Thermal Physics
1/53
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
54 Terms
Distuingishing features of solids
Definite shape and definite volume, can not flow or be compressed
Distuingishing features of liquids
No definite shape but have definite volume, can flow to take shape of container but can not be compressed
Distuingishing features of gasses
No definite shape or volume, can flow to take shape of caontainer and can be compressed
1) melting, 2) boiling, 3) freezing 4) condensing
Solid arrangement
Regular pattern
Liquid Arrangemen
Irregular arrangement
Gas Arrangemen
Irregular arrangement,
Solid separation
Very closee togethor
Liquid separation
close togethor (touching)
Gas separation
Widely separate
Solid movement
Vibrate arround fixed positions
Liquid movement
Move around each other
Gas movement
Move quickly in all directions
Which 3 things affect the properties of solids liquids and gasses?
Forces and Distance and motion
What happens to the speed of the particles as temperature increases
Increase in kinetic energy so move faster
What is absolute 0
-273 degrees celcius where particles are stationary
What causes an increase in pressure in gasses and why?
Increase in temperature as the particles gain kinetic energy so collide with the walls of the container with more force per unit area
Describe Kinetic Particle Theory/ Brownian Motion
Random collisions between the microscopic particles in a suspension and the particles of the gas or liquid
Evidence for kinetic particle model of matter
Random motion of microscopic particles in a suspension
What causes microscopic particles to move
collisions with fast moving molecules
Microscopic Particles
Something too small to be seen with naked eye but can be seen with light microscope, made of billions of atoms, eg dust pollen
Formula between Kelvin and Celsius
T ( in kelvin) = θ (in celcius) +273
How does increase in temperature effect the pressure with fixed volume
Increases the pressure as the increase in temperature means the particles have more kinetic energy so collide with greater force causing a greater force per unit area
How does a change of volume effect pressure with a constant temperature
If volume increases it causes a decrease in pressure as there is more space in between the particles so there will be less frequent collisions causing less force per unit area.
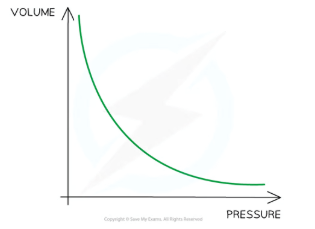
Equation linking pressure and volume with constant temperature
pV = constant
Graphical display of pV = constant
Why does thermal expansion happen?
The molecules move around faster as they gain kinetic energy This causes them to collide more often and push each other apart
Order how much thermal expansion effects each state of matter
Solids expand the least, then liquid then gas
Why do solids expand less in thermal expansion
The low energy molecules cant overcome the intermolecular forces holding them togethor
Why do liquids expand the amount they do in thermal expansion?
Thry have enough energy to partially overcome the intermolecular forces holding them togethor
Why do gasses expand the most in thermal expansion
The high energy molecules have enough energy to completely break intermolecular forces
Everyday applications of Thermal Expansion
1) Liquid in glass thermometer 2)Temperature activated switches
How do liquid in glass thermometer work?
The thin glass capillery contains a liquid which expands with temperature In one end there is a glass bulb containing a large amount of the liquid which will be expanded into the narrow tube The scales along the side allow the temperature to be measured based on the length of liquid in the tube
How do temperature activated switches work
There is a bimetallic strip which is made of 2 metals which expand at a different rate and bend by a predictable rate and will close the circuit when heated
Effect of thermal expansion
It can cause solid substance to expand if given to much heat so objects built this way have gaps built in to allow expansion to happen without causing damage. ege railway tracks, bridges, roads
Which energy does a rise in temperature in an object increase?
Internal energy
What is an increase in temperature in an object
Increase in the average kinetic energies of all the particles in the object
Define Specific Heat Capacity
Energy required per unit mass per unit temperature increase
Specific heat capacity formula
c = ∆E /(m∆θ), θ is temperature
Plan an experiment investigating specific heat capacity
Place beaker on digital balance and press 0 Add of water and record the mass of water using the digital balance Place immersion heater and thermometer in water Connect circuit with the ammeter in series with the power supply and immersion heater and voltmeter in parallel with the immersion heater Record the initial temperature of the water at 0s Turn on the power supply and start stopwatch Record the voltage from the voltemeter and the current from the ammeter Continue to record temp volt and curretn every 60 sec for 10 min Repeat using solid block of aluminium Calculate specific heat capacity of both
Calculate specific heat capacity using Current Potentail difference and time
c =( IV∆t) /(m∆θ) with I as current and v as p.d and θ as temp
Melting/boiling definition
Energy input without change in temperature
When does water melt and boil
Water boils at 100 and melts at 0 degrees C
Condensation in terms of particles
When a gas cools it loses energy away from the substance and kinetic energy decreases until the boiling point, then energy transferred away reduces potential energy so particles have not enough energy to overcome intermolecular forces and can now only flow over each other
Solidification in terms of particles
When a liquid cools it loses energy away from the substance and kinetic energy decreases until the melting point, then energy transferred away reduces potential energy so particles have not enough energy to overcome intermolecular forces and are now bound to each other nad can onlly vibrate around a fixed point
Evaporation definiton
The escape of more energetic particles away from the surface of a liquid
How does evaporation effeect the liquid left behind
it cools it
Boiling vs Evaporation
Boiling only happens at a fixed temperature (boiling point) while evaporation happens at a range of temperatures below the boiling pointBoiling happens throughout the liquid while evaporation happens at only the surfaceBoiling is a fast process while Evaporation is a slow processIn boiling heat is usually provided by an external source while in evaporation energy is taken from the surroundings
What effects evaporation
Temperature, Surface area and air movement
How does the temperature of a liquid effect its evaporation
Increased temp - increased kinetic energy, molecules at surface more likely to break free of intermolecular forces
How does surface area effect the rate of evaporation
Molecules escape from the surface of a liquid in evaporation so bigger S.A means more area for the liquid to escape from
How does air movement effect the rate of evaporation
Air carries vapour of evaporated liquid away so there is dryer air which accepts water more easily so increasing evaporation
How does being in contact with evaporating liquids affect the temperature of the object and why
Cools the object as the cool liquid absorbs heat from object