Part 2: Chemicals and the Environment
1/153
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
154 Terms
Ecotoxicology
the effects of chemical contaminants on ecosystems (plants, animals, and other life forms)
Ecotoxicology major considerations
What are the contaminants
How do they disperse in the environment (Water, air, soil, etc)
How do the contaminants degrade
How are living species affected
How is the contamination detected, monitored and remediated?
Environment health toxicology
the effects of chemicals (usually contaminants) found in the environment on human health
Populations
A group of interbreeding individuals of the same species within a community (ex. A population of large mouth bass (Micropterus salmoides) in a lake; all humans (Homo sapiens) on this planet).
Community
a naturally occurring assemblage of populations of plants and animals that live in the same environment, are mutually sustaining and interdependent, and the are constantly fixing, utilizing, and dispensing energy
Ecosystem
Consists of communities of living organisms together with their habitat ( the abiotic environment) and includes the interaction among these compounds. Major ecosystems include deserts, forests, grasslands, tundra, and several types of aquatic environments
Contain biotic (living) and abiotic (non-living) components)
Terrestrial Ecosystems
Forests, Grasslands, Deserts, Tundra
Forests
The largest ecosystem, covering about 31% of land. They include Tropical Rainforests (the most biodiverse), Temperate Deciduous Forests, and Boreal Forests (Taiga).
Grasslands
Dominated by grasses rather than large trees, including Savannas(tropical) and Prairies (temperate)
Deserts
Arid regions with extremely low rainfall, ranging from hot subtropical deserts like the Sahara to cold deserts,
Tundra
Cold, treeless regions found in the Arctic or at high altitudes, characterized by low-stature vegetation like mosses and lichens.
Biome
A regional community of species that are adapted to live in similar conditions
Bigger than an ecosystem
Chemicals in different biomes
behave differently
Types of aquatic ecosystems
Freshwater, marine, estuarine
Freshwater Ecosystems Types
Lentic, Lotic, Wetlands
Lentic
Still or slow-moving (lakes, ponds, pools)
Lotic
Fast-moving (rivers, streams, creeks)
Wetlands
Saturated or inundated soil (marshes, swamps, bogs).
Marine ecosystems
High salt content (oceans, seas, coral reefs, estuaries)
Estuarine
Where fresh and saltwater mix (bays, river mouths)
Key components of aquatic ecosystems
Biotic (Living): Producers (algae, aquatic plants), consumers (fish, amphibians, insects), decomposers (bacteria, fungi).
Abiotic (Non-living): Water, temperature, sunlight, nutrients, dissolved gases, salinity
Chemicals move in our environment in 4 phases
air, water, soil, bioosphere
Air
contaminants enter via evaporation, stack emissions and tron the other phases. Transport within air is rapid.
water
contaminants enter via direct application, spills, wet and dry deposition.
Soil
contaminants enter via direct application, spills, wet and dry deposition and from other phases. Microbial degradation can be significant.
Biosphere
Chemicals move within Food webs, in some cases the chemical is very stable => increased concentrations, in other cases the chemical is unstable => decreased concentrations.
Movement and storage of chemicals in the environment
movement of chemicals from air, soil, and water into the biosphere (living organisms) depends on the chemical’s bioavailability
Bioconcentration
Directly non-dietary uptake of chemicals from the external environment
Uptake (in fish) through their gills
Bioaccumulation
uptake of chemicals from the external environment via food
Biomagnification
increasing chemical concentrations at higher trophic (feeding) levels
Sublethal effects on individual organisms
Thankfully, most (but not all) chemical contaminants currently exist in relatively low levels or unavailable forms.
Chronic (sublethal) effects in populations
Reproductive success, Inhibition of growth rate, Predator avoidance behavior, Foraging behavior, Migration and homing, Alter breeding behavior, Nest-building and courtship,Territorial defense
These effects may occur even though
there is no measurable chemical present (e.q., rapid metabolism)
Mixtures of chemicals (common in the environment) may result in
effects not predictable from the effects of the individual chemicals.
