ISOMERISM - GEOMETRICAL ISOMERISM
1/8
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
9 Terms
conditions for gi
restricted rotation
x and y groups on same atom shouldn't be the same
which of the following geometry and bonds do not show gi?
trigonal planar, tetrahedral, single bond ands triple bonds
GI IN CYCLOALKANES DUE TO DOUBLE BONDS
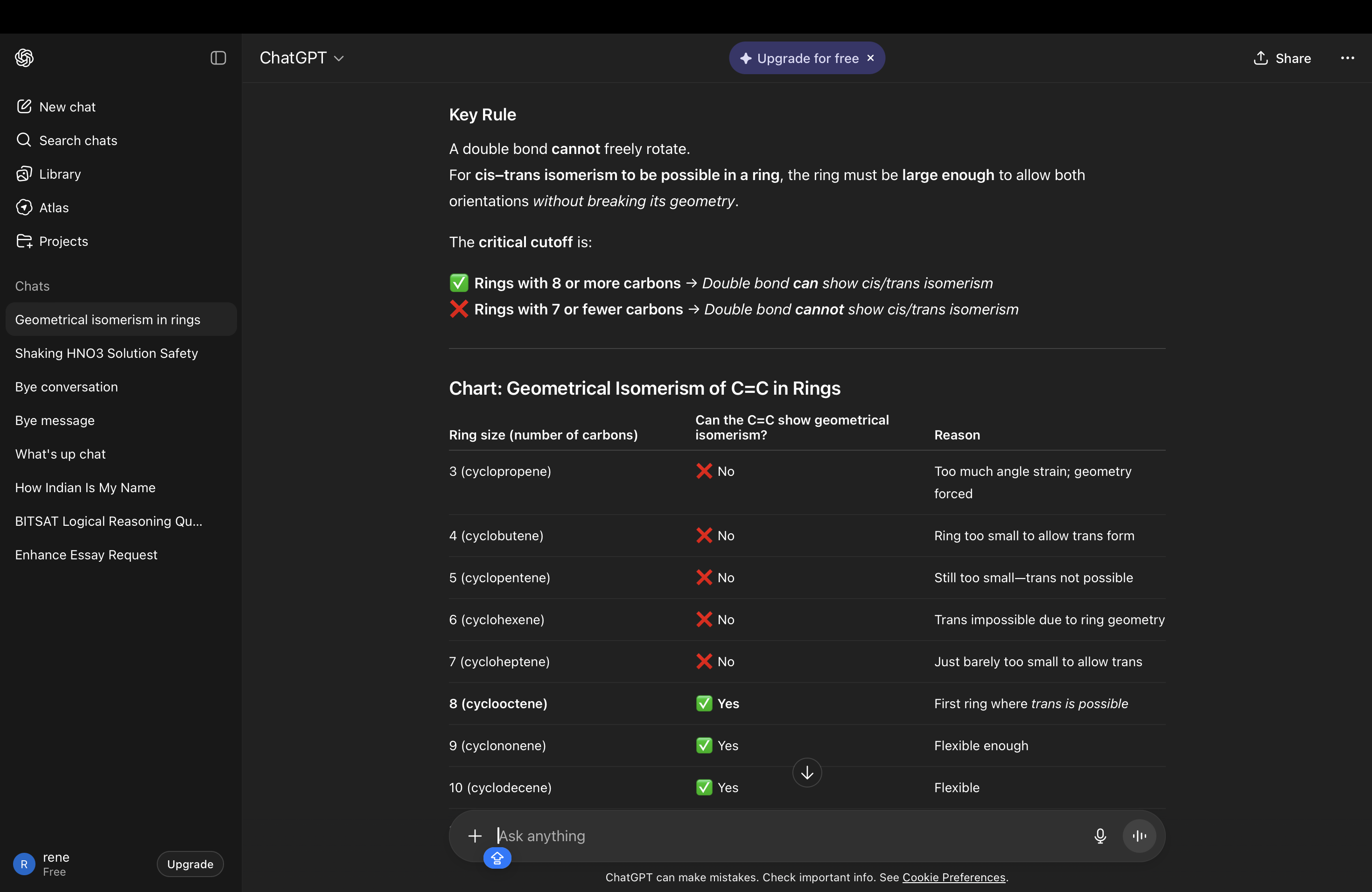
PLANARITY OF COMPOUNDS
why do even Spiro rings and cumuelens not show gi? answer in 2 lines
