BIOL 2056 - Vesicle transport, cargo delivery and cytoskeletal components
1/26
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
27 Terms
vesicle transport pathways
helps to compartmentalise the cell —> regulates transcription, translation, modification, trafficking of proteins
mechanism or turning on/off signal transduction
pH important in compartmentalisation as it alters the affinity of molecules for their receptors
SECRETORY PATHWAY
flow of membrane bound or soluble proteins destined for certain organelles
ENDOCYTIC PATHWAY
plasma membrane capture of EC components and internalisation of membrane proteins into vesicles
recycling of receptors or degradation of lysosome contents
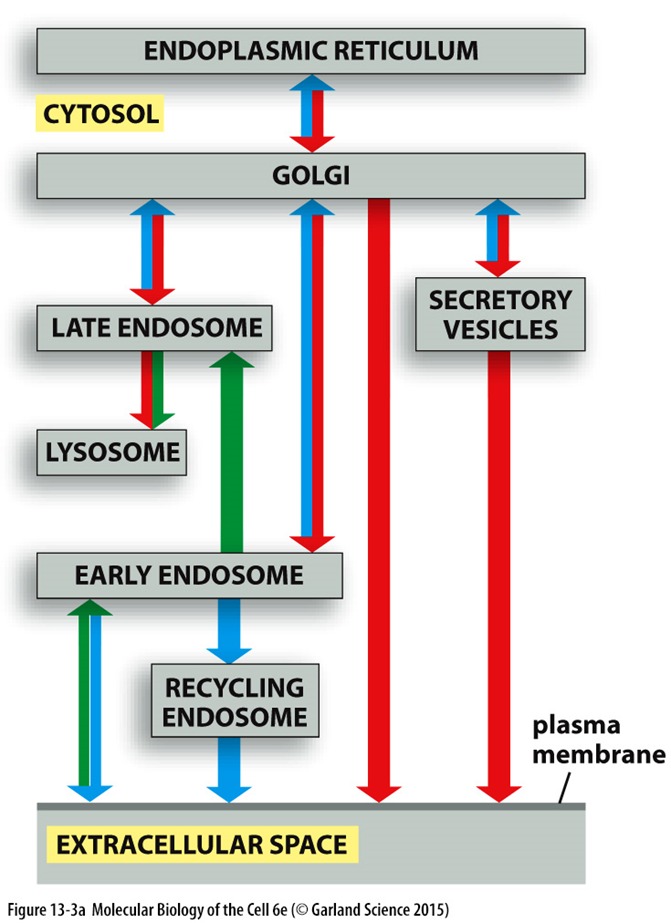
requirements of vesicle transport
BIDIRECTIONAL FLOW
cells dont change size, there’s a constant flow
lipid and protein components maintained
ID OF SPECIFIC CARGO
SORTING OF VESICLES AND ASSOCIATED CARGO
TRANSPORT
TRANSFER OF MATERIAL
fission
tethering
fusion
vesicle coat protiens
CLATHRIN
to the golgi from plasma memb
found on the early endosome, matures into the lysosome where a protein goes to a lysosome or to the trans golgi face is an adaptor p protein
ARF GTPase
COP 1
golgi complex to the ER (retrieval)
heptameric coatamers and ARF GTPase
COP 2
goes from the Er to the golgi
sec 13/14 and sec 23/24 w associated GTPase
protein interactions with membranes
may be anchored with amphipathic alpha helix
may be post translational modification of lipid anchor
may be non covalent interactions w other memb
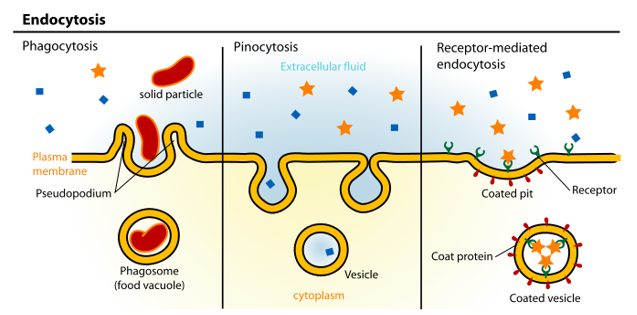
types of endocytosis
receptor mediated —> triggered by a receptor embedded into the membrane
clathrin dependent
caveolin dependent
clathrin caveolin independent
phagocytosis
pinocytosis
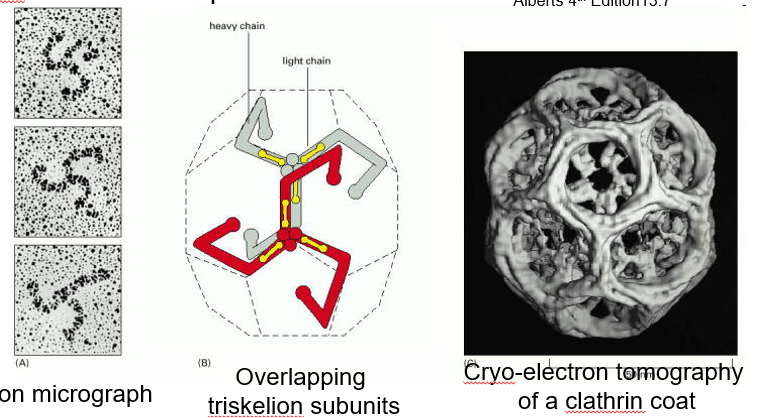
clathrin coat
CLATHRIN COAT
made of 3 large chain and 3 light chain polypeptides that assemble in triskeletons of the trans golgi network or at the plasma membrane
these clathrins then form an outer protein lattice
FORMATION
mediated by adaptor protein complex —> required for clathrin recruitment, coat assembly and eventual budding
lipid modification sites of phosphoinositides on the cell membrane allow for attachment of APs.
