Carboxylic Acids Part 1
1/43
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
44 Terms
Carboxylic acid general formula
CnH2nO2
What are the 4 carboxylic acids derivatives?
Esters
Acyl chlorides
Acid anhydrides
Amides
What is the suffix used when naming carboxylic acids?
-anoic acid
What is the suffix used when naming acyl chlorides?
-oyl chloride
What is the suffix used when naming amides?
-anamide
What is the suffix used when naming esters?
alkyl alk-anoate
What is the suffix used when naming acid anhydrides?
-oic anhydride
Carboxylic acid displayed formula
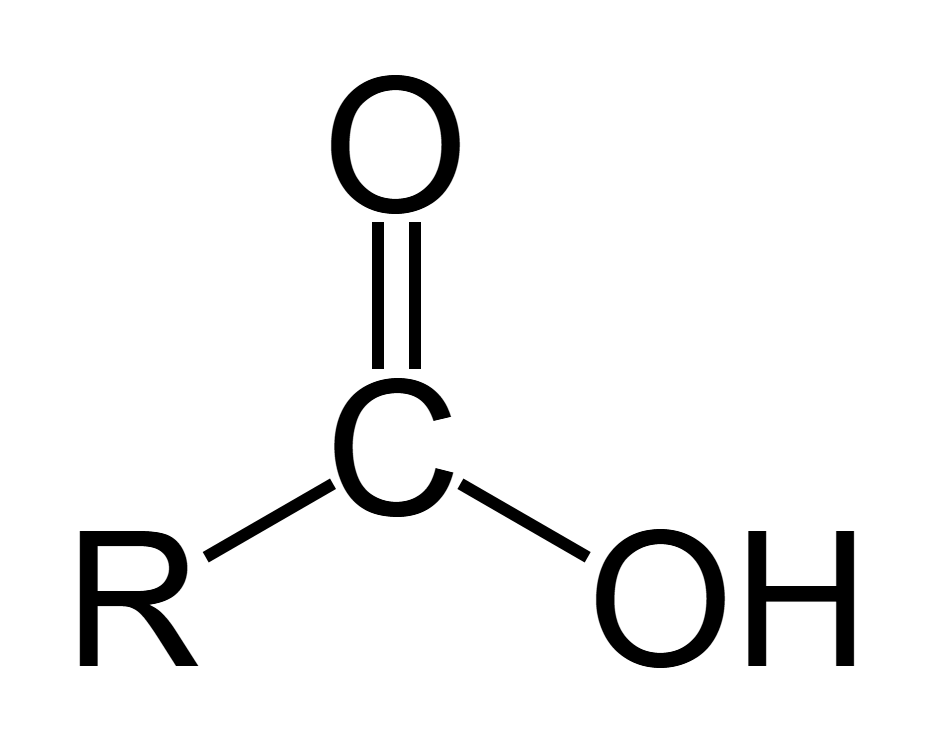
Ester displayed formula
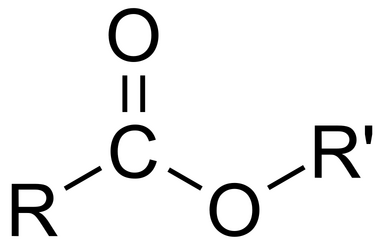
Acyl chloride displayed formula
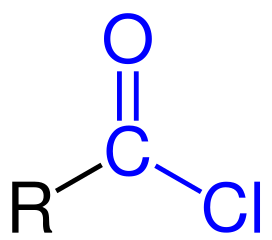
Acid anhydride displayed formula
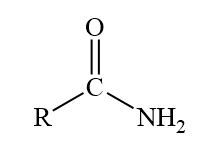
Amide displayed formula
Acid definition
A substance that donates H+ ions
Why do carboxylic acids have relatively high melting and boiling points?
Hydrogen bonds
acid + base →
salt + water
acid + carbonate →
salt + water + carbon dioxide
acid + metal →
salt + hydrogen
Ester general formula
CnH2nO2
What are 3 properties of esters?
Sweet-smelling
Due to not forming hydrogen bonds:
Lower boiling points than carboxylic acids
Less soluble in water than carboxylic acids
What are 4 uses of esters?
Perfumes
Food flavourings
Solvents
Plasticisers (Allow materials to become flexible and smooth)
What are the 3 methods and equations of preparing esters?
Carboxylic acid + Alcohol → Ester + Water
Acid anhydride + Alcohol → Ester + Carboxylic acid
Acyl Chloride + Alcohol → Ester + Hydrochloric acid
Condition and catalyst for Carboxylic acid + Alcohol → Ester + Water
Catalyst: conc H2SO4
Condition: Reflux
Conditions for Acid anhydride + Alcohol → Ester + Carboxylic acid
Gently warm
Conditions for Acyl Chloride + Alcohol → Ester + Hydrochloric acid
Room temperature
Condensation reaction
A chemical reaction which two molecules are joined together with the elimination of a water molecule
What are the 3 laboratory methods of preparing esters involving condensation reactions?
From a carboxylic acid and an alcohol
From an acid anhydride and an alcohol
From an acyl chloride and an alcohol
What is aspirin prepared from?
2-hydroxybenzoic acid (salicylic acid)
Ethanoic anhydride
Why are esters prepared with Ethanoic anhydride as opposed to Ethanoyl chloride (3 reasons)
Less corrosive
It does not react with water as readily
It is safer as the by-product of its reaction is ethanoic acid (weak acid) rather than HCl (strong acid)
Hydrolysis
Breaking of a bond using water
What is the general equation for Acid hydrolysis of esters?
RCOOR’ + H2O ⇌ RCOOH + R’OH
What are the products and conditions for acid hydrolysis of esters?
Products
Carboxylic acid
Alcohol
Conditions
Reflux with dilute H2SO4 or dilute HCl
What is the general equation for Alkaline hydrolysis of esters?
RCOOR’ + NaOH → RCOO-Na+ + R’OH
What are the products and conditions for alkaline hydrolysis of esters?
Products
Carboxylate salt
Alcohol
Conditions
Reflux with dilute NaOH(aq) or dilute KOH(aq)
Where do triglycerides occur naturally?
Animal fats
Vegetable oils
What is a triglyceride?
A triester of glycerol and 3 fatty acids
What is a fatty acid?
A long chain carboxylic acid
What is the difference between a fat and oil? What is the difference between the triglycerides in fats and oils?
A fat is a solid at room temperature whereas an oil is a liquid at room temperature
Triglycerides found in fats often contain saturated fatty acids, whereas triglycerides found in oil often contain unsaturated fatty acids
Hydrolysis
The breaking of a bond using water
What conditions can triglycerides be hydrolysed in and why?
Alkaline conditions
Contain ester groups
What are the conditions and products of hydrolysing a triglyceride with NaOH?
Products
Glycerol
Sodium salt
Conditions
Reflux
What are sodium salts formed in the hydrolysis of esters used as?
Soaps
Biodiesel
A mixture of methyl esters of long-chain fatty acids produced from vegetable oils or animal fats
What is the name of the process where biodiesel is produced from triglycerides?
Transesterification
What are the reactants and catalyst in transesterification?
Reactants
Vegetable oils
Methanol
Catalyst
KOH