materials exam 2
1/20
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
21 Terms
Diffusion
Mass transport by atomic motion
Solids Diffusion (vacancy diffusion)
substitutional
self diffusion: atomic migration in a pure metal
Solids (interstitial diffusion)
impurity diffusion
diffusion of atoms of one material into another material
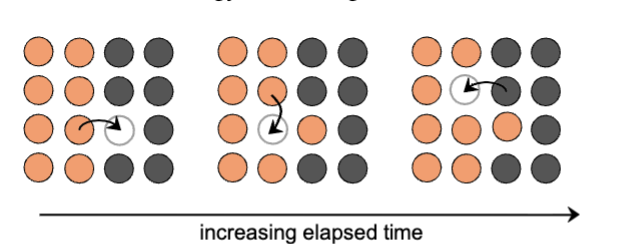
Diffusion Mechanism 1: Vacancy diffusion
substitutional
atoms and vacancies exchange positions
applies to host and subs impurity atoms
diffusion rate depends on # of vacancies & activation energy to exchange
Diffusion Mechanism 2: Interstitial Diffusion
small atoms move from one interstitial position to an adjacent one
more rapid than vacancy diffusion
Self- Diffusion
migration of host atoms in pure metals
labeled atoms moving to another position
Interdiffusion (impurity)
atoms tend to migrate from high concentration to low concentration
Case Hardening
outer surface selectively hardened by diffusing carbon atoms into surface
carbon atoms makes iron harder
improves wear resistance of gear & fatigue failure
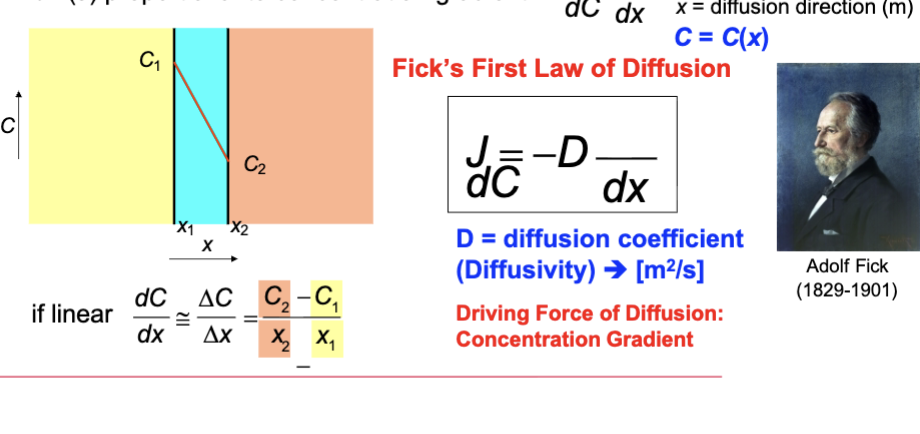
Rate of diffusion (J)
usually time dependent
rate of mass transfer
Measure mass of diffusing species (M) that pass through the sheet over time period (t)
Steady State Diffusion
flux is independent of time
J flux proportional to concentration gradient
Effect of Temp on Diffusion 1
Diffusion coefficient increases with T

Non-steady diffusion
the concentration of diffusing species is a function of time and position (C= c(x,t))
the eqn assumes D is independent of concentration
chp 7: Metals Dislocation Characteristics
quite deformable
dislocation motion: relatively easy
metallic bonding- non-directional
close-packed planes & directions for slip
Ceramics Dislocation characteristics (Covalent Bonding)
fragile and much less deformable
Dislocation motion difficult
covalent bonding- directional
Ceramics Dislocation characteristics (Ionic Bonding)
dislocation motion: relatively difficult
few slip systems

Plastic Deformation by Dislocation motion
called slip too
applied shear stress can cause extra half-plane of atoms
atomic bonds broken and reformed along slip plane as dislocation moves
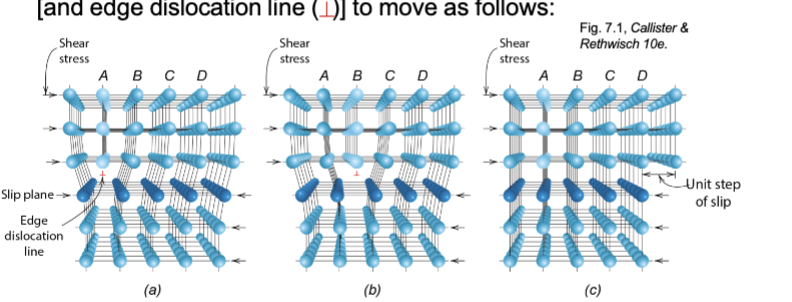
Dislocation motion
movement of extra plane of atoms by breaking/reforming interatomic bonds
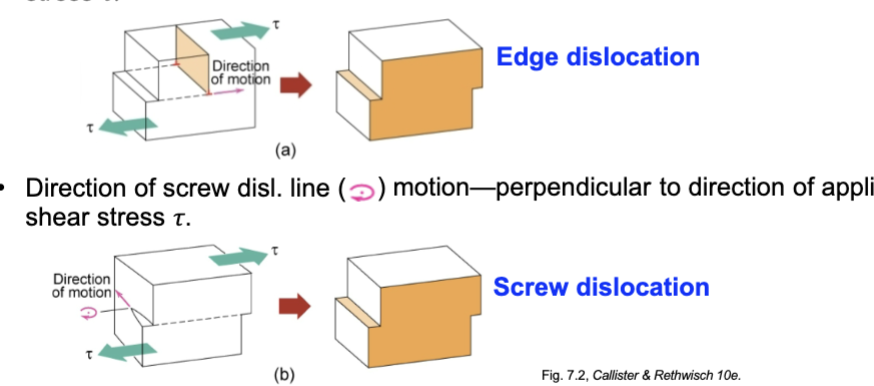
Motion of Dislocations
Edge dislocation line parallel to direction of applied shear stress
screw dislocation line perpendicular to direction of applied shear stress
Single Crystal Slip
parallel slip steps form on surface of crystal
result from motion of large #s of dislocations on same slip plane
Critical Resolved Shear Stress
crystal doesn’t start deforming until shear stress on a specific plane reaches ciritcal value
the applied stress needed depends on orientation
slip occurs when it reaches the cirtical value → plastic deformation
in a single crystal: multiple slips/ variety orientations
Polycrystalline Materials- Slip
many grains, random crystallographic orientations
slip orientation: vary from grain to grain