Biochem Exam 3 Cards
1/111
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
112 Terms
What transcription factor facilitates anaerobic glycolysis?
Hypoxia-indueible transcription factor (hif-1)
.what can activate hypoxia-inducible transcription factor (hif-1)?
Hypoxia caused by rapid tumor growth or anerobic exercise training
How does hypoxia-inducible factor tie into glucose metabolism?
Proteins as part of glucose metabolism are encoded for by genes regulated by HIF-1
what is the activation of HIF-1 protein in tumor cells called?
the Warburg Effect
where do the reactions catalyzed by PDH occur?
the mitochondrial matrix
what is the net reaction for the pyruvate dehydrogenase complex?
Pyruvate + CoA + NAD+ → Acetyl CoA + CO2 NADH
is the PDH complex reaction overall spontaneous or non-spontaneous
spontaneous (ΔG<<0)
there are two types of coenzymes attached to PDH, what are they?
catalytic and stoichiometric
what is the difference between catalytic and stoichiometric coenzymes?
catalytic are permanantly bound as prostetic groups, stoichiometric are not
about what is the size of Pyruvate Dehydrogenase?
VERY large - 5000-10000 kd, 60 subunites, 3 enzymes, and 5 coenzymes
what are the catalytic coenzymes in the PDH complex?
Thiamine (B1) Pyrophosphate
Lipoic Acid
FAD (FAD/FADH2)
what are the stoichiometric coenzymes in the PDH complex
CoA and NAD
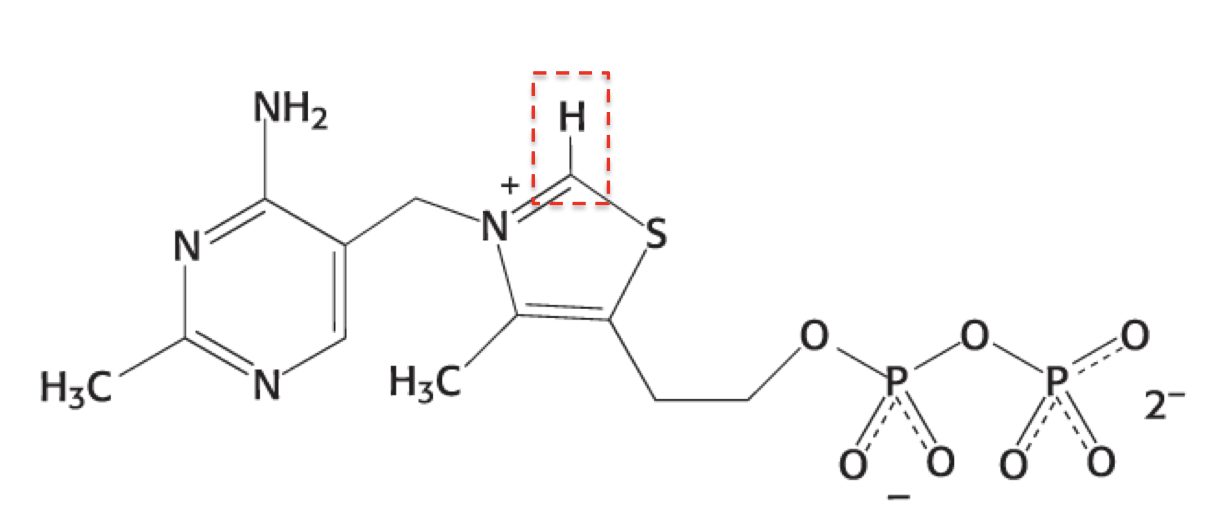
What is this structure?