Population and community effects
Abundance, age structure of the population, etc. Any effects on a population may ultimately have effects on the community since communities are collections of populations.
Community diversity, community composition
Bioconcentration - trophic levels and bioaccumulation
Build up of toxins in an animal due to passive exposure to toxins through gills or skin
Example: Fish take up toxins through gills due to living in contaminated water
Bioaccumulation - trophic levels and bioaccumulation
Build up of toxins in a single organism over time
Example: Deer accumulate toxins over time by eating contaminated plants
Biomagnification - trophic levels and bioaccumulation
Biomagnification = increase in toxin concentration in species at higher trophic levels
Example: Insects take up toxins from sediment, fish take up toxins from contaminated insects, eagles take up toxins from contaminated fish. Toxin levels increase up the food chain
DDT (insecticides) example
Bioconcentration, Bioaccumulation and Biomagnification of DDT, DDD & DDE (metabolites - affects breakdown) —> calcium in eggs —> thinner shells —> smaller population. Similar processes occur for mercury in fish and other persistent chemicals (PCBs and many others) in our environment.
Chemodynamics
the study of chemical release, distribution, degradation and fate in the environment
PCB uses
Electrical, heat transfer and hydraulic equipment
Plasticizers in pains, plastics and rubber products
Pigments, dyes and carbonless copy paper
Other industrial applications
PCB effects
manufactured from the 1920s and banned in 1979 by the EPS
cause cancer, immune effects (ex: killer whales), reproductive effects (ex: killer whales), endocrine effects
persist in the environment for decades
Housatonic River (PCB contamination, GE facility in Pittsfield, MA) - PCBs will degrade only over hundreds of years
From the CT Department of Environmental Protection (DEEP) - tells how to avoid PCB’s in fish, high risk vs low risk , what fish accumulates more PCB’s, what fish is safe to eat
Chemicals in the Environment and Human Health
Humans have most of the same cellular and subcellular processes as do plants and animals. We live in the same environment and are exposed to many of the same contaminants.
Environmental health therefore predicts effects on human health.
Concerns for Human Exposure risk
increased risk of various cancers, neurological problems and endocrine effects (EDCs) caused by the direct effects of relatively low levels of environmental contaminants that were used in the past or are still being used
Considerable toxicological research is directed toward
understanding the sublethal chronic direct health effects of these compounds on humans and ecosystems.
Why is causal linking of contaminant exposures to adverse effects in human population difficult?
Particularly when effects are not identified for many years and the causative agents) is/are no longer present. Since humans have a much longer life span than most other animals, low dose, chronic exposures may be significant.
Monitoring Water Quality
Testing water for chemicals of concern: streams, rivers, drinking water, well water, industrial water effluent, wast treatment effluent
Other monitoring
soil samples, air samples
using sentinel organisms for risk monitoring
Species can be collected and analyzed for toxic compounds
Useful species
Plentiful, easy to sample, prone to bioaccumulation of chemicals
Testing for exposure - catch and release strategies
Biomarkers and biomarkers of exposure
Biomarkers
Attempting to relate the presence of a hazardous chemical to a prediction of risk in an individual animal/plant, population, community or ecosystem.
Biomarkers of exposure
the presence of a chemical or its metabolite in an organism can be classified as a biomarker of exposure. In general, biomarkers of exposure are used to predict the dose received by an individual.
Convenient biomarkers of exposure and what they’re used for
exhaled air, urine, feces, blood, milk, hair, feathers —> drgu testing, breath testing, mercury in bird feathers
Biomarkers of animal heatlh
Biomarkers of effect
Biomarkers of effect
Measurable biochemical, physiological, behavioral, or other alterations within an organism that can be recognized as causing some type of toxicity
Biomarkers of effect examples
eggshell thinning by DDT/DDE, suppression of the immune system, hypersensitivity, high glucose levels, inability to capture prey, low birth rates, high infant mortality, altered migration, courtship behavior, etc.
