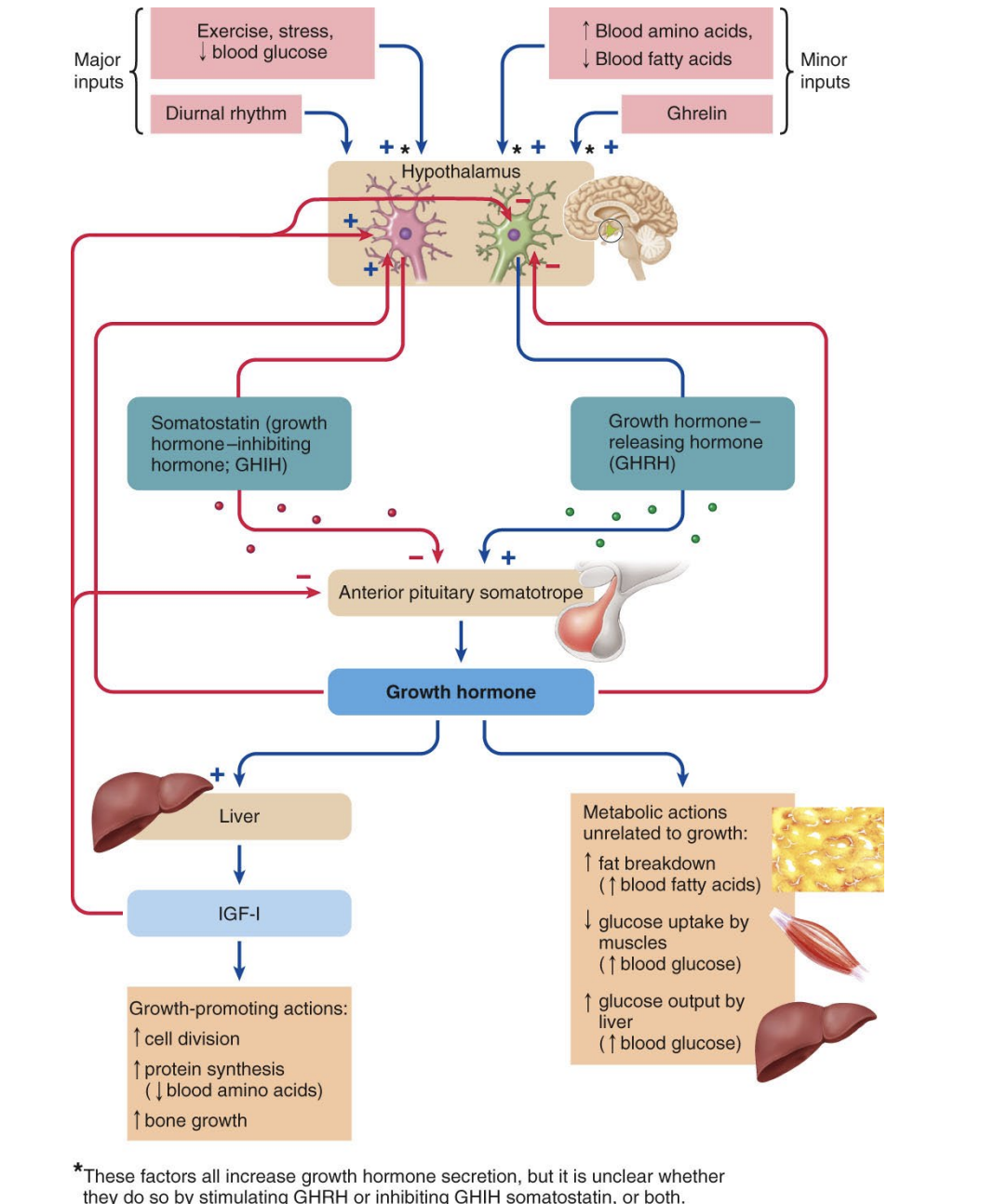
GH regulation
Regulated by two hypothalamic hormones
Growth hormone-releasing hormone (GHRH)
Somatostatin
GHRH
stimulates release of GH
secretion increases during exercise, fasting, and stress, and after ingestion of a protein-rich meal
GH release is inhibited by hypothalamic hormone somatostatin
GH and diurnal rhythm
GH levels low and constant during the day
GH levels increase sharply after onset of deep sleep (5X daytime levels)
GH levels then drop over the next few hours back to daytime levels
Factors affecting GH secretion
Levels of GH are increased when energy demands are greater than available glucose reserves
Low blood glucose
Exercise
Stress
GH increased under these conditions to
Conserve glucose for use by the brain
Make fatty acids available for use as an energy source for muscles
Factors affecting GH secretion
Levels of GH are increased:
After a high protein meal
Amino acids used for protein synthesis
When blood fatty acids levels decline
Metabolises fat – releasing fatty acids – keeps blood levels constant
Factors affecting GH secretion
Regulating levels of GH aimed at controlling levels of
Amino acids
Fatty acids
Glucose
Normal growth also requires
Insulin
thyroid hormone
parathyroid hormone and calcitonin
reproductive hormones
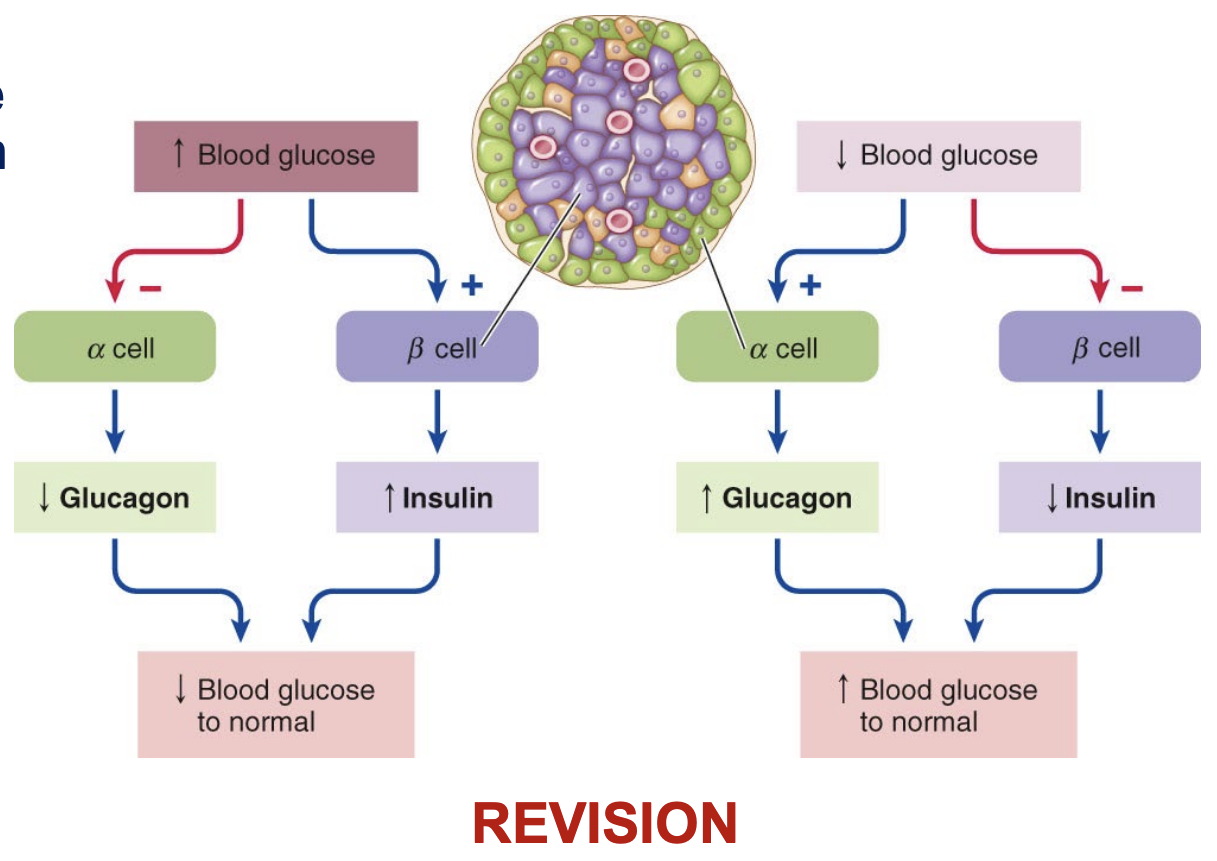
Insulin
Produced by β-cells of the pancreas
Decreases blood glucose levels when they get high
Promotes movement of glucose through the cell membranes
Stimulates the storage of both glucose and fats
Stimulates glycogen and protein synthesis
Regulation of insulin is via negative feedback mechanisms
Thyroid hormone
Consists of an amino acid core bound to either 3 (triiodothyroxine, T3) or 4 (thyroxine, T4) iodine atoms
Both T3 and T4 are physiologically active but T3 activity is greater
T4 is commonly converted to T3 in some target tissues
Both T3 and T4 enter target cell nucleus
they bind with receptors that either activate or inhibit specific gene transcription
Effects of thyroid hormone
Almost every cell in body contains thyroid hormone receptors
makes their effects widespread
