BC.04 Atomic and molecular orbitals
0.0(0)
Card Sorting
1/10
Earn XP
Description and Tags
Last updated 12:27 PM on 1/9/23
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
11 Terms
1
New cards
What is a wave function?
A wave function (Ψ) is a function that relates the location of an electron at a given point in space (x, y, and z coordinates) to the amplitude of its wave, which corresponds to its energy.
2
New cards
What is electron density (Ψ^2)?
It is the probability of finding an electron at a point, which leads to a distribution of probabilities in space.
3
New cards
Draw a graph of wave function, electron density and radial electron distribution for a 1s orbital. Note the most likely distance to find an electron on the radial electron distribution graph.
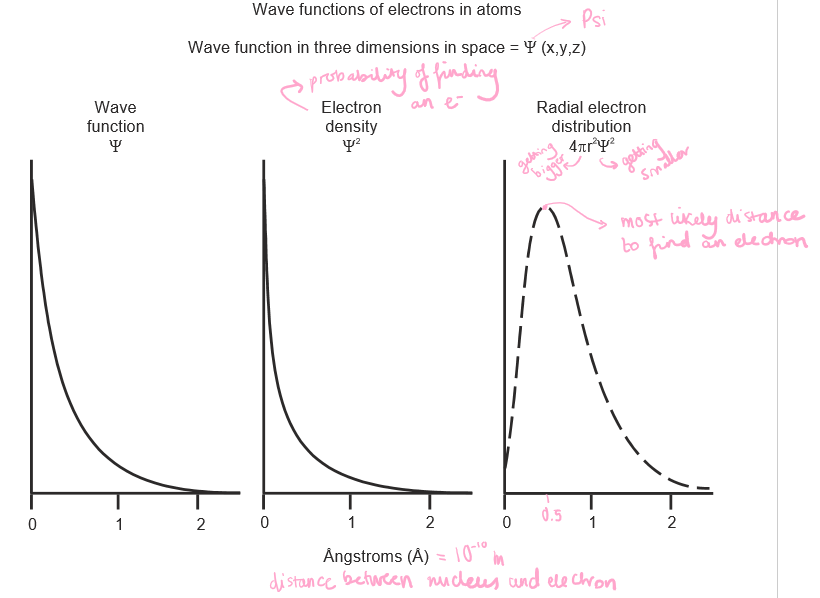
4
New cards
What is the most likely distance to find an electron in a 2s orbital?
2Å
5
New cards
What is Coulomb’s Law?
Where q is charge and r is separation distance.
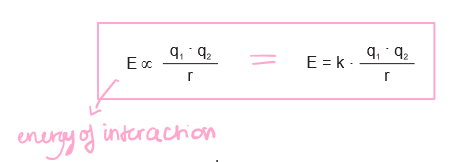
6
New cards
What is the Aufbau principle?
Electrons placed in orbitals starting with lowest energy and working up
7
New cards
What is the Pauli exclusion principle?
Two electrons per orbital, spins paired
8
New cards
What is Hund’s rule?
When multiple orbitals of same energy are available, electrons are distributed among them, spins parallel
9
New cards
When the electrons are in phase, a ??? forms. This is an energetically favourable interaction.
Bonding orbital (σ)
10
New cards
When the electrons are **not** in phase, a ??? forms. This is an energetically unfavourable interaction (in fact more unfavourable than if the electrons were just separate from each other).
Anti-bonding orbital (σ\*)
11
New cards
Why doesn’t Helium exist as a diatomic molecule?
Because 2 electrons exist in both bonding and anti-bond orbitals, which is unfavourable to form.