chem231 - ch.10 - radicals & alkyl halides
1/20
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
21 Terms
radical
a reactive intermediate w/ a single unpaired e-
heterolysis
2 e- movement
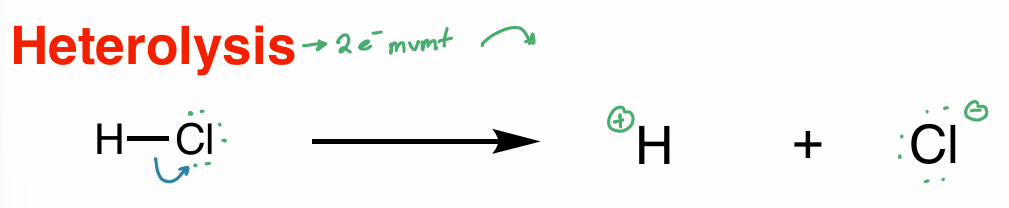
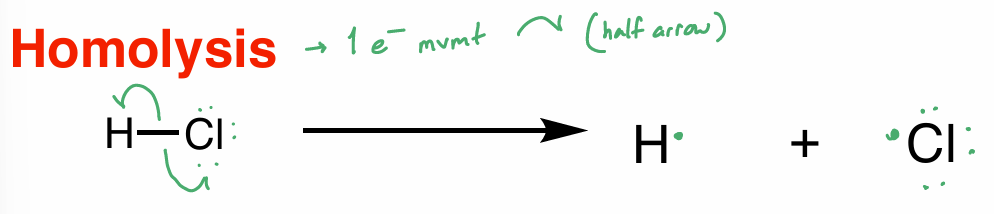
homolysis
1 e- movement
half arrow
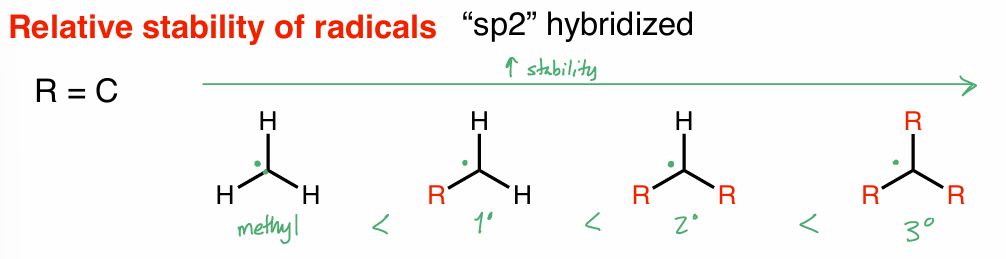
radical stability: hybridization
R = C
want 3°
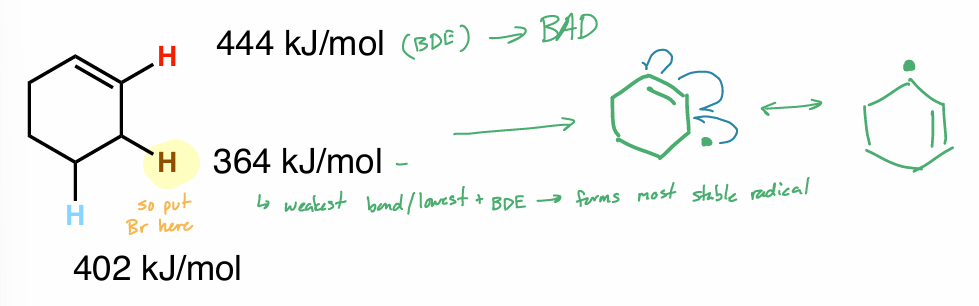
weakest/lowest BDE → most stable/fastest forming radical
radical stability: resonance