AP2 adaptor protiens bind to specific phospholipids and cause a conformational change which allows:
binding to cargo receptors on cells surface
triggers membrane curvature
recruitment of clathrin
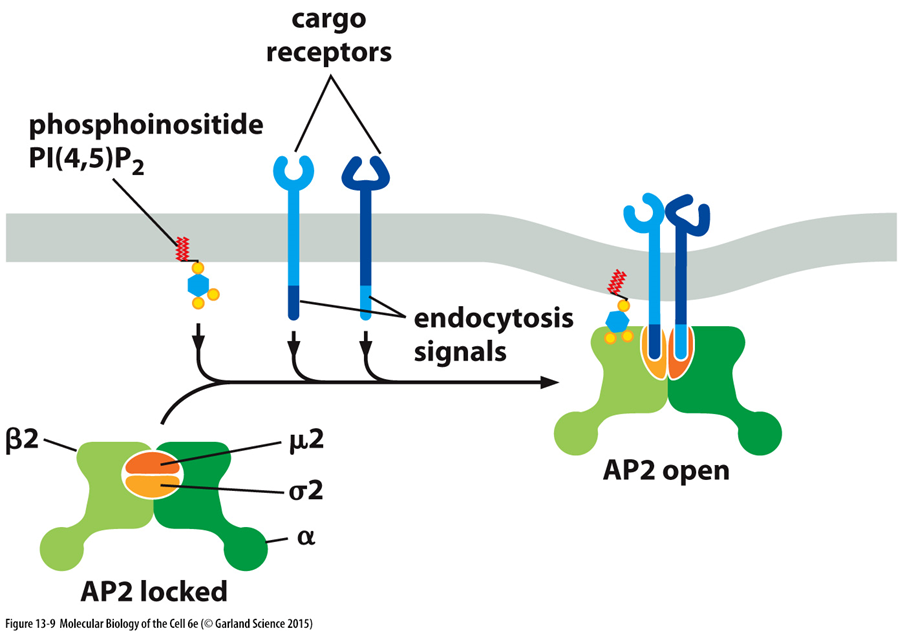
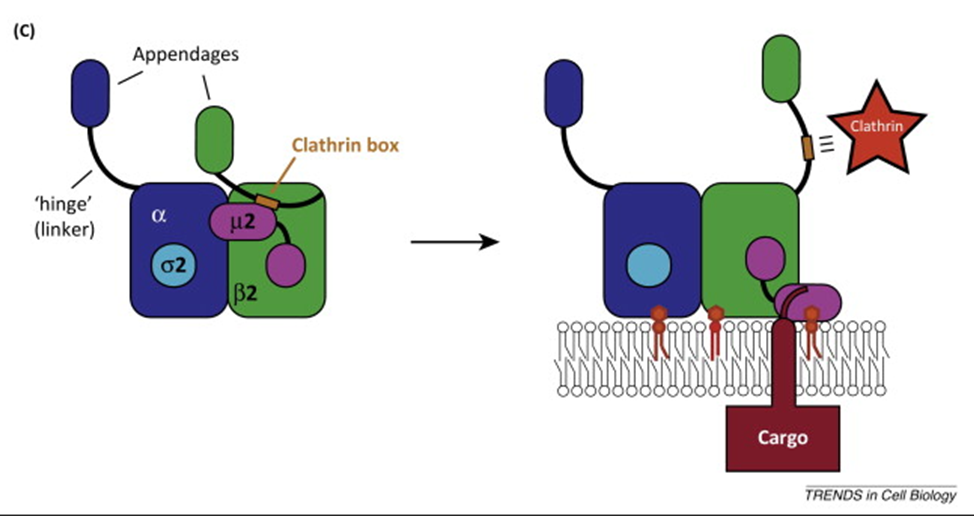
AP2 adaptor protein complex
AP2 ADAPTOR PROETIN COMPLEX
heterotrimeric subunit
a adaptin
b2 adaptin
sigma 2 adaptin
mu 2 adaptin
AP1 adaptor complex found on the vesciles coming from teh golgi
AP2 on clathrin coated vesicles originating from plasma membrane
can recognise specific peptide motifs on the cargo receptor (endocytosis signal)
interacts w plasma membrane lipids, cargo and clathrin —> link between clathrin and cargo
ACTIVATION
always present but kept in closed state in the cytoplasm —> internal interactions so binding domain buried
activation of CSM receptors causes clustering of receptors and PIP2 formation
PIP2 binds to AP2 and holds it at the membrane so that it can bind to a receptor —> only occurs when there’s enough PIP2 at the membrane
activation of AP2 with PIP2 at the membrane allows the clathrin binding site to be exposed
mu 2 subunit is displaced which stabilises the AP2 complex in the open formation
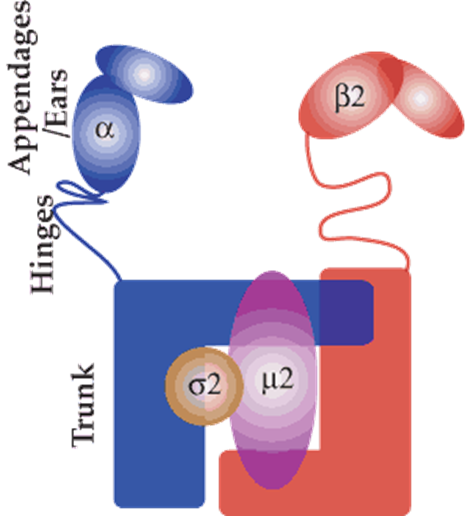
summary of assembly of clathrin
binding of cargo to receptor
recruitment of adaptr complex