thiamine pyrophosphate
___________ of lipoate functions ad an oxidant
disulfide
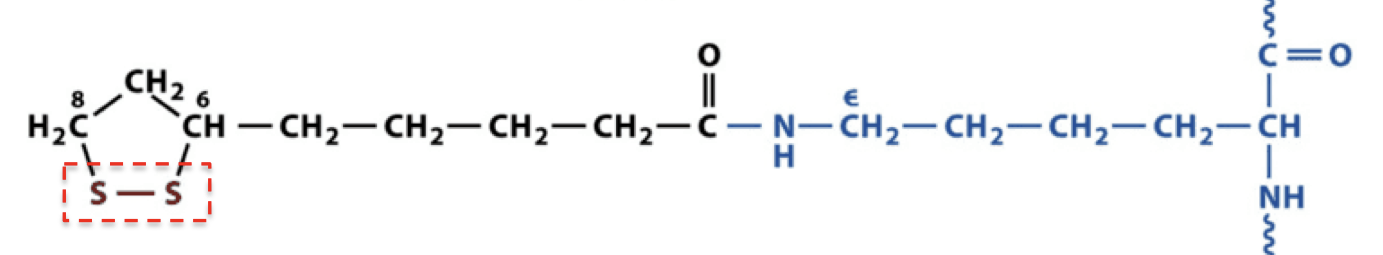
what structure is this
lipoamide
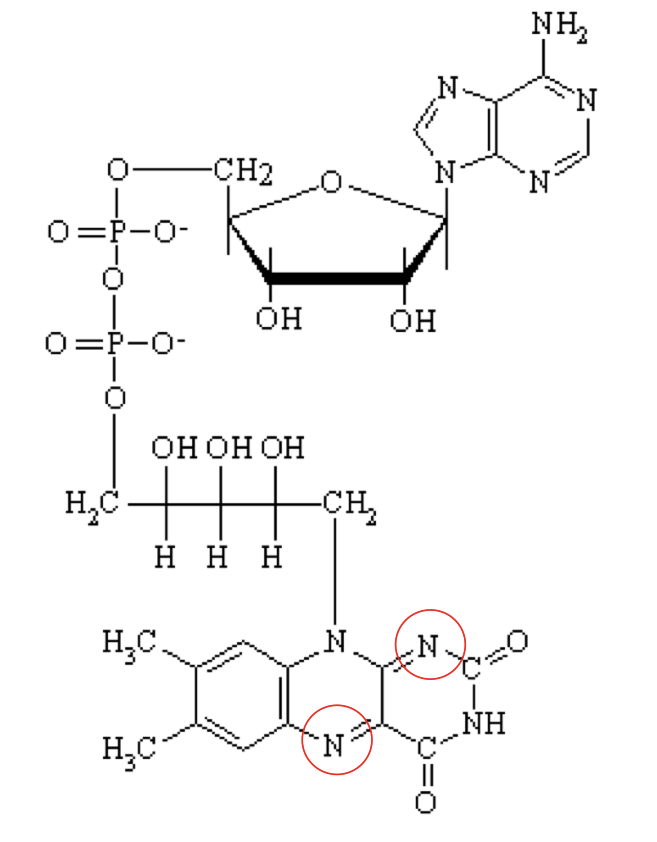
what structure is this?
Flavin Adenine Dinucleotide
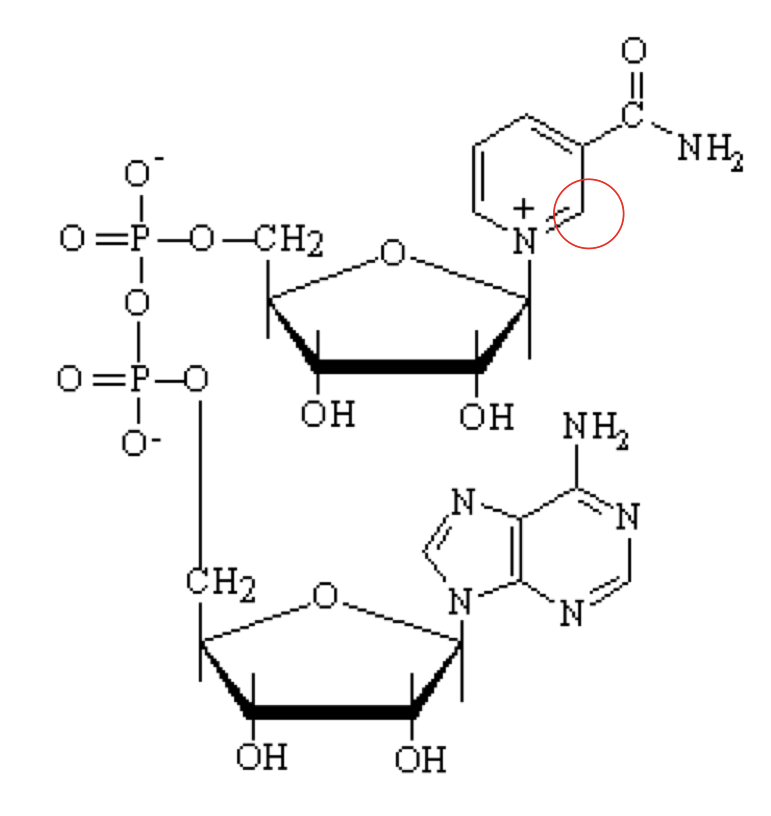
what structure is this?
