unit 1 not on sac
1/20
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
21 Terms
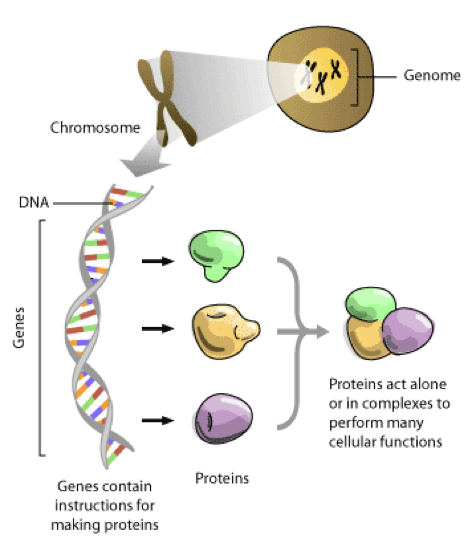
The Proteome
uThe proteome is the complete set of proteins expressed by the genome of an individual cell or organism at a given time.
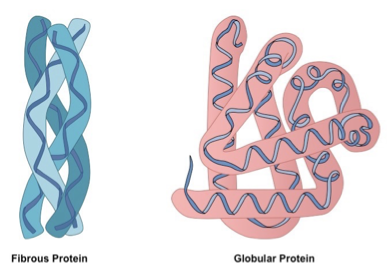
Types of Proteins
Fibrous proteins – generally elongated and insoluble, used for structure e.g. collagen in connective tissue, keratin in har and nails.
Globular Proteins – compactly folded and coiled into spherical tertiary and quaternary structures, generally soluble, have