pH, Electrolytes, ECF & ICF
1/14
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
15 Terms
What does pH stand for?
Potential Hydrogen
What does pH measure?
pH is a measure of how acidic or basic a solution is
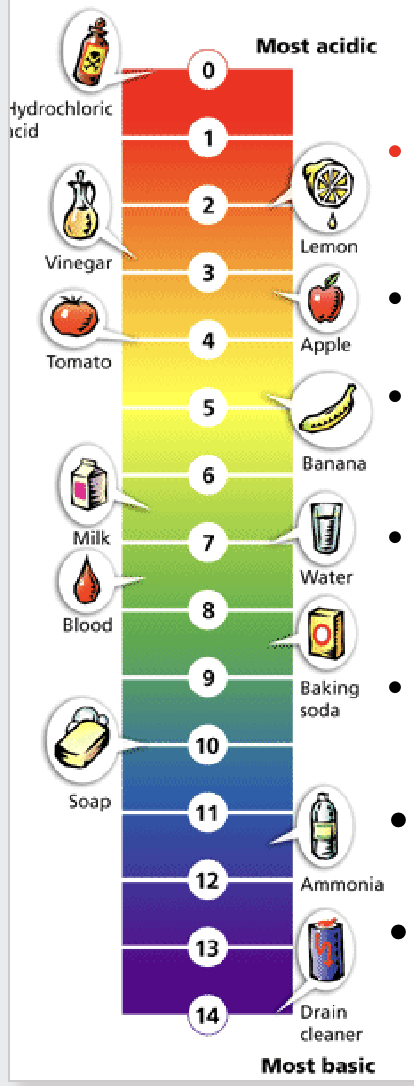
What are the pH intervals?
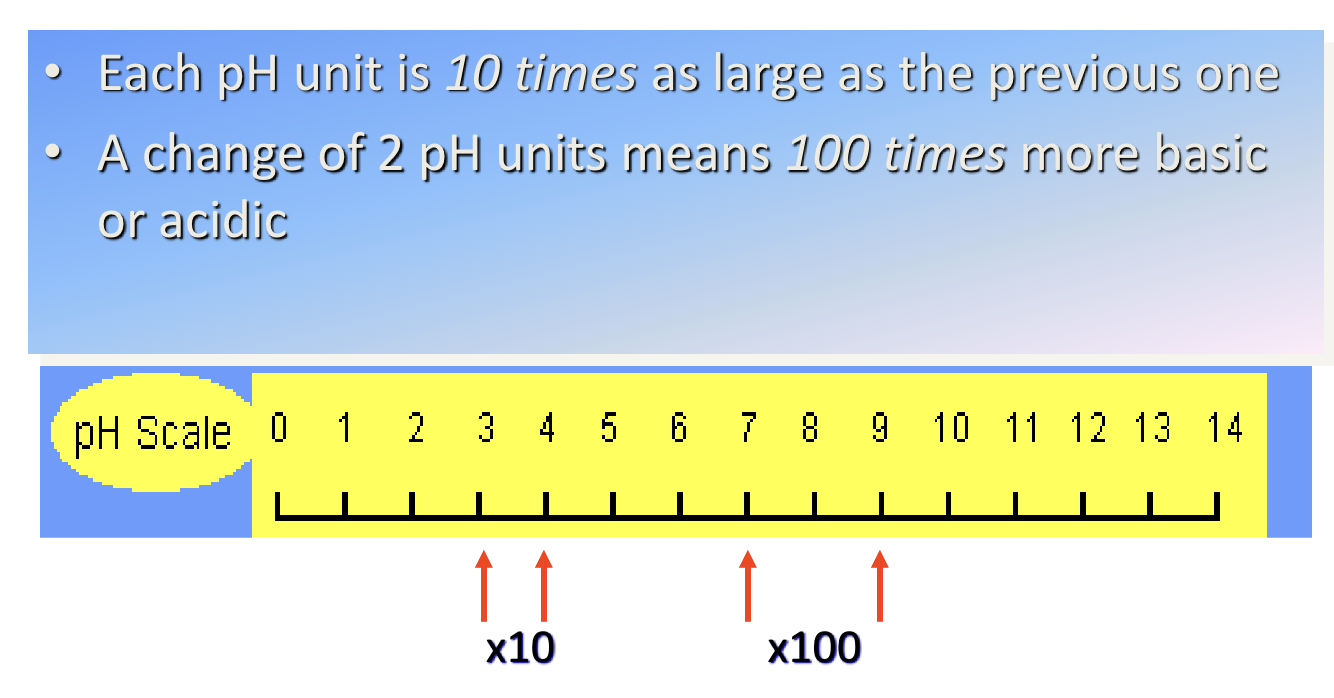
Each pH interval is 10x larger than the previous one, meaning 2 pH intervals are a change of 100x
What is an electrolyte?
