Week 9-Immunology and Serology
1/41
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
42 Terms
immune system
provides defense against foreign substances (Ag)
comprised of
Bone marrow
Thymus
Lymph nodes
Spleen:
Bone marrow
lymphocytes develop from stem cells into B and T lymphocytes
Thymus
site where T lymphocytes develop
Lymph nodes
site where antigens are presented to B lymphocytes
Spleen
B–cells and T–cells meet antigen presenting cells
The Immune response
Antibodies bind to antigens (foreign bodies) and mark them for destruction
Antibodies are proteins that are produced in response to foreign substances (antigens)
Neutrophils and monocytes (macrophage) also involved in cellular response
• Engulf pathogens through phagocytosis
Immune Reaction properties
Specific – recognize and remembers different Ag
Recognition – can distinguish between self and foreign substances
Specificity – antibody will only react with its matching antigen
Memory – once antibodies develop, they are formed for life
– remembers long after initial exposure
Class of immunoglobulins (antibodies)
IgM,
IgG,
IgA,
IgD,
IgE
IgM
Large molecule; cannot cross into the placenta
5 IgM monomers bound together by a protein
A mother’s IgM cannot attack the RBCs of her fetus having a different ABO group
Antibody to blood group antigens A and B
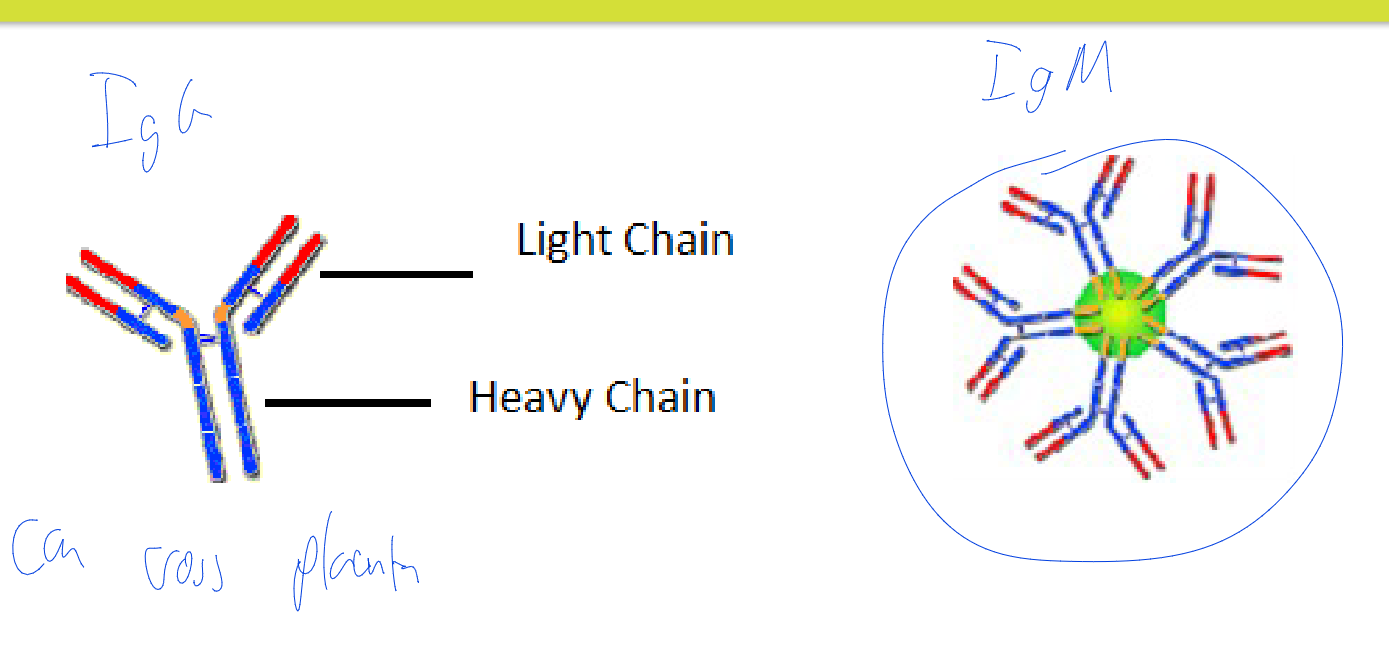
IgG (Gamma globulin or Immune globulin)
Small antibody that can cross the placental barrier and provides protection to the fetus
Highest concentration in the blood
Develop after exposure to antigens
– Including blood antigens other than A and B
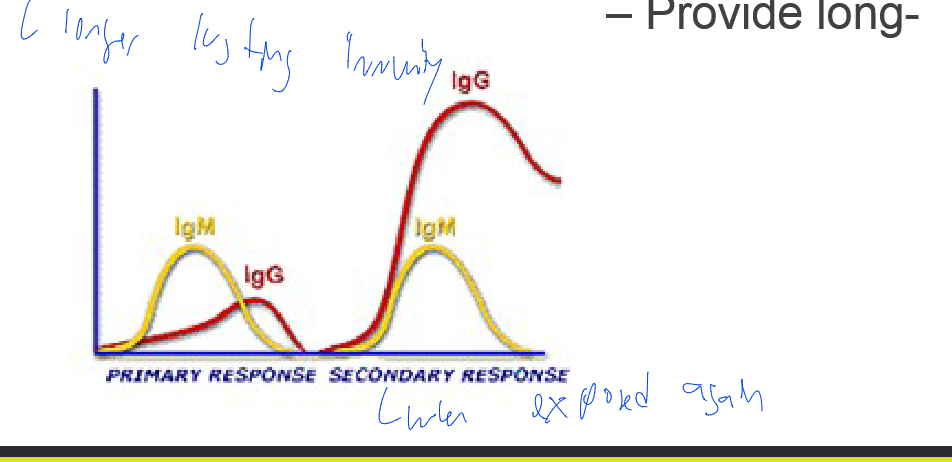
