dissociation curves
1/19
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
20 Terms
haemoglobin structure
4 pp chains
iron ion haem grp
prosthetic grp attached to each chain via ionic disulphide bridges
why is haemoglobin able to bind w/ oxygen so well
it has a high affinity for oxygen
affinity
strength of binding of haemoglobin to oxygen
what do oxygen and hemoglobin join when formed and how ?
form oxyhaemoglobin in a reversible reaction so it can also unload and disassociate oxygen able to diffuse into body cells for respiration
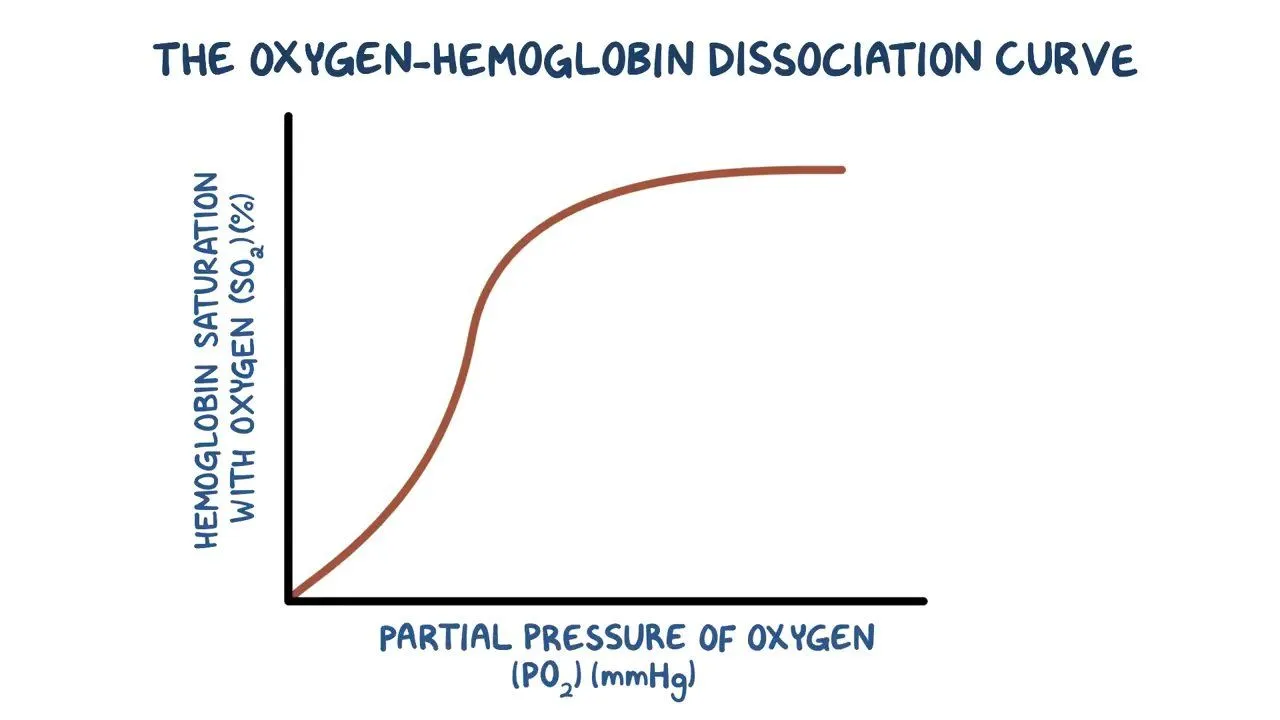
what happens when haemoglobin binds to its first oxygen ?
