Operative One -- Dental Composite Material- Biocompatibility: Vidal
1/50
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
51 Terms
Biocompatibility: Craig's textbook
Ability of a material to elicit an appropriate biological response in a given application in the body
Biocompatibility: Schmalz and Arenholt-bindslev
Ability of a material to perform with an appropriate host response when applied as intended
A single material may not be
biologically acceptable in all applications
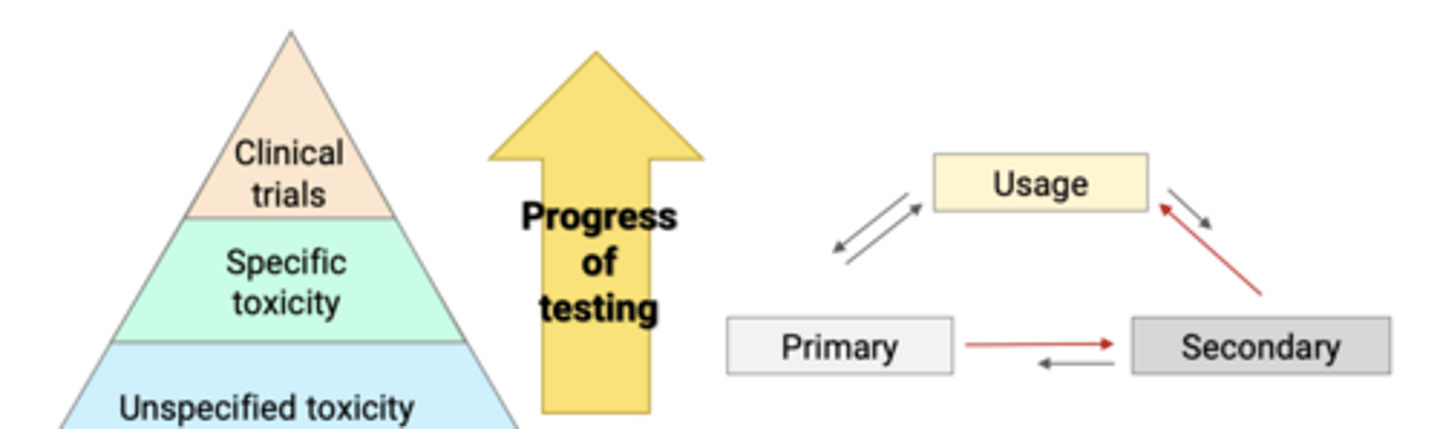
Measuring Biocompatibility
In Vitro tests
Animal Tests
Usage tests or clinical trials

In vitro tests
Require placement of a material in contact with a cell, enzyme, or some other isolated biological system
Primary cells or cell lines
Direct or indirect (barrier) contanct
Direct:
- Material physically present with the cells
- Extract of the material contacts the cell system
Primary cells
Taken directly from an animal/tissue and cultured
Grow for a limited time
Retain many characteristics of cells in vivo
More relevant to measureing cytotoxicity of materials
Limited genetic variability, may harbor viral or bacterial agents that alter their behavior, loose their in vivo functionality once placed in culture
Cell lines
Have been transformed to allow them to grow indefinitely in culture
Do not retain all in vivo characteristics but consistently exhibit retained features
Genetically and metabolically stable, contribute for standardizing assay methods
Examples: HeLa (cancer cells- 1st cell line, obtained in 1951)
Examples of in vitro tests
Cell cytoxicity tests, membrane permeability tests, cell immune function test, changes in cell cycle or activation of complement systems
Mutagenesis tests: assess the effect of a biomaterial on a cell's genetic material
Animal tests
Involve mammals (mice, rats, hamsters, guinea pigs, etc)
Material is NOT placed in the animal with regards to its final use
WHY?
Allow for the complex interactions between a material and functioning, complete biological system to occur
More comprehensive and relevant than in vitro tests
Disadvantages in animal tests
More expensive, time consuming, often involve significant ethical concerns and oversight, difficult to interpret and quantify
Example of animal tests:
Mucous membrane irritation test
Determine whether a material causes inflammation to mucous membrane or skine
Example of animal tests:
Skin sensitization
Materials are injected intradermally, followed by secondary treatment with adhesive patches containing the test substance
Example of animal tests:
Implantation test
Evaluate materials that will contact the subcutaneous tissue or bone (common for periodontal and endodontic treatment materials)
Usage tests
Involve animals (dog, min-swine, monkeys) or humans
Material is placed in a situation IDENTICAL to its intended clinical use
Ability to mimic the clinical use of the material in every regard, including time, location, environment, and placement techniques
usually employ larger animals (similar oral environment to humans)
When human is used = clinical trial
Gold stand tests, highly relevant
VERY expensive, time consuming, ethical/legal concerns, difficult to interpret and quantify results
Usage tests:
Dental pulp irritation tests
Materials are placed in Class V cavity preparation in intact non-carious teeth
Teeth are extracted and sectioned for microscopic examination
Necrosis and inflammation are classified according to their intensity
Usage tests:
Intraosseous implant test
Materials are inserted into the jaw of test animals
Primates, dogs, miniature pigs, guinea pigs, and rats
Tissue reaction is assessed histologically as well as the tissue in contact with the implant
Usage test:
Mucosa and gingival usage tests
Material is placed in cavity preparation with subgingival extensions
Responses are characterized according to the number of mononuclear inflammatory cells in the epithelium and adjacent connective tissues
- Difficulties: Presence of existing inflammation, rough margins of restorative materials, over- or under- contour of the restoration
Correlation among tests?