Nicotinamide Adenine Dinucleotide
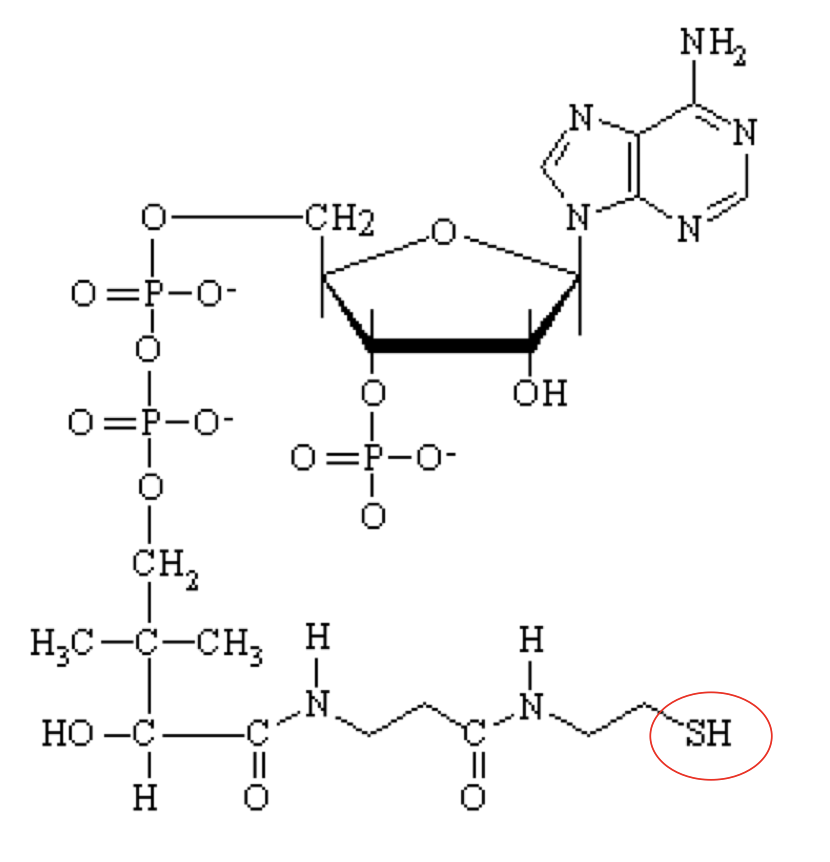
what structure is this?
Coenzyme A (CoASH)
what is step 1 of the PDH Complex cycle
Decarboxylation of pyruvate
2H+ are added to pyruvate and 1 CO2 is removed, then the pyruvate attaches to TPP on E1 of PDH
what happens in step two of the PDH complex cycle?
oxidation of ethyl to acetyl
the acetyl group on the TPP is transferred to one of the lipoamide arms, breaking the disulfide bond, done by E2
what happens in step 3 of the PDH cycle
moving from one lipamide to the next lipoamide
the acetyl group gets transferred to the OTHER lipoamide arm
done by E2 on PDH
what happens in step 4 of the PDH cycle
esterification of acetyl group to CoA
the acetyl group leaves the lipoamide arm and binds to CoA-SH, creating acetyl CoA. Done by E2
what is the structure of Acetyl CoA?
