CHEM 2131 | SN1 and E1
1/9
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
10 Terms
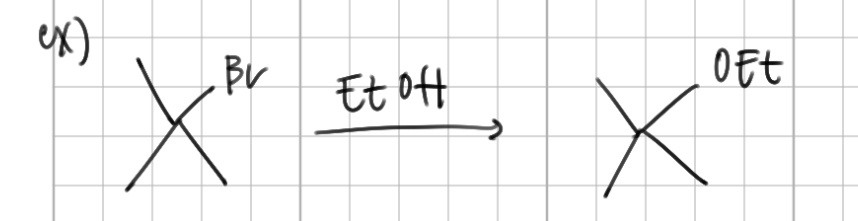
a SN1 mechanism has a substrate that is _____ ______ and a ____ nucleophile
sterically hindered, weak
in SN1, the nucleophile can attack the carbocation from ____ side(s), creating ____ and ____ of configuration, which is a _____ mix
both, inversion, retention, racemic
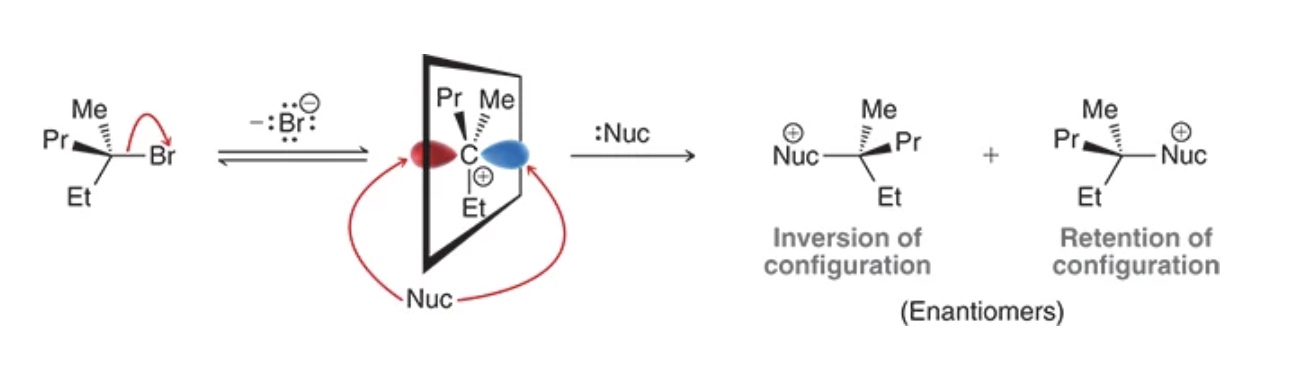
SN1 rearrangement
carbocation rearranges to be more stable before nucleophilic attack
hydride shift occurs when a _____ carbocation is next to a _____ carbon
secondary, tertiary
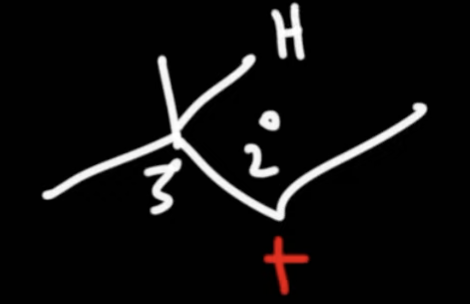
racemization occurs at a ____ ____
chiral center
if there is no chiral center in the final product of SN1, there is no ______ overall
racemization
E1 regiochemistry: the major product is the more/less substituted product
more (Zaitsev)
(T/F) for an E1 reaction, we are allowed to borrow from ANY beta proton (geometry does not matter/the beta proton does not have to be antiperiplanar
true
hyperconjugation only occurs on ______ carbons
neighboring
show a hydrogen (implicitly/explicitly) during hydride shift
explicitly