Three main categories of effects
Regulation of metabolic rate and thermoregulation
Promotion of growth and development
Synergism with sympathetic nervous system
Regulation of metabolic rate and thermoregulation
Thyroid hormones set basal metabolic Promotion of growth and development (amount of energy required by body at rest) by
increasing rate of ATP consumption
increasing gluconeogenesis
initiating energy-requiring reactions in these same target cells
Heat is generated
critical for core body temperature homeostasis
Promotion of growth and development
Thyroid hormones are required for
normal bone growth
muscle growth
nervous system development
Reproductive capabilities
Enhances protein synthesis and lipid breakdown
Synergism with sympathetic nervous system
Increases in thyroid hormone levels act on target cells of sympathetic nervous system
increase (up-regulate) receptors for sympathetic neurotransmitters
affects regulation of blood pressure, heart rate, and other sympathetic activities
PTH and calcitonin
Promotes the absorption of calcium salts (PTH) and deposition in bone (calcitonin)
In the absence of adequate levels of PTH and calcitonin
Bones can still enlarge
Poorly mineralized , weak flexible
Reproductive hormones that effect growth
Testosterone
anabolic
stimulates bone growth
stimulates growth in length
increases muscle mass and weight
Controlled by negative feedback mechanisms
Estrogen
Not involved in growth stimulation
Causes growth plates to close
Stops growth in length
Controlled by negative feedback mechanisms
Puberty
Stage of physical maturation in which an individual becomes physiologically capable of sexual reproduction
Onset varies among individuals
May occur anytime from age 10 to 15
Usually begins a year earlier in females than in males
Lasts 3 – 5 years
Physical changes of puberty
female secondary sex characteristics
male secondary sex characteristics
Biological changes of puberty
Neurosecretory factors and /or hormones
Modulation of somatic growth
Initiation of development of the sex glands
Major hormones of pubery
GnRH
LH and FSH
Estrogen and progesterone
testosterone
Physiology of puberty
