Period 3 elements
1/35
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
36 Terms
Equation for Na reacting with water and observations
2Na + 2H2O→ 2NaOH + H2
Effervescence
Large temp increase
Equation for Mg reacting with cold water and observations
Mg + 2H2O → Mg(OH)2 + H2
Gentle fizzing
Equation of Mg reacting with steam and observations
Mg + H2O → MgO + H2
White solid
White flame
Equation for Na reacting with oxygen and observations
4Na + O2 → 2Na2O
Yellow flame
White solid
Equation and observations for Mg reacting with Oxygen
2Mg + O2 → 2MgO
White flame
White solid
Equation and observations for Al reacting with oxygen
4Al + 3O2 → 2Al2O3
White flame (Less bright than Mg)
White solid
Equation and observations for P reacting with oxygen
P4 + 5O2 → P4O10
White flame
White solid
Equation and observation of S reacting with oxygen
S + O2 → SO2
Blue flame
Fumes of pungent gas
What are the structures of Na2O, MgO and Al2O3
Giant ionic lattice
Structure of silicon SiO2
Giant covalent
Structure of P4O10 and SO3
Simple molecular
Why does SiO2 have a high melting point
Has a giant covalent structure
Strong covalent bonds between atoms
A lot of energy is required to overcome these bonds
Why do simple molecular substances tend to have low MPs
Weak IM forces between molecules
Very little energy needed to overcome VDWS forces between molecules
(VDWS forces increase with size of molecule therefore P4O10 has a higher MP than SO3)
Explain why Na2O has a higher MP than SO3
It exists as a giant ionic lattice with strong ionic bonding
SO3 is simple molecular
Electrostatic forces of attraction between Na+ and O2- in lattice are much stronger than VDWS forces between SO3 molecules
What do the reactions of P3 oxides with water depend on
The type of bonding
Ionic oxide examples and how they react with water
Na2O and MgO
Contain oxide ions which react with water to produce OH ions
Forming alkaline solutions
Insoluble oxides examples and how they react with water
Al2O3 and SiO2
Little to no reaction with water
Examples of molecular oxides and how they react with water
P4O10 and SO2 and SO3
Form acidic solutions with water
Equations for both Na2O and MgO reacting with water
Na2O + H2O → 2NaOH
MgO + H2O → Mg(OH)2
Why is NaOH solution more alkaline than Mg(OH)2
Sodium hydroxide is more soluble (i.e dissociates more)
[OH-] is greater
Ph is higher
Equations of how the 3 non-metal oxides react with water
SO2 + H2O → H2SO3 / Weak acid
SO3 + H2O → H2SO4 / Strong acid
P4O10 + 6H2O → 4H3PO4 / Weaker than sulfuric acid stronger than H2SO3
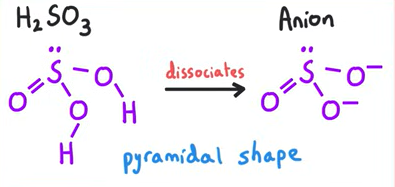
Structure of H2SO3 and its anion
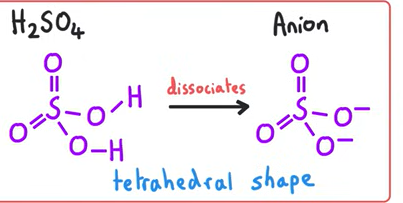
Structure of H2SO4 and its anion
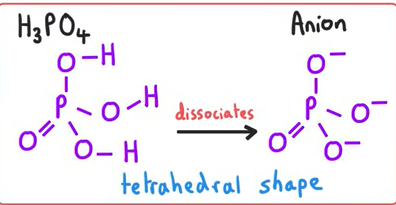
Structure of H3PO4 and its anion
What will happen to the Ph of water when Al2O3 or SiO2 is added
Will stay 7
Why is SiO2 insoluble
Has strong covalent bonds between atoms
H2O cannot supply enough energy to break the strong covalent bonds
Why is Al2O3 insoluble
Is an Ionic oxide
Al3+ has a very high charge density
Leads to additional covalent character
Therefore very strong attractions within the lattice that H2O cant overcome
3 tests to distinguish between aq solutions of sulfur trioxide and sulfur dioxide

List basic oxides and acidic oxides
Basic- React with acids
Na2O
MgO
Acidic-React with bases
SiO2
P4O10
SO3
SO2
What is special about Al2O3
It is amphoteric
Meaning it reacts with acids and bases
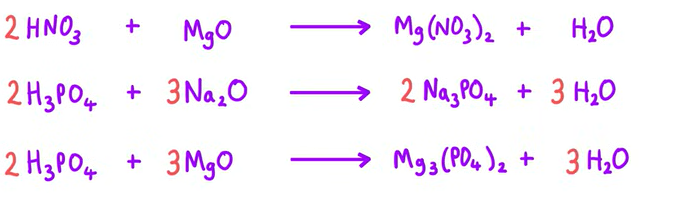
Equation of Nitric acid reacting with MgO
Equation of Phosphoric acid reacting with Na2O
Equation of Phosphoric acid reacting with MgO
Equation for SO3 reacting with KOH
Equation of P4O10 reacting with NaOH

Equation for Al2O3 reacting with HCL and H2SO4 (acids)
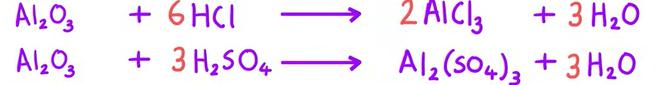
Equation for Al2O3 reacting with NaOH (base)
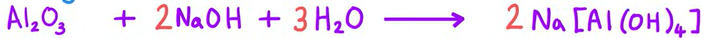
General equation for P3 oxides reacting with each other
Acid + Base → Salt
No water
Equation for P4O10 reacting with MgO
Equation for SO3 reacting with Al2O3
P4O10 + 6MgO → 2Mg3(PO4)2
3SO3 + Al2O3 → Al2(SO4)3