1.3 - Volumetric Analysis
1/18
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
19 Terms
To calculate the mass and number of moles…
n = m/M and m = Mn
The calculate the concentration and amount of moles in a solution…
n = CV / C = n/v
-
-
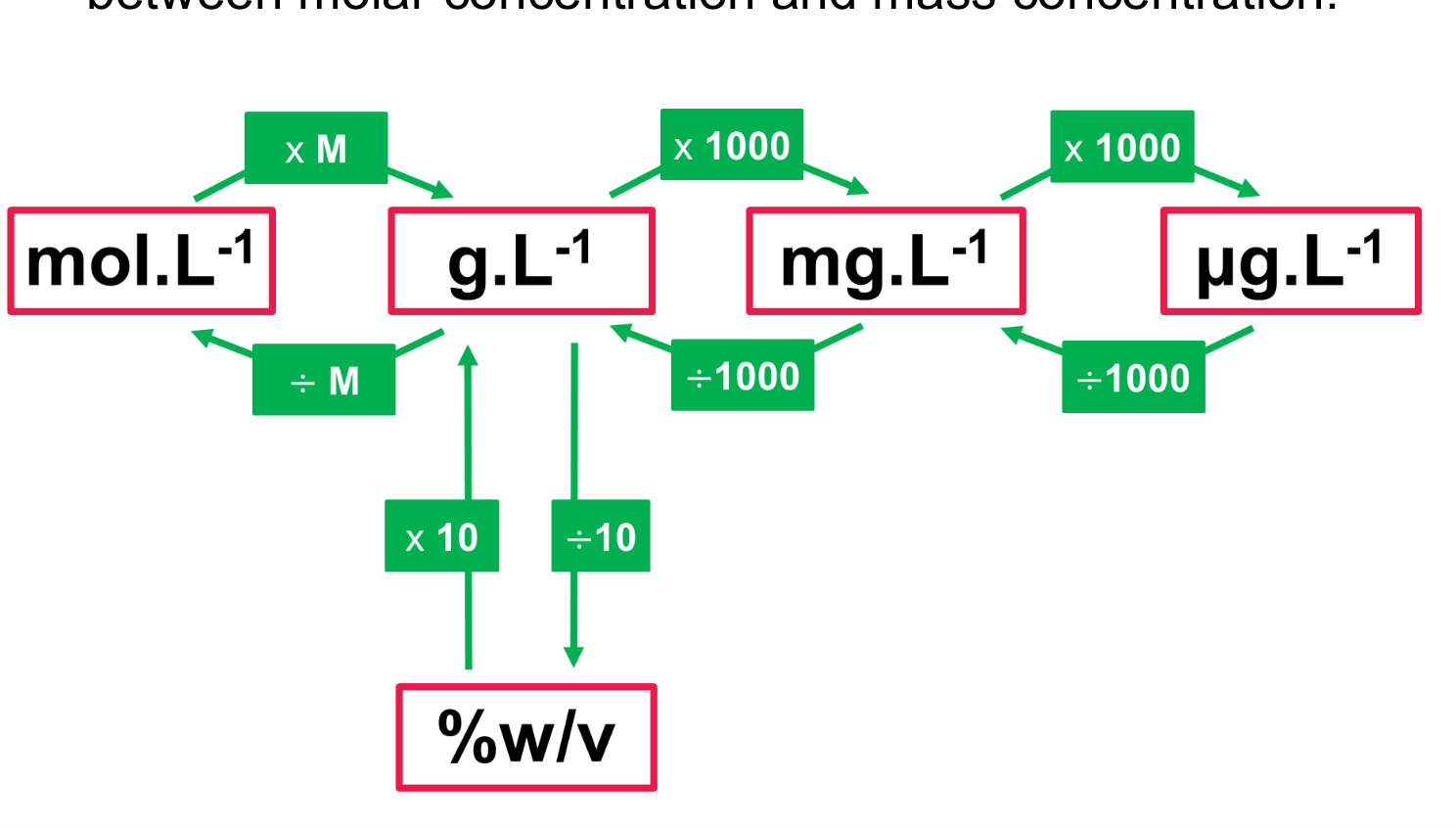
What does the conversion table look like for molar, mass, and percentage concentrations.
What are the 3 rules for significant figures
Non-Zero = Significant
Sandwiched 0’s = Significant
After or Before 0’s = Non-Significant
What is Stoichiometry, and how do you determine unknown quantities.
Stoichiometry utilizes a ratio taken from balancing an equation to determine an unknown quantity from a known quantity.
1) Check that the equation is balanced.
2) Convert units of a given substance to moles. [n=m/M / n = cv]
3) Use mole ratio of the targeted unknown and original substance (r:p) and multiply it the number of moles of the original substance.
4) Convert number of moles of desired substance to the units required.
The limiting reagent looks at… (and what is the limiting and excess reagent)
How the quantity of one reactant can affect the consumption of other reactants within a reaction.
Limiting Reagant = Reactant that is consumed first
Excess Reactant = The reactant that is still left over and partially consumed.
What is the recipe to calculate the limiting reagant?
-
What is Titration?
An experimental method that is used to identify the concentration of an unknown substance by reacting it with a known solution.
What is the substance in the known and unknown solution known as?
Known = Titrant
Unknown = Analyte
How do you calculate the concentration of an unknown solution using titration?
-
What’s the first step of titration?
-
What’s the second step of titration?
-
What are 4 ways to minimise error in titration?
Taking measurements (miniscus) - Make sure to look at bottom of the miniscus at eye level.
Equivalence and Endpoint - Endpoint is reached when the equivalence is reaches and a pale colour is shown when the unknown solution is full reacted and neutralized ( pH 7).
Concordant Titres - Make sure to calculate the average of 3 titres that are less than 0.1mL difference of each other.
Rough Titre (First Titre) - Do a first titre with less care to gauge the rough endpoint so you can be careful in the next few titres.
Direct Titration -
Is the most common form of titration where the concentration of the analyte is found by directly titrating the standard solution into the stock solution.