its tertiary structure shape alters uncovering another haem binding state making it easier for other molecules to join
shown by the steep middle in the curve
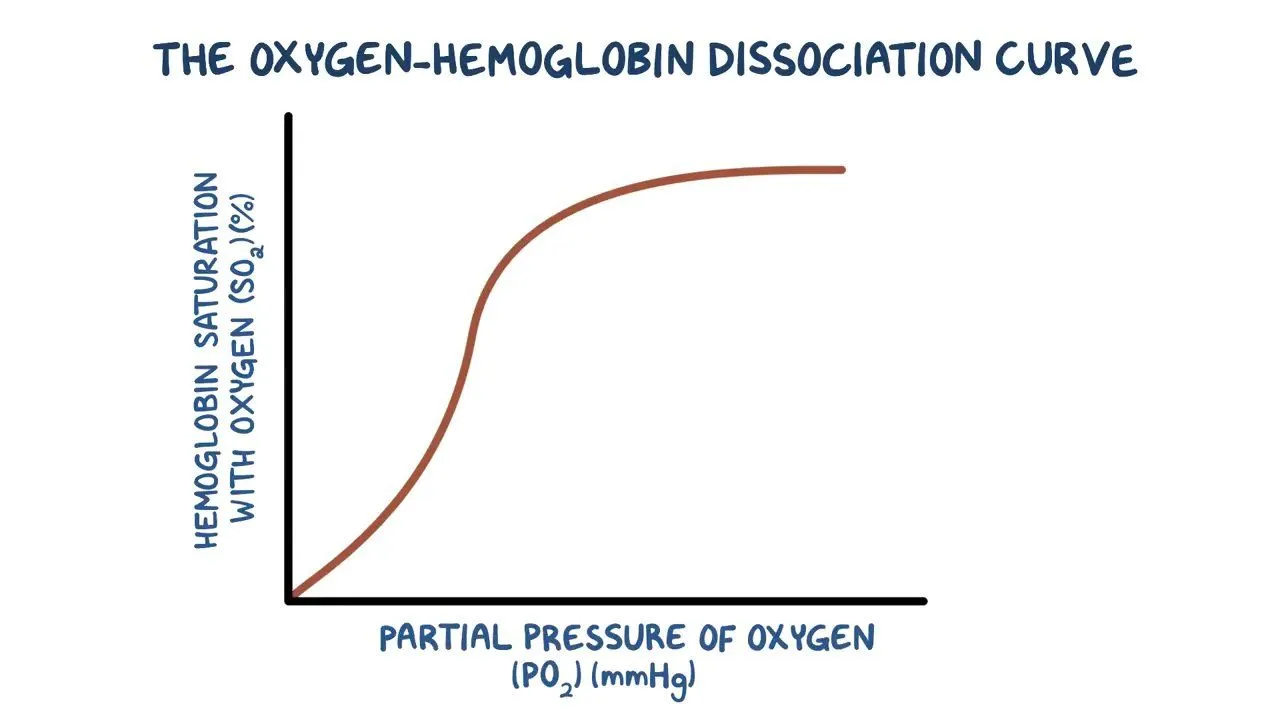
how is a more rapid intake of oxygen ensured in the lungs (refer to the graph)?
a small change in pO2 can cause a big change in the vol of O2 carried by the Hb
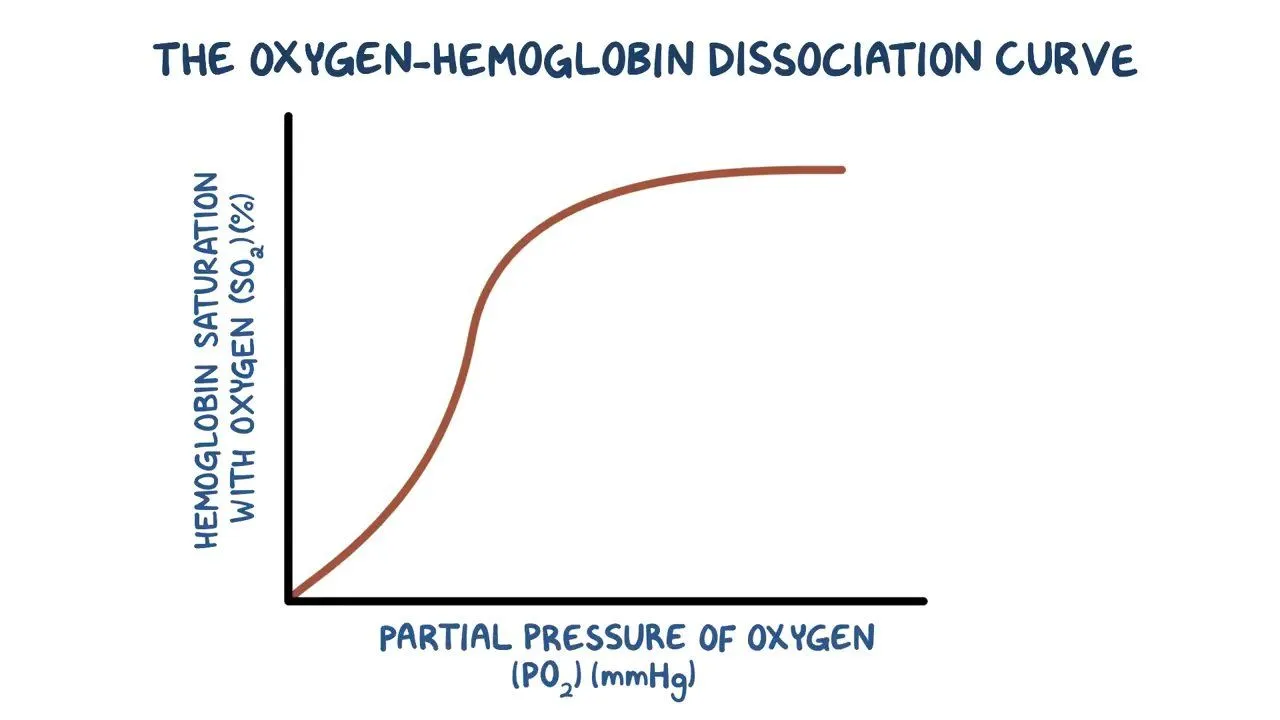
why does the graph begin to flatten towards the end ?
as the Hb becomes more saturated it becomes harder for the O2 to join onto it as most of the binding sites are taken
however Hb will still always load O2 even at high concs
partial pressure of oxygen
the conc of dissolved oxygen in cells
how is Hbs affinity for oxygen affected by the partial pressure of oxygen increasing ?
affinity incs
Hb loads more readily than unloads and dissociates so becomes more saturated