Activation of the hypothalamic-pituitary-gonadal axis
Induces ovarian and testicular sex hormone secretion
Responsible for the biological, morphological and psychological changes during puberty
Sex steroid production
Appearance and maintenance of sexual characteristics
Capacity for reproduction
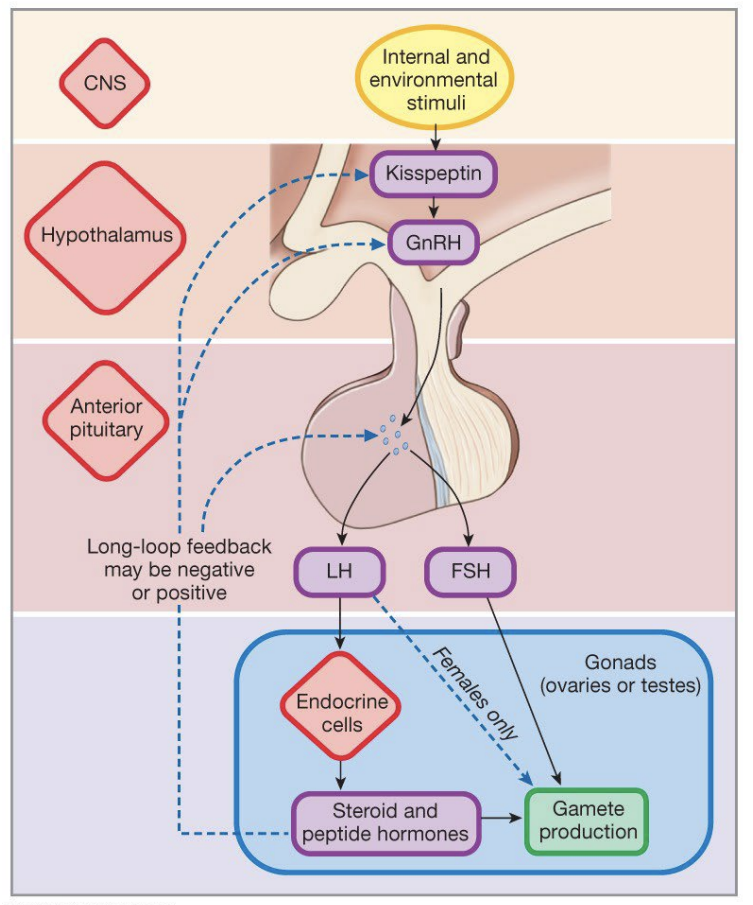
Hypothalamic-pituitary gonadal axis
Major physiological function in both males and females
Development of primary and secondary sexual characteristics
Control of gametogenesis and reproduction
Hypothalamic-pituitary gonadal axis is active when?
Active in males and females during three main periods of life
Midtrimester of the fetal period
Early in the neonatal period
From puberty throughout the reproductive years
Fetal period of Hypothalamic-pituitary gonadal axis
Testosterone production is essential for sexual differentiation in males
Elevated levels of FSH contributes to folliculogenesis in females
Silenced towards term because of the negative feedback effects mediated by the placental hormones
Neonatal period of Hypothalamic-pituitary gonadal axis
Increased activity due to decreased placental hormone levels at birth – disinhibits the hypothalamo-pituitary system
Puberty to adulthood period of Hypothalamic-pituitary gonadal axis
Increased pulsatile release of GnRH occurs mostly at night in early puberty
Required for FSH and LH secretion
Later – pulsatile release of GnRH occurs throughout the 24 day
Initiation of puberty
Factors unclear in humans
Leptin - produced by adipocytes - thought to play a role in females
Postulated that melatonin stimulates onset
Melatonin is anti-gonadotropic (inhibit effect of gonadotropic hormones)
Some evidence of reduced melatonin secretion at puberty in humans – particularly at night – when GnRH peaks
Male reproductive system
Consists of: testes, duct system, accessory glands, and penis
From testis, sperm travels within male reproductive duct system (epididymis, ductus deferens and urethra, before leaving the body via the penis)
Accessory glands – seminal glands, the prostrate and the bulbourethral gland secrete fluids into the duct system.
