ap chem unit 7 equilibrium
1/24
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
25 Terms
what are some examples of reversible reactions? how can you tell that a reaction is reversible? what are some examples of non-reversible reactions?
reversible reactions: solid dissolving then crystalizing (Ksp), electrons being lost/gained in redox reactions (K), and H+ ions being exchanged in acid-base reactions (Ka and Kb)
often accompanied by an observable event (ex: color change, gas release, etc.) that will allow you to see the process is reversible
non-reversible reactions: combustion of a hydrocarbon, neutralization reactions, rusting, cooking, etc.
what is K?
K is the equilibrium constant, which is the ratio of the concentrations of products to reactants for a reversible reaction at equilibrium. each concentration is raised to the power of its stoichiometric coefficient and is calculated by K=[products]/[reactants]. K has no units
what is dynamic equilibrium?
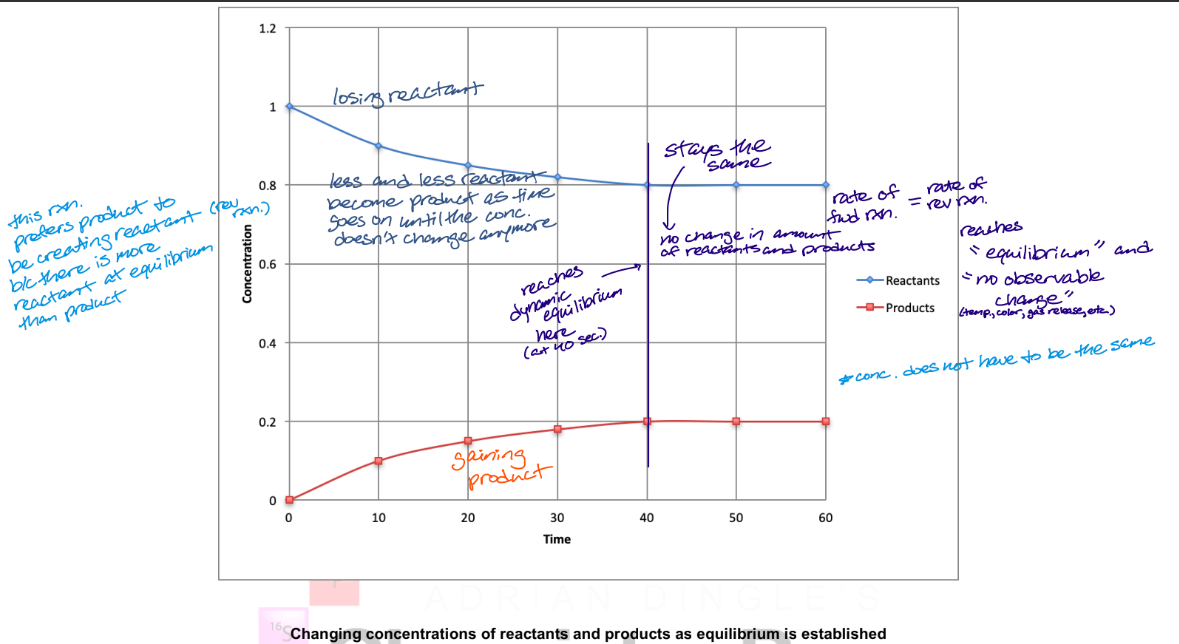
occurs when the rate of the forward reaction = rate of the reverse reaction, resulting in no net change in the concentration of reactants and products
rate affected by concentration and temperature
constant but not necessarily equal concentrations at equilibrium
occurs after a certain amount of time
at the same speed reactants are becoming products, products are becoming reactants
the E being released/absorbed in the forward reaction is being absorbed/released in the reverse reaction
describe what occurs at equilibrium in terms of concentrations and rates
at the beginning of the rxn., there is a high concentration of reactants and a low concentration of products
rate of forward reaction > rate of reverse reaction at this point because higher concentration of reactants than products
as rxn. proceeds, concentration of reactants decrease and concentration of products increase
rate of reverse rxn. increases as concentration of products increases
increase/decrease of rates occurs until rates become equal (dynamic equilibrium established)
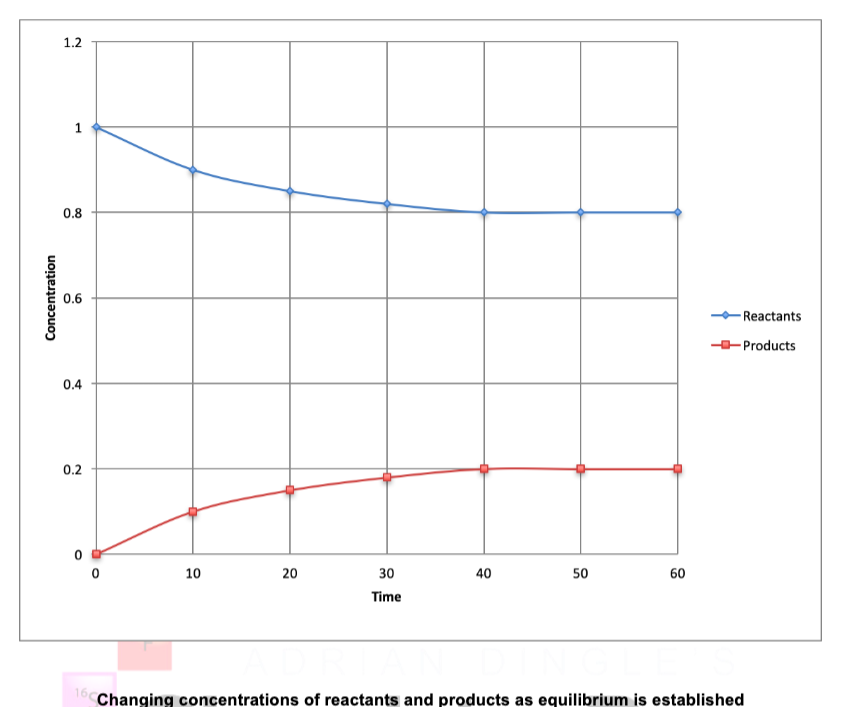
describe what this graph shows
what happens if you add more reactants or products to a reaction?
