Chem 110 Terms Quiz 2
1/86
Earn XP
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
87 Terms
Conservation of Energy Law
Matter or energy can not be created or destroyed
Thermal energy
Kinetic energy measured by finding the temperature of an object
Heat
The amount of thermal energy transferred from one object to another as a result of a temp difference
Dalton's Law of Partial pressure
Total pressure exerted by a mixture of gases in a container is equal to the sum of the pressures of each individual gas
Molarity:
number of a substance dissolved in a liter of solution (M)
Solution
homogenous mixture
Solute
dissolved substance in a solution
Precipitation
Process where soluble reactants yield to an insoluble product that falls out of solution
Acid base neutralization
an acid reacts with a base to yield water and a salt
Oxidation reduction (redox)
where one or more electrons are transferred between the reaction
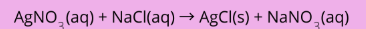
Molecular equation
all substances in the chemical reaction
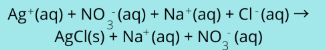
Complete ionic equation
where all the components are broken apart

Net ionic equations
only the ions undergoing change are written
Spectator ions
Ions that undergo no change in the reaction and are the same on both sides of the arrow
HNO2
Nitrous Acid
HNO3
Nitric Acid
H3PO4
Phosphoric Acid
H2SO3
Sulfurous Acid
H2SO4
Sulfuric Acid
HClO
Hypochlorous acid
HClO2
Chlorous Acid
HClO3
Chloric Acid
HClO4
Perchloric Acid
NO2-
Nitrite Ion
NO3-
Nitrate Ion
PO43-
Phosphate Ion
SO32-
Sulfite Ion
SO42-
Sulfate Ion
ClO-
Hypochlorite Ion
ClO2-
Chlorite Ion
ClO3-
Chlorate Ion
ClO4-
Perchlorate Ion
Oxidation
The loss of one or more electrons
Reduction
The gain of more electrons
Redox reaction
The process in which electrons are transferred from one component/compound to another
NH4+
Ammonium
CH3CO2-
Acetate
CN-
Cyanide
ClO-
Hypochlorite
ClO2-
Chlorite
ClO3-
Chlorate
ClO4-
Perchlorate
H2PO4-
Dihydrogen Phosphate
HCO3-
Hydrogen Carbonate
HSO4-
Hydrogen Sulfate
OH-
Hydroxide
MnO4-
Permanganate
NO2-
Nitrite
NO3-
Nitrate
CO32-
Carbonate
CrO42-
Chromate
Cr2O72-
Dichromate
O22-
Peroxide
HPO42-
Hydrogen Phosphate
SO32-
Sulfite
SO32-
Sulfate
S2O32-
Thiosulfate
PO43-
Phosphate

Exceptions to soluble compounds: Li+, Na+, Rb+, Cs+ (Group 1 Cations)
None
Exceptions to soluble compounds: NH4+ (ammonium ion)
None
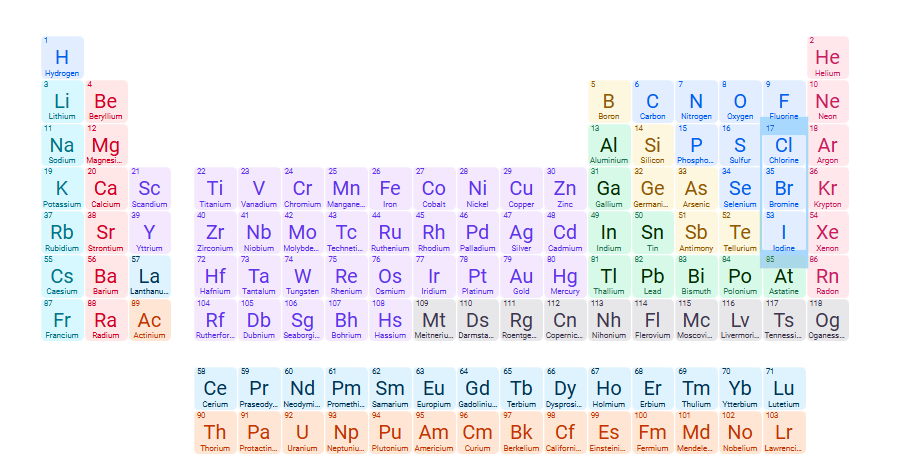
Exceptions to soluble compounds: Cl-, Br-,I-