External genitalia – scrotum that enclose the testes, urethra and penis
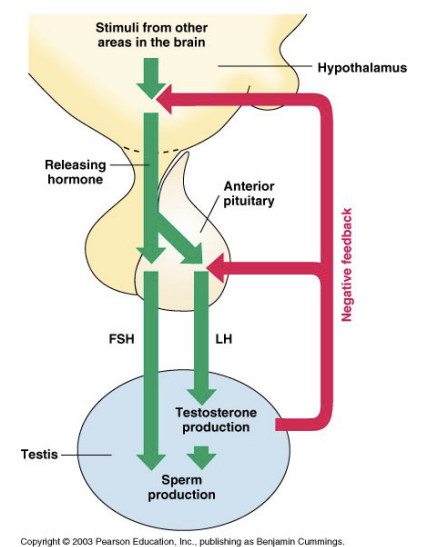
Hormones and male reproductive function
GnRH
Produced by the hypothalamus
Stimulates the release of LH and FSH by anterior pituitary
LH and FSH act on separate components of the testes
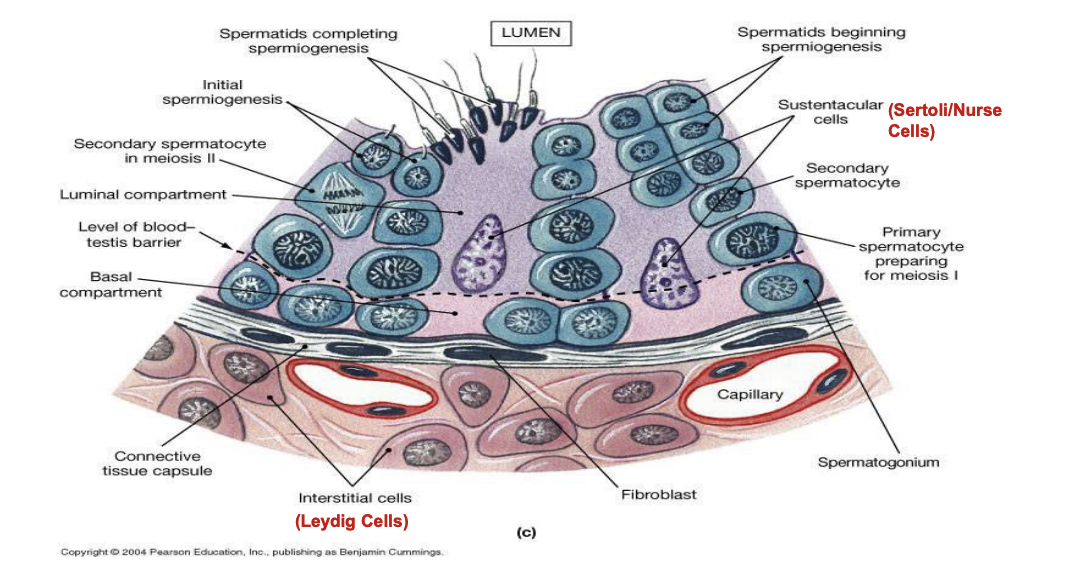
Seminiferous tubule
sertol/nurse cells are important
leydig cells are important aka interstitial cells #
Hormones and male reproductive function
LH
Acts on interstitial (Leydig) cells
Regulates testosterone secretion
Interstitial cell-stimulating hormone (ICSH) – alternate name in males
FSH
Acts on seminiferous tubules – specifically Sertoli cells
Enhances spermatogenesis
Brain-testicular axis
Hormonal regulation of spermatogenesis and testicular androgen production involves interactions between
Hypothalamus
Anterior Pituitary
Testes
Relationship called brain-testicular-axis
Hormones involved in brain-testicular axis
Testosterone
main hormone involved in regulation of spermatogenesis and male reproductive physiology
regulated by multi-tiered negative feedback loop
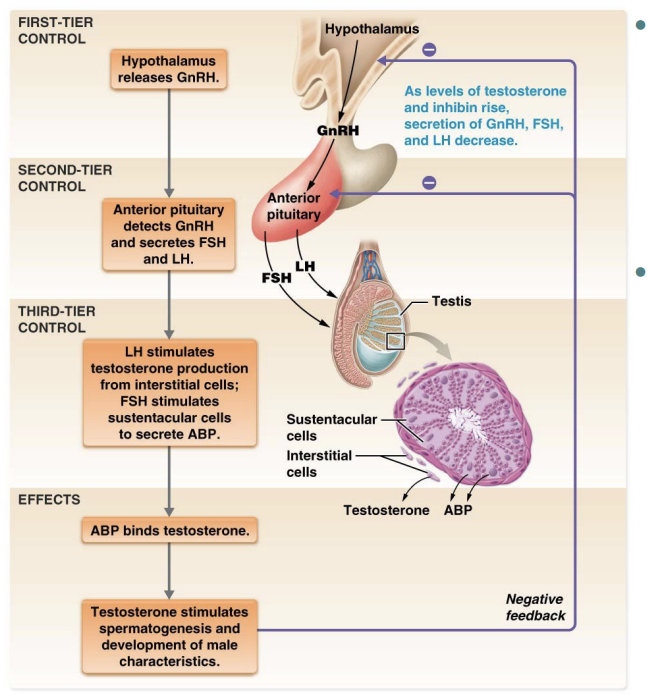
Brain-testicular-axis
regulates hormones involved in testosterone production and testicular function
First-tier control – Gonadotropin-releasing hormone (GnRH), secreted by hypothalamus
Second-tier control – anterior pituitary detects GnRH; stimulates secretion of gonadotropins
Third-tier control – in testes: LH, FSH, stimulates secretion of inhibin, androgen binding protein, testosterone
Brain-testicular axis: FSH
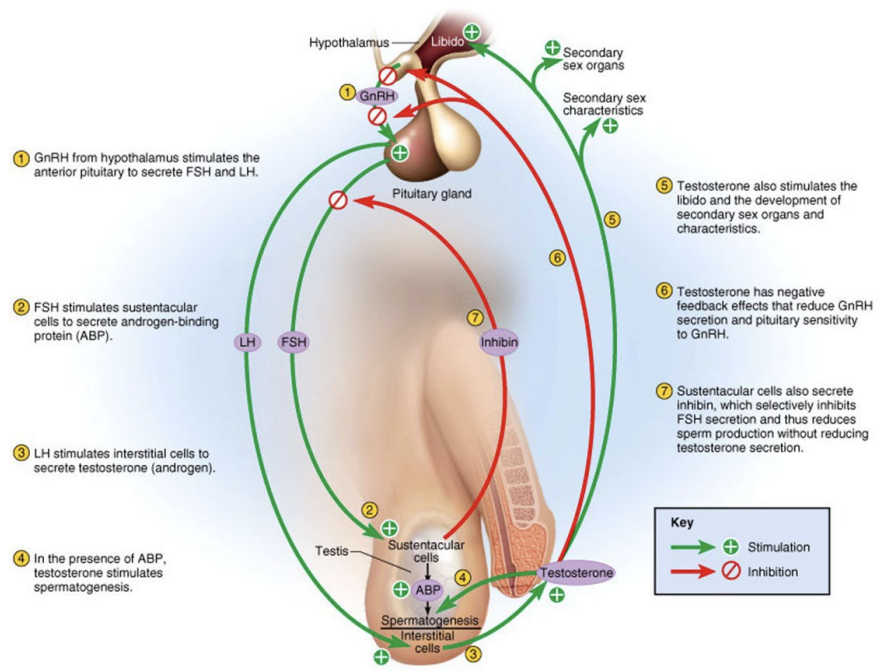
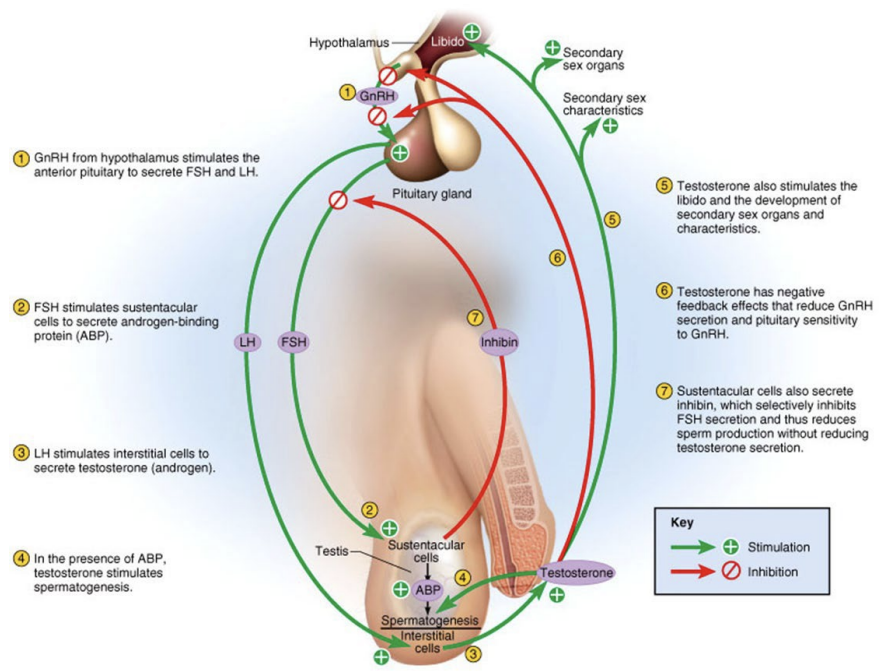
FSH stimulates spermatogenesis indirectly