recruitment of clathrin
clathrin causes memb curvature
vesicle now needs to be excised from memb
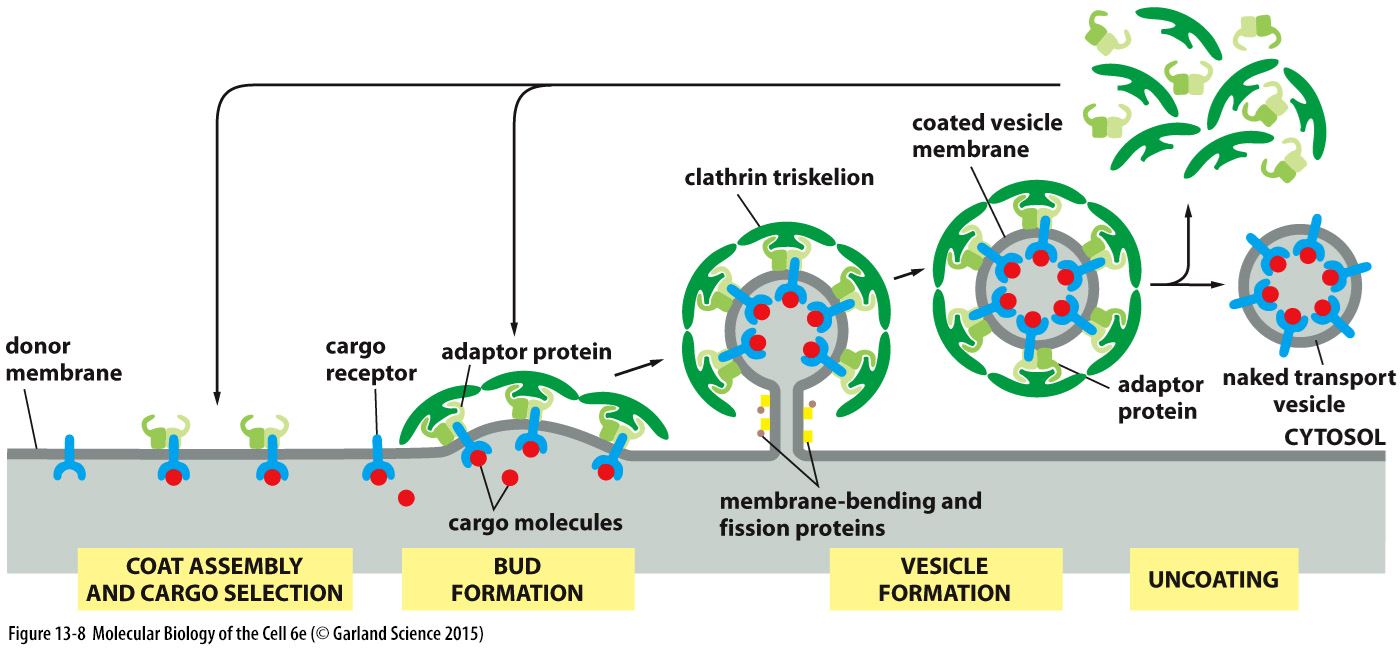
dynamin
cuts off the bud from the membrane
is a GTPase
coat is then lost after the vesicle buds off —> components recycled and the proteins needed for a fusion event are exposed
hydrolysis of GTP causes a conf change in dynamin which helps to break the 2 membranes
oligomerises to form a helical ring around teh neck of the band
then will recruit other proteins and will thether itself via a lipid binding motif
GTP hydrolysis results in the lengthwise extension of helix and fission of the memb
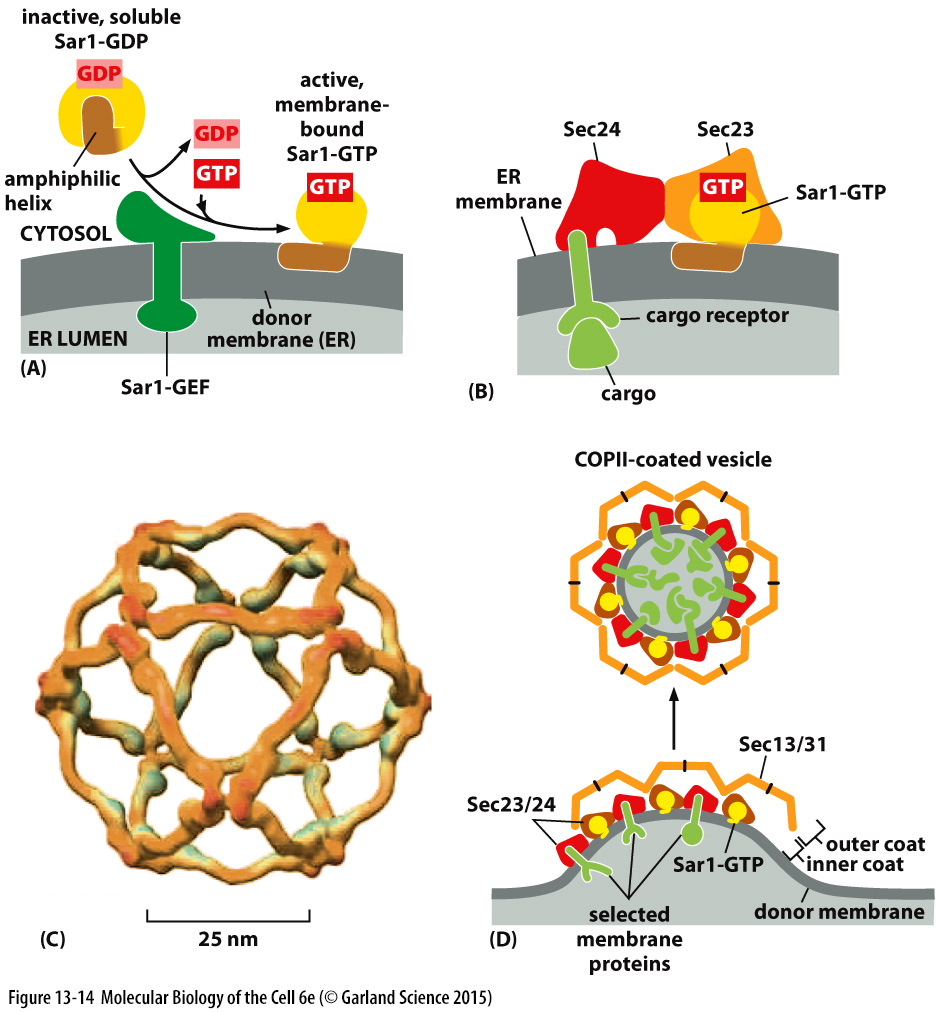
formation of cop 2 vesicles
cop 2 has 5 protein subunits which has an associated sar 1 GTPase
sec 12 is a receptor in the ER membrane which can switch on sar 1 allowing for the conversion of GTP
cop 2 vesicles carry bulk proteins but also
enzymes for golgi processing
docking and fusion proteins
integral proteins that bind to specific targets
the GTP bound GTPase embeds in the membrane and acts as a marker to recruit the coat complex
sec23 & sec24 act as the inner coat whilst sec 13/14 act as the outer coat
hydrolysis of GTP disassembles the coat and allows the vesicle to move onwards
cargo receptors ensure the correct packaging of protein by interaction with the coat complex.