CH3-(C=O)-SCoA
what happens in step 6 of the PDH cycle?
oxidation of sulfhydryl groups on lipoamide by FAD, when the acetyl group leaves the disulfide bond remains broken and bound to hydrogen (Reduced), FAD takes the hydrogens to become FADH2 and reforms the disulfide bridge
what happens in step 6 of the PDH cycle?
Oxidation of FADH2 by NADH, done by E3 the NAD comes in and takes the H2 off the FAD, turning into NADH + H. the enzyme is them fully reset to go through another cycle
what are the allosteric regulators of PDH?
acetyl CoA and NADH
what are the covalent modifications for regulation of PDH
PDH kinase/PDH phosphatase
is PDH active or inactive when phosphorylated?
inactive
what might cause phosphorylation of PDH?
NADH/NAD+ - more NADH means higher ability to make ATP = higher energy potential = PDH-P to lower activity
acetyl CoA/CoASH - ACoA in high amounts mean cells have enough ATP so PDH is phosphorylated/inhibited
ATP/ADP or energy charge - High energy charge so phosphorylate PDH and lower activity
insulin stimulates [phosphorylation / dephosphorylation] of pdh - why does this make sense?
dephosphorylation - insulin is released to signal the cells to take in glucose - when cell glucose concentration increases aerobic metabolism, PDH gets dephosphorylated to increase the Krebs cycle to that cell glucose gets processed and turned into energy
when in high energy charge, what regulates PDH
NADH, ATP, and Acetyl CoA all negatively inhibit PDH
when in low energy charge, how is PDH regulated
Pyruvate and ADP inhibit PDH-kinase so that PDH is dephosphorylated and active
what are the 6 steps in the PDH cycle mechanism
pyruvate is decarboxylated at the active site of E1 forming the hydroxyethyl-TPP intermediate. CO2 leaves as the first product
E2 inserts the lipoamide arm of the lipoamide domain into the deep channel in E1 leading to the active site. E1 catalyzes the transfer of the acetyl group to the lipoamide
the acetylated arm then leaves E1 and enters the E2 cube to visit the active site of E2, located deep in the cube at the subunit interface
the acetyl group is then transferred to CoA, and the second product, the Acetyl CoA leaves. The reduced lipoamide arm then swings to the active site of E3
at the E3 active site, the lipoamide is oxidized by FAD. The reactivated lipoamide is ready to begin another reaction cycle
the final product NADH, is produced with the reoxidation of FADH2
what is the difference between a synthase and a synthetase?
Synthase - catalyzes a synthetic process without ATP - comes under lyase or transferase classification - eg HMG-CoA synthase, ATP synthase
Synthetase - requires ATP (syntheTase Takes ATP) - comes under ligase classification - eg succinyl-CoA synthetase, Glutamine synthetase
what is the first step of the carboxylic acid cycle?
Oxaloacetate + Acetyl CoA → Citryl CoA →(H2O → CoA)→ Citrate
what enzyme in the CAC converts oxaloacetate + Acetyl CoA → Citrate
citrate synthase
what is the ΔGº’ of the reaction catalyzed by citrate synthetase? what does that mean?
-7.5 kcal/mol - hydrolysis of thioester bond is highly favorable
citrate synthase prevents ___________. How?
prevents undesireable reactions.
Citrate Synthase exhibts induced fit (like hexokinase), since oxaloacetate binding induces structural changes in the enzyme that lead to the formation of the acetyl CoA binding site
formation of the reaction’s intermediate —Citryl CoA— causes a dramatic structural change that completes active site formation, enabling cleavage of thioester linkage
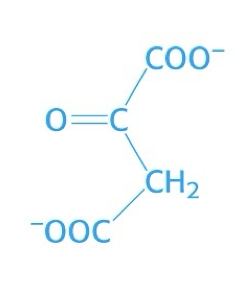
what is this structure
oxaloacetate
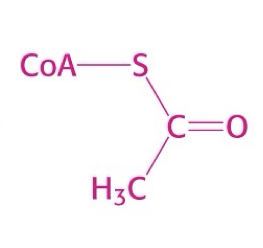
what is this structure?