Stimulates Sertoli cells to release androgen-binding protein (ABP)
ABP binds to and concentrates testosterone in spermatogenic cells
ABP-testosterone complex stimulates spermatogenesis
FSH directly makes Sertoli cells receptive to stimulatory effect of testosterone
androgen = male hormone
Brain-testicular axis: LH
LH binds to interstitial cells
Stimulates them to secrete testosterone
Locally testosterone is the final trigger for spermatogenesis
Testosterone entering the blood-stream exerts a number of effects at other body sites
Brain-testicular axis: Inhibin
Inhibin – peptide hormone secreted by the Sertoli cells
High sperm count – increases inhibin release
Inhibits anterior pituitary release of FSH
Increased levels of inhibin decreases spermatogenesis
Sperm count < 20 million/ml – inhibin secretion declines steeply
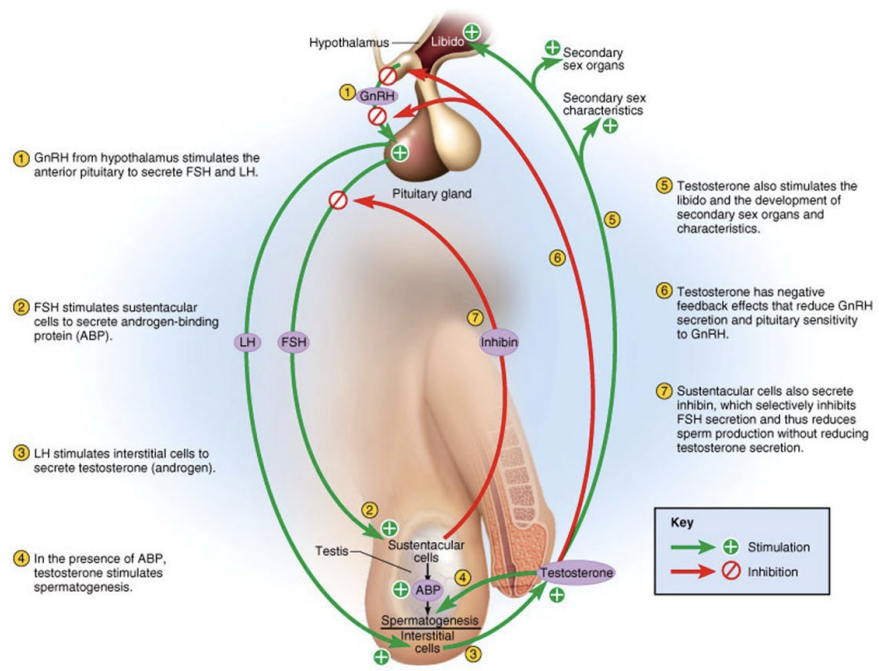
Brain-testicular axis: Testosterone
Testosterone: negatively inhibits LH in TWO ways
Acts on the hypothalamus
inhibits GnRH
Indirectly decreases LH and FSH secretion by the anterior pituitary
Acts directly on the anterior pituitary
Reduces response of LH secretory cells to GnRH
Exerts a greater inhibitory effect on LH than FSH
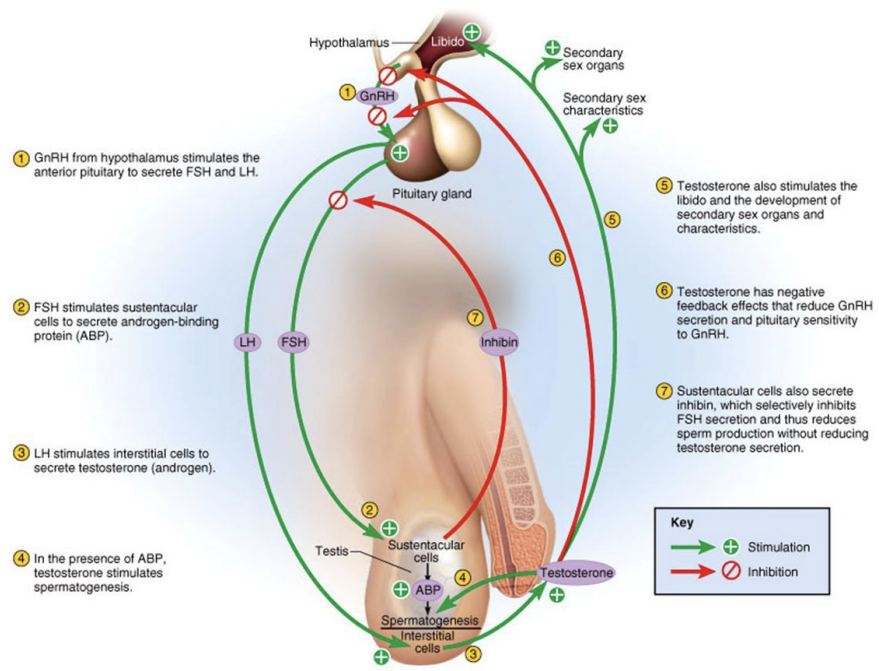
Three sets of hormones that balance amount of testosterone and sperm produced
GnRH – indirectly stimulates testes via effects on LH and FSH release
Gonadotropins (LH and FSH) – directly stimulate the testes
Testosterone and inhibin – exert negative feedback controls on the hypothalamus and anterior pituitary
In absence of GnRH and gonadotropins
testes atrophy
sperm and testosterone production ceases
Testosterone
synthesised from cholesterol
~98% circulated in blood
bound to one of two transport proteins – protected from metabolism in liver