cop 1 vesicles
STRUCTURE
function as a retrieval pathway
ER resident enzymes have a specific KDEL motif at the C terminus which allows resident Er proteins to be retrieved
ER has higher pH than golgi so in golgi the KDEL has high affinity for receptor —> low affinity in Er so is deposited
is a heptamer in the cytoplasm
it is not formed at the vesicle
driven by an ARF1 GTPase for coatomer recruit
clathrin and cop 1 have the coat proteins bought to the membrane where the coat is formed unlike cop 1
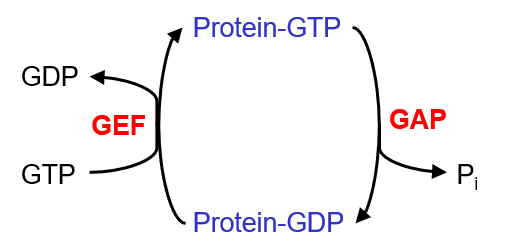
the importance of GTPases
switch for vesicle transport
activation w GTP allows for interaction w effector proteins
rab family of GTPases provide compartments with identity
rab family part of teh ras family
RAS FAMILY
lipidated - allows membrane association
small
conserved regions which allows for selectivity of effector proteins
5 major subfamilies, one of them being rab
various PT modifications
RAB FAMILY
61 members
intracellular transport
localised lipid binding motif
PT addition of prenyl lipid group
effector proteins allow vesicle formation, budding, transport, vesicle tension
NOTE: GEF = guanine exchange factor, GAP = GTPase activating proteins
maturation of vesicles
vesicle carrying cargo originating from plasma membrane may require rab 5
this may transition into early endosome w rab 4/11
maturation of vesicle is changing of location via turning off/on molecular switch
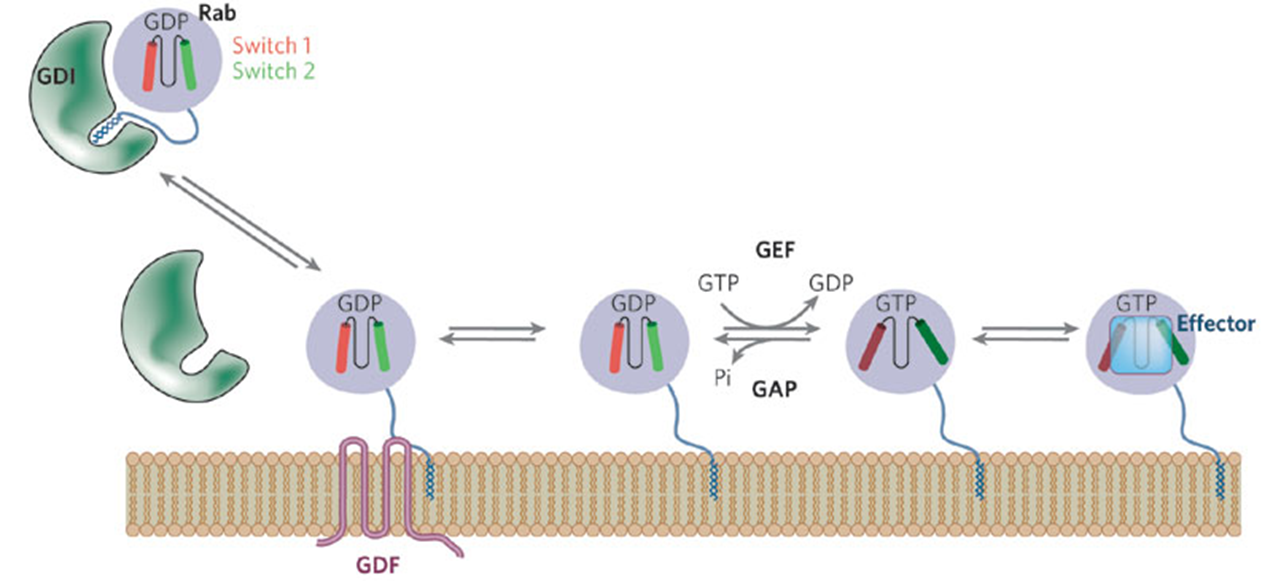
recruitment of Rab GTPases
Rabs kept inactive in the cytoplasm
interaction of GDI masks the prenyl lipid group which inactivates it
GDI = Guanine dissociation inhibitor
when lipid group exposed can embed in the membrane and recruit proteins
