Ideal gasses
1/42
Earn XP
Description and Tags
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
43 Terms
Define a mole in terms of mass
The amount of substance that contains as many elementary entities as there are atoms in 0.012 kg (12 g) of carbon-12.
Define a mole in terms of number of atoms
The amount of substance that contains 6.02×1023 elementary entities. This number is called the Avogadro constant and has symbol NA
What is the unit of molar mass?
kg mol-1
Define the Avogadro constant in words
The number of atoms in 0.012 kg (12 g) of carbon-12
This equals the number of molecules in one mole of a substance
What is the value of the Avogadro constant?
6.02×1023
What is the symbol for the Avogadro constant?
NA
Define Molar Mass
The mass of one mole of a substance
What is the molar mass of CO2 in kg? (C = carbon-12, O = oxygen-16)
1×12 + 2×16 = 44 → Molar mass of CO2 is 44g mol-1 = 0.044 kg mol-1
What symbol represents the number of moles?
n
What symbol represents the number of particles?
N
What equation links the number of moles and the number of particles?
N = n × NA
What are the five assumptions made in the kinetic theory of gasses?
There are a large number of particles in random motion with random speeds
Volume of particles is negligible compared to volume of gas
Collisions between particles and container walls are perfectly elastic
Time of collisions between particles is negligible compared to the time between collisions
Negligible electrostatic forces exist between particles except during collisions
State the assumption in the kinetic theory of gasses about the movement of particles
There are a large number of particles in random motion with random speeds
State the assumption in the kinetic theory of gasses relating to volume
Volume of particles is negligible compared to volume of gas
State the assumption in the kinetic theory of gasses about the type of collisions
Collisions between particles and container walls are perfectly elastic
State the assumption in the kinetic theory of gasses about the time of collisions
Time of collisions between particles is negligible compared to the time between collisions
State the assumption in the kinetic theory of gasses relating to forces
Negligible electrostatic forces exist between particles except during collisions
What is an ideal gas?
A model of a gas including assumptions that simplify the behaviour of real gases.
An ideal gas obeys the gas laws under all conditions.
How does the kinetic theory of ideal gases explain gas pressure on a container?
The atoms or molecules in a gas are always moving, and when they collide with the walls of a container the atoms or molecules exert a force on the walls, which we call pressure.
What is the change in momentum in a collision with a wall?
Innitial velocity = u, so innitial momentum = mu
Velocity after collision = -u (speed doesn’t change but directions is opposite), so momenutum after collision = -mu
∆p = (-mu) - (mu) = -2mu
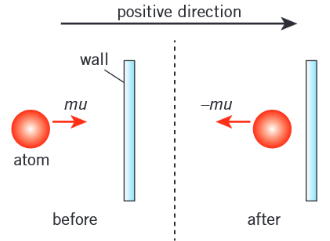
How do you derive pressure from the collision momentum?
Atoms in the gas bounce between the container walls, making frequent collisions.
According to Newton's second law, the force acting on the atom Fatom = ∆P / ∆t, where ∆p = -2mu and ∆t is the time between collisions with the wall. From Newton's third law, the atom also exerts an equal but opposite force on the wall.
A large number of atoms collide randomly with the walls of the container. If the total force they exert on the wall is F, then the pressure they exert on the wall is given by p = F / A where A is the cross-sectional area of the wall.
What is Boyle’s Law?
Pressure and volume of a gas are inversely proportional at constant temperature. This means:
pV = constant
p1V1 = p2V2
What must remain constant for Boyle’s Law and how is this achieved?
Mass and temperature of gas must remain constant
Temperature can me kept roughly the same by slowly compressing the gas
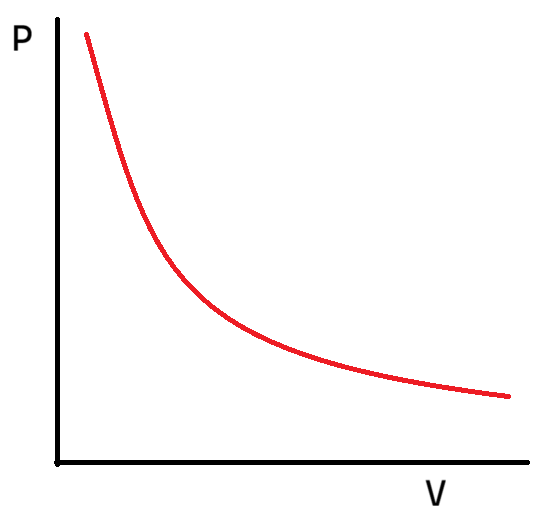
What is the graph of P against V (pressure against volume)?
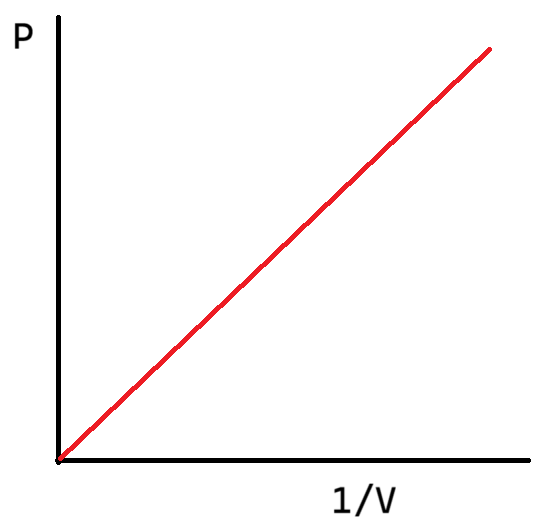
What is the graph of P against 1/V (pressure against 1/volume)?