Sex hormone binding globulin (SHBG) [~44%]
Serum Albumin (~54%)
Exerts it effect by activating specific genes
Testosterone: effects on reproductive organs
Targets accessory reproductive organs – causes them to grow and assume adult size and function
Ducts
Glands
Penis
In adult males – normal plasma levels of testosterone maintains these organs
When testosterone is deficient or absent
All accessory organs atrophy
Semen volume decline
Erection and ejaculation impaired
Sterility and impotence
Treated with testosterone replacement therapy
Testosterone and male 2nd sex characteristics
Appearance of pubic, axillary and facial hair
Enhanced hair growth on the chest and other body areas (in some men)
Deepening of the voice as the larynx enlarges
Skin thickens and becomes oilier
Testosterone and somatic effects
Thickening and strengthening of the bones
Skeletal muscle increases in size and mass
Epiphyseal plate closure occurs late in puberty due to increased estrogen levels (bone growth stops)
Testosterone and metabolic effects
Anabolic
Stimulates hematopoiesis (production of blood cells)
Enhances basal metabolic rate
Testosterone and neural effects
Responsible for libido in males
Masculinizes the brain (e.g. differences in male and female brain areas in response to sexual arousal)
Promotes aggressiveness
Hormones and the female reproductive cycle
Regulatory pattern more complicated than in males
Interplay of secretions from pituitary and gonads control the female reproductive cycle
Coordinates both the ovarian and uterine cycle
Infertility results if cycles not coordinated
Female reproductive cycle (leptin)
Onset of puberty linked to adiposity