Resident GEFs in the membrane can activate Rab so it can hydrolyse GTP
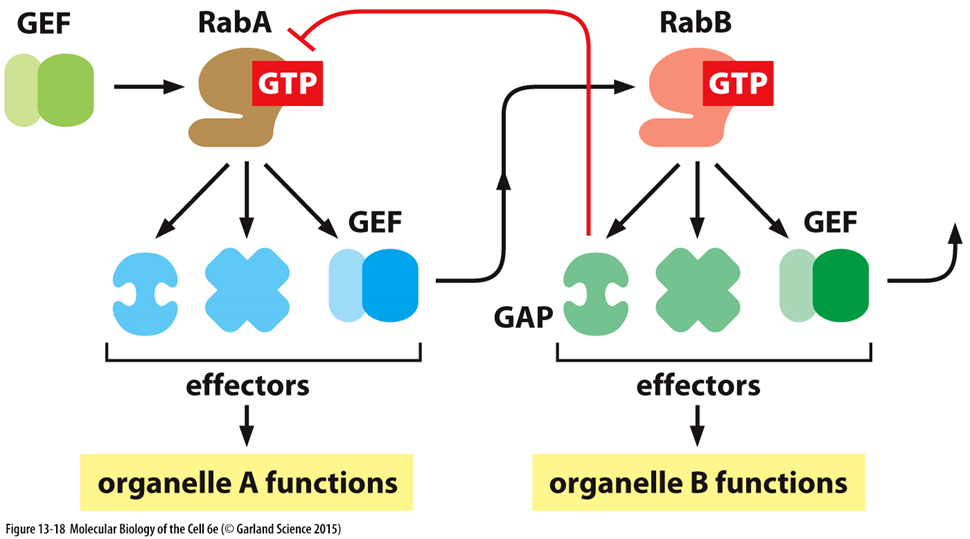
direction of vesicles and vesicle identity
IDENTITY
changing/maturation of vesicles determined by switching on of GEFs to actuvate next rab whist the previous GAP turns off the previous rab
DIRECTION
budding off of vesicles causes uncoating to reveal proteins on the surface
uncoating driven by GTPases
vesicle now exposed to snare proteins/other Rabs
SNARE proteins help with the fusion of membranes and delivery of cargo
Rab GTpases can bring the vesicles close to target membranes so snares can interact
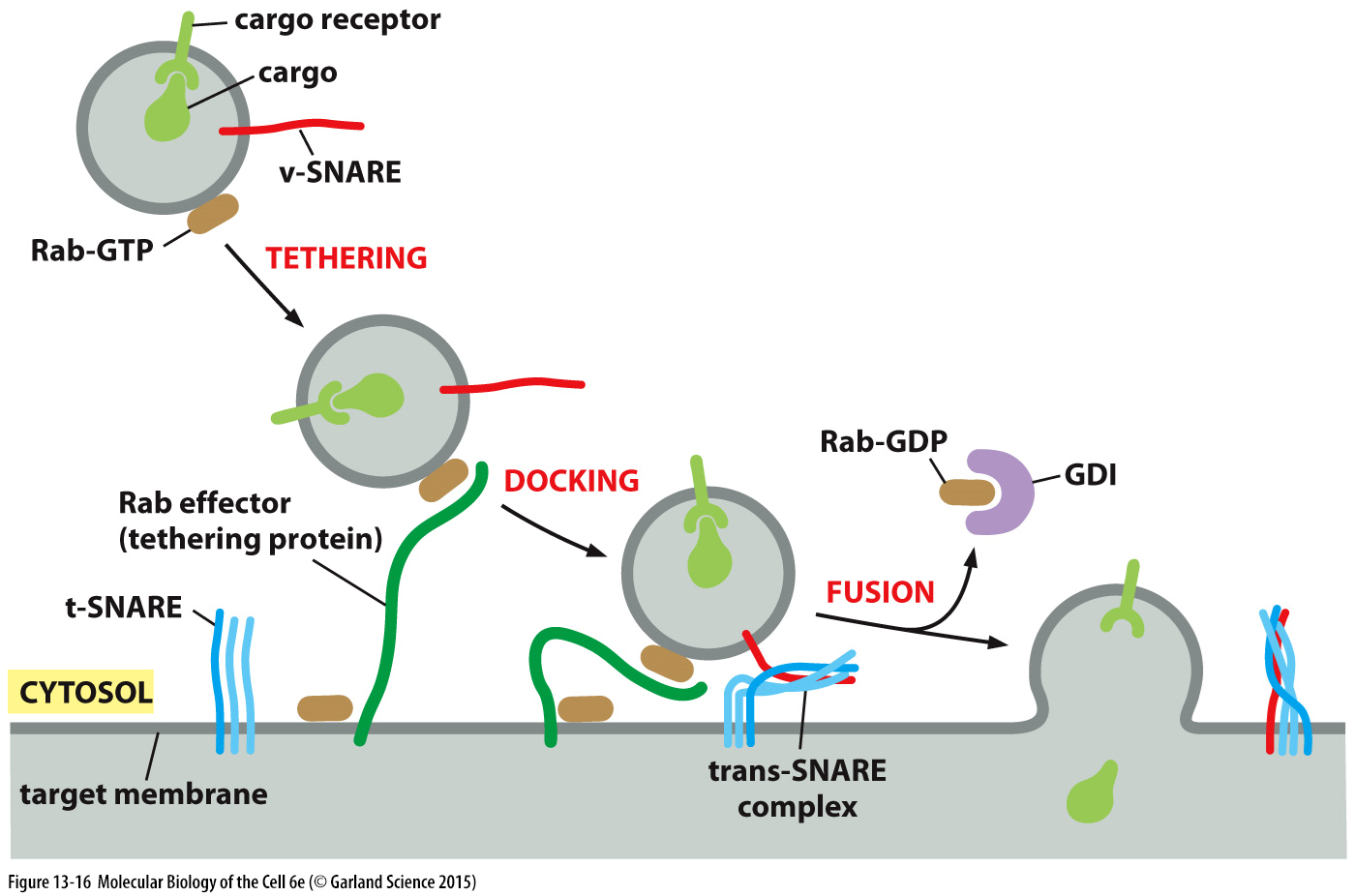
id of target membrane and vesicle docking
ID OF TARGET MEMBRANE
different snare proteins exist in pairs
vsnare on surface of vesicles
tsnare on membrane of target
v and t snares have helices which interact with one another and dock the vesicle to the target membrane
initiated by a sepcific Rab GTPase
can also be used for binding of vesicles which are intended for extracellular space
DOCKING
Rab GTP protein on surface binds to specific Rab effector in target memb (VAMPS)
this brings v and t snares in close proximity so they can form coiled coils called a trans snare complex
this exerts an inwards force that bringd the two membranes close together
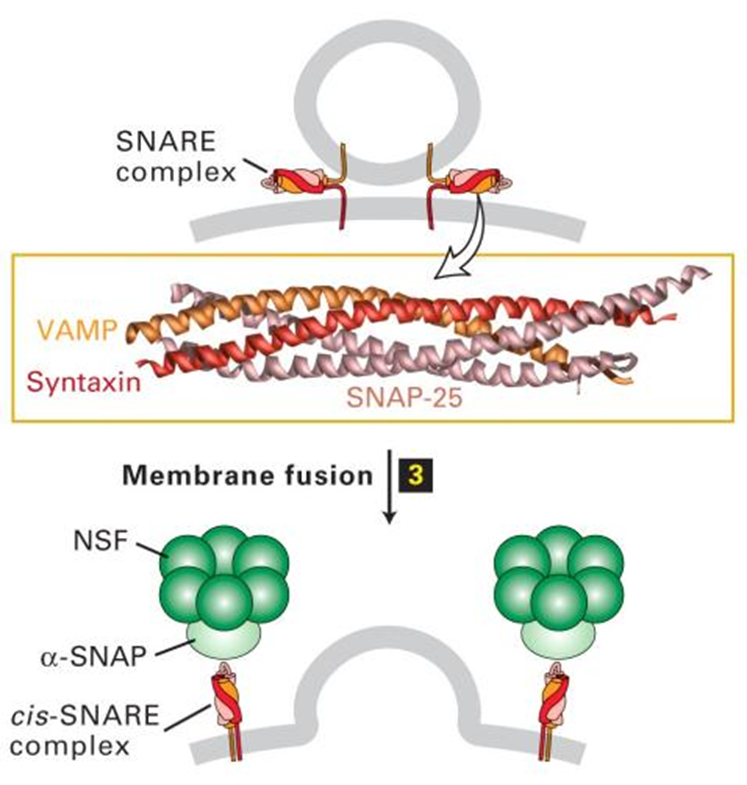
membrane fusion
the two membranes are slightly repulsive so need to be forced together
the alpha helices bring these together to release cargo
a cis snare complex is formed on one membrane —> this will drive selectivity
cis snare complex formation driven by alpha snap and NSF —> also disaasembles cis snare complex and allows for components to be recycled