outer hydrophilic region and inner hydrophobic core, most enzymes and hormones are globular.
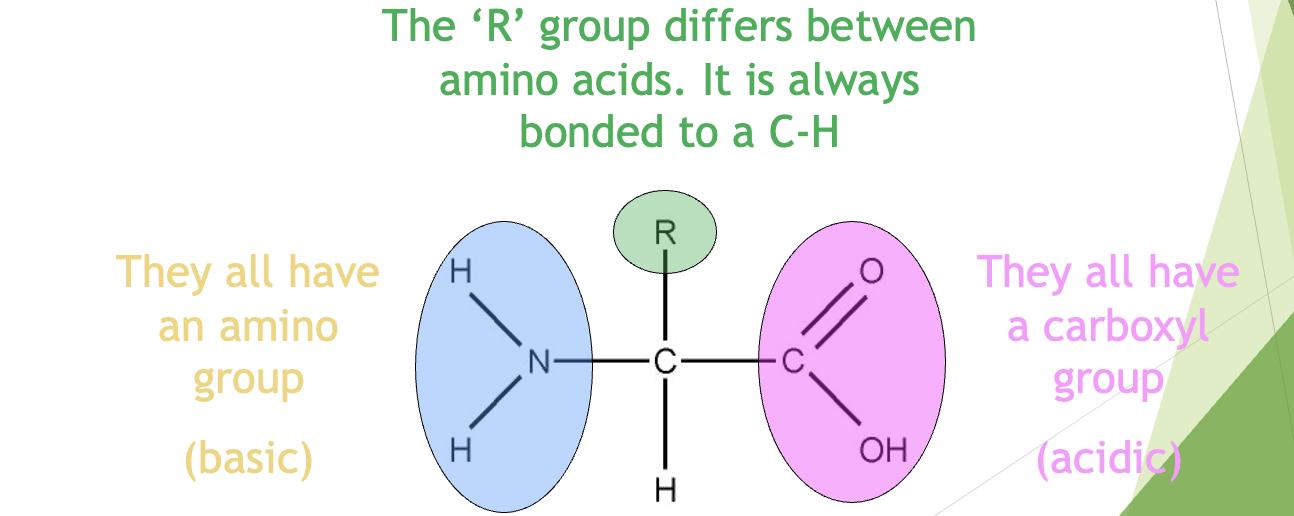
The Monomer – An Amino Acid
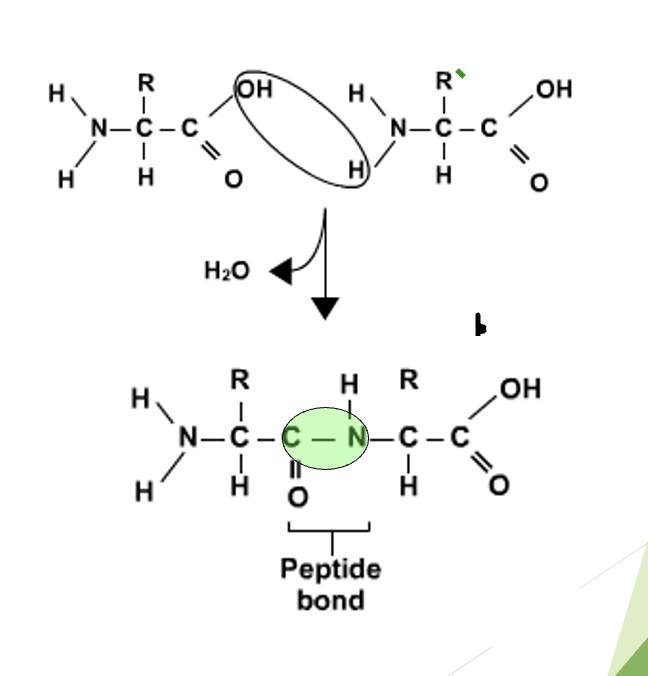
Building a Polypeptide Chain
Process called condensation polymerisation. One amino acid bonds to another (hydrogen and oxygen from the carboxyl group of one amino acid join), one molecule of water is released (condensation). A peptide bond is formed between them, and the molecule is now called a dipeptide. Many amino acids joined together forms a polypeptide chain.