increasing the concentration of reactants will increase the rate of the forward reaction
increasing the concentration of products will increase the rate of the reverse reaction
pictured as a spike on a graph
do reactions ever truly stop at equilibrium?
on a macroscopic scale, it may look like it has stopped
closer inspection on the microscopic scale confirms that it is still occuring
if two solutions have an observable difference (like color), what will it look like at equilibrium
a mixture of the two
if the soln. on the LHS is red and the RHS is blue, and the forward reaction is preferred, it will look more blue than red at equilibrium
if the soln. on the LHS is red and the RHS is blue, and the reverse reaction is preferred, it will look more red than blue at equilibrium
you know dynamic equilibrium is reached because color doesn’t change anymore because the concentrations of the products and reactions are no longer changing
what does it mean when we say that a reaction favors the products? the reactants? what are the K values for each?
favors products: the concentration of products is higher than the concentration of reactants at equilibrium
favors the forward reaction
K>1
favors reactants: the concentration of the reactants is higher than the concentration of products at equilibrium
favors the reverse reaction
K<1
what is Kc and what is it used for?
Kc is an equilibrium constant at a given temperature (if temp. changes, so does Kc)
it is the ratio of product concentration to reactant concentration at equlibrium
Kc has no units
ONLY for aqueous or gaseous substances, never solid or liquid
assume 1L container if not given
why aren’t solids or liquids considered in Kc and Kp?
Kc: concentrations of solids and liquids are considered constant (unchanging)
Kp: vapor pressures of solids and liquids are considered constant
what is the difference between Q and K? what do differences in these numbers tell us?
K is the ratio of product concentration to reactant concentration at dynamic equilibrium, while Q is the ratio of product concentration to reactant concentration at any point within the reaction (not necessarily equilibrium)
if Q=K, the reaction is at equilibrium
If Q>K, too much product, too little reactant (reverse reaction)
if Q<K, too much reactant, too little product (forward reaction)
how do you calculate Kp?
step 1: figure out moles at equilibrium (use ICE table if necessary)
step 2: find mole fraction of each gaseous substance by dividing the moles of each by the total amount of moles at equilibrium
step 3: find the partial pressures of each by multiplying the mole fraction by the total pressure (assume 1 atm if not given)
step 4: calculate Kp=(products)/(reactants)
each raised to the power of stoichiometric coefficient
what is the relationship between Kp and Kc?
Kp = Kc(RT)∆n
T is temperature: if you increase the temperature, Kp increases
if you decrease the temperature, Kp decreases
∆n is the stoichiometric number of moles of gaseous products minus the stoichiometric number of moles of gaseous reactants
if the number of moles of reactants is greater than the number of moles of products based on coefficients, Kp will be smaller because of a negative exponent
opposite is true
what is le châtlier’s principle?
states that in any equilibrium system, when a stress is placed on a system, such as a change in temperature, pressure, or concentration, then there is a shift in the position of the equilibrium to oppose that stress
ex: if you increase the pressure by compressing the container, rxn. will try to decrease the pressure
ex: if temperature increases, rxn. will try to decrease it
ex: if more reactant is added, rxn. will make more product
determine what happens when there is an increase in pressure, temperature, concentration of reactants, and concentration of products for the following reaction:
N2(g) + 3H2(g) ⇌ 2NH3(g)
ΔH = -92 kJ/mol
increase in pressure: shift towards RHS bc there’s less mols on that side, leading to less particles in the container and a decreased pressure
increase in temperature: shift towards LHS (endothermic process); absorbs E and decreases temperature in container
added N2 and H2: shift towards RHS
added NH3: shift towards LHS
determine what happens when there is an increase in pressure, temperature, concentration of reactants, and concentration of products for the following reaction:
H2(g) + I2(g) ⇌ 2HI(g)
ΔH = +51 kJ/mol
increase in pressure: no change bc equal amount of mols on both sides
increase in temperature: shift towards RHS to decrease temperature in container
added H2 and I2: shift towards RHS
added HI: shift towards LHS
how do you use K with hess’s law?
multiplying the reaction by x: Kx
when you flip/reverse the reaction: inverse of K (1/K)
adding reactions together: multiply K values by one another (K1)(K2)
what is the range of K?
very large (10-40 - 10120) but is ALWAYS positive
how do you use ∆G with K?
∆Gº = -RT(lnK)
R: gas constant -8.314 J/(mol • K)
T: temperature in Kelvin
if ∆G is negative, it is a spontaneous reaction
what does it mean for K if -∆Gº has a very large magnitude?
very large K value
much greater than 1
favors products
what does it mean for K if +∆Gº has a very large magnitude?
very small K value
much lower than 1
favors reactants
what does it mean for K if ∆Gº is close to 0?
K will be closer to 1
reactants and products will be at similar concentrations at equilibrium
+∆H
endothermic
-∆H
exothermic