how is Hbs affinity for oxygen affected by the partial pressure of oxygen decreasing ?
affinity decs
Hb unloads and dissociates more readily than loads so becomes less saturated
effect of partial pressure in the lungs
alveoli have a high pO2
Hbs affinity incs readily loads than unloads
higher saturation
effect of partial pressure at the body cells
rbcs deliver the oxyhaemoglobin to the respiring tissues which have used the O2 up (low pO2 )
affinity decs
Hb readily unloads and dissociates
lower saturation
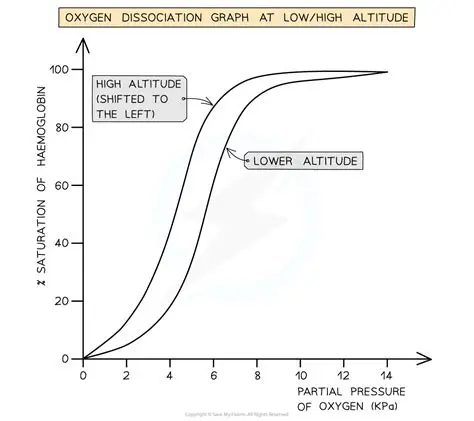
curve for an active animal
curve to the right
lower affinity + saturation
Hb unloads and dissociates more readily than loads and is less saturated at any given partial pressure
more O2 to cells so more respiration
what happens as the partial pressure of CO2 increases ?