Trophic levels in ecosystems
Decay detrivores (get energy from food) —> producers —> herbivores, primary predators, secondary predators
Autotrophs
Plants - make their own energy
Heterotrophs
Herbivores, primary predators, secondary predators - get energy from food
How have humans changed metals?
Have changed distribution and biochemical form
Interest no longer focused on preventing acute toxicity, but
chronic long-term effects of metal exposure
Convenient biomarkers of exposure to metals
blood, urine, hair
Metals of special toxic concern
Cr, Cd, Hg, Pb, As
Where do metals come from?
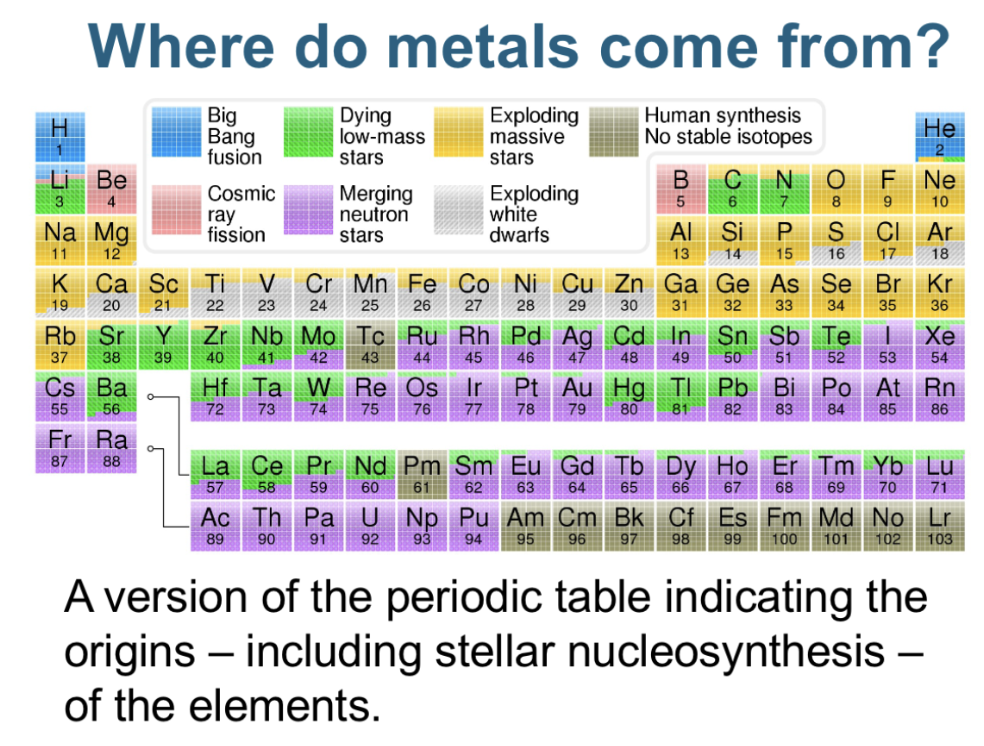
Essential metals
Essential calcium (Ca), phosphorus (P), potassium (K), sodium (Na), iron (Fe), iodine (I), copper (Cu), manganese (Mn), zinc (Zn), cobalt (Co), molybdenium (Mo), selenium (Se), nickel (Ni), vanadium (V), silicon (Si) - metalloid
Why are essential metals needed for normal functioning? - Ex: Iron
Protein hemoglobin - carries oxygen in blood, heme groups (4) - contain iron atom - can bind to/carries oxygen or carbon dioxide
Toxic essential metals