acetyl coA
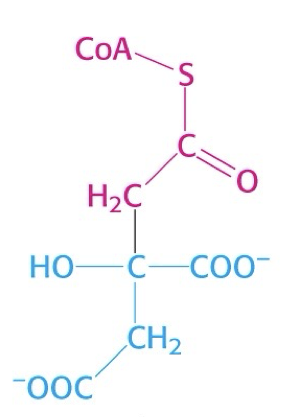
what is this structure?
citryl CoA
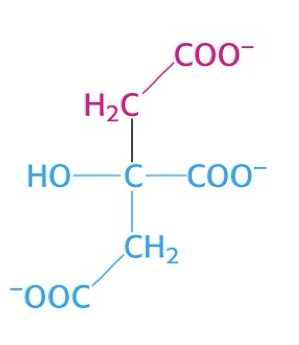
what is this structure?
citrate
what is the second step of the CAC
citrate → cis-Aconitate → isocitrate
what enzyme catalyses the reaction from Citrate → isocitrate?
aconitase
other than citrate, cis-aconitate, and isocitrate, what other molecule is involved in the reaction catalyzed by aconitase? how?
water, h2o is removed (deydration) to get from citrate to cis-aconitate, then added (hydration) to get from cis-aconitate to isocitrate
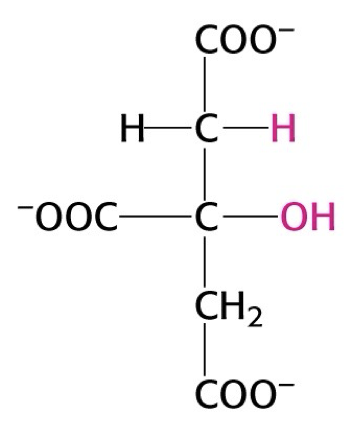
what structure is this?
citrate
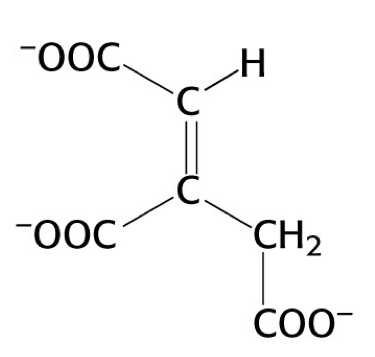
what structure is this?
cis-aconitate
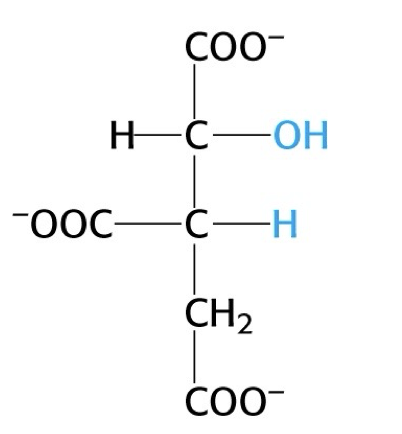
what structure is this?
isocitrate
what is the third step in the CAC
isocitrate → oxalosuccinate → alpha-ketoglutarate
what enzyme catalyzes the reaction from isocitrate → alpha-ketoglutarate
isocitrate dehydrogenase
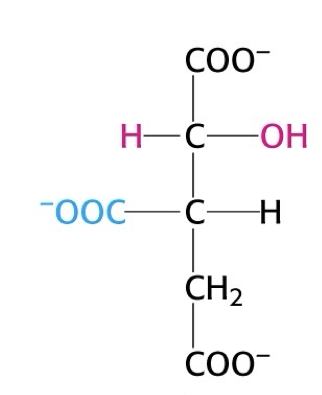
what structure is this?
isocitrate
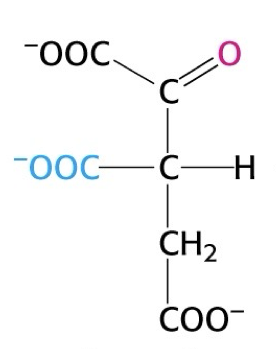
what structure is this?