Hierarchical Structure of Proteins
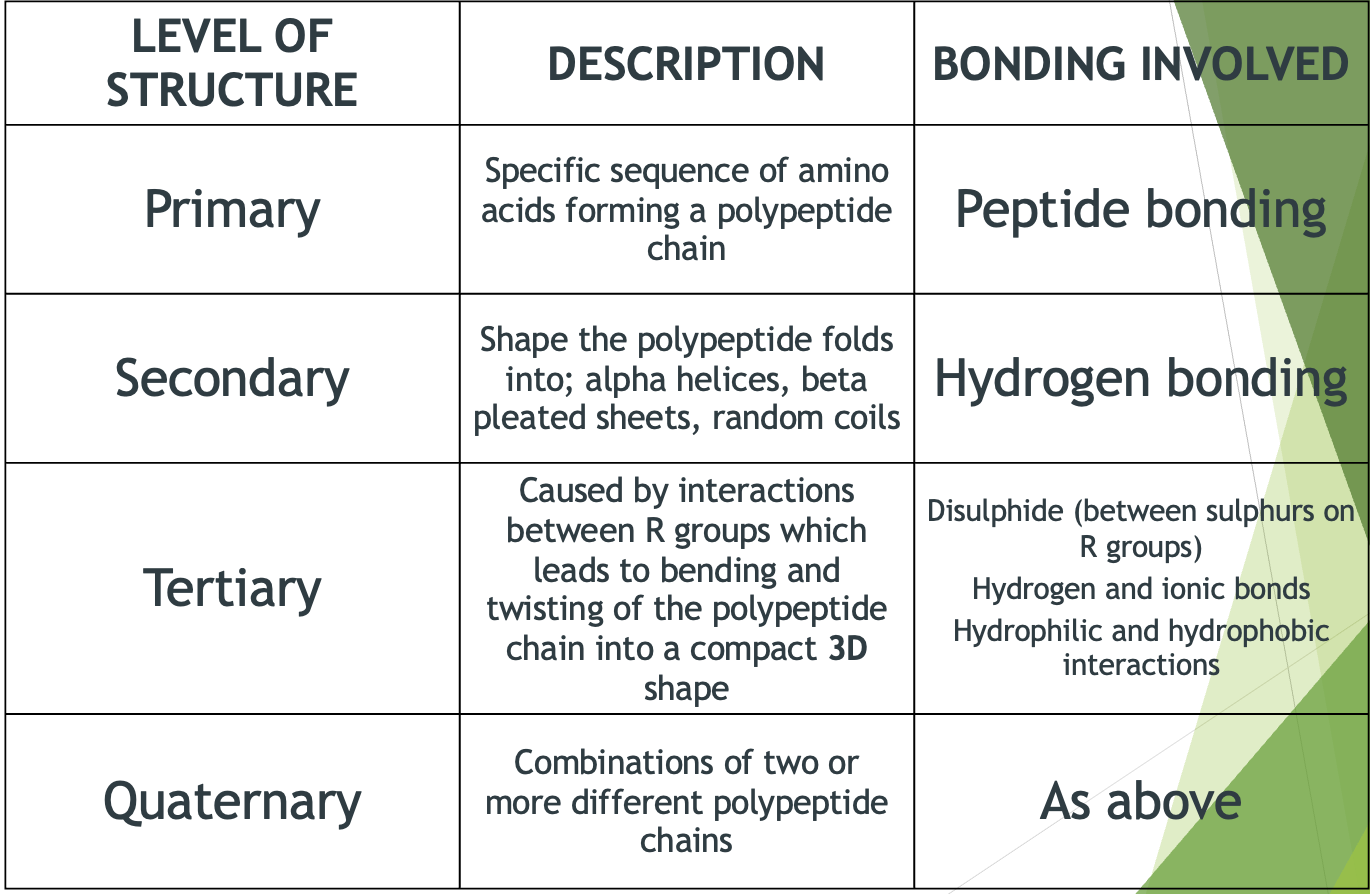
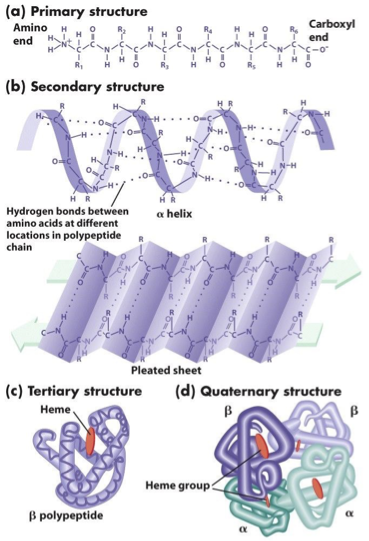
3D shape depends on
primary structure because the amino acid side groups respond differently to water, altering the shape.
Hydrophobic – water repelling. A hydrophobic side group will turn inwards, away from the water, towards the centre of the protein molecule.
Hydrophilic – water attracting. A hydrophilic side group will turn outwards, forming hydrogen bonds with the surrounding water molecules.
plasma membrane and Phospholipids
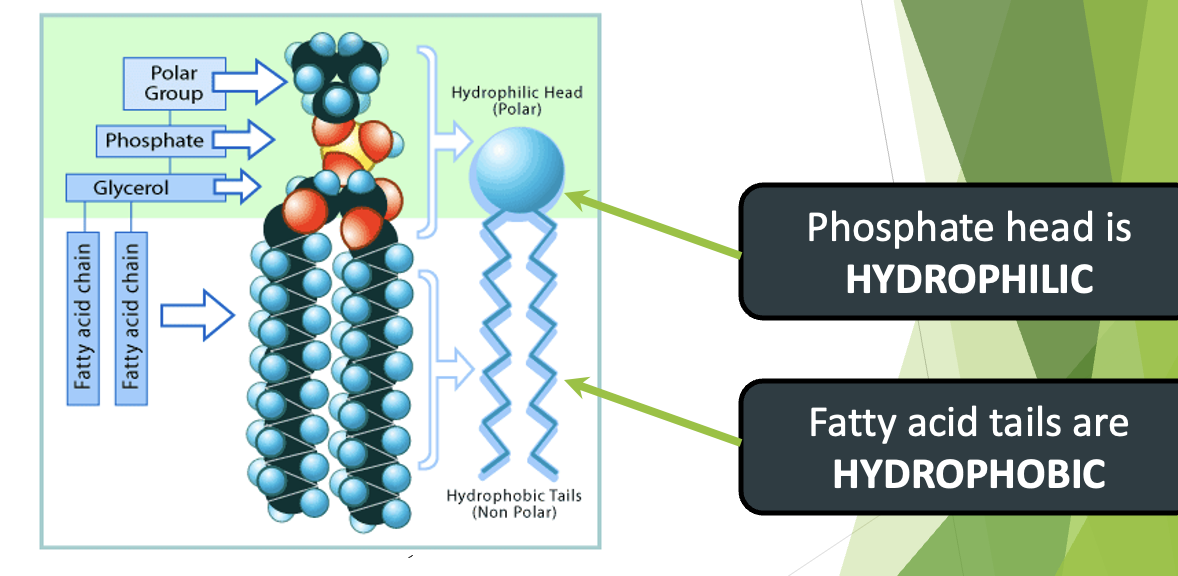