Cobalt
Non essential
everything else - lead, mercury, arsenic - metalloid, chromium, cadmium
Toxic non essential metals
Lead, Mercury, Arsenic, Chromium, Cadmum
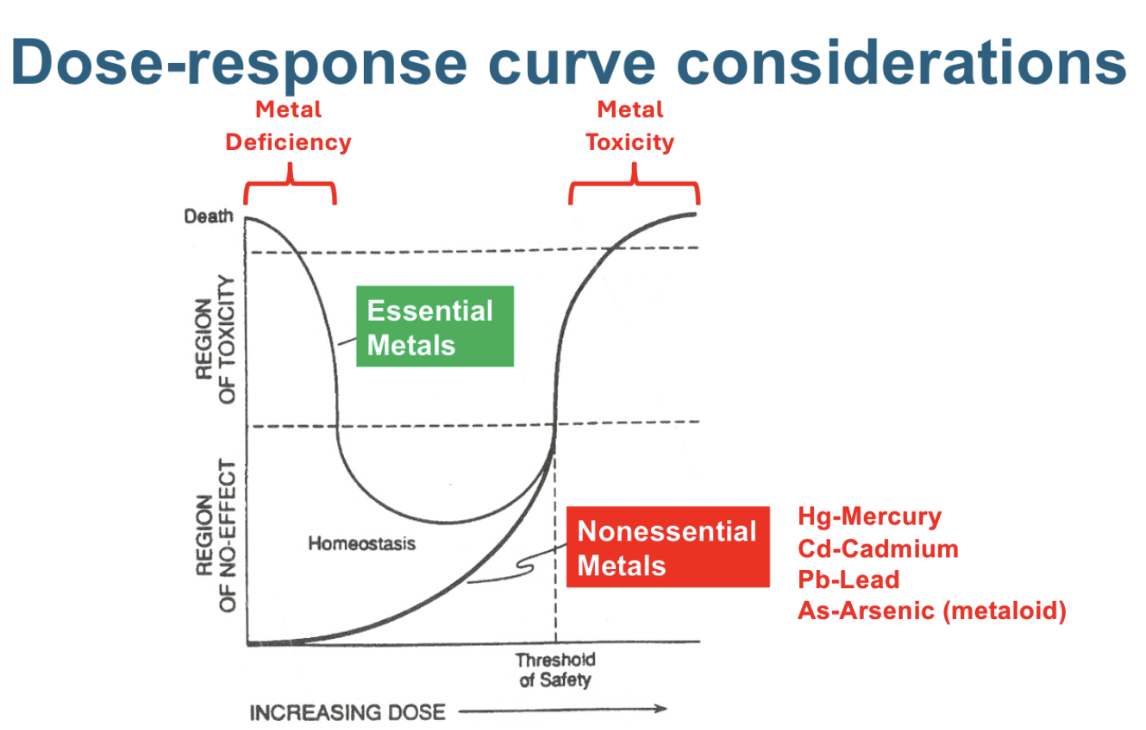
Essential metals are ____, whereas non-essential metals are _____, but __
Needed for life, common, toxic
Shape of dose response curve - essential metal deficiency and toxicity
Cobalt (Co)
Important in vitamin B12
Copper (Cu)
Important for many enzymes
Iron (Fe)
Iron deficiency is very common in the US, affecting older infants, young children and women of childbearing age, causes anemia, impaired brain development, decreased resistance to infections
Accidental ingestion of iron-containing medicines in children
Ingestion of > 2.5 g of Iron sulfate can result in acute toxicity - acute iron poisoning
Magnesium (Mg)
Important for many enzymes. Toxicity when magnesium containing drugs (usually antacids), are ingested over long periods of time.
Zinc (Zn)
More than 200 enzymes require Zn and Zn is essential for development and normal function of the nervous system.
Most Zn is from food.