oxalosuccinate
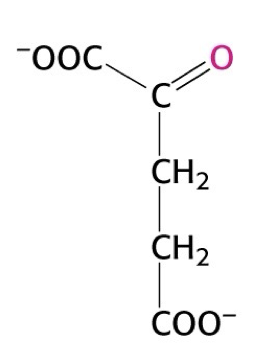
what structure is this
alpha ketoglutarate
what is step 4 of the CAC
alpha - ketoglutarate → succinyl CoA
what enzyme catalyzes the reaction from alpha-ketoglutarate → succinyl CoA
alpha-ketoglutarate dehydrogenase
what other molecules are involved in the reaction catalyzed by alpha-ketoglutarate dehydrogenase
NAD+ and CoA
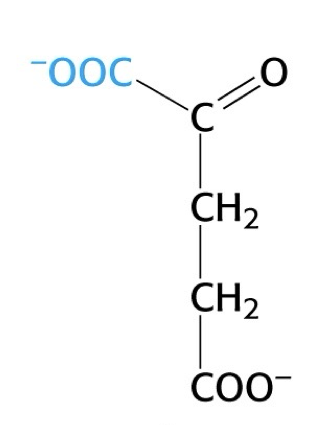
what is this structure
alpha - ketoglutarate
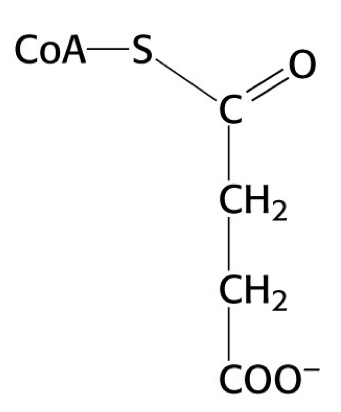
what is this structure
succinyl CoA
what is step 5 in the CAC
succinyl CoA → succinate
what enzyme catalyzes the reaction from succinyl CoA → succinate
succinyl CoA synthetase
what other molecules are involved in the reaction catalyzed by Succincyl CoA synthetase
Pi and GDP → CoA + GTP``
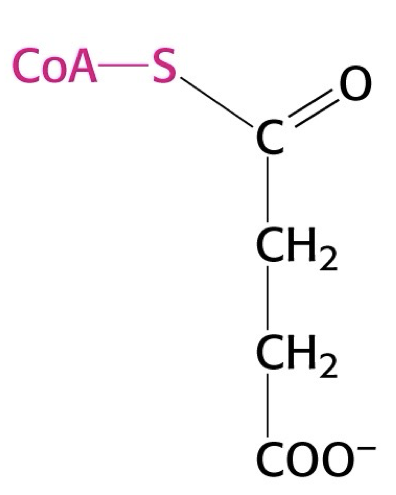
what structure is this?
succinyl CoA
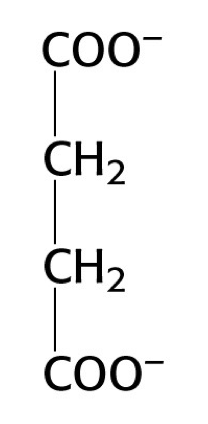
what structure is this?
succinate
what are the uses for GTP?
2 GTP are used for a peptide bond - ATP is recycleable energy so creates bonds that can be broken which would be bad for our proteins, GTP makes peptide bonds more stable
GTP + ADP ←> GDP + ATP - catalyzed by nucleoside diphosphokinase
what is step 6 in the CAC
succinate → fumarate
what enzyme catalyzes the reaction of succinate → fumarate
succinate dehydrogenase
what other molecule is involved in the reaction catalyzed by succinate dehydrogenase
FAD
why is FAD used as the electron acceptor in the reaction of succinate → fumarate instead of NAD?
free energy change for the reaction would be insufficient to reduce NAD
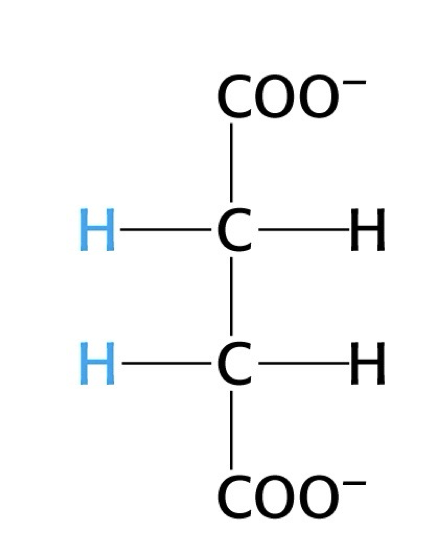
what structure is this?
