Molecular interactions, Physical property and IMF strength
1/16
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
17 Terms
Intermolecular interactions between chemical species can have more than one type of WHAT that can be present at the same time in a WHAT or WHAT species
Intermolecular interactions between chemical species can have more than one type of INTERMOLECULAR FORCES (IMFS) that can be present at the same time in a PURE or MIXTURE species
Intermolecular interactions between chemical species explaining WHAT
Intermolecular interactions between chemical species explaining PHYSICAL PROPERTIES
Interactions used to explain physical properties of WHAT or WHAT chemical species
Interactions used to explain physical properties of LIQUID or GASEOUS chemical species
Intermolecular forces and physical properties
WHAT
WHAT
WHAT
WHAT
WHAT
Intermolecular forces and physical properties
Miscibility
Solubility
Boiling point
Viscosity
Surface tension
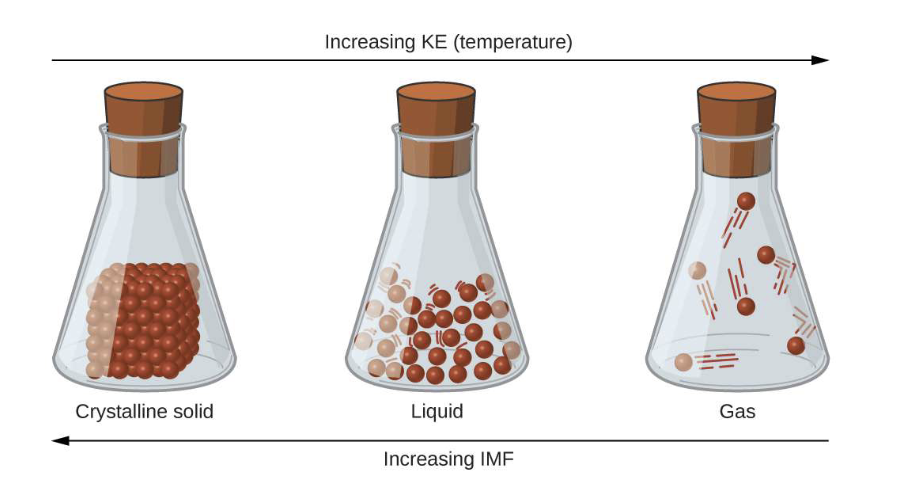
The LESS the molecules are moving the WHAT the intermolecular forces (IMFs)
The LESS the molecules are moving the STRONGER the intermolecular forces (IMFs)
Miscibility
Ability of liquids to mix and form HOMOGENEOUS (i.e single phase, solutions)
Solubility
Ability of a solid solute to dissolve in a liquid solvent
General rule:
One species will mix with another species if the interactions it has with the other species are WHAT or WHAT than the interactions it has with WHO
General rule:
One species will mix with another species if the interactions it has with the other species are EQUAL or GREATER than the interactions it has with ITSELF
Boiling point
WHAT at which a molecule leaves the liquid phase to enter the gas phase
At that temperature enough WHAT provided to WHAT interactions between molecules in the liquid
The stronger the interaction, the WHAT the boiling point
Boiling point
TEMPERATURE at which a molecule leaves the liquid phase to enter the gas phase