the Bohr effect
Hbs affinity decs
unloads and dissociates more readily than loads
O2 saturation of Hb decs
more O2 is released to allow for aerobic resp and prevent anaerobic resp
less chance of lactic acid build up
curve shifts right
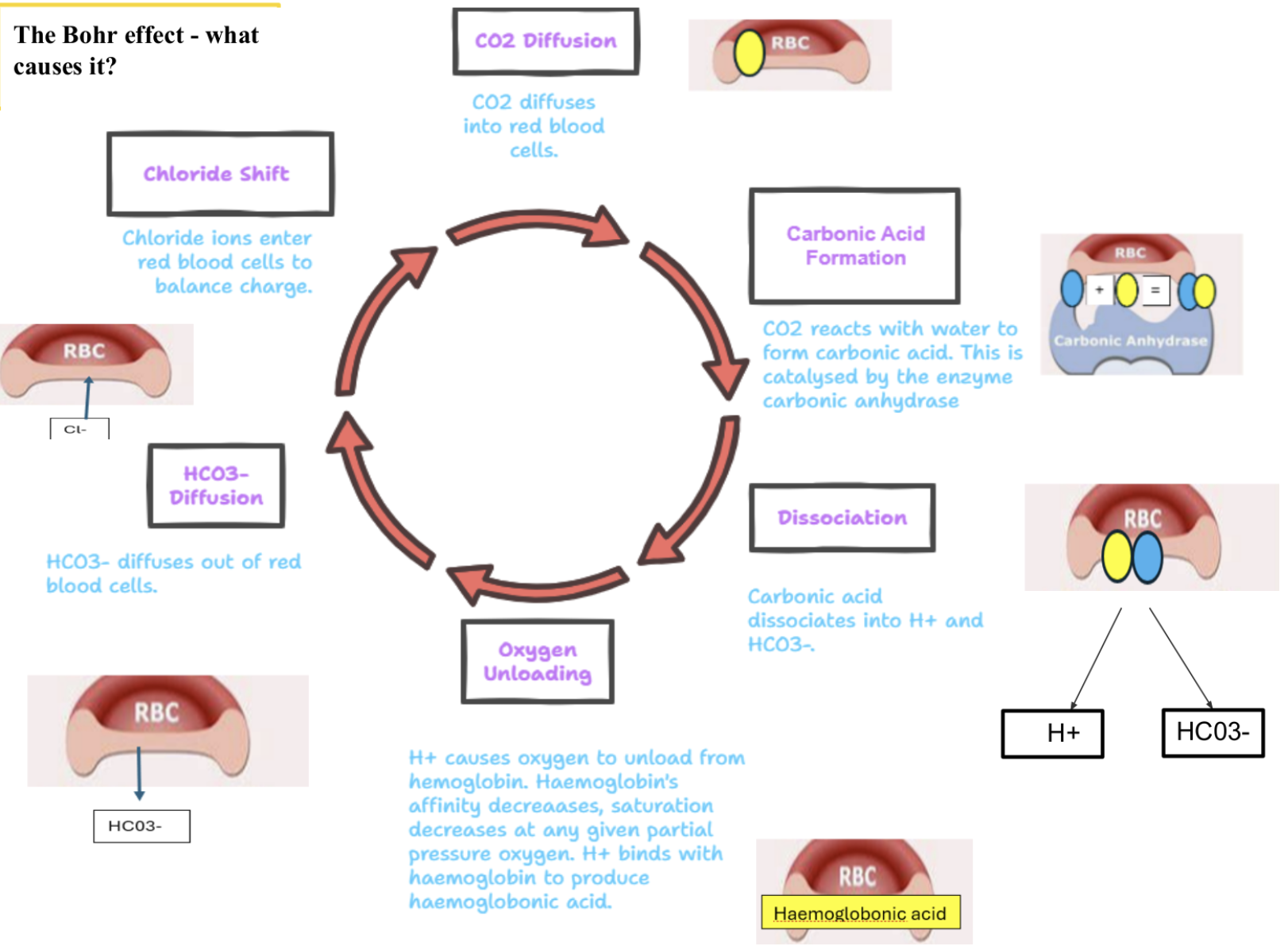
what causes the Bohr effect
CO2 diffuses into rbcs
CO2 reacts w/ water to form carbonic acid catalysed by carbonic anhydrase
carbonic acid dissociates into H+ and HCO3-
H+ causes O2 to unload from Hb - affinity decs - saturation decs at any given pO2
H+ binds w/ Hb to produce haemoglobonic acid -decs blood pH
HCO3- diffuses out of rbcs
chloride shift - Cl- ions enter rbcs to balance charge
how is CO2 made for exhalation ?