more resonance stabilized → more stable
e- coming together to form double bond
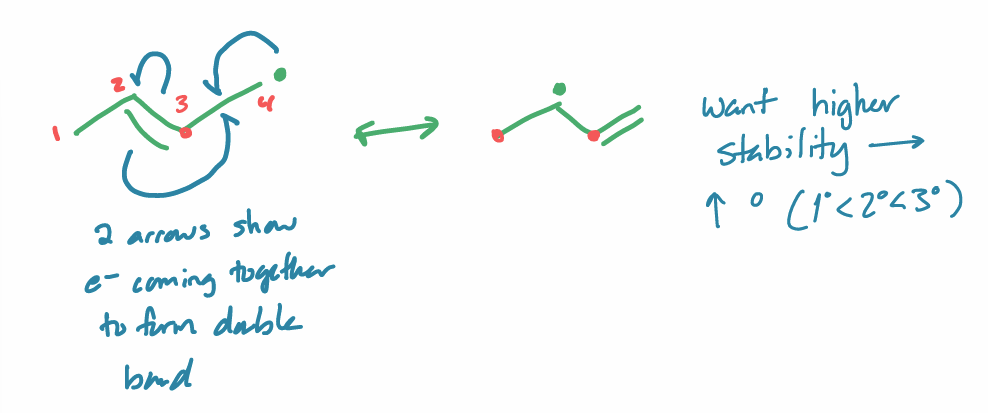
radical stability example
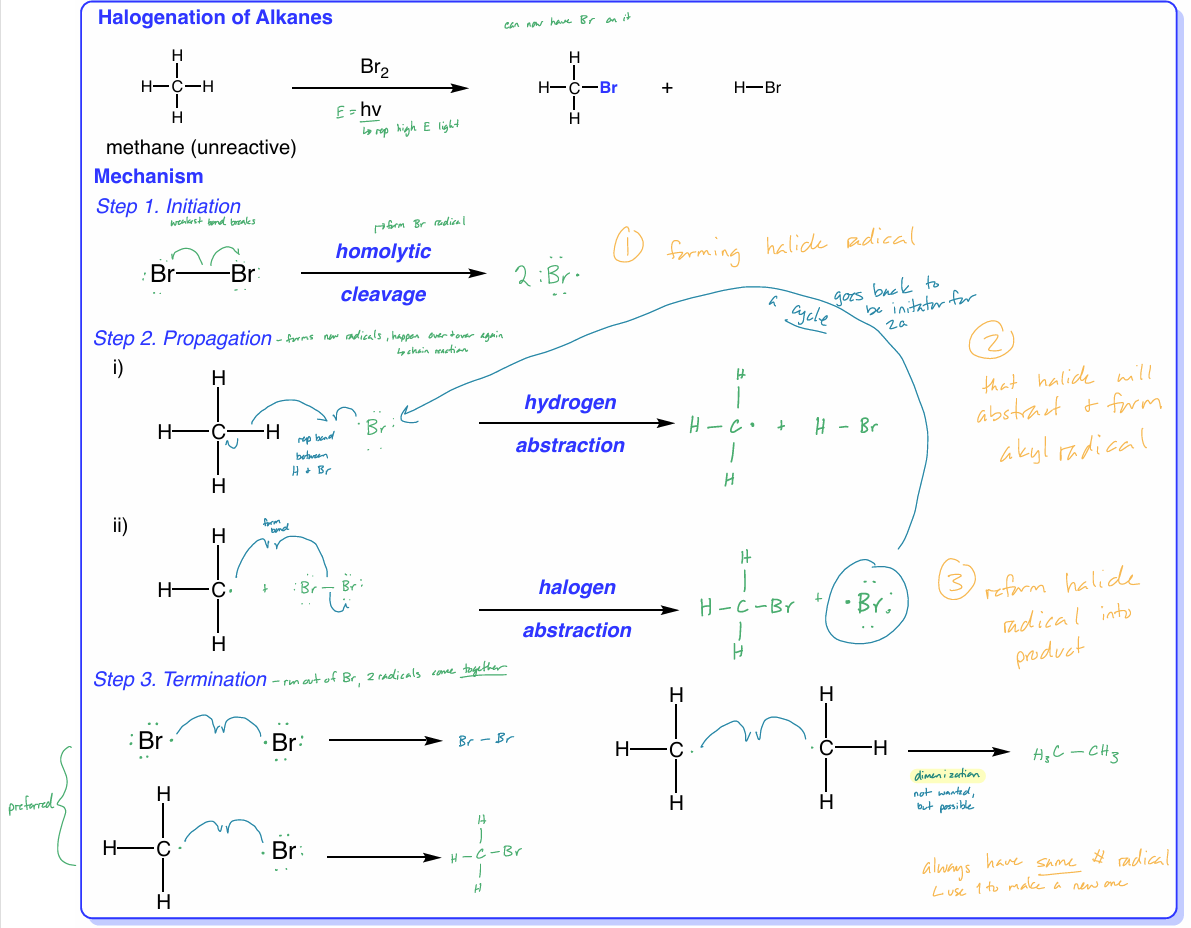
alkane halogenation
alkanes react w/ halogen (Cl2 or Br2), + heat/light (hv), H atom of alkane is replaced by halogen atom to form alkyl halide product
initiation → form halide radical
propagation → that halide will abstract & form alkyl radical
termination → reform halide radical into product
always have same # of radicals
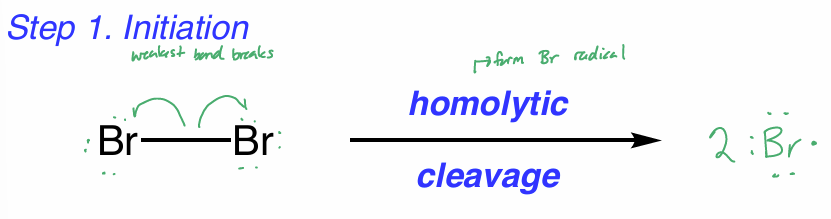
