CHEM 2301, Final Exam, Simple Alkyne Mechanisms, Aromatic Compounds, Conjugation of Allylics and Dienes
1/31
Earn XP
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
32 Terms
Simple Alkyne Mechanism for Hydrohalogenation
-C≡C- → -CH2-CHX2
Simple Alkyne Mechanism for Halogenation
C≡C → X2C-CX2
Simple Alkyne Mechanism for Hydrogenation
C≡C → H2C-CH2
Simple Alkyne Mechanism for Ozonolysis
C≡C → HO-C=O + HO-C=O
Simple Alkyne Mechanism for Hydration
C≡C → H2C-C=O
Simple Product Alkyne Mechanism for E2
XHC-CHX → C≡C
Simple Product Alkyne Mechanism for Alkylation
C-C≡C- → C-C≡C-C
The reagent of alkyne Hydrohalogenation
HX (slower than alkene)
The reagent of alkyne Halogenation
X2
The reagent of alkyne Hydrogenation
H2 w/ a catalyst (Pt, Pd, CaCO3
The reagent of alkyne Ozonolysis
1) O3, 2) H2O
The reagent of alkyne Hydration
H2O w/ Hg2+ or H2SO4
The reagent of alkyne E2
1) NaNH2, 2) H2O
Rxn MUST run to completion
The reagent of alkyne Alkylation
1) NaNH2, 2) H3C-X
MUST be 1° or methyl
The selectivity of alkyne Hydrohalogenation
Markovnikov
The selectivity of alkyne Halogenation
Anti
The selectivity of alkyne Hydrogenation
Syn
The selectivity of alkyne Hydration
Markovnikov
____ and ____ are possible mechanisms with benzene rings.
hydrogenation, @ high temperature, and ozonolysis, from O3’s high energy.
Aromatic Ring Physical Properties
no polarity
low m.p. and b.p.
reflects visible light
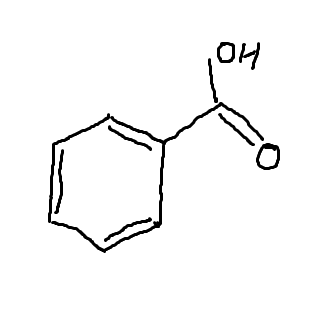
Benzaldehyde

Benzoic Acid
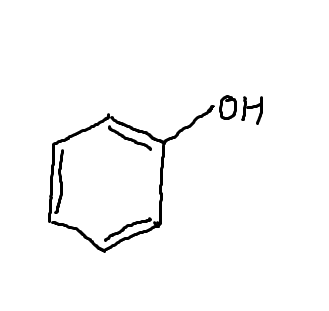
Phenol
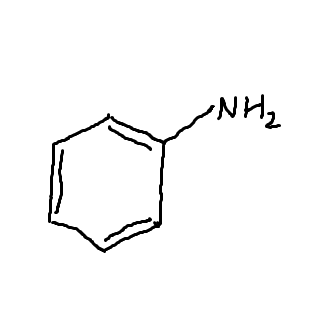
Aniline
Aromatic compounds must have…
2, 6, 10, 14… electrons.
These are abnormally stable and their intermediates form easily. Avoids rxn’s where aromaticity is disrupted.
Anti-Aromatic compounds
4, 8, 12, 16… electrons
These are abnormally unstable
Allylic intermediates are ____ and ____ stable compared to other intermediates.
faster and more stable
Structure of Dienes
C=C-C=C
the middle C-C bond is shorter than a single bond.
Prefers to form E1/E2
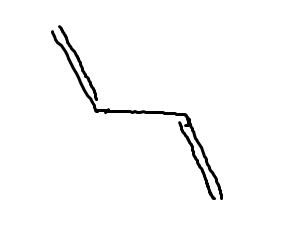
Alternative Dienes
-C=C=C-, with double bonds immediately connected, this is less stable.
C=C-C-C=C, double bonds aren’t close enough. Acts like normal alkenes.
1,2-Diene Addition
kinetic product
H-C-CX-C=C
forms faster @ low temperatures
Monosubstituted
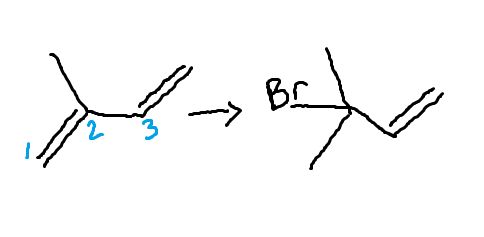
1,4-Diene Addition
thermodynamic product
forms slow @ low temperatures speeds up @ 40°C.
Trisubstituted, more stable
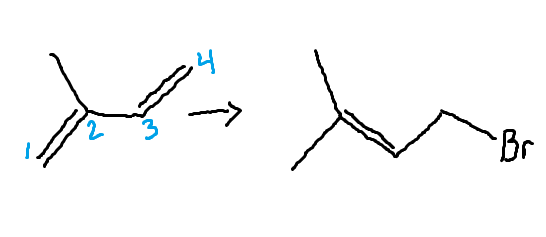
The following counts as 2 electrons
a lone pair, an anion, double bond