- Not always possible
- Test conditions affect the results
- Most accurate and cost effective = in vitro + animal + usage
- No single test can completely characterize biocompatibility
Biocompatibility is assessed by
various experts according to specific guidelines in which a comparison with products already on the market plays an important role
there are lots of different ways to assess biocompatibility, thus
evaluations may not generate identical results
- it is the dentists obligation to not rely on these assessments blindly but rather to question them critically
Standards that regulate biocompatibility tests
Regulatory agencies require biocompatibility testing and provide guidance on how the tests should be performed
International organization for standardization (ISO), the US Food and drug administration (FDA) and the conformité Européenne (CE)
The FDA recognized human health substances as what
only two categories
- drugs or devices
Dental restorative materials are classified as
devices
Preclinical evaluation of biocompatibility of medical devices used in dentistry - Test methods for dental materials
Initial tests:
- Assess biological reaction to materials (cytotoxicity, sensation, and systemic toxicity). Some done in vitro, other in animals
Supplementary tests:
- Assess chronic toxicity, carcinogenicity, biodegradation (many in animals)
Manufatueres evaluate the device to
determine if it could cause an adverse biological response
Manufactures perform
biocompatibility testing
The FDA reviews the testing results before
authorizing the device for market sale
Biocompatibility of dental materials
Reactions of pulp
Reactions of other soft tissues to restorative materials
Reactions of bone and soft tissues to implant materials
Reactions of pulp
Dentin bonding/bonding agents
Resin-based materials
Amalgam and casting alloys
Glass ionomers
Liners, varnishes, and non-resin cements
Bleaching agents
Reactions of pulp: Dentin bonding/bonding agents
Factors that determine/affect the biocompatibility of adhesive material
1. Smear layer is removed in etch and rinse protocol --> increases dentin permeability --> acids used for etching can be irritating (etch and rinse vs self etch)
2. Toxicity of monomers: HEMA and Bis-GMA
- High cytotoxicity when applied in contact with cells
- Less cytotoxicity when dentin barrier is present (at least 05 mm)
- In vivo evidence of HEMA cytotoxicity when <0.1 mm of dentin
- HEMA diffuses through dentin (via dentinal tubules) --> may cause pulpal irritation

Reactions of pulp: Resin-based materials
reduced cytotoxicity reported in test using dentin barrier
low pulpal inflammatory response when RBC is placed in cavities with at least 0.5 mm of remaining dentin. Reactions are diminished within 5-8 weeks
Factors that determine/affect biocompatibility of resin-based materials
Incomplete polymerization of adhesives
Resin-based restorative materials release unbound free monomers immediately after setting and later (degradation/hydrolysis) -> cytotoxic for pulp and gingival cells
Reactions of pulp: Resin-based materials
Microleakage and nanoleakage =
adhesive interfaces do present permeability and/or gaps-> bacterial infiltration --> pulp irritation
Good _____ _____ is key to avoid pulpal irritation, but material selection is also important
marginal sealing
restorative materials can alter
pulpal responses and dentin repair
Concerns about biocompatibility resin-based materials
Estrogenicity of biphenol A (BPA)
BPA can be a degradation product of some methacrylate used in resin composite and sealants, but levels of BPA release are extremely low (diet is the main source of BPA exposure in most countries)
BPA release can be minimized by proper light curing the material, using rubber dam isolation, remove
3M clinpro dental sealant does not contain BPA
Amalgam and Casting alloys: Cell culture tests
High-copper amalgam is toxic, low-copper amalgam that has set for 24h does not inhibit cell growth
Amalgam and Casting alloys: Implantation tests
low-copper amalgam are well tolerated, high-copper amalgam causes severe reactions
Amalgam and Casting alloys: Usage tests
Minimal pulpal irritation when placed in shallow cavities or in deep cavities using a liner
Amalgam and Casting alloys: when used in deep cavities with no liner =
pain (thermal and electric conductivity)
Amalgam and Casting alloys: Alloys used for restorations crown and dentures may have
adverse effects on cells if released
Metal ions released from alloys are likely to
contact the gingival and mucosal tissues than the pulp (more likely to affect by the cement retaining the restoration)
Pulp biocompatibility to Glass ionomers is attributed to
the weak nature of and high molecular weight of the polyacrylic acid - unable to diffuse through detnin
Pulp biocompatibility is pretty good
Glass ionomers: usage tests
mild pulp reactions, absent after 1 month
Glass ionomers: sensitivity to pain is increased after placing GI in cervical cavities possibly related to
increased permeability after conditioning the dentin
Alkaline liners
Used in exposed pulps but caused necrosis, coagulation of hemorrhagic exudate of the superficial pulp
After necrosis, neutrophils infiltrate the subnecrotic zone, which undergoes dystrophic calcification after week/months
not used anymore
Calcium hydroxide-based materials containing resin
less irritating and able to stimulate dentin bridge formation quicker than calcium hydroxide (no zone of necrosis)
Common brands:
- TheraCal LC = resin-modified calcium silicate cement
- Biodentine= tricalcium silicate-based material
MTA
Calcium silicate-based cement
Calcium hydroxide is the main soluble component in MTA
Stimulates formation of a thick compact dentin bridge with no inflammatory response
Stimulates dentin remin through solubilization of non-collagenous proteins from the dentin
Induces cell proliferation, migration, and differentiation to odontoblast-like cells
SUMMARY: Biocompatibility depends on the
material composition, location, and interactions with the oral cavity
SUMMARY: Diverse reactions can occur if
Material release their components and whether those components are toxic, immunogenic, or mutagenic