halogenation: initiation
weakest bond breaks
homolytic cleavage to form Br radical
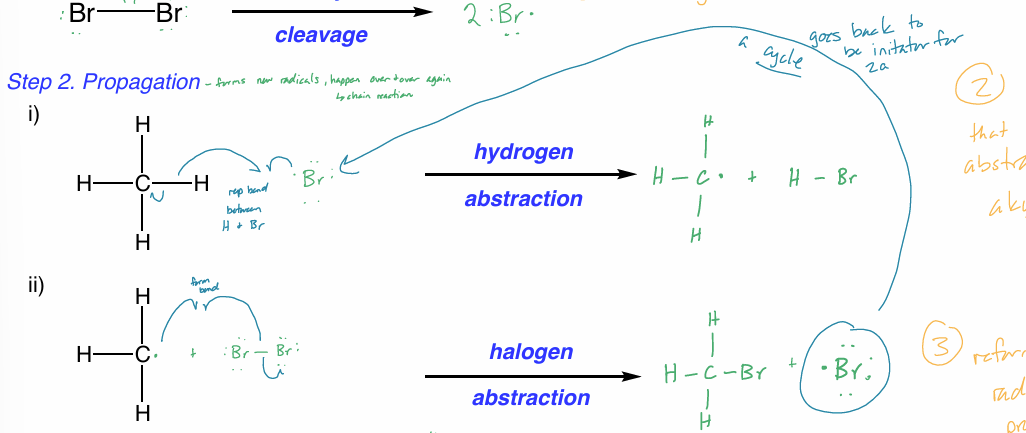
halogenation: propagation
forms new radicals
hydrogen abstraction
Br radical makes bond w/ 1 e- from H bond
other H e- goes to C
halogen abstraction
1 e- from Br—Br form bond w/ newly made carbon radical
other e- from Br—Br goes to Br → new Br radical
Br radical can be used as reactant in hydrogen abstraction
happens over & over again (chain reaction)
the product of the previous step is used as the reactant in the next step