Adipose tissue produces leptin – which acts on the hypothalamus
Leptin stimulates the hypothalamus to secrete GnRH
Low blood levels of lipids and leptin delays puberty
Female reproductive cycle
GnRH stimulates the anterior pituitary to produce LH and FSH
LH and FSH stimulate the ovaries to produce estrogen and progesterone
Hormones interact to produce the cyclic events occurring in the ovaries
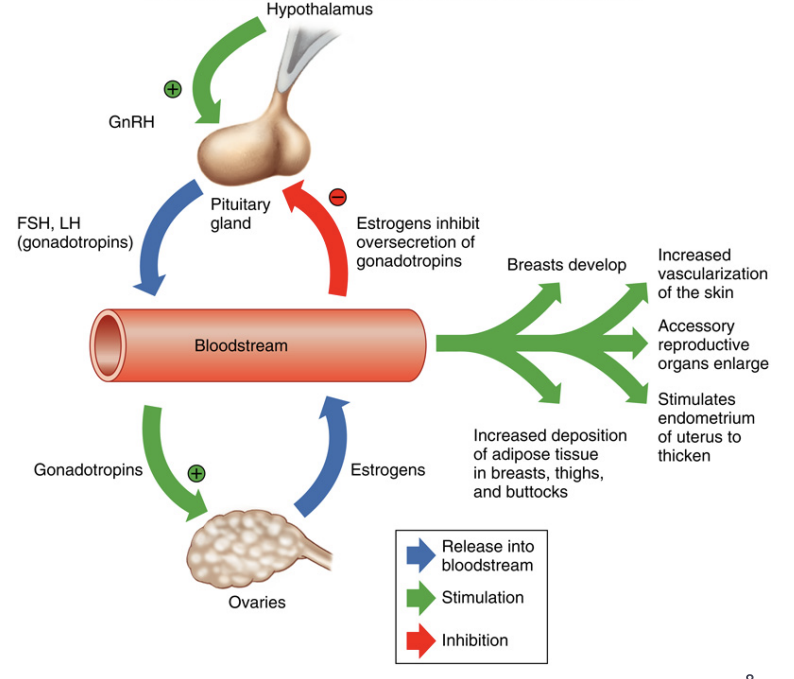
Female reproductive cycle (ovary)
Ovary has TWO related endocrine units
Estrogen-secreting follicle during the first half of the cycle
Follicular Phase
Corpus luteum which secretes both progesterone and estrogen during the second half of the cycle
Luteal Phase
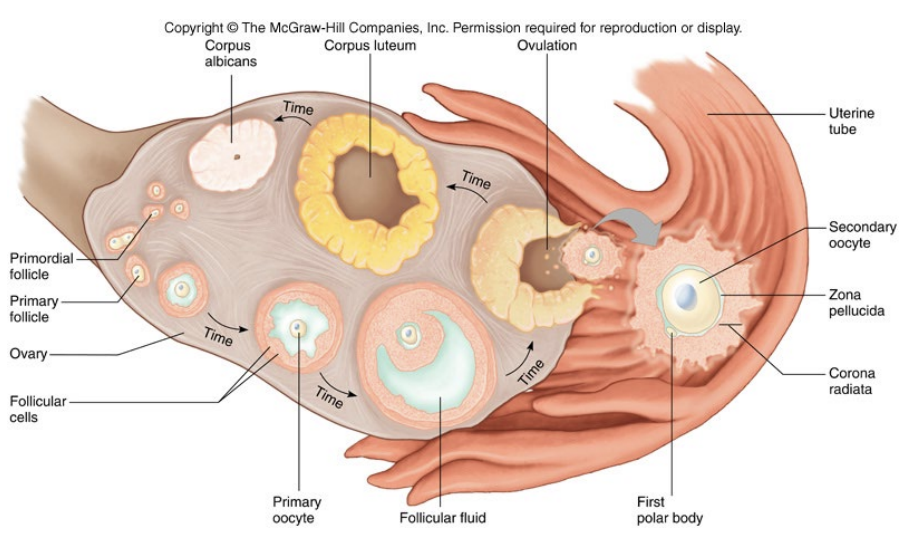
Phases of the ovarian cycle
Follicular Phase
Development of the follicle
Secretion of estrogen from follicle
Ovulation
Occurs at mid-cycle
Ejection of egg from ovary
Luteal Phase
Secretion of estrogens and progesterone from the corpus luteum (previously the follicle) after ovulation
Early follicular phase
Early in the follicular phase
Estrogen levels are low
GnRH pulse is low
FSH secretion > LH secretion by anterior pituitary
Estrogens released by developing follicles inhibits LH secretion
As secondary follicles develop
FSH levels decline due to negative feedback effects of inhibin and estrogen
Follicular development and maturation continue
Supported by combination of estrogens, FSH and LH
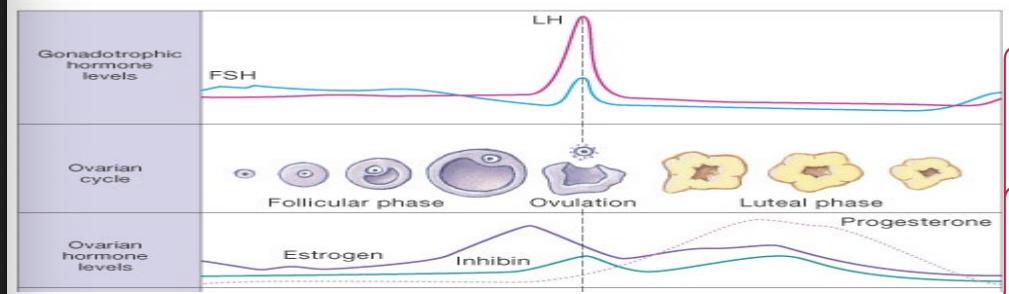
Late follicular phase
As tertiary follicles begin forming
Concentration of estrogens rises sharply
GnRH pulse frequency increases and stimulates LH secretion
At roughly day 10 of the cycle – effect of estrogen on LH changes from inhibition to stimulation