Because in spiro and cumulated (allene-type) systems, the substituents lie in mutually perpendicular or fixed planes, restricting rotation. This fixed 3-D arrangement creates two distinct, non-interconvertible geometries
necessary conditions for gi in cuemles and Spiro compounds

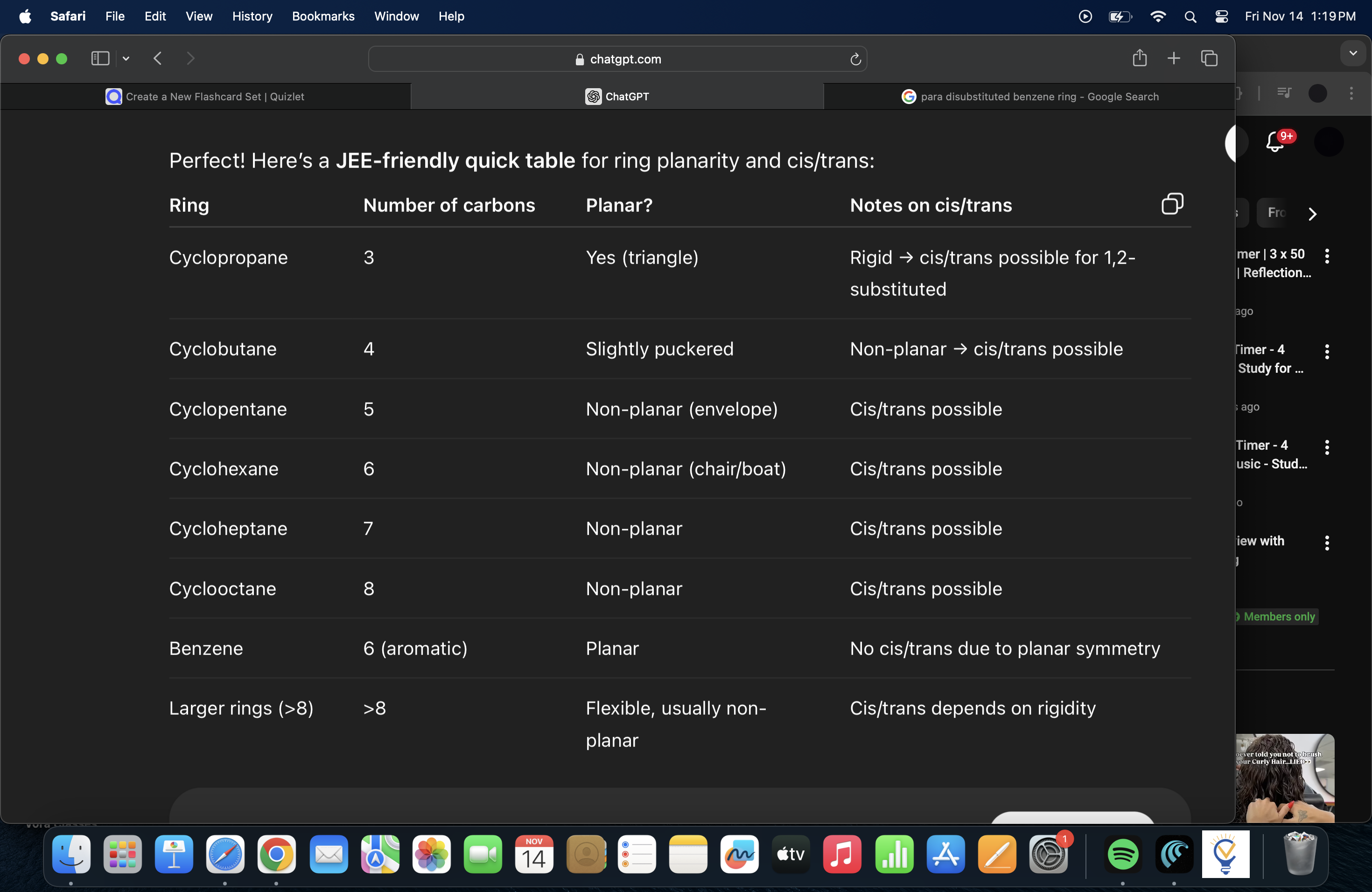
necessary conditions for gi in ring
minimum two sp3 carbons with different groups on carbon should be present
gi calculation formulas

gi non formula ones are
sq planar, tricycloalkane,
chain w branches in which branches having stereo centers if similar ends - case banao
non similar ends - 2^n
