Unit 19: Chemical Reactions
1/23
Earn XP
Description and Tags
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
24 Terms
What is an acid?
Proton (H ion) donor, reacts with a base to form salt and water
What is a base?
Proton (H ion) acceptor, reacts with acid to form salt and water
What is an alkali?
Soluble base
Acid + Metal →
Salt + H2
Acid + Metal Carbonate →
Salt + H2O + CO2
Acid + Metal Oxide →
Salt + H2O
What is a solute?
Substance that dissolves to make a solution
What is a solvent?
Substance that dissolves a solute
What is a solution?
Combination of a solute and solvent
How does dissolving work?
Particles of solvent collide with solute; solute is surrounded and moved away until evenly spread through solvent
What is a saturated solution?
Solution where no more solute can dissolve in an amount of solvent
What does solubility of gases depend on?
Pressure and temperature
As pressure increases, more gas molecules will
dissolve into the solvent
Why do fizzy drinks go ‘flat’ quicker on a hot day?
Gases tend to be less soluble as temperature of solvent increases
What is a precipitate?
Insoluble product that forms when two solutions mix and react
What is an exothermic reaction?
Energy given out (exited) to surroundings:
1. Heats up surroundings (i.e. heat ↑ energy ↓)
2. Heats up chemicals in reaction
a. Bond forming
Examples of exothermic reactions
Anything burning (combustion) e.g. wood, petrol
Respiration
Neutralisation i.e. acid + base
> Hand warmers
What is an endothermic reaction?
Energy enters (taken in) from surroundings
(i.e. heat ↓ energy ↑)
Examples of endothermic reactions
Photosynthesis
Thermal decomposition of calcium carbonate
Lightning
Ice packs
Reaction profile diagram
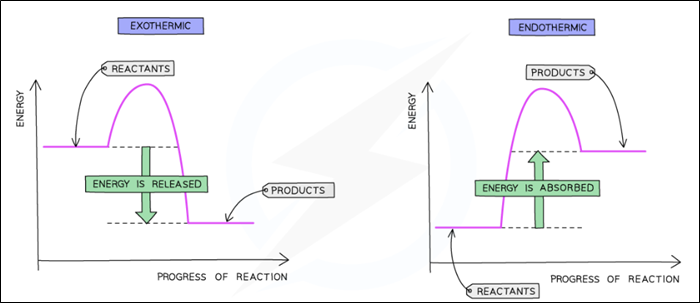
Energy level diagram
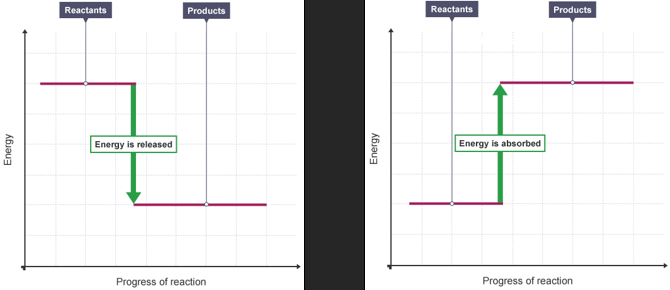
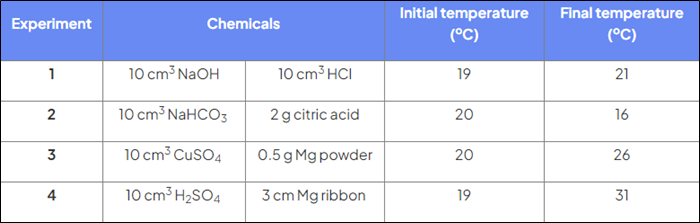
Which reactions are exothermic?
Reactions 1, 3, 4
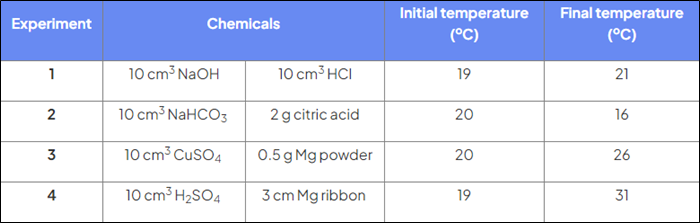
Which reactions are endothermic?
Reaction 2
What is temp. in terms of particles?
measure of average speed of particles (kinetic energy)