succinate
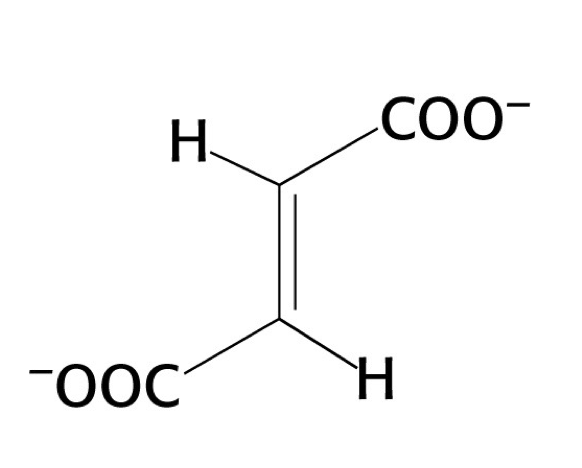
what structure is this?
fumarate
what is step 7 of the CAC
fumarate → malate
what enzyme catalyzes the reaction from fumarate → malate?
fumarase
what other molecules play a role in the reaction catalyzed by fumarase?
h2o (hydration)
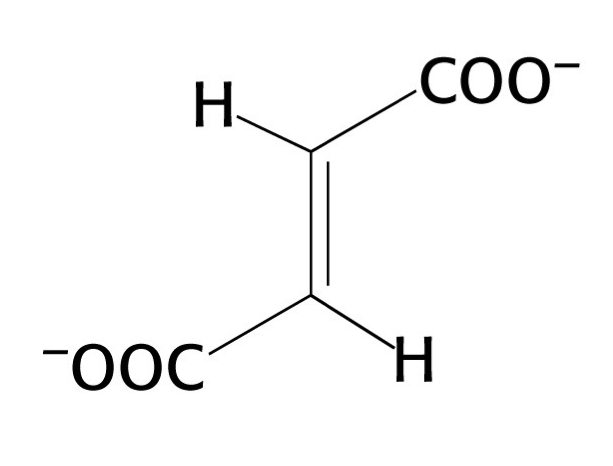
what structure is this?
fumarate
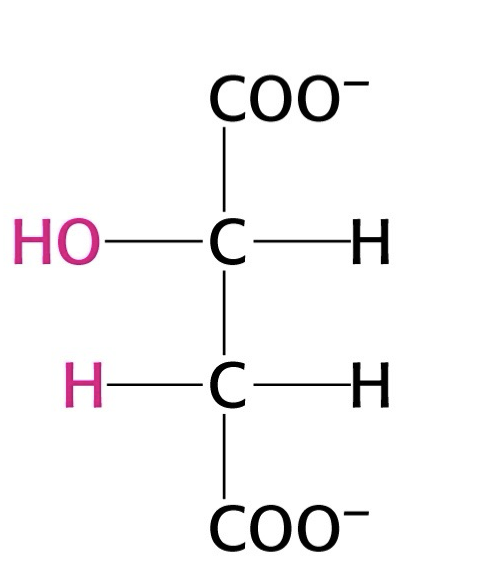
what structure is this?
malate
what is step 8 in the CAC
malate → oxaloacetate
what enzyme catalyzes the reaction from malate → oxaloacetate
malate dehydrogenase
what is the ΔGº’ for the reaction catalyzed by malate dehydrogenase? what does that mean?