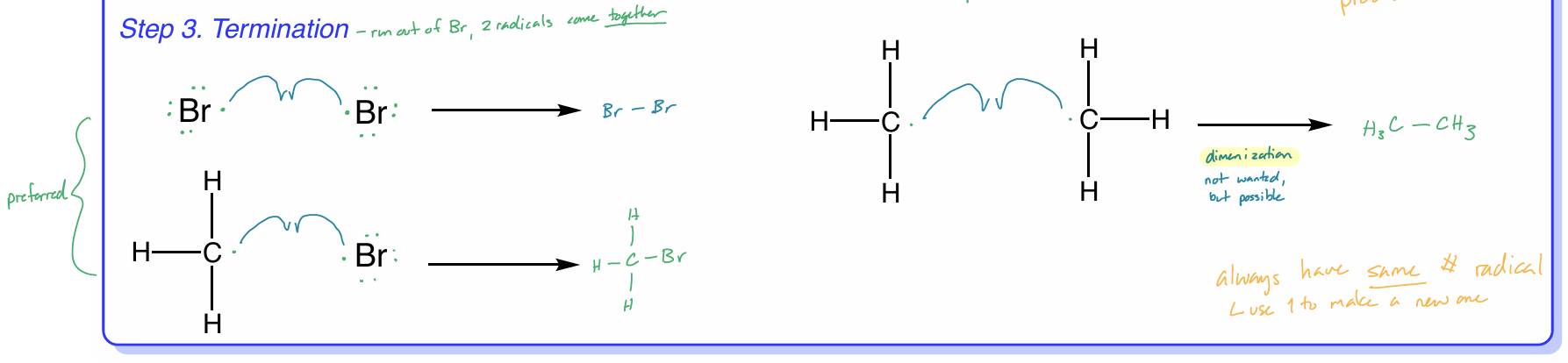
halogenation: termination
when you run out of Br, the 2 radicals come together
3 options
2 Br radical e- come together & form Br—Br bond
H3C radical + Br radical come together → H3CBr
these 2 preferred
2 H3C radicals come together → H3C—CH3
dimerization - not wanted, but possible
radical indicators
peroxides - oxygen—oxygen bond is weak
radical forms w/ heat
hv (light) - light E can break X—X bond → radicals

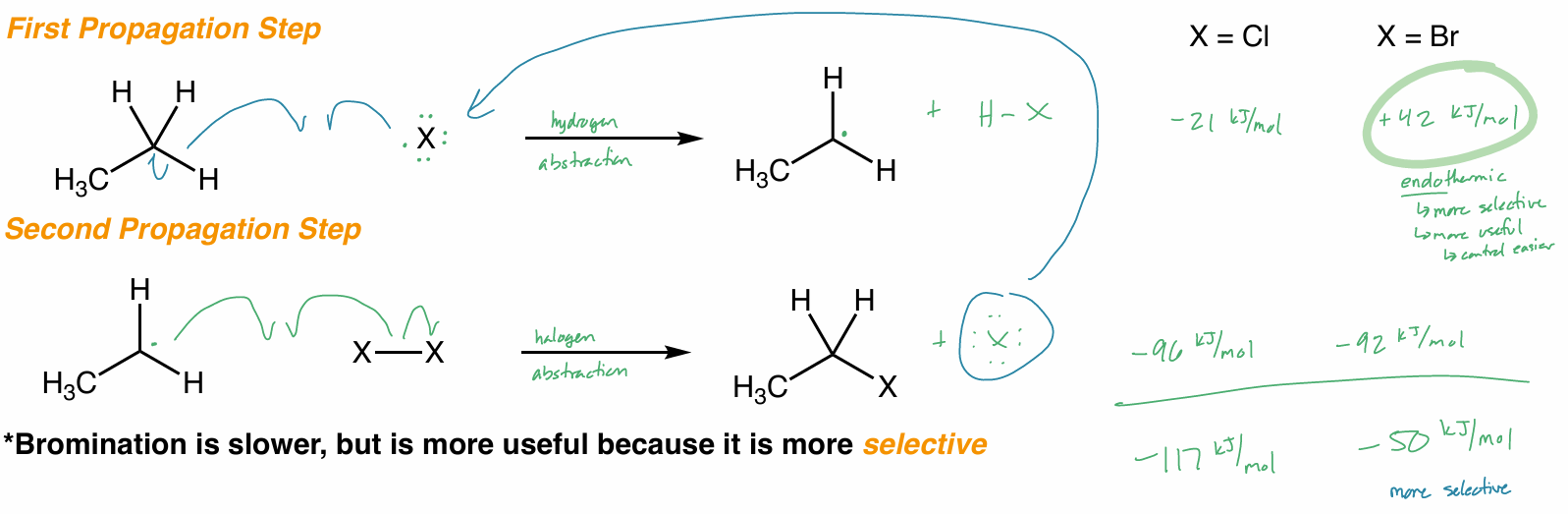
radical thermodynamics
only Cl2 & Br2