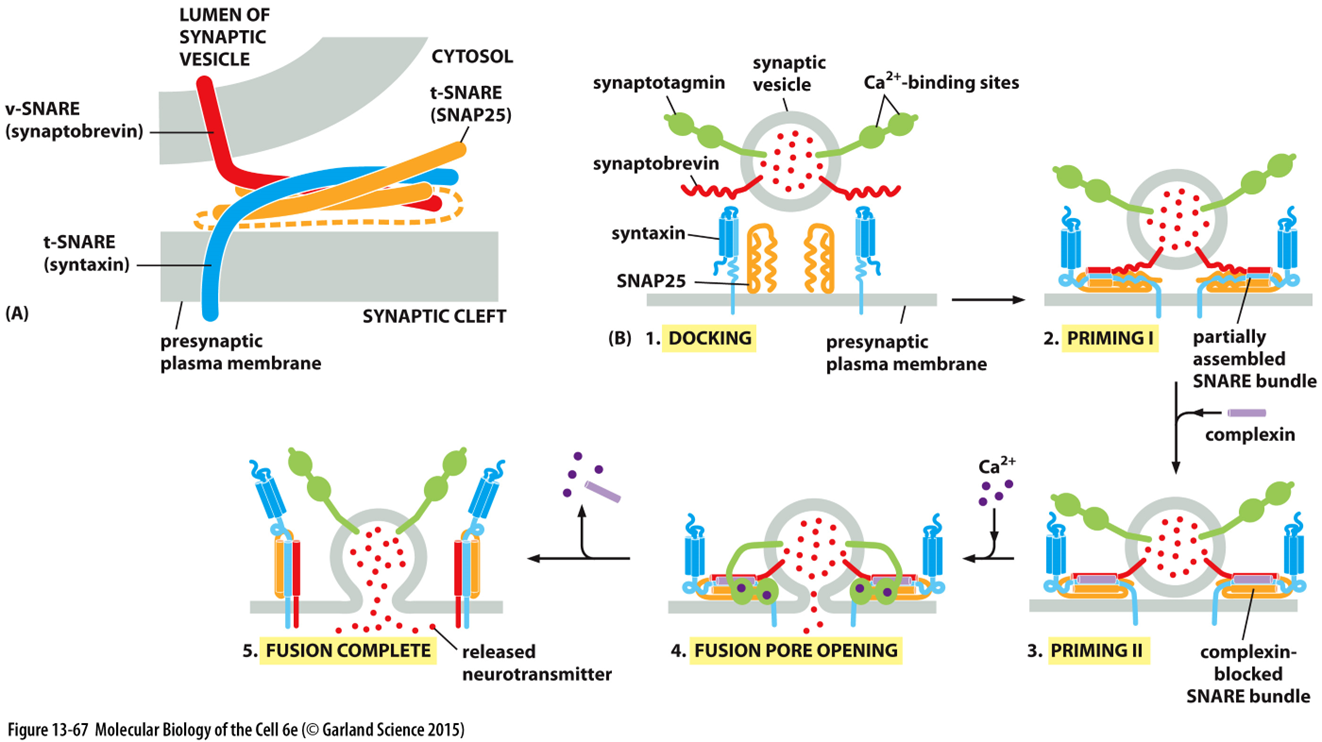
vesicles in neurotransmission
synaptic membrane is recycled to form vesicles
in neurotransmission vesciles must be primed so their release is not random
v snare - synaptobrevin
t snare - syntaxin and snap 25
complexin helps with docking - keeps it in the locked position where its held close to the membrane
ca2+ causes synaptotagmin to undergo conf change and kick out complexin so vesicles can fuse at the same time
removal of one complexin causes the removal of all the complexin
endocytosis
receptors on the plasma membrane get taken up by vesicles which go to an early endosome
receptors sometimes recycled back to the membrane
also a method of uptake for ligand bound receptor
eventually goes to lysosome via a multivesicular body (intermediate organelle)
maturation of the lysosome is required so must acquire proteins which facilitate sorting, transfer, transport and vesicle fusion
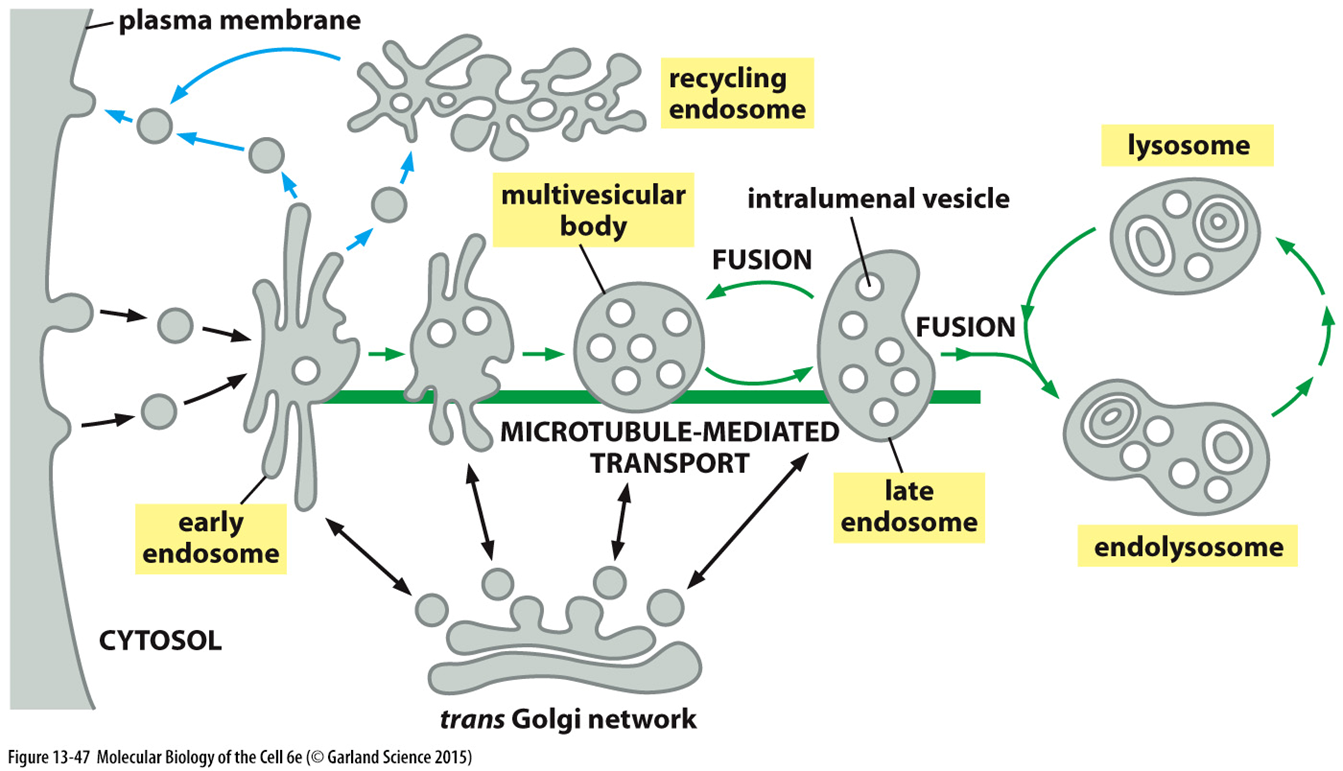
late endosome
receives both the early endosome from the plasma membrane and receptors from the golgi which need to fuse with the plasma membrane
many different sub domains which have diff target locations
some of this trafficking done by Rab GTPases
multivesicular body
low pH driven by proton gradient caused by ATPases
intralumenal vesicles which shield the cytoplasmic tails of receptors so they are unable to drive signalling
requires a specialised set of machinery to form intralumenal vesicles
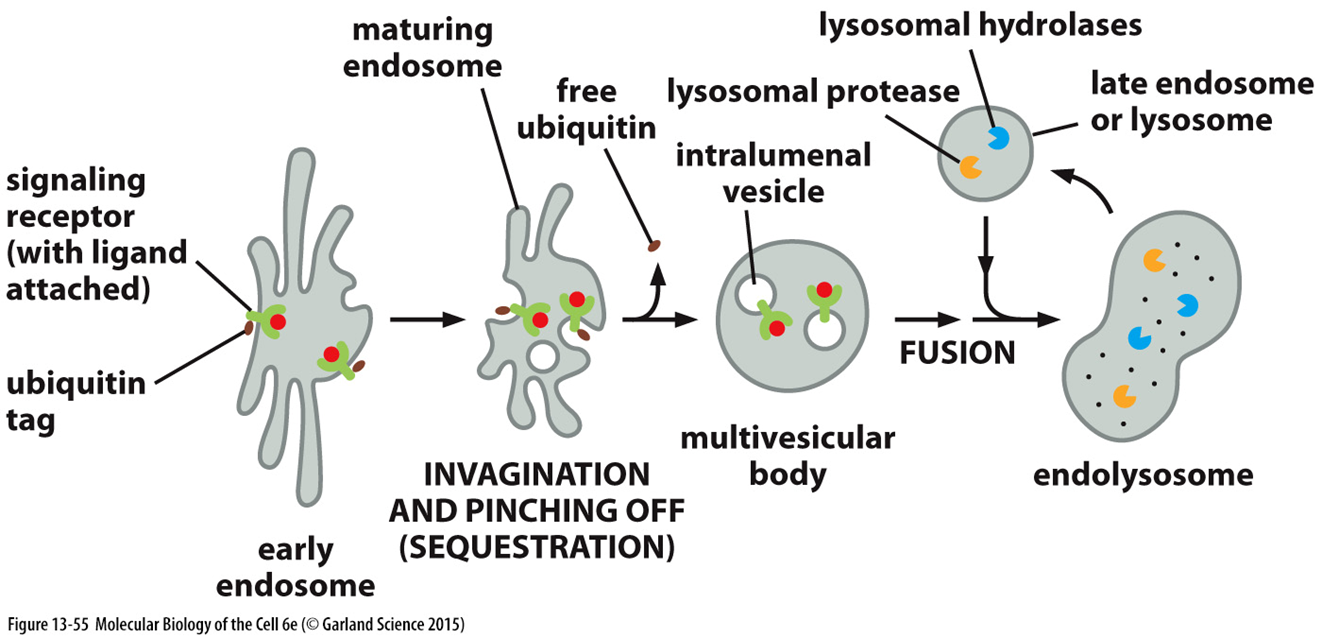
receptor signalling
activation of receptors drives vesicle formation