Antibody production after exposure to an antigen
1º response: IgM is produced first
2º response: IgG at an increased level due to memory response
– Provide long- lasting immunity
IgA
found in tears ,saliva, breast milk
IgD
found in blood in small amounts
IgE
east found (trace) in blood. Involved in allergies such a Hey fever and food allergies
Serology
Study of antigens and antibodies in serum
– Use of Ag/Ab reaction to diagnose a disease
– Identify blood group
– Identify cell markers
General procedure – combine a source of Ag with a source of Ab+
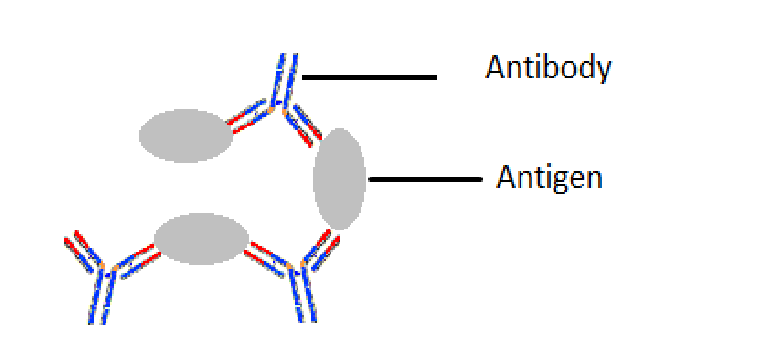
Agglutination
– Visible clumping of cells/antigens due to reaction with specific antibodies
– Example
ABO and Rh typing
Agglutination, Precipitation (Types of Immunological/Serological Tests)
Antibody mixed with antigen causes clumping (insoluble)
Florescent Ab stain (Types of Immunological/Serological Tests)
Bacterial smear is prepared
Smear is stained with a fluorescent dye fixed to an antibody to the target organism.
ex VDRL for syphilis
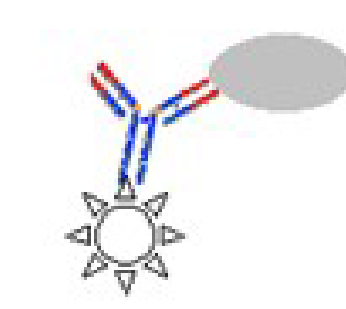
Enzyme Linked Immunosorbent Assay (EIA or ELISA) (Types of Immunological/Serological Tests)
• Antibody to target antigen is fixed to a microtiter plate
• Sample is added → antigens bind to antibody
• Non-specific unbound antigen is removed by rinsing
• Antibody linked to an enzyme is added →antibody binds to the antigen
• Unbound antibody-enzyme is removed by rinsing
• A colourless substrate is added a coloured product is formed indicating a positive test
used to measure the presence and/or concentration of an
antigen,
antibody,
peptide,
protein,
hormone,
or other biomolecule in a biological sample.