Br2 is slower, but more selective → more useful
both exothermic
F2 too explosive
I2 too slow, NR
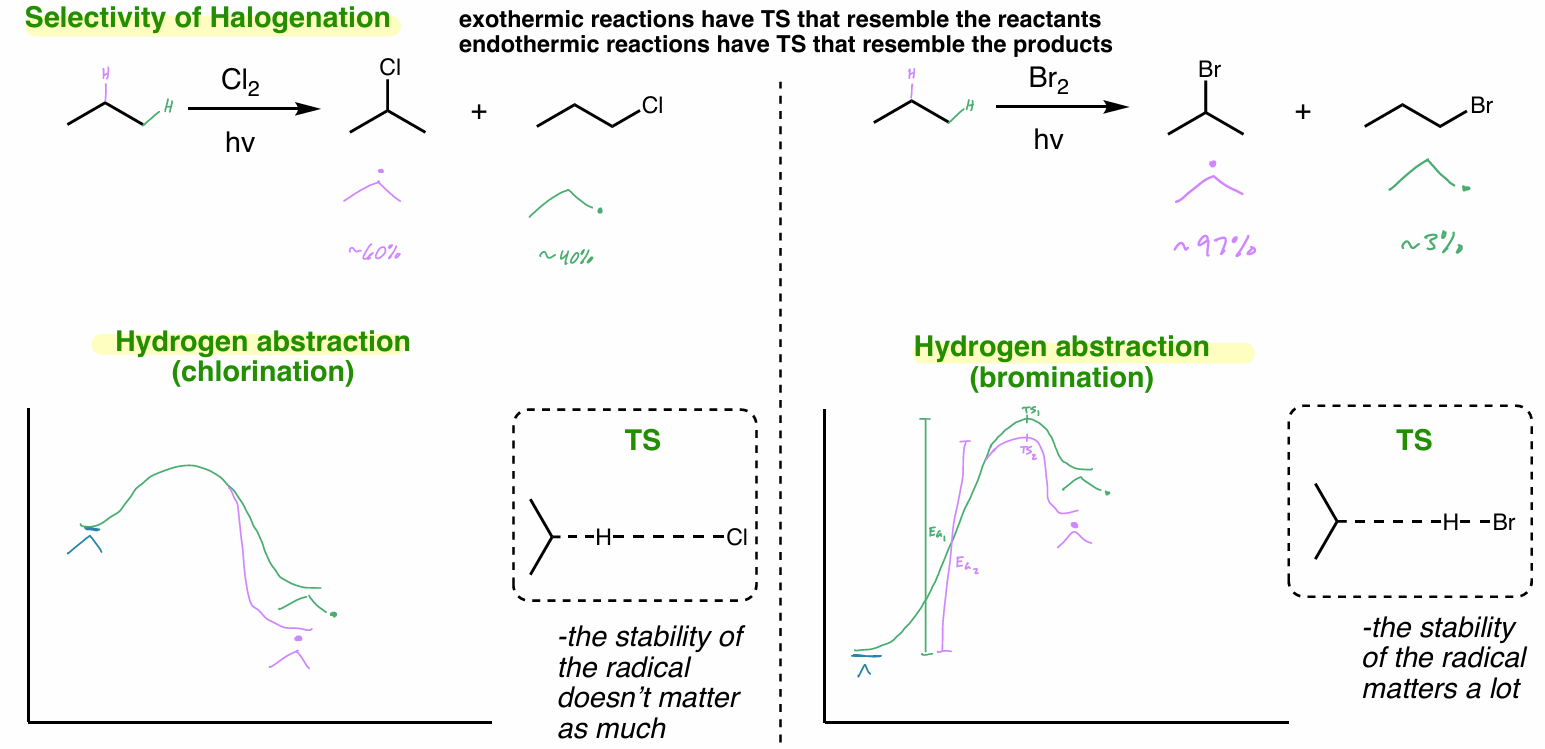
halogenation selectivity
exothermic reactions → TS resemble reactants
endothermic reactions → TS resemble products
don’t get good selectivity for better radical b/c exothermic
racemic
solid lines in skeletal
represent both dash & wedge (50:50)
implied w/ lines
must write if dash/wedge present
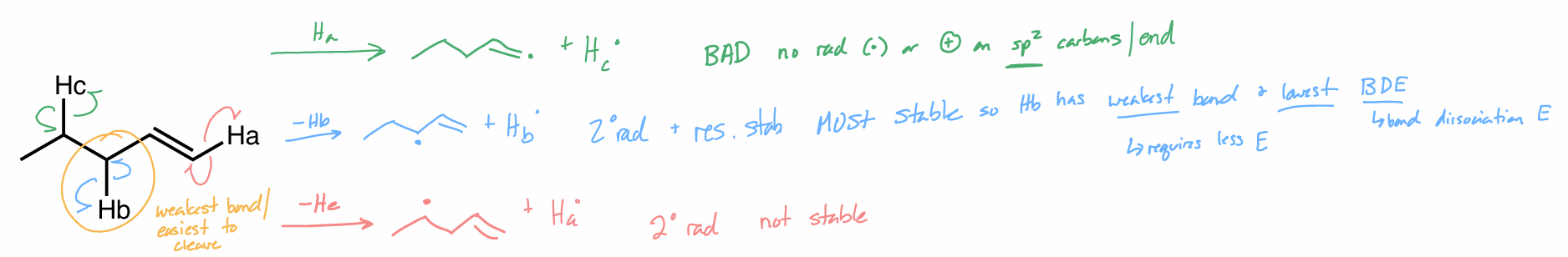
allylic bromination
best C—H bond to be abstracted