Deficiency results in delayed growth and sexual maturation, dermatitis, and degenerative disorders of the nervous system
Anthropogenic and Natural metals in the environment
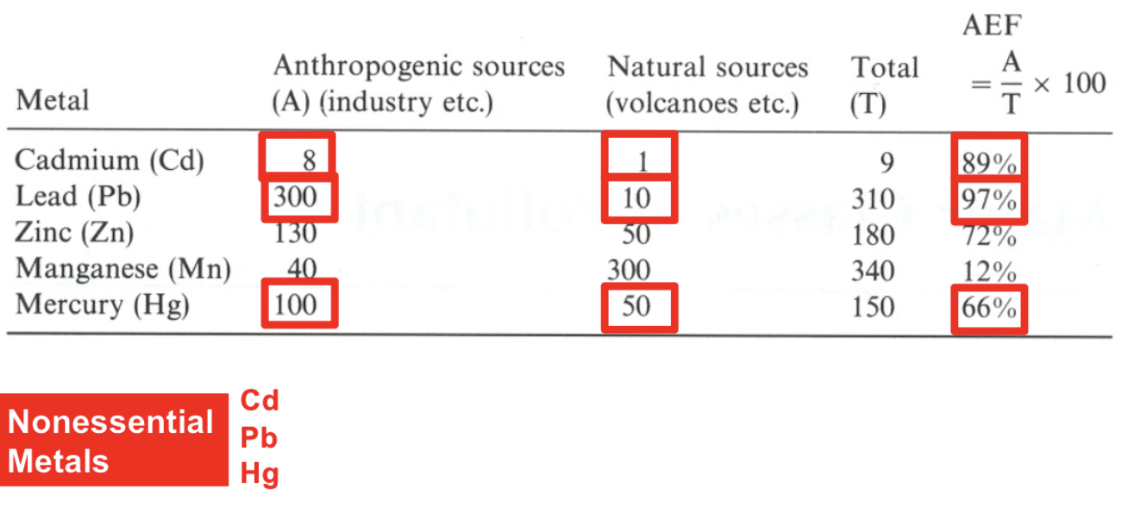
Metals vs compounds
Humans cannot create or destroy metals (and other elements) except through nuclear chemistry.
However, humans can influence the amount of chemicals in the environment (by release through industrial processes)
In contrast, humans can readily create compounds through chemistry.
Compounds are composed of elements, and chemistry can rearrange the order of elements within compounds
Interactions with other essential metals
Occurs when the body uses a non-essential metal (Hg, Cd, Pb) in place of an essential metal. For example, Lead (Pb) is similar to calcium and interferes with the calcium-dependent release of neurotransmitters (so it is a neurotoxin) and can also bioaccumulate in bone.
Age and stage of development
Major exposure to children is via food, high milk diet (high lipid content) increases metal absorption; also accidental ingestion by children (e.g., lead paint chips).
Lifestyle factors and diet
Cigarettes contain cadmium (Cd) and smoking alters pulmonary effects of metals.
Alcohol alters diet and reduces essential metal intake.
Occupational exposure (miners etc.)
Immune status
Metals that can cause immune reactions include Au, Pt, Be, Cr, Ni 5.
For many metals (Cd, Cr, Hg, Li) mechanisms of toxicity are
not entirely known
Lead toxicity
Nervous system-cognitive (IQ), hearing acuity and behavioral defects seen in chronically exposed children
Acts by impairing neuronal cell-cell connections, interferes with neurotransmitter function, competes for uptake of Ca by mitochondria.