extremely sensitive, capable of detecting low antigen concentrations
ability to detect the interactions between a single antigen-antibody complex
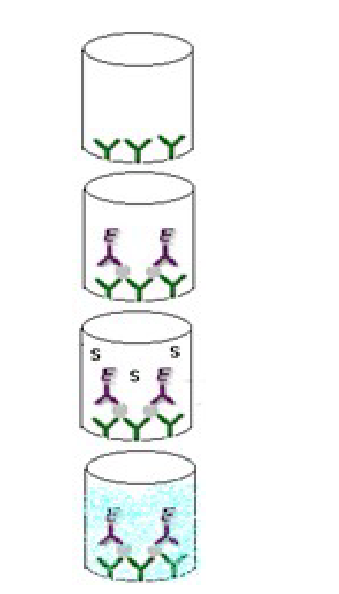
The indirect ELISA
is frequently used to determine the outcome of an immunological response, such as measuring the concentration of an antibody in a sample
is commonly used to measure the amount of antibodies in serum or in the
supernatant of a hybridoma culture.
The general procedure for the indirect ELISA assay is:
Coat wells with antigens
Add serum or hybridoma culture supernatant containing antibody (primary or 1° antibody)
Incubate and wash
Add secondary (or 2°) enzyme-conjugated antibody
Incubate and wash
Add substrate
1. Indirect ELISA
An indirect ELISA is one where the primary antigen-specific antibody is recognized by a secondary conjugated antibody. The following protocol is an example of an indirect ELISA method, where the serum samples of of influenza A virus (IAV)-infected mice are tested for the presence of IAV-specific IgG antibody. One strength of this example is that different secondary antibodies can be used that recognize all antibody isotypes or specific isotypes (e.g., IgG).
Coating antigen to the microplate
Coat the wells of a 96-well ELISA plate with purified antigen by pipetting 50 µL of purified antigen (2 mg/mL of purified A/PR/8 Influenza A virus in 0.05M Tris-HCl buffer (pH 9.5)) into each well of the plate.
Cover the plate with an adhesive cover and incubate it overnight at 4°C to allow the antigen to bind to the plate.
Upon incubation completion, remove the coating solution by flicking the plate over a sink.
Blocking
Block the remaining protein-binding sites in the coated wells by adding 200 µL blocking buffer, 5% donkey serum in 1X PBS is used here, per well. Alternative blocking reagents include 5% non-fat dry milk or BSA in PBS or normal serum from an animal in which the secondary antibody was generated.
Incubate for at least 2 hours at room temperature or overnight at 4°C.
Following the incubation, remove the blocking buffer by flicking the plate and then wash plate with PBS containing 1% Tween-20.
Incubation with the primary antibody
Prepare a serial dilution of the serum sample, which contains the primary antibody, to obtain a dilution range of 1 to 204,800, using 1X PBS. To do this, first dilute the serum 1:12.5 and then perform a 4X dilution (dilution range - 1:12.5 to 1:204,800).
Add 100 µL of the serially-diluted serum samples to the wells.
Cover plate with adhesive cover and incubate at room temperature for 1-2 h.
Following the incubation, flick the plate over a sink and wash plate with PBS containing 1% Tween-20.
Incubation with the secondary antibody
Add 100 µL of an enzyme-conjugated secondary antibody, horseradish peroxidase, HRP-conjugated donkey anti-mouse secondary in this experiment, to each well.
Incubate the plate for 1 hour at room temperature.
Following the incubation, flick the plate over a sink and then wash plate with PBS containing 1% Tween-20.
Detection
Add 100 µL of the indicator substrate (3,3',5,5'-tetramethylbenzidine (TMB)) at a concentration of 1 mg/mL to each well.
Incubate the plate with the substrate for 5-10 min at room temperature.
After 10 min, stop the enzymatic reaction by adding 100 µL 2N Sulfuric acid (H2SO4).
Within 30 min of adding the stop solution, read the plate using a microplate reader at 405 nm to determine the absorbance of the wells.
Indirect ELISA Strength and Weakness
Strength
1) High sensitivity due to the fact that multiple enzyme-conjugated secondary antibodies can bind to the primary antibody
2) Many different primary antibodies can be recognized by a single enzyme-conjugated secondary antibody
giving the user the flexibility of using the same enzyme-conjugated secondary antibody in many different ELISA
(regardless of the antigen being detected)
3) Best choice when only a single antibody for the antigen of interest is available
Weakness
1) High background signal may occur because the coating of the antigen of interest to the plate is not specific
(i.e., all proteins in the sample will coat the plate)
The sandwich ELISA
is best suited for analyzing complex samples, such as
tissue culture supernatants
or tissue lysates,
where the analyte,
or antigen of interest, is part of a mixed sample
differs from the indirect ELISA assay in that the method does not involve coating the plates with a purified antigen.
a "capture" antibody is used to coat the wells of the plate.
