4.2 - Aromaticity
1/38
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
39 Terms
Kekule structure of benzene
Six membered ring containing single and double bonds
Why the Kekule structure of benzene is incorrect
benzene does not undergo addition reactions unlike compounds with double bonds
Each C-C bond length is equal (less than C-C and greater than C=C)
Enthalpy of hydration is less than expected to more thermodynamically
molecular formula of benzene
C6H6
geometry of benzene
Planar
Bond angle in benzene?
120 between 3 adj carbon atoms
Kinds of bonds benzene contains
Sigma and pi
How sigma bonds are formed in benzene
Each C atom is bonded to two other carbon atoms and a hydrogen atom by sigma bonds
How pi bonds are formed in benzene
Fourth outer shell electron is in a 2p orbital above and below the plane of the carbon ring. These p orbitals overlap sideways, forming pi delocalised electron system structure above and below the plane of the carbon ring
Stability of benzene
The more electrons delocalised, the more stable the compound
Name for extra stability in benzene
Delocalisation/resonance energy
Kind of mechanism benzene undergoes
Electrophilic substitution
Why benzene does not undergo addition reactions
This would disrupt the stable delocalised pi electron system and the resulting product would be less stable
Why benzene is more susceptible to electrophilic attack
Delocalised ring of electrons is an area of high electron density
Makes it susceptible to attack by an electrophile
If a H atom is replaced by an electrophole, the delocalisation must be maintained
Three reactions that benzene undergoes which involve electrophilic substitution
Nitration
Halogenation
Friedel-Crafts Alkylation
Reagent used in nitration of benzene
Conc. HNO3 (aq)
Catalyst used in nitration of benzene
Conc. H2SO4 (aq)
Electrophile in nitration of benzene
NO2^+
Equation for formation of the electrophile in nitration of benzene

Mechanism for nitration of benzene

Overall equation for nitration of benzene

Equation for reforming the catalyst in nitration of benzene

Reagents and conditions in nitration of benzene
Conc. H2SO4 catalyst
50•C
Reagent used in halogenation of benzene
Br2 or Cl2
Catalyst used for halogenation of benzene?
FeBr3 or AlCl3
Equation for formation of the electrophile in halogenation of benzene?

Mechanism for halogenation of benzene
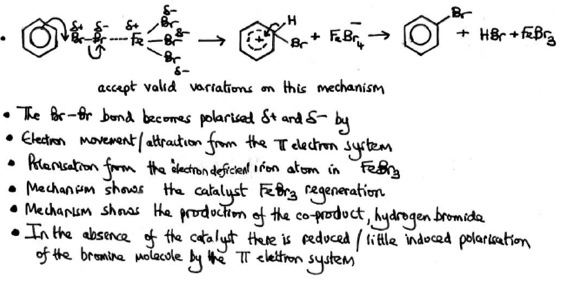
Overall equation for halogenation of benzene
Equation for reforming the catalyst in halogenation of benzene

Reagents and conditions for halogenation of benzene
FeBr3 or AlCl3
Room temp
Reagent for alkylation of benzene
Chloroalkane (R-Cl)
Catalyst for alkylation of benzene
AlCl3
Equation for formation of the electrophile in benzene
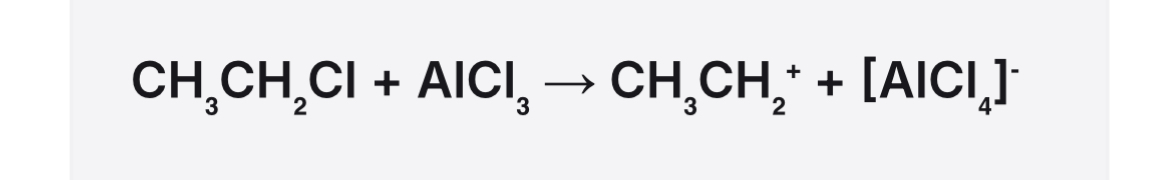
Mechanism for alkylation of benzene
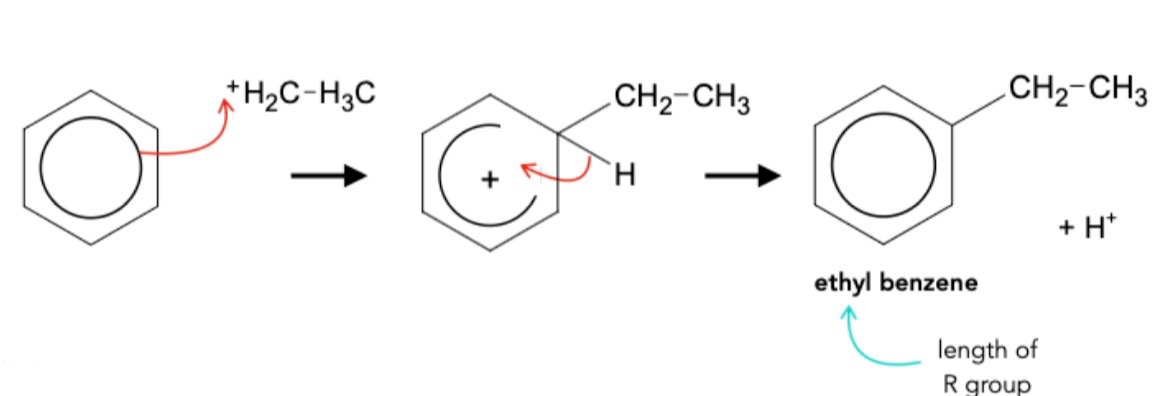
Electrophile in alkylation of benzene
CH₃CH₂⁺ (ethyl carbocation)
Overall equation for alkylation of benzene

Equation for reformation of the catalyst in alkylation of benzene
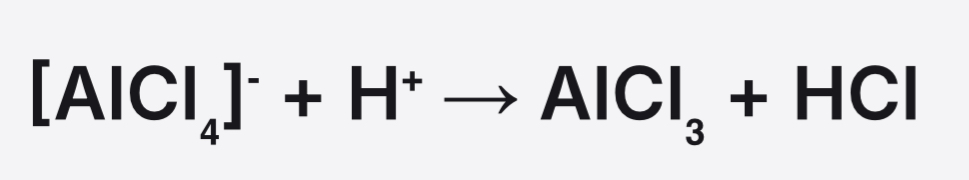
Reagents and conditions for alkylation of benzene

Compare C-Cl bond strength in chlorobenzene to that of a chloroalkane
greater bond strength in chlorobenzene
Overlap between non bonding p electron pairs on the chlorine and the delocalised pi electron ring system
Gives pi character to the bond
Strengthens the bond
Much more energy is needed to break it
Compare the reaction of chlorobenzene with aqueous sodium hydroxide with that of a chloroalkane
chloroalkanes react with aqueous sodium hydroxide in a nucleophilic substitution reaction to give alcohols
Chlorobenzene does not react with aq NaOH
Resistant to nucleophilic substitution because nucleophiles would be repelled by the stable pi system of electrons