Current FDA permissible levels in baby foods are:
10 ppb for fruits, vegetables, mixtures, meats
20 ppb for root vegetables
20 ppb for dry cereals
Lead is a possible
carcinogen
Lead in commercial products
Sure Grip Paint Brushes Recalled by Early Childhood Resources Due to Violation of Lead
Paint Standard
United Scientific Recalls Magnets Due to Violation of Lead Paint Standard
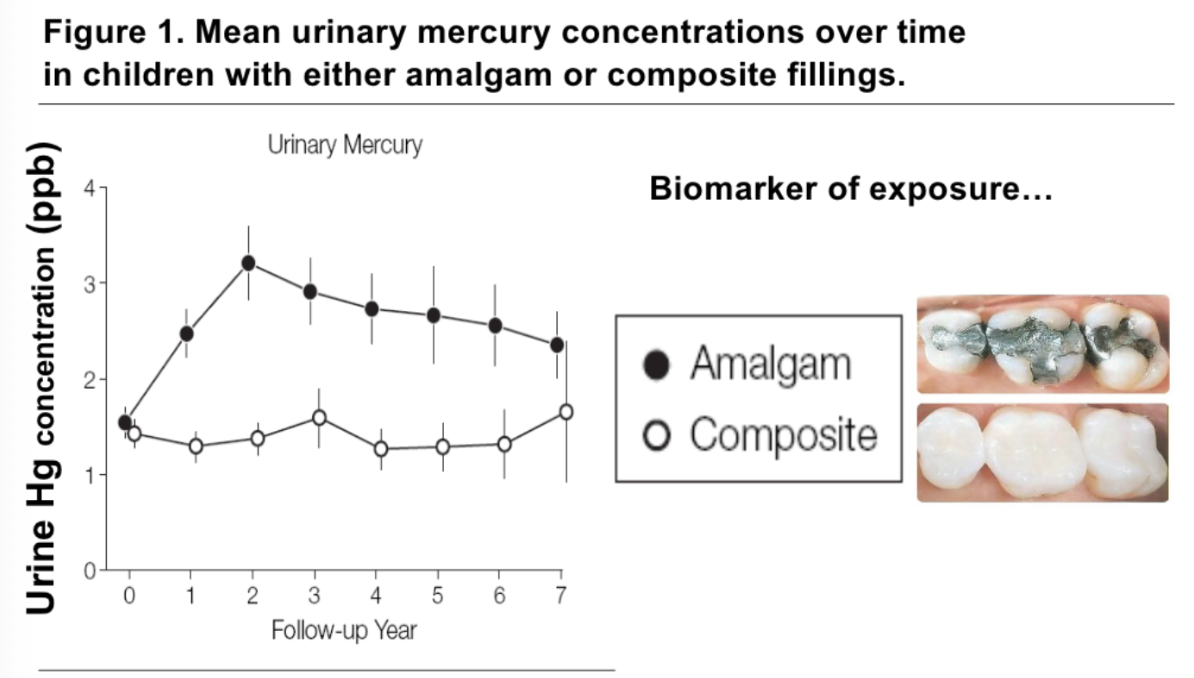
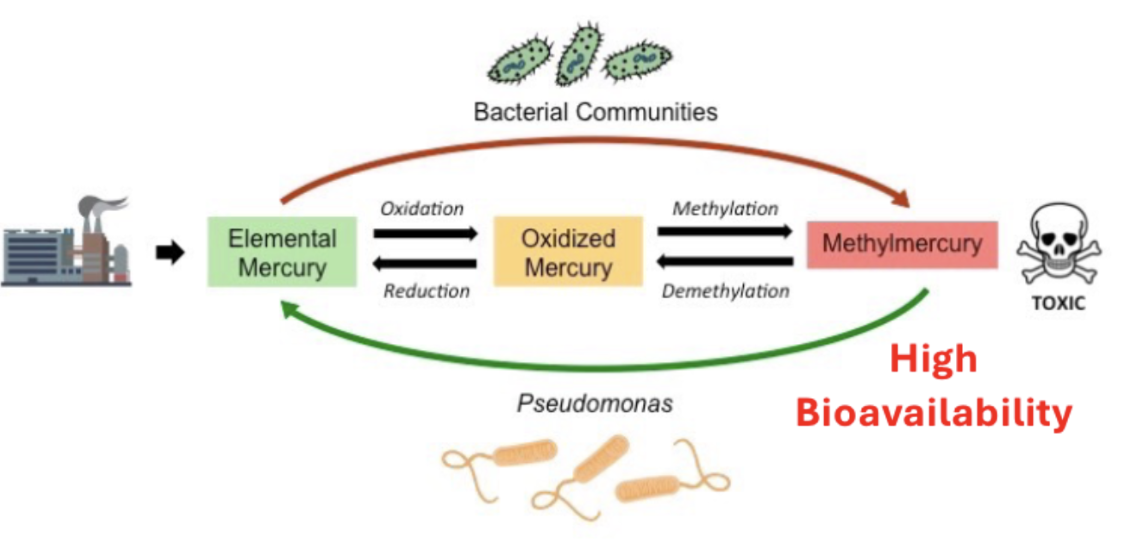
Mercury toxicity
Hg distributes globally as a vapor in the atmosphere.