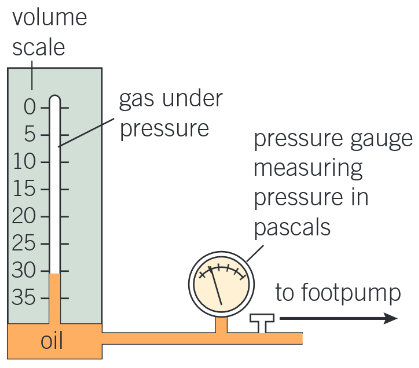
What apparatus is used to test Boyle’s Law?
What is the graph of P against T (pressure against temperature)?
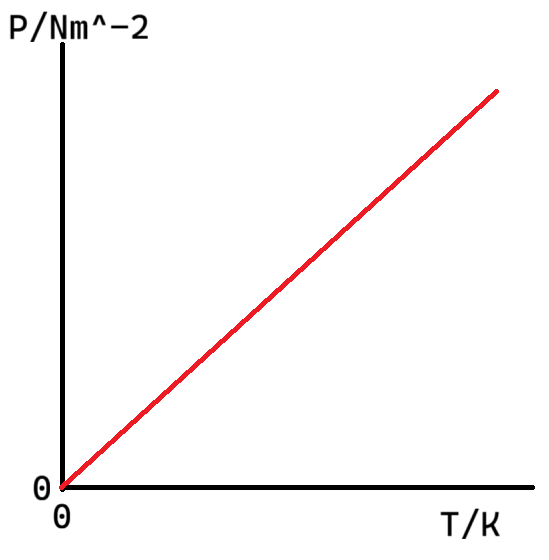
Describe the Pressure Law (relationship between P and T) in words
If the volume and mass of gas remain constant, the pressure of an ideal gas is directly proportional to its absolute (thermodynamic) temperature in kelvin.
Pressure Law
The pressure and temperature of a gas are directly proportional at a constant volume. This means:
p/T = constant
p1/T1 = p2/T2
What needs to remain constant for the Pressure Law? How do we achieve this?
The volume and mass of the gass must remain constant
Use container of fixed volume
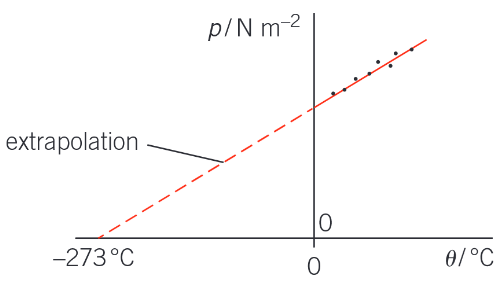
How can the experiment used to test the Pressure Law be used to find an estimate for absolute zero?
Plotting a graph of pressure against temperature θ in Celsius from the experimental results gives a line that can be extrapolated back to a point where the pressure is zero. At absolute zero the particles are not moving so the pressure of the gas must be zero.
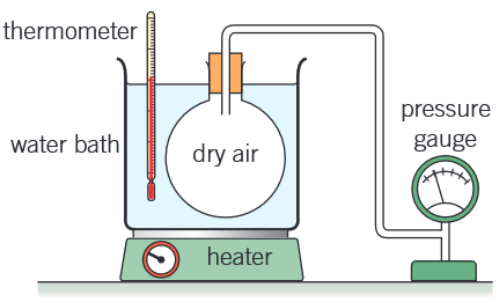
Sketch and label the apparatus used to test the Pressure Law
What is the ideal gas equation involving n and what do the terms mean?
pV = nRT
p is pressure
V is volume of gas
n is the number of moles of gas
R is the molar gas constant
T is the temperature of the gas in kelvin
What is the ideal gas equation involving N and what do the terms mean?
pV = NkT
p is pressure
V is volume of gas
N is the number of molecules in the gas
k is Boltzmann constant (given in formula book)
T is the temperature of the gas in kelvin
What is Charle’s Law?
The volume of a gas is directly proportional to its temperature at a constant pressure. This means:
V/T = constant
V1/T1 = V2/T2
Charle’s, Boyle’s and Pressure Law combined into a single equation
p1V1/T1 = p2V2/T2
What is the internal energy of an ideal gas?
The sum of the randomly distributed kinetic energies of the molecules in an ideal gas.
Does not include the potential energy as this is assumed to be zero in an ideal gas
Define internal energy (U)
The sum of the randomly distributed kinetic energies and potential energies of all the particles in an object
The constant that relates the average kinetic energy of particles in a gas to the absolute temperature of the gas.
k = 1.38 x 10-23 J K-1
Mean square speed
The mean of the squared velocities of all the particles in a gas
Equation linking boltzmann constant to avogadro and molar gas constant?
k = R / NA
boltzmann constant = molar gas constant / avogadro constant
The constant of proportionality in the ideal gas equation relating the pressure, volume and absolute temperature of an ideal gas to the amount of gas in moles.
R = 8.31 J mol-1 K-1
Molecular mass
The sum of the relative atomic masses of all the atoms in a molecule. As it is a relative quantity, it has no units.