High estrogen levels ↑ gonadotrope sensitivity to GnRH
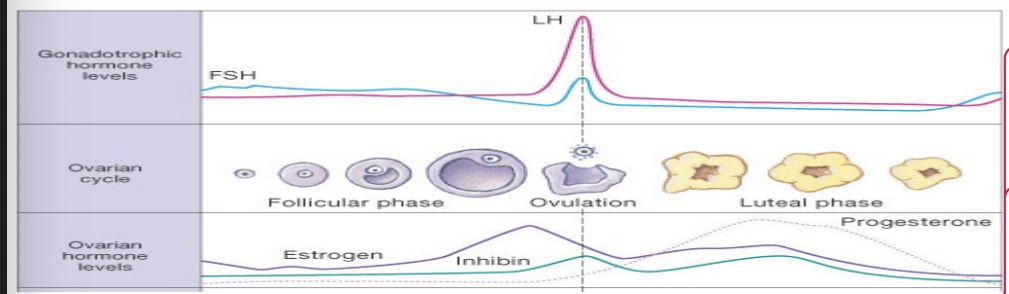
Ovulation
At about day 14
Estrogen levels peak
Gonadotropes are at maximum sensitivity
GnRH pulse frequency is high
Result
Massive release of LH from anterior pituitary gland
Sudden surge of LH triggers
Rupture of the follicular wall
Ovulation
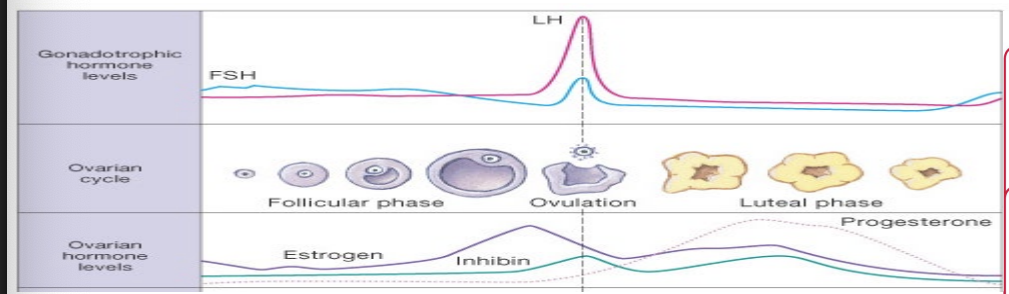
Luteal phase
High LH levels that trigger ovulation
Formation of the corpus luteum
Corpus luteum secretes progesterone, estrogen and inhibin
As progesterone and estrogen levels rise
Negatively feeds back on anterior pituitary and hypothalamus
GnRH pulse frequency declines to very low levels and stimulates LH secretion more than FSH secretion
LH (low levels) maintains the structure and secretory function of the corpus luteum
Progesterone - Main hormone of the luteal phase
Primary function - Prepare the uterus for pregnancy
Progesterone levels remains high for a week
If pregnancy does not occur - Corpus luteum begins to degenerate 12 days after ovulation
Estrogen and reproductive organs
Promote oogenesis and follicle growth in the ovary
Exert anabolic effects on the female reproductive tract – grow larger and become functional – to support pregnancy
Uterine tubes
Uterus
vagina
Enhanced motility of the uterus and uterine tubes
Vaginal mucosa thickens – and external genitalia mature
Estrogen and 2nd sexual characteristics
Growth of breasts
Increased deposit of subcutaneous fat (hips and breasts)
Widening of pelvis
DHEA (adrenal androgen) and not estrogen causes growth of axillary and pubic hair
Estrogen and growth
Not involved in stimulating growth
Adrenal androgen DHEA causes female growth spurt
Stops growth in length by causing growth plates to close