Eventually returns in rainwater, then methylated by microbes to produce methylmercury, then enters aquatic food chains and can become bioaccumulated and biomagnified, reaching our food supply (mostly protein and fish).
Also, occupational exposure (extraction of gold) and in dental amalgams (fillings).
Concerns about amalgam fillings
Neurobehavioral Effects of Dental Amalgam in Children: A Randomized Clinical Trial
Amalgams contain 50% liquid Hg (+ silver (~27%), tin (~15%) & copper (~8%))
Mercury persistence
Industrial Mercury Poisoning
Vinyl Chloride and acetaldehyde plants used HgCh as catalyst.
700 cases of poisoning & >70 deaths
Minamata disease victim
Mercury and methylmercury
Mercury in ecosystems
methylmercury: bioaccumulation and biomagnification
Mercury accumulation and trophic levels
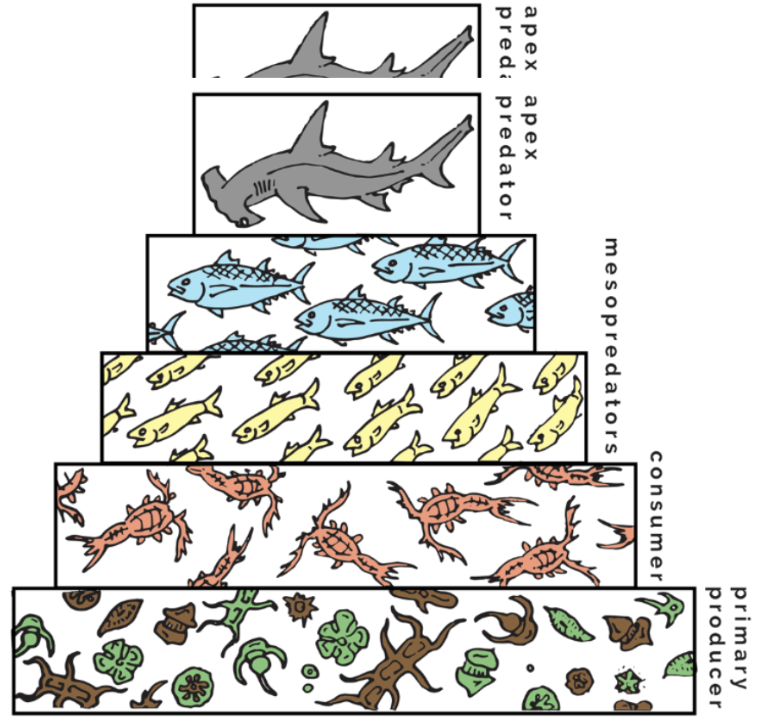
____ fish accumulate mercury
Older/larger
Arsenic (As)
5 to 20 ug/day in our food and water (and some inhalation (cigarettes)); excretion is relatively rapid (a few days) & mainly via urine. Probably an essential metal in trace levels.
Arsenic (at high concentrations) is a mitochondrial toxin that acts by blocking the production of ___ and increases ___.
ATP, ROS
70 to 180 mg Arsenic as a single dose
may be fatal
Symptoms:
Chronic exposure (>1 mg/day?) may lead to fever, anorexia, cardiac arrhythmia, neurotoxicity.
Known skin and lung carcinogen (dose???)