An electrolyte is a compound that releases ions when in water
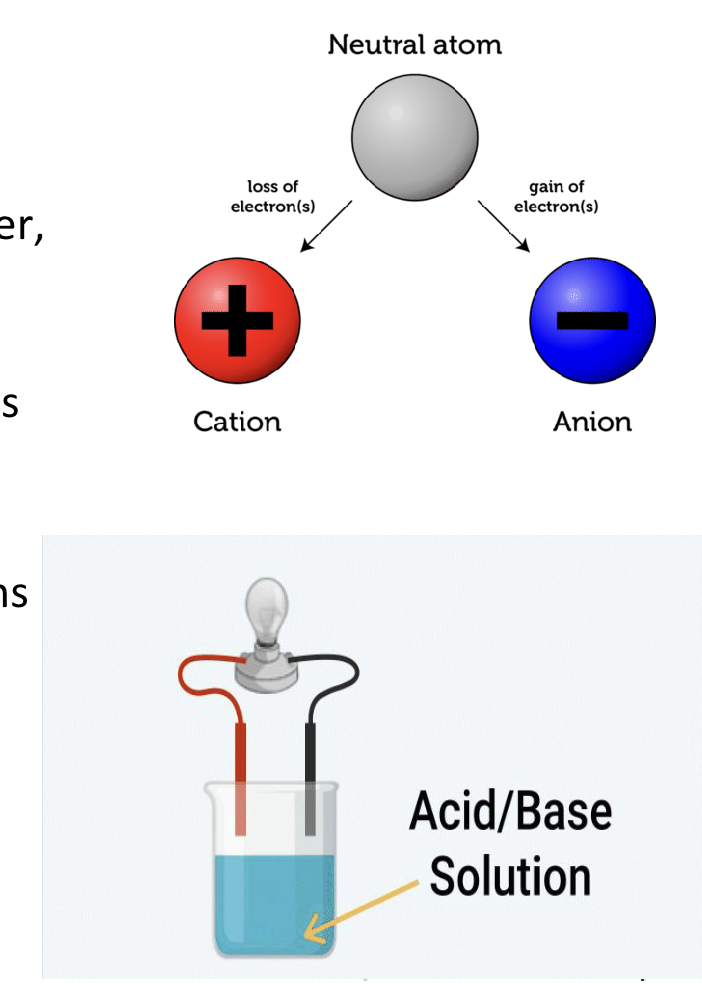
What do strong electrolytes do?
Dissociates in water producing positive and negative ions as well as conducting a strong electric current
What do non electrolytes do?
Dissolves in water as molecules and does not produce ions or electric current
What are the functions of electrolytes?
Used as cofactors for optimal enzyme activity
Maintenance of fluid balance (osmosis)
Maintenance of acid-base balance (pH)
Synaptic transmission (carries electrical currents)
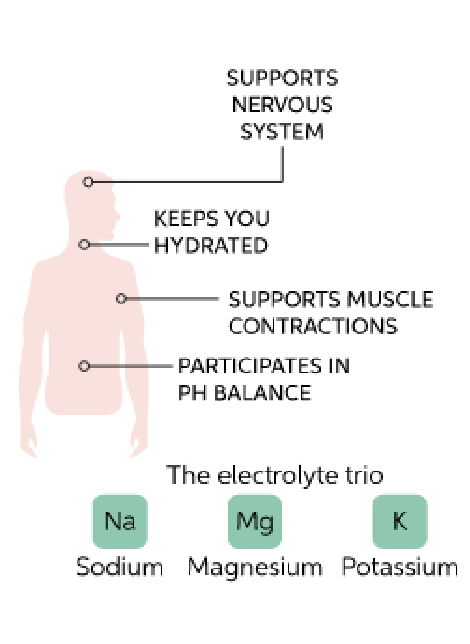
Which body fluids contain electrolytes?
All body fluids contain electrolytes
In Extracellular fluid, what is the major cation?
Na+ (sodium) is the major cation in ECF
In Extracellular fluid, what is the major anion?
Cl- (chloride) is the major anion in ECF
In Intracellular fluid, what is the major cation?
K+ (Potassium) is the major cation in ICF
In Intracellular fluid, what is the major anion?
Phosphate is the major anion in ICF
In the ECF, what is sodium’s function?
In the ECF, sodium’s function is impulse transmission, muscle contraction, and fluid & electrolyte balance
In the ECF, what is chloride’s function?
In the ECF, chloride’s function is regulating osmotic pressure and forming HCl in gastric acid
In the ICF, what is potassium’s function?
In the ICF, potassium’s function is resting membrane potential, action potentials of nerves and muscles, maintaining intracellular volume, and regulation of pH