+7 kcal/mol - extremely enderdonic, needs LOTS of energy to make it happen
how can we make the reaction from malate → oxaloacetate more spontaneous?
by decreasing the [OAA][NADH] and increasing the [malate][NAD+]
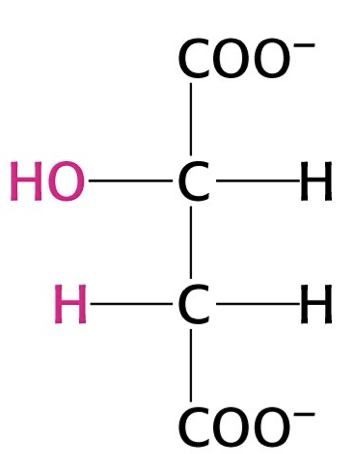
what structure is this?
malate
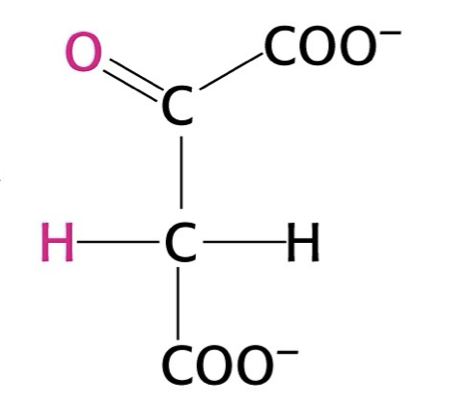
what structure is this?
oxaloacetate
per one glucose, what is yielded in the CAC
6 NADH, 2 FADH2, 2 ATP (GTP), 4 CO2
each NADH produced in the cytoplasm will be worth ____ ATP equivalents
1.5
each NADH produced in the mitochondiral matrix will be worth _____ ATP equivalents
2.5
each FADH2 produced in the mitochondrial matrix will be worth ____ ATP equivalents
1.5
what is the net yield per glucose of glycolysis?
2 ATP and 2 NADH
what are the three non-equilibrium enzymes in the CAC that are targeted for regulation?
Citrate synthase
isocitrate dehydrogenase
a-ketoglutarate dehydrogenase complex
what are the prosthetic groups on the alpha-ketoglutarate dehydrogenase complex?
lipoid acid, TPP, FAD
what molecules regulate citrate synthase? do they inhibit or induce?
citrate - inhibit, succinyl CoA - inhibit, NADH - inhibit, ATP - inhibit
what molecules regulate isocitrate dehydrogenase? do they induce or inhibit?
NADH - inhibit, ATP - inhibit, ADP - induce
what molecules regulate alpha-ketoglutarate dehydrogenase? do they induce or inhibit?
succinyl CoA - inhibit, NADH - inhibit, high energy charge - inhibit, Ca2+ - induce
why would calcium stimulate alpha-ketoglutarate in muscle tissue?
to increase muscle contraction, the calcium concentration must increase, if there is lots of muscle contraction happening you would also needs lots of ATP for energy
what molecules regulate pyruvate dehydrogenase? do they induce or inhibit?
ATP - inhibit, acetyl coA - inhibit, NADH - inhibit, ADP - induce, pyruvate - induce
defects in the citric acid cycle can contribute to what serious illness?
cancer
the defects in the enzymes succinate dehydrogenase, fumarase, and isocitrate dehydrogenase contribute to….?
cancer growth
how would mutations in succinate dehydrogenase or fumarase contribute to cancer growth?
activation of HIF-1 - enhanced anaerobic glycolysis (even when there is enough O2) - warburg effect
how would mutations in isocitrate dehydrogenase contribute to the development of cancer?
cause synthesis of 2-hydroxyglutarate, which modifies methylation pattersn in DNA. These modifications can alter gene expression and cause rapid cell growth
what anabolic roles does the CAC have?
alpha-ketoglutarate and OAA - amino acid synthesis
succinyl CoA - porphyrin synthesis
citrate - fatty acid and sterol synthesis via citrate lyase
→in the cytoplasm, citrate lyase catalyzes the reaction: citrate + CoASH + ATP → acetyl CoA + OAA + ADP + Pi
(acetyl CoA is the precursor for fatty acid / sterol synthesis)
how are metabolites in the CAC maintained?
anaplerotic reactions
what are some examples of anaplerotic reactions that maintain metabolite concentrations for the CAC
pyruvate carboxylase - catalyzes pyruvate + CO2 + ATP + H2O → OAA + ADP + Pi + 2H+
acetyl CoA stimulates pyruvate carboxylase
Glutamate → alpha ketoglutarate
beta oxidation of fatty acids → acetyl CoA