controlled by receptor clustering —> this means that when receptors are clustered on the surface of vesicles they can drive signalling
receptor internalisation can turn off signalling pathways
UBIQUITYLATION
activated receptors at the plasma membrane are internalised and trafficked via an endosome
activated receptors are ubiquitylated which adds a signal for receptor sequestration within an intralumenal vesicle
receptors will then be degraded when they fuse with the lysosome
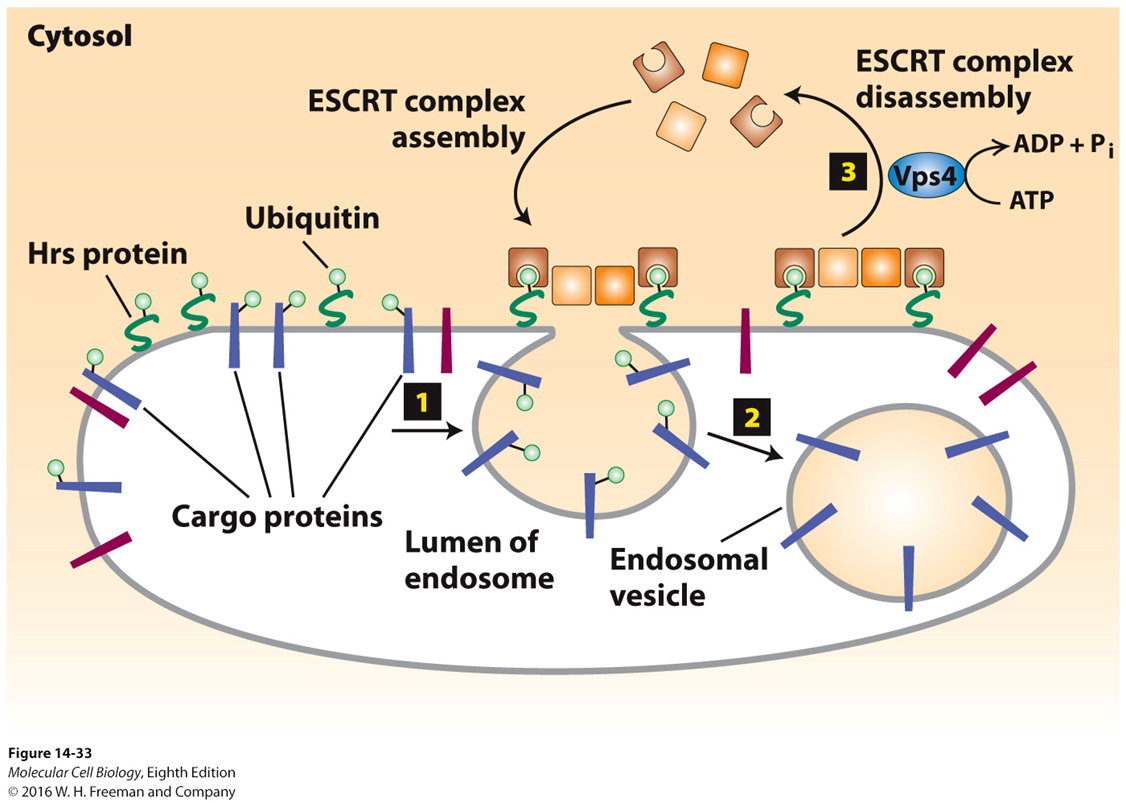
ESCRT complex
ESCRT complex required for intralumenal vesicle transport
identifies cargo for intralumenal vesicles
ESCRT has binding domains for:
ubiquitin which interacts with ubiquitinated receptor cargo
also contains interaction for PIP3
multiple ESCRT proteins shape the membrane to form an invagination for the budding of the intralumenal vesicle
note this forms a negative membrane curvature which requires this different set of machinery as opposed to the adaptor proteins
selectivity of vesicle formation is driven by different phospholipids which are present in the membrane
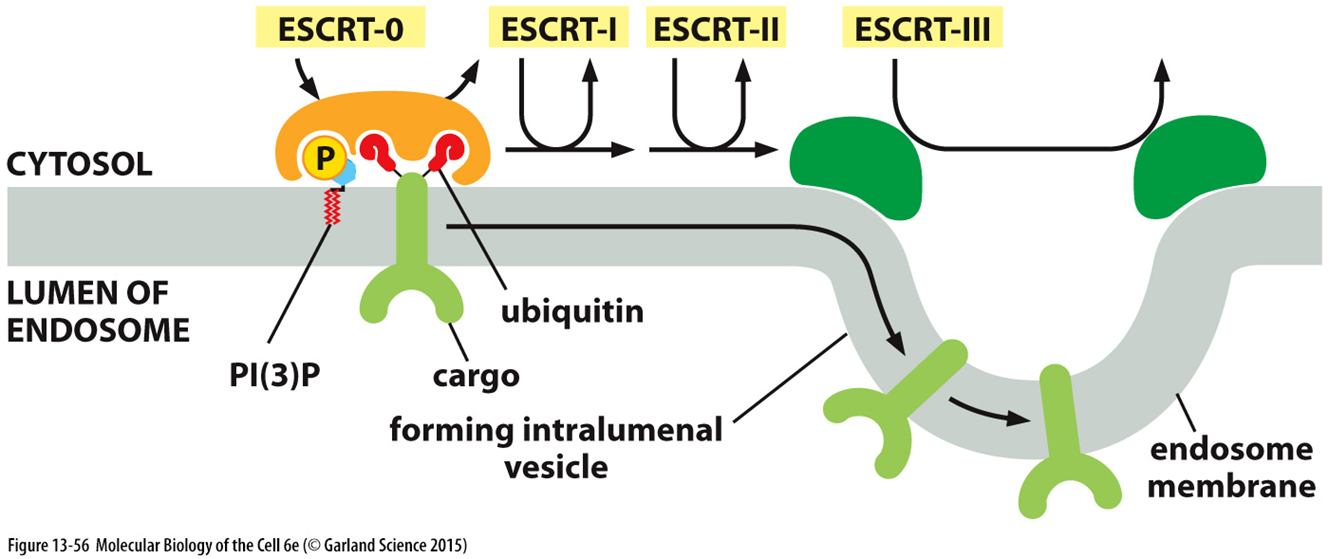
ESCRT proteins
form tetramers or dimers depending on the proteins
ESCRT 1 and 2 help cluster the cargo once its been recognised
ESCRT 3 and Vsp 4 help with budding and excision
ESCRT 3 is a cytosolic monomer but forms a filamentus structure
VSP4 is a cytosolic monomer but is a transient hexamer
Vsp4 is an ATPase and wraps around ESCRT 3 to form the inward budding
ATP hydrolysis of Vsp 4 will then cause disassembly of the complex
this results in a uniform vesicle in which receptors are unbound to their cargo
viral shedding using ESCRT
virions use ESCRT/ Vsp4 machinery