The antigen is "sandwiched" between the capture antibody and a second "detection" enzyme-conjugated antibody
- where both antibodies are specific for the same antigen, but at different epitopes
By binding to the capture antibody/antigen complex, the detection antibody remains in the plate.
Either monoclonal antibodies or polyclonal antisera can be used as the capture and detection antibodies.
The main advantage of the sandwich ELISA is that the sample does not have to be purified before analysis.
Moreover, the assay can be quite sensitive (4).
Many commercially available ELISA kits are of the sandwich variety and use tested, matched pairs of antibodies.
The general procedure for the sandwich ELISA assay is:
Coat wells with capture antibody
Add test samples containing antigen
Incubate and wash
Add enzyme-conjugated detection antibody.
Incubate and wash
Add substrate
2. Sandwich ELISA
In this ELISA version, the experimental sample is "sandwiched" between an unconjugated capture antibody and a conjugated detection antibody, both of which are specific to the same protein but at different epitopes. In the following sandwich ELISA example, concentration of human TNFα was determined in unknown sample using a standard curve generated from 2.5X serial dilution of a known standard, recombinant human TNFα (stating at concentration of 75 pg/mL).
Coating capture antibody to the microplate
Coat the wells of a 96-well ELISA plate with purified capture antibody by adding 100 µL of capture antibody (1-10 µg/mL range) to each well of the plate.
Cover plate with an adhesive plate cover and incubate it overnight at 4°C.
After incubation, remove the coating solution from plate by flicking the plate over a sink.
Blocking
Block the remaining protein-binding sites in the antibody coated wells by adding 200 µL blocking solution, 5% nonfat dry milk containing PBS, to the wells.
Incubate for at least 2 h at room temperature or overnight at 4°C.
Following the incubation, remove the blocking buffer by flicking the plate and then wash plate with PBS containing 1% Tween-20.
Add antigen containing test samples
Add 100 µL of the test sample to the wells. Seal the plate with an adhesive cover.
Incubate for 1-2 h at room temperature or overnight at 4°C.
After incubation, remove the samples by flicking the plate over the sink and then wash the wells with 200 µL 1X PBS containing 1% Tween-20.
Add enzyme-conjugated detection antibody
Add 100 µL of enzyme-conjugated detection antibody to the wells at a preoptimized concentration.
Seal the plate with an adhesive cover and incubate at room temperature for 2 h.
Remove the unbound detection antibody by flicking the plate over a sink and wash the wells with 200 µL 1X PBS containing 1% Tween-20.
Detection
Add 100 µL of the indicator substrate at a concentration of 1 mg/mL. Any bound enzyme-conjugated detection antibody will convert the substrate to a detectable signal.
Incubate the plate for 5-10 min at room temperature.
After 5-10 min, stop the enzymatic reaction by adding 100 µL 2N H2SO4 to the wells. Within 30 min of adding the stop solution, read the plate using a microplate reader to determine the absorbance of the wells.
ELISA Sandwich Strength and Weakness
Strength
The use of antigen-specific capture
and detection monoclonal antibody
increases the sensitivity and specificity of the assay
(compared to the indirect ELISA)
Optimizing the concentrations of the capture and detection monoclonal antibodies can be difficult
(especially for non-commercial kits)
Weakness
Optimizing the concentrations of the capture and detection monoclonal antibodies can be difficult
(especially for non-commercial kits)
the competitive ELISA is most often used
when there is only one antibody available to detect the antigen of interest.
are also useful for detecting a small antigen with only a single antibody epitope that cannot accommodate two different antibodies due to steric hinderance.
Most commercially available sandwich ELISA kits come with enzyme-conjugated detection antibodies.
In cases where an enzyme-conjugated detection antibody is not available, a secondary enzyme-conjugated antibody specific for the detection antibody can be used.
The enzyme on the secondary antibody performs the same role,
which is to convert the colorless substrate to a chromogenic or fluorescent product.
The above-mentioned secondary enzyme-conjugated antibody would more like to be used in a "homemade" sandwich ELISA
developed by an investigator who has generated their own monoclonal antibodies, for example.