in the lungs :
pO2 is much higher causing haemoglobonic acid to dissociate - H+
H+ binds to HCO3- ions that diffuse back into the rbcs making carbonic acid
carbonic anhydrase catalyses this reversible reaction back to water and CO2
CO2 is exhaled
Hb is now free to bind w/ O2
how does fetal haemoglobin differ from adult haemoglobin
fetal Hb has a higher affinity for oxygen at any given partial pressure bcs :
adult Hb unloads and dissociates at the placenta due to low pO2
Hb saturation decs
O2 diffuses frm adult to fetal Hb
fetal Hb loads and becomes more saturated at this given pO2
O2 is then delivered to cells of fetus for resp
energy released for cell division - growth
myoglobin
dark red muscle pigment found in muscle cells
no role in O2 transport
used as an O2 store
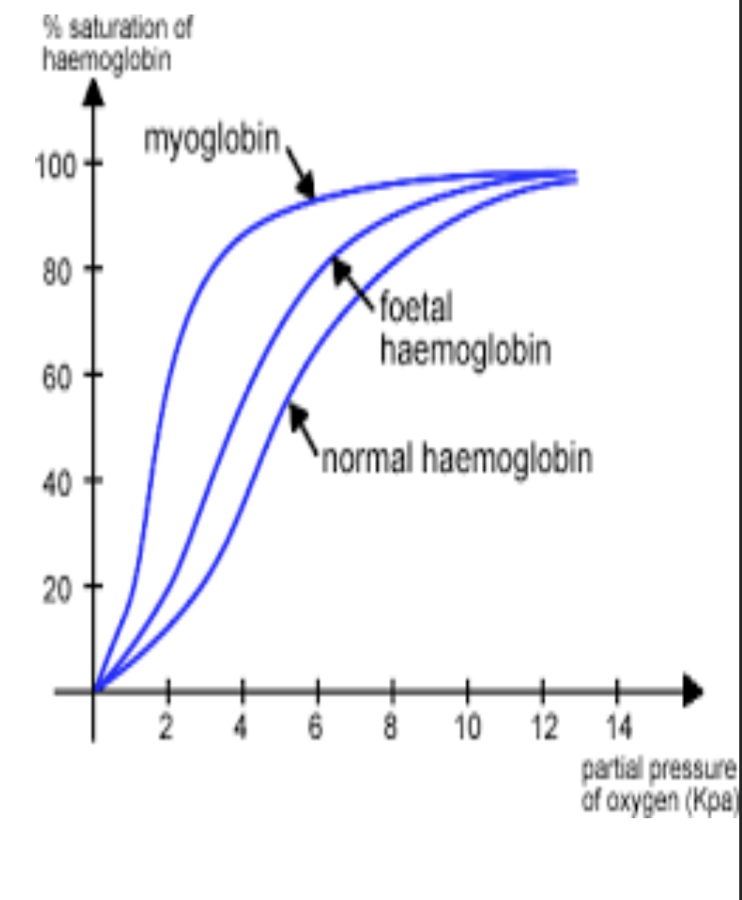
what is the role of myoglobin?
high affinity for O2
readily picks up O2 but will only give it up when O2 conc dips to vv low lvls
in vv active muscles the O2 delivered by the blood may not be enough and the O2 conc drops below 0.5kPa
myoglobin releases its O2 providing a reserve supply to keep the muscle going
eventually the myoglobin will have no more O2 to give and the muscle has to respire anaerobically
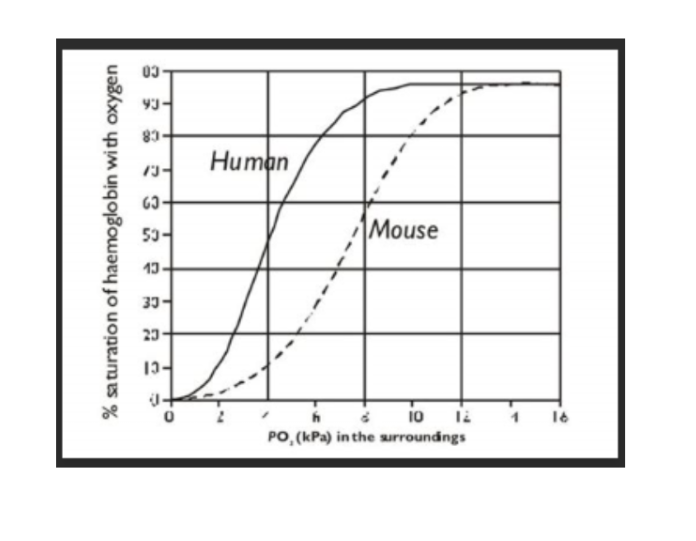
how does size affect the curve
smaller mammals have a a higher sa:v ratio
they lose heat vv quickly
so they have a high metabolic rate to keep warm
high O2 demand so lower affinity to O2
curve shifts to the right