BSC1010 Quiz 3
1/25
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
26 Terms
What was the first discovered enzyme called?
Diatase
The hydrolysus of starch by diatase was seen in 1835 and acknowledged as a _________ reaction.
catalytic
When was the first pure form enzyme obtained, and where?
1926 at Cornell University
Enzymes are biological ________ that lower the _________ energy of a reaction
catalysts; activation
How do enzymes lower activation energy of a reaction?
They bind to a substrate
How does an enzyme binding to a substrate lower the activation energy”?
The enzyme binds in a way that allows the reaction to occur more efficiently (helps reaction go quicker)
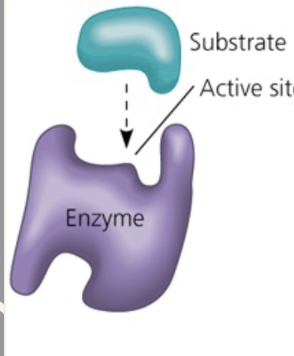
What is the active site?
The area where the substrate binds
When an enzyme binds to a substrate, the ______-________ complex is formed
Enzyme-substrate
Enzymes are typically ________ (biological molecule), but some _____ (RNA/DNA) molecules can also act as enzymes
proteins; RNA
T or F: Enzymes are specific to their substrate
True. They may only have one or two that they react with
What factors affect enzyme activity? (4)
Temperature, enzyme concentration, pH, and substrate concentration
T or F: Enzymes do NOT need an optimal pH level to operate
False. Enzymes have their own optimal pH and a change in pH outside of this optimum will often slow reactions. In the case of an extreme pH, it will cause reactions to stop
T or F: The relationship between temperature and reaction rate is proportional.
True. When the temperature goes up, the reaction rate goes up (think of molecules moving faster, they will collide more and increase likelihood of reaction). When temperature goes down, reaction rate goes down.
T or F: Extreme temperatures will not denature enzymes
False. Extreme temperatures denature enzymes and cause reactions to stop
*Denature = unraveling
When enzyme concentration ______ (increases/decreases) the reaction rate _______ (increases/decreases)
Increases; increases
*As long as there is substrate to bind to, this relationship is true
When the substrate concentration increases, what will happen to the reaction rate?
It will also increase
What are the two types of inhibition?
Competitive and noncompetitive
In ___________ (competitive/noncompetitive) inhibition, a molecule that is structurally similar to the substrate tricks and enzyme and competes for an active site
competitive
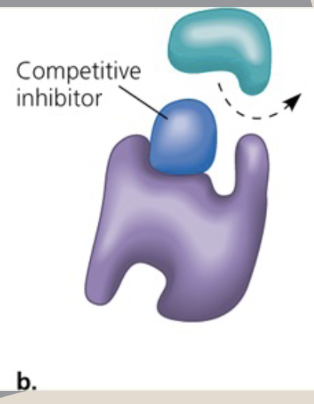
What is competitive inhibition?
When a molecule that is structurally similar to the substrate tricks the enzyme and competes for an active site on the enzyme
T or F: There is room on the substrate of a reaction to bind when a competitive inhibitor binds
False. There is no room for the substrate to bind
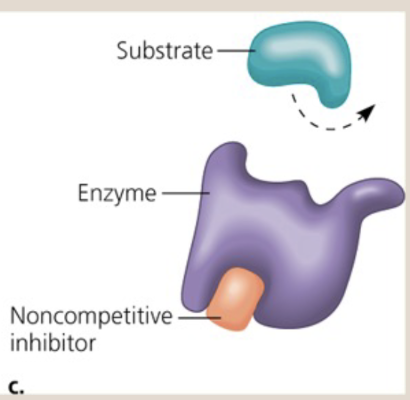
In _________ (noncompetitive/competitive) the inhibitor molecule blocks the active site of the enzyme or binds to another part of the enzyme and changes its shape
noncompetitive
What is noncompetitive inhibition?
The inhibitor blocks the active site of the enzyme or binds to another part of the enzyme and changes the shape of the protein so that no other substrates may bind to it
T or F: In noncompetitive inhibition, the structure of the inhibitor is not similar to the substrate
True. It does not compete for the active site because it is not similar to the structure of the substrate
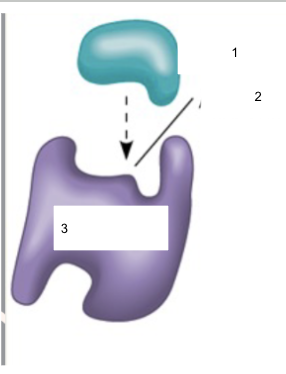
Name parts 1, 2, and 3
1.) Substrate
2.) Active site
3.) Enzyme
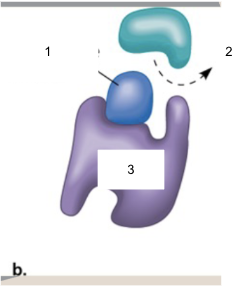
Name parts 1, 2, 3
1.) Competitive inhibitor
2.) Substrate
3.) Enzyme
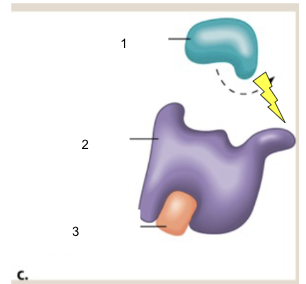
1.) Substrate
2.) Enzyme
3.) Noncompetitive inhibitor
*Note that at the lightning bolt, the shape of the enzyme is warped because of the noncompetitive inhibition