Key Terms Chapter 4
1/36
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
37 Terms
Wave like properties of light
Light is perceived in the electromagnetic spectrum in a range of wavelengths
Key terms in waves
Wavelength (l) - the distance between two successive crests
Frequency (n) - the number of waves passing a certain point in 1 s (in s-1 or Hz)
Amplitude - the maximum displacement of the wave from its midpoint/centre (intensity)
Particle properties of light
Light comes in packets of energy called photons, energy of light depends on its frequency and intensity
What did the photoelectric effect show
Showed how energy of light depended on its frequency and intensity
How do you calculate the energy of a photon
Its proportional to its frequency and planck’s constant

Electron kinetic energy

Photon emissions (ground state, excited state process)
When an atom emits a photon, it returns to ground state
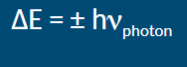
What happens when an atom absorbs a high energy photon
Can cause the atom to lose one of its electrons, implies an energy gain for an electron in the atom

Quantization of energy
Each energy level is restricted to specific energy values (quantised), can be described by the following equation, where n is the energy level

Quantum number 1
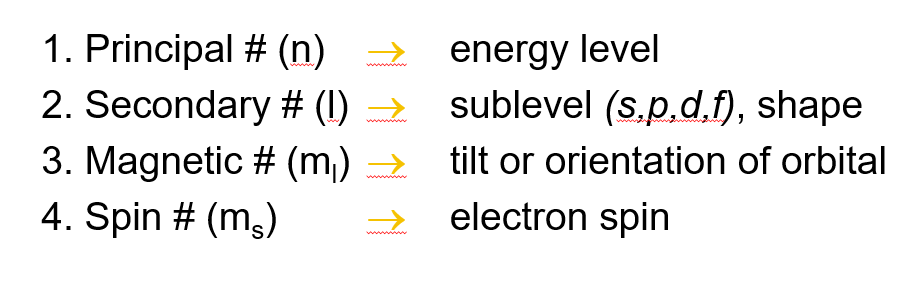
Principle quantum number, symbol n
What does the principal quantum number indicate
Indicates the energy level and distance from nucleus of an electron (it is the period number, which row of the periodic table you are on)
What are the values of the principal quantum number
n can be 1,2,3… to infinity, total electrons allowed in the orbital is 2n²
Quantum number 2
Secondary/angular momentum quantum number, symbol l (fancy l)
What does the secondary quantum number indicate
It describes the shape of the orbital (subshell number)
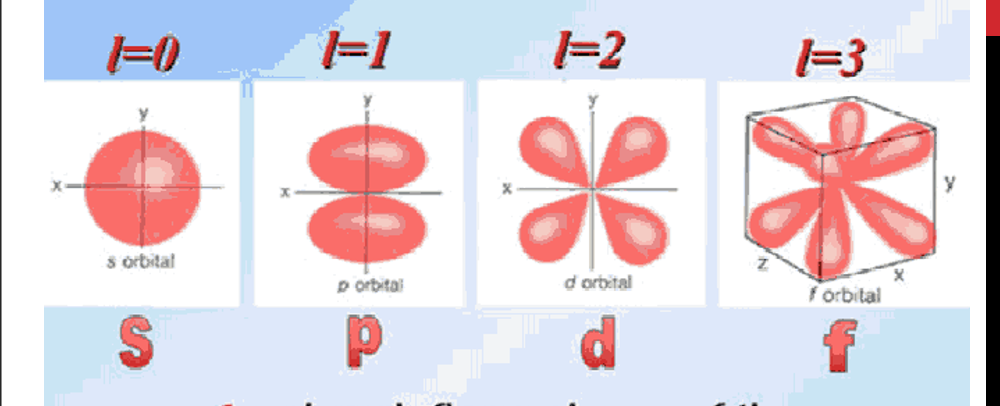
What are the values of the secondary quantum number
allowed values from l=0 to (n-1)
What orbital does each l value correspond to
l=0 is s, l=1 is p , l=2 is d, l=3 is f

Quantum number 3
Magnetic quantum number, symbol ml
What does the magnetic quantum number indicate
Describes the orientation of the orbital
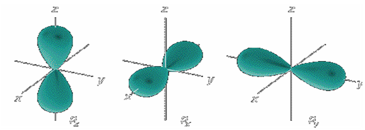
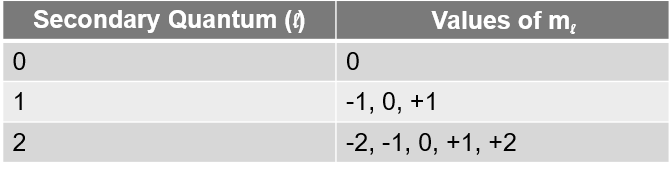
What are the values of the magnetic quantum number
Values of ml range from -l to +l
Quantum number 4
Spin quantum number, symbol ms
What does the spin quantum number indicate
Describes the spin of an electron, either clockwise or counterclockwise
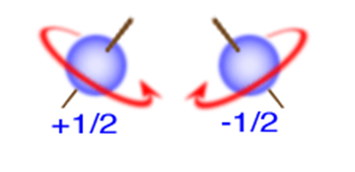
What are the values of the spin quantum number
Allowed values are plus or minus 1/2
Summary of the quantum numbers and what they indicate
Aufbau’s principle
electrons fill atomic orbitals of the lowest available energy levels before occupying higher energy levels

Pauli exclusion principle
No two electrons can have the same four quantum numbers
Hund’s rule
for a given electron configuration, the lowest energy state (ground state) is achieved when electrons occupy degenerate orbitals (orbitals with the same energy, like or ) singly before pairing up
Or simply: Electrons remain unpaired for as long as possible
Exceptions to electron configurations (know first 2 for sure)
Cr: 1s²2s²2p63s²3p63d54s1
Cu: 1s22s22p63s23d104s1
Ag: 1s22s22p63s23d104s23d104p64d105s1
Au: [Xe] 4f¹⁴ 5d¹⁰ 6s¹
What is an atom/ion with all electrons paired called (magnetism wise)
Diamagnetic
What is an atom/ion with unpaired electrons called (magnetism wise)
Paramagnetic
Nuclear shielding/screening
Occurs when valence electrons experience repulsion from deeper core electrons, resulting overall charge is called effective nuclear charge (z*)
Covalent radius
When atoms of the same element are bonded together, the covalent radius is half the distance between the atomic nuclei
Metallic radius
In metallic elements, half the distance between the nuclei of adjacent atoms in a crystal
Periodic trends, atomic radius
A measure of the radius of an atom, increases as you go down and decreases as you go across
First ionization energy (IE1)
The minimum amount of energy needed to remove the outer most electron from a neutral atom, increases as you go across and decreases as you go down
Ionic radius
Radius of ions, cations are always smaller than neutral atoms and anions are always larger
Electronegativity
The ability on an element to attract bonding electrons to itself, increases as you go across and decreases as you go down
Electron affinitiy
The energy change (released) when an electron is added to an atom in the gas phase, increases across and decreases down