At that temperature enough ENERGY is provided to BREAK interactions between molecules in the liquid
The stronger the interaction, the HIGHER the boiling point
Viscosity
Ease of WHAT of species in the WHAT phase
The stronger the interactions of the molecule with their neighbouring molecules, the WHAT distance it can travel and the WHAT the molecules move within the liquid
Viscosity
Ease of MOVEMENT of species in the LIQUID phase
The stronger the interactions of the molecule with their neighbouring molecules, the LESS distance it can travel and the SLOWER the molecules move within the liquid
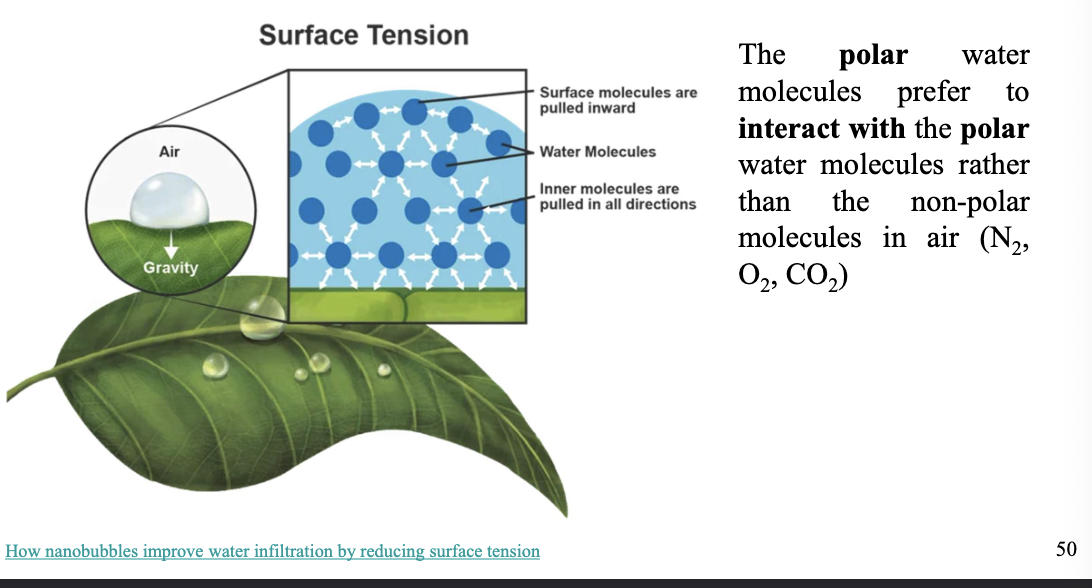
Surface tension
The force that causes the molecules on the surface of a liquid to be pushed together and form a layer
example: The POLAR water molecules prefer to interact with the POLAR WATER molecules than the NON-POLAR molecules in the air *N2, O2, CO2)
Strength of intermolecular forces
WHAT
WHAT
WHAT
WHAT
WHAT
Strength of intermolecular forces
Shape
Size
Functional group
Position of function groups
Number of interactions
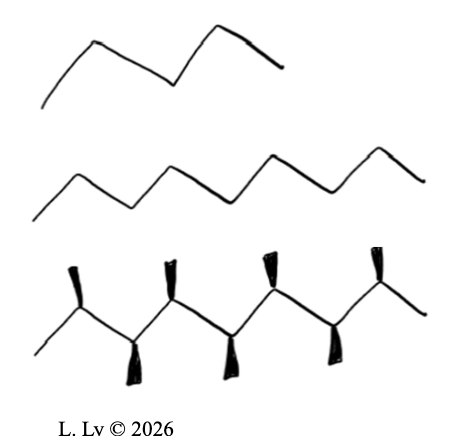
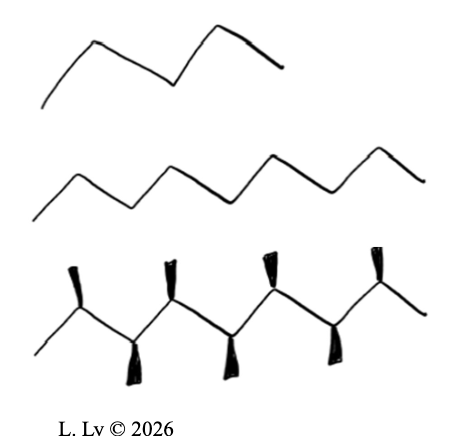
Shape
molecules with MORE branches: WHAT
molecules with MORE branches: LESS contact points for interactions
Size
LARGER molecules: WHAT
Size
LARGER molecules: MORE available contact points for interactions
Functional group: Order of decreasing IMF
Hydrogen bonding (Examples: C(O)OH, OH, NH, NH2,)
Dipole (example: any other groups that contain O, N)
Dipole-dipole
Dipole-induced dipole
LDF (Examples: C – C, C – H)
Position of function groups
A functional group located INSIDE a bulky molecular structure LESS accessible for interactions compared to the one at the END of a chain
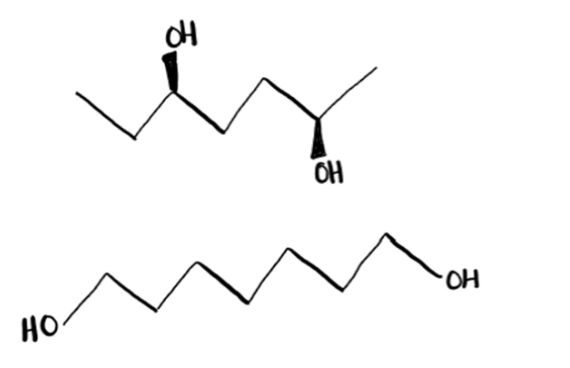
Number of interactions
A LARGER number of weak IMFs can end up being STRONGER than a small number of STRONG IMFs.
In this case, the overall strongest IMF is the weak IMF and it will define the PHYSICAL PROPERTY of the molecules