One drawback to using a secondary enzyme-conjugated antibody is to be sure it only binds to the detection antibody,
and not the capture antibody bound to the plate.
This would result in a measurable product in all wells, regardless of the presence or absence of antigen or detection antibody.
Finally, the competitive ELISA assay is used to detect soluble antigens.
It is simple to perform,
but it is only suitable when the purified antigen is available in a relatively large amount.
The general procedure for the competitive ELISA assay is:
Coat wells with antigen
Incubate and wash
Preincubate test sample with enzyme-conjugated primary antibodies
Add mixture to well
Incubate and wash away any unbound enzyme-conjugated primary antibody
Add substrate
Name comes from the fact that more antigen in the test sample used in step 3 will result in less antibody available to bind to the antigen coating the well.
Thus, the intensity of the chromogenic/fluorogenic product in the well at the end of the assay is inversely related to the amount of antigen present in the test sample.
3. Competitive ELISA
The steps of a competitive ELISA are different from those used in indirect and sandwich ELISA, with the main difference being the competitive binding step between the sample antigen and the "add-in" antigen. The sample antigen is incubated with the unlabeled primary antibody. These antibody-antigen complexes are then added to the ELISA plate, which has been pre-coated with the same antigen. After an incubation period, any unbound antibody is washed away. There is an inverse correlation between the amount of free antibody available to bind the antigen in the well and the amount of antigen in the original sample. For example, a sample with abundant antigen would have more antigen-primary antibody complexes, leaving little unbound antibody to bind to the ELISA plate. An enzyme-conjugated secondary antibody specific to the primary antibody is then added to the wells, followed by the substrate.
Coating antigen to the microplate
Coat the wells of a 96-well ELISA plate with 100 μL of purified antigen at a concentration of 1-10 μg/mL.
Cover plate with an adhesive plate cover and incubate the plate overnight at 4°C.
Following incubation, remove the unbound antigen solution from the wells by flicking the plate over a sink.
Blocking
Block the remaining protein-binding sites in the coated wells by adding 200 μL of blocking buffer to each well, which can be either 5% non-fat dry milk or BSA in PBS.
Incubate the plate for at least 2 h at room temperature or overnight at 4°C.
Incubation sample (antigen) with the primary antibody
While blocking the wells, prepare the antigen-antibody mixture by mixing 150 μL sample antigen and 150 μL of primary antibody for each well in the assay.
Incubate this mixture for 1 h at 37°C.
Add antigen-antibody mixture to the well
Now, remove the blocking buffer from the wells by flicking the plate over a sink.
Then, wash the wells with 1X PBS containing Tween-20.
Add 100 μL of the sample antigen-primary antibody mixture.
Incubate the plate at 37°C for 1 h.
Remove the sample mixture by flicking the plate over a sink.
Then, wash the wells with 1X PBS containing 1% Tween-20 to remove any unbound antibody.
Add the secondary antibody
Add 100 μL of an enzyme conjugated secondary antibody, which in this case is AP-conjugated antibody, to each well.
Incubate the plate for 1 h at 37°C.
Following incubation, wash the plate with 1X PBS containing 1% Tween-20.
Detection
Add 100 μL of the substrate solution to each well.
Wait for 5-10 min.
After 10 min, stop the enzymatic reaction by adding 100 μL 2N sulfuric acid to the wells. Then, measure the absorbance in a microplate reader within 30 min of adding the stop solution
ELISA Competitive Strength and Weakness
Strength
1) Impure samples can be used
2) Less sensitivity to reagent dilution effects
3) Ideal for detecting small molecules (such as a hapten)
Weakness
1) Requires a large amount of highly pure antigen to be used to coat plate
drawbacks to any ELISA
One is the uncertainty of the amount of the protein of interest in the test samples.
If the amount is too high or too low, the absorbance values obtained by the microplate reader may fall above or below the limits of the standard curve, respectively.
This will make it difficult to accurately determine the amount of protein present in the test samples.
If the values are too high,
the test sample can be diluted prior to adding to the wells of the plate.
The final values would then need to be adjusted according to the dilution factor.
As mentioned, homemade kits often require careful optimization of the antibody concentrations used to yield a high signal-to-noise ratio.