also useful during cytokinesis
this is useful for when the membrane must be cut from the outside
autophagy
cytoplasmic degradation pathway
housekeeping function that sends cytoplasmic cargo to the lysosome for turnover
cytoplasmic contents are soluble and not memb bound
always ends up in a lysosome
requires an autophagosome
2 FORMS:
nutrient starvation
Active pathway during starvation or stress areas of the cytoplasm turned over to harvest AAs
mtorc acts as an energy sensor
is AAs low or ATp low mtorc inactivated which causes activation of autophagy
captures random peices of cytoplasm to stay alive
selective pathway
take up organelles when they produce damage signals
depends on the cargo selected how the signals put on
multiple ubiquitins in specific conformation is recognised by autophagy receptors
autophagy receptors bind to the ubiquitin or LC3 protein and select for damaged cargo like this
can take muliple forms such as damaged cell components, tau proteins, damaged organelles
LC3
plays a role in the selective pathway
present in the cytoplasm but becomes lipidated and can insert in memb
acts as a platform for other proteins to bind which may be involved in cargo recognition or transport or curving the membrane
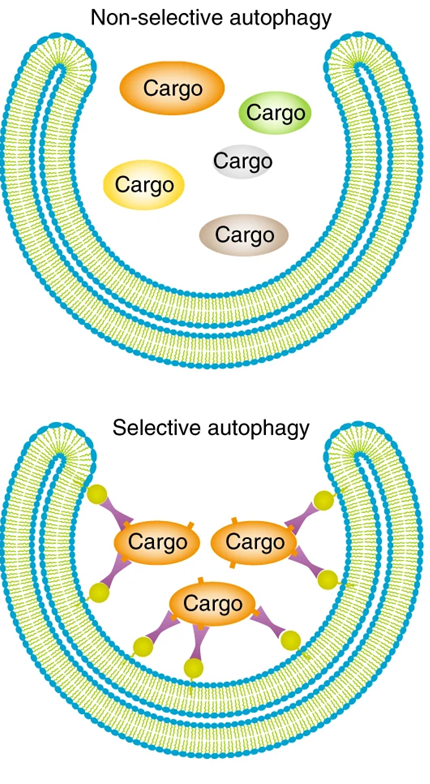
autophagosome
double membrane bound
turnover bulk portions of cytoplasm or target specifc structures
derived from other intracellular membranes
eventually gets sealed, pH lowered and transported to the lysosome
MATURATION
must fuse with other organelles, some of which are in the endocytic pathway
reduction in internal pH is required for fusion
after closure and fusion w endosomes and MVBs —> forms an amphisome