Molarity And Moles
5.0(1)
1
Card Sorting
1/3
There's no tags or description
Looks like no tags are added yet.
Last updated 11:16 PM on 11/15/25
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
4 Terms
1
New cards

Moles (mol)
Moles = mass (g) / molar mass (g/L)
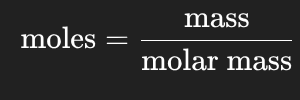
2
New cards

Molarity (M)
M = moles / volume (L)
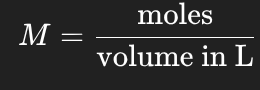
3
New cards

You dissolve 8 g of KBr (molar mass = 119 g/mol) in 400 mL of water.
What is the molarity of the solution?
Moles = molar mass / mass = 1198 = 0.06723 mol
M = volume in L / moles = 0.6723 / 0.400 = 0.168 M
4
New cards

What are the units for molar mass?
g/mol