Radioimmunoassay (RIA) (Types of Immunological/Serological Tests)
Use antibodies that are labelled with a radioactive isotope
used in some chemistry departments
Antibody Titre
• Serum is diluted until test is negative
• Titre is inverse of highest dilution at which test is still positive
result = 1/dilution
Infectious Mononucleosis (Serological Tests)
• Epstein-Barr virus (EBV)
• Cause fatigue, fever, sore throat and lymphadenopathy
• Tests include: Heterophile antibody screen (Monospot test)
Detect heterophile Ab in the patients’ blood
Heterophile antibody titre (Paul Bunnell)
• Sample: serum or plasma
Newer Rapid Testing Systems (Serological Tests)
• Can detect the IgM heterophile antibodies in serum, plasma or whole blood
• Color Immuno Chromatographic Assay
• Have built in control
• Fast
• Positive test- blue line
• Negative test – no blue line
QC:
• Store reagents at 2-8ºC
• Do not mix reagents from different kits
• Warm to RT before use
• Other tests: EBV Test (Epstein Barr Virus)

Rheumatoid Arthritis (RA / RF)
• Arthritis: - caused by autoimmune antibodies
• Rheumatoid factor is an antibody to human IgG
• Serum containing RF (auto antibodies) is mixed with IgG coated particles
• RF’s bind to IgG causing agglutination
• Tests: - RF Latex, RF Slide
• Sample: - Serum
• Controls - Positive and Negative
• QC: - Store reagents at 2-6ºC; warm up to RT before using
• Other tests: - CRP (C-reactive protein), ESR
Systemic Lupus Erythematosus (SLE)
an autoimmune disease that cause inflammation
Test: antinuclear antibody (ANA)
Method:
tissue slide flooded with serum;
then stained with anti human globulin bound to fluorescent dye
Sample:
SST,
spin and separate immediately
Other tests:
ANA panel
(anti-DNA, anti-Sm, anti-RNP, anti- Ro, anti-La)
SLE Prep/Collection
(use heparinized sample, must process immediately)
Thyroid antibodies
Used to test Hypothyroidism
may be due to antibodies to thyroid gland or to thyroid hormones
testing T3, T4, and TSH
Tests:
anti-Thyroglobulin Ab:
cause thyroid cell destruction
Anti- Microsomal Ab
destroy microsomal component of thyroid cells
Hepatitis A Testing method using ELISA
test for Anti-HAV
Hepatitis B Testing method using ELISA
test for
HBs Ag,
HBe Ag,
anti-HBc,
anti-HBs,
anti-HBe
Hepatitis C Testing method using ELISA
Anti-HCV
Syphilis
Venereal disease caused by Treponema pallidum
Tests
VDRL and RPR
are nonspecific tests with false positives
FTA and MHA
are specific test and are used to confirm positives
Sample – Serum
• Sent to MOH with standard MOH form; positive results are reported
HIV Testing
• Laboratory Testing: ELISA test, repeated if positive, confirmed by a second method
• POCT: rapid test kits using ELISA; positives sent to lab to confirm
even if just a screening test, if neg, still test if was exposed
• Specimen serum sent to MOH with standard MOH forms
Specimen and MOH form can be identified with a coded number to ensure no identifying information on the label
now specific tests for HIV
Cold Agglutinins
Antibodies to Mycoplasma (atypical pneumonia) causes RBCs to agglutinate at cold temperatures
Specimen: blood is allowed to clot in 37ºC water incubator, centrifuged and separated immediately
allowed to clott in incubator
Method
Serum mixed with RBCs, incubated at 4ºC and examined for agglutination
– Repeat using diluted sample to determine titre
– Need cells and serum
Primary protein structures
Amino acids' unique sequence in a polypeptide chain
2d structure
Secondary protein structures
The local folding of the polypeptide in some regions gives rise to the secondary structure of the protein
most common are the α-helix and β-pleated sheet structures
Both structures are held in shape by hydrogen bonds
The hydrogen bonds form between the oxygen atom in the carbonyl group in one amino acid and another amino acid that is four amino acids farther along the chain
Tertiary protein structures
The polypeptide's unique three-dimensional structure is its tertiary structure
This structure is in part due to chemical interactions at work on the polypeptide chain
the interactions among R groups create the protein's complex three-dimensional tertiary structure.
All of these interactions, weak and strong, determine the protein's final three-dimensional shape.
When a protein loses its three-dimensional shape, it may no longer be functional.
Quaternary protein structures
In nature, some proteins form from several polypeptides, or subunits, and the interaction of these subunits forms the quaternary structure.
Weak interactions between the subunits help to stabilize the overall structure