↑ stable radical → ↓ bond + ↑ resonance stabilized
don’t put e- on double bond (DB)
N-bromosuccinimide (NBS)
if molecule has double bond you do not want to react w/ Br2, use NBS + hv to do radical halogenation
generates small amt of Br2
small [Br2]
![<p>if molecule has double bond you do <strong>not</strong> want to react w/ Br<sub>2</sub>, use NBS + hv to do radical halogenation </p><ul><li><p>generates small amt of Br<sub>2</sub> </p><ul><li><p>small [Br<sub>2</sub>]</p></li></ul></li></ul><p></p>](https://assets.knowt.com/user-attachments/9ca736fa-7335-416d-b445-782c51b4e6e1.png)
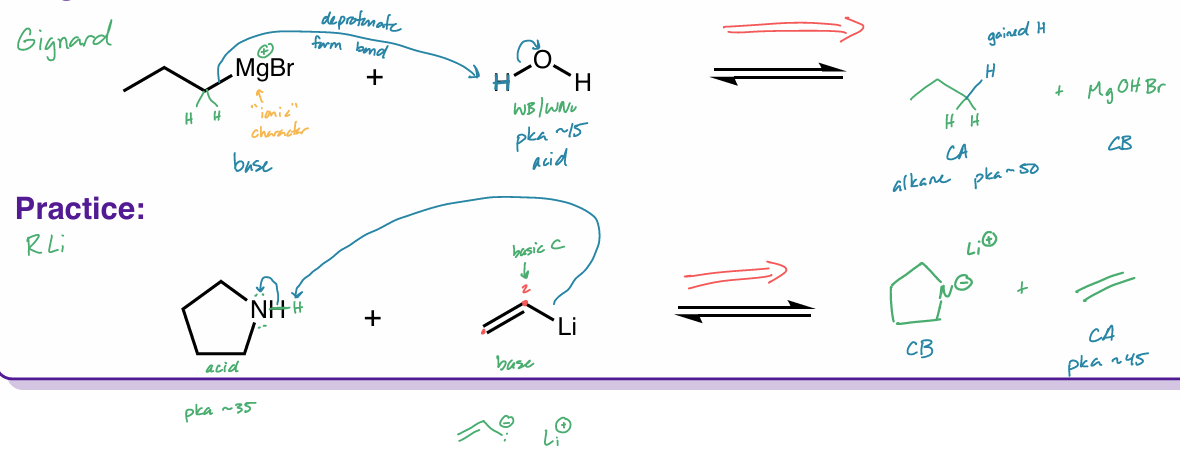
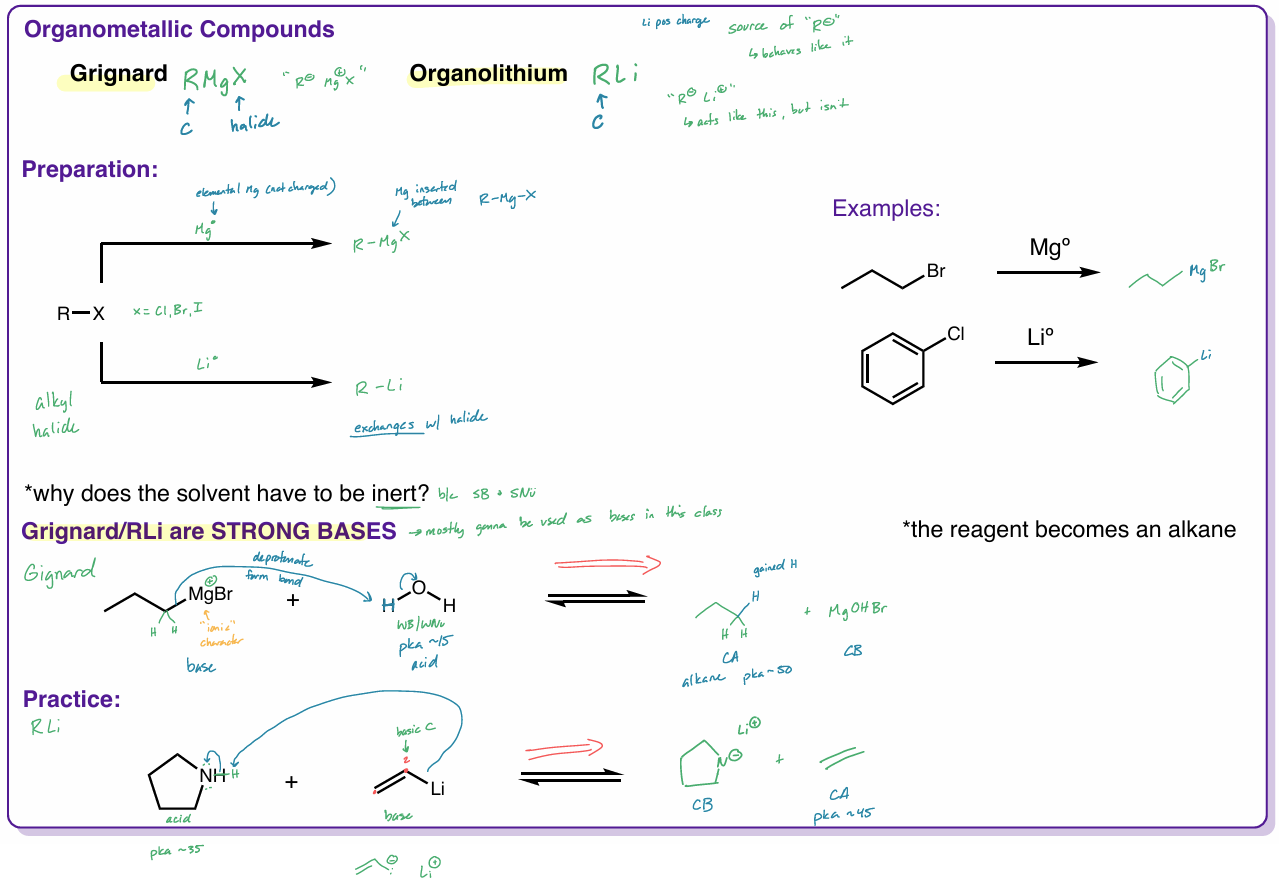
organometallic compounds
grignard - RMgX
organolithium - RLi
R = C
solvent has to be inert → b/c SB + SNu
are strong bases
reagents becomes an alkane
elemental form
represented by “ ° “ next to element
not charged
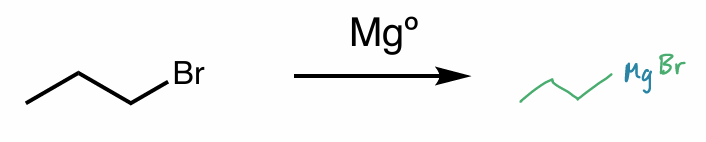
grignard
RMgX
Mg is “inserted” between R—X
X = halide
“ R- Mg+X “
organolithium
RLi
“ R- Li+ “
acts like this, but isn’t
Li exchanges w/ halide
organometallic compound examples