Halides of Ag+, Hg22+, Pb2+
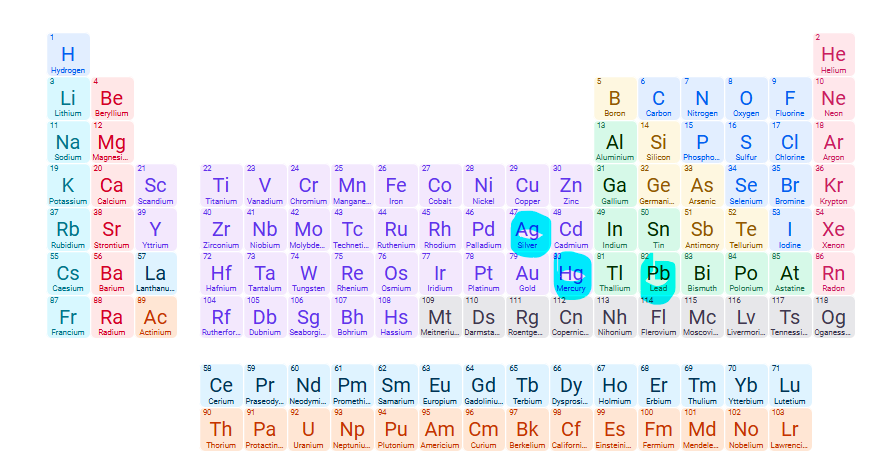
Exceptions to soluble compounds: NO3- (Nitrate)
None
Exceptions to soluble compounds: ClO4- (perchlorate)
None
Exceptions to soluble compounds: CH3CO2- (acetate)
None
Exceptions to soluble compounds: SO42- (Sulfate)
Sulfates of Sr2+, Ba2+, Hg22+ , Pb2+
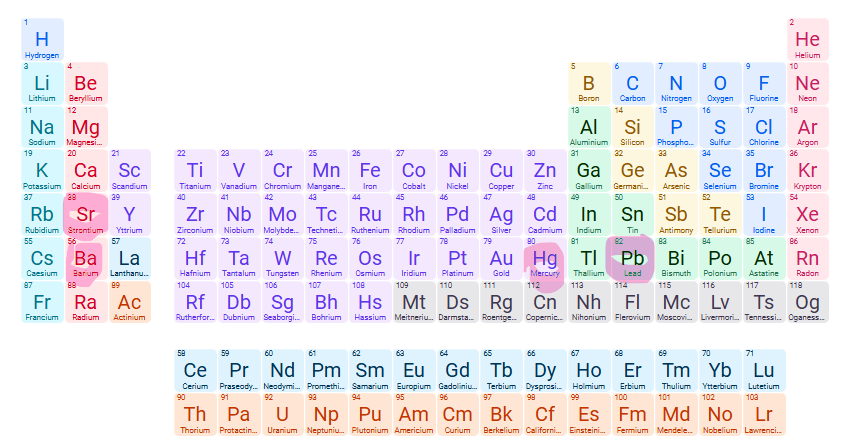
Exceptions for INsoluble compound: CO32- (carbonate)
Carbonates of Group one cations, NH4+
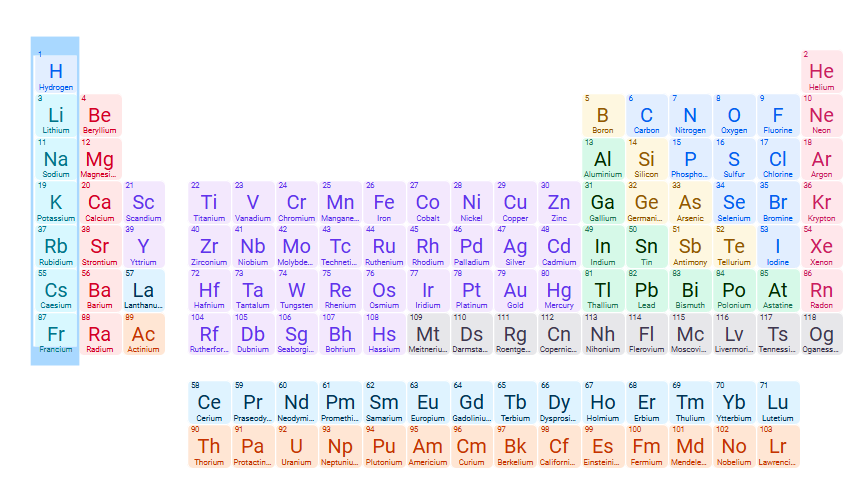
Exceptions for INsoluble compound: S2- (sulfide)
Sulfides of group 1A cations, NH4+, Ca2+, Sr2+ , Ba2+
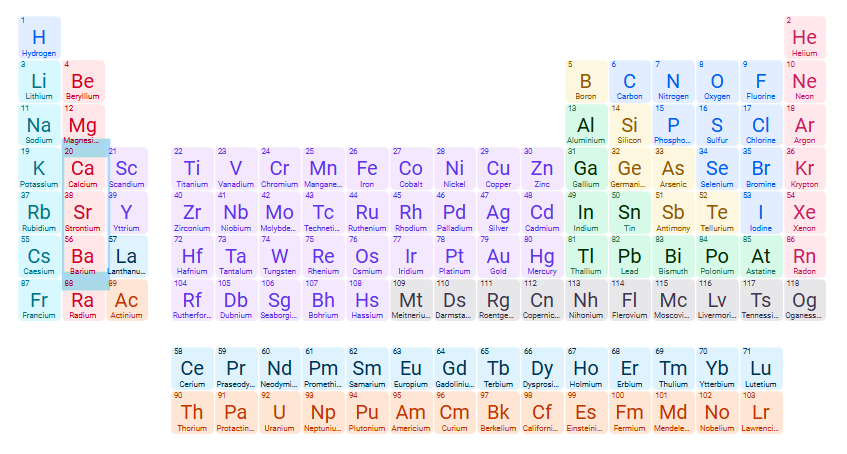
Exceptions for INsoluble compound: PO43- (phosphate)
Phosphates of group 1A cations, NH4+
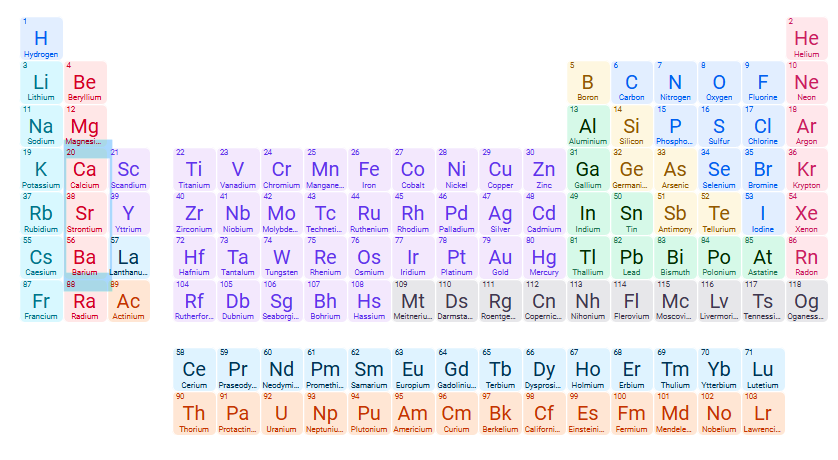
Exceptions for INsoluble compound: OH- (Hydroxide)
Hydroxides of group 1A cations, NH4+, Ca2+, Sr2+ , Ba2+
Ionic Bonds
When a nonmetal bonds with a metal
Covalent bonds
When a nonmetal bonds with a nonmetal
Metallic Bonds
When a metal bonds with a metal
Solvent
the factor that you have the MOST of, what things are getting dissolved into
Solute
The element that is getting dissolved
Aqueous Solution (aq)
When something is dissolved into water