Transition metals
1/32
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
33 Terms
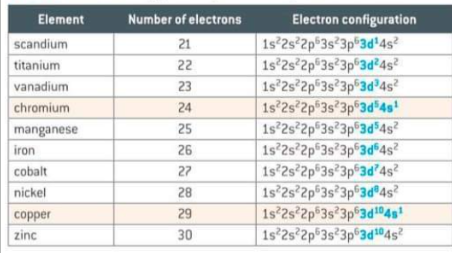
What is the special case of chromium and copper in electron configuration?
The electron configurations do not follow the expected principle for placing electrons singly in orbitals before pairing. A half filled d5 and fully filled d10 give more stability
What happens when the d-block elements form positive ions?
they lose their 4s electrons before losing any of their 3d electrons.
When forming an atom in the d block element which subshell fills first 3d or 4s?
4s
When forming an ion in the d block element which subshell empties first 3d or 4s?
4s
What are transitions elements?
d-block elements that form at least one ion with a partially filled d-orbital. Although scandium and zinc are d-block elements they do not match this definition and are not classed as transition elements
Why are scandium and zinc exceptions?

What are the three characteristic properties of transition metals?
they form compounds in which the transition element has different oxidation states
they form coloured compounds
the elements and their compounds can act as catalysts
Formation of coloured compounds - how do transition metals form coloured compounds?
the colour of the solution is linked to the partially filled d-orbitals of the transition metals. The colour of a solution can vary with different oxidation states

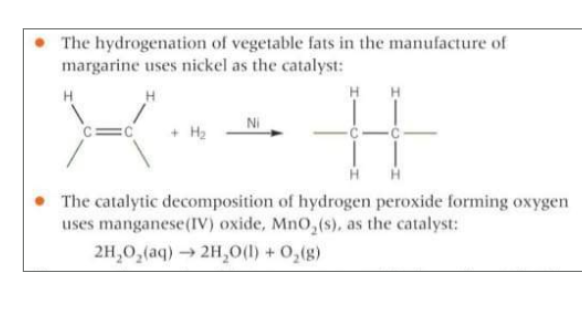
When is iron a catalyst when is vandium a catalyst?

When is nickel a catalyst, when is MnO2 a catalyst
true or false transition metals can form both heterogeneous and homogeneous catalysts
true
Describe the reaction when Fe2+ ion act as a homogenous catalyst

When is a complex ion formed?
when one or more molecules or negatively charged ions bond to a central metal ion, these molecules or ions are known as ligands
What is a ligand?
a molecule or ion that donates a pair of electrons to a central metal ion to form a coordinate bond or a dative covalent bond
What is the coordination number?
The number of coordinate bonds attached to the central metal ion
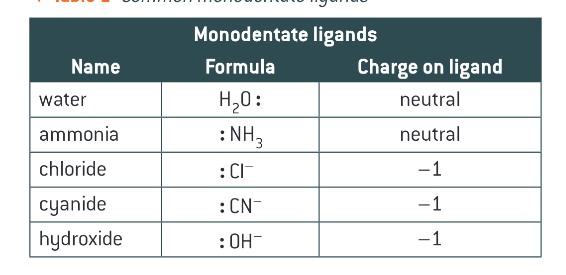
What is a monodentate ligand?
a ligand that is able to donate 1 par of electrons to a central metal ion
What is a bidentate ligand?
one that can donate 2 lone pairs of electrons to the central metal ion, forming 2 coordinate bonds
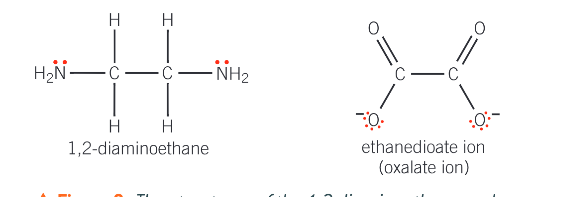
What does the shape of the complex ion depend on?
the coordination number
If a molecule has a coordination number of 6 what is the shape of the moleucle?
octahedral
What is the shape of molecules which a coordination number of 4?
tetrahedral and square planar
Where does the square planar shape occur?
in complex ions of transition metals with 8 d-electrons in the highest energy d-sub-shell. Platinum (II), Palladium (II) and gold (III) fall in this category. Bond angle of 90degrees
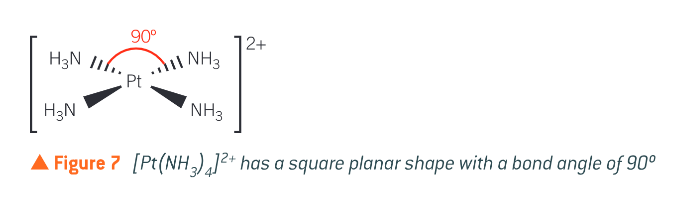
What are steroisomers? What are the 2 types complex isomers can display?
same structural formula different arrangement of atoms in space
cis/trans isomerism
optical isomerism
What types of complex ions show cis/trans isomerism?
some four coordinate and six coordinate complex ions containing 2 different monodentate ligand
What types of complex ions show optical isomerism?
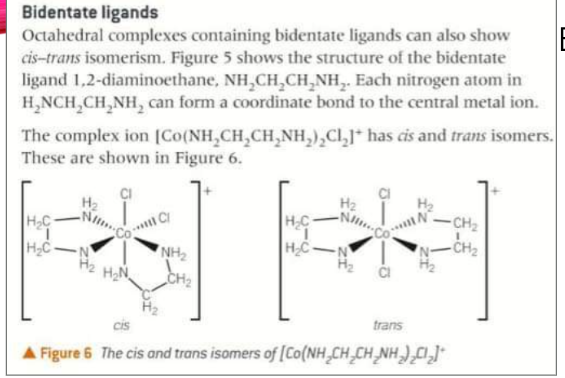
some six coordinate complex ions containing monodentate and bidentate ligands can show both cis-trans and optical isomerism
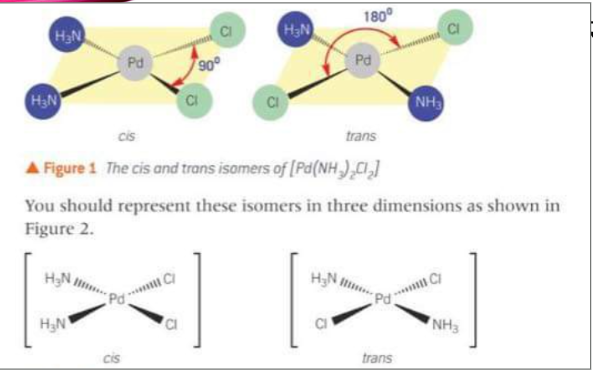
What do the cis-trans isomers of [Pd(NH3)2Cl2] look like?

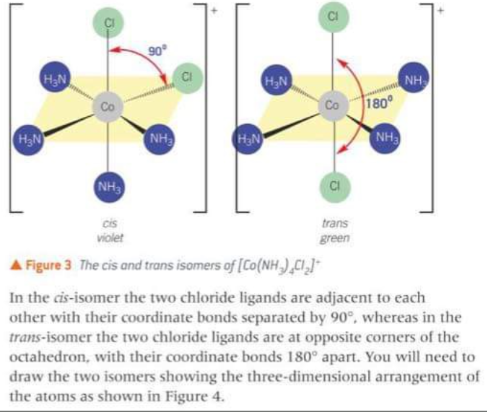
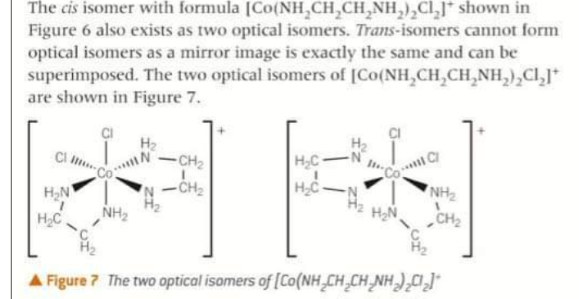
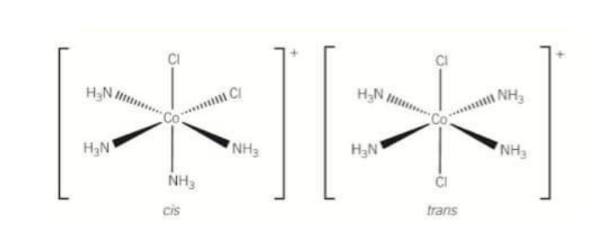
What does the cis/trans optical isomerism of [Co(NH3)4Cl2]+ look like

What are optical isomers (enantiomers)?
non-superimposable mirror images
Where does optical isomerism occur in complex ions?
in octahedral complexes containing 2 or more bidentate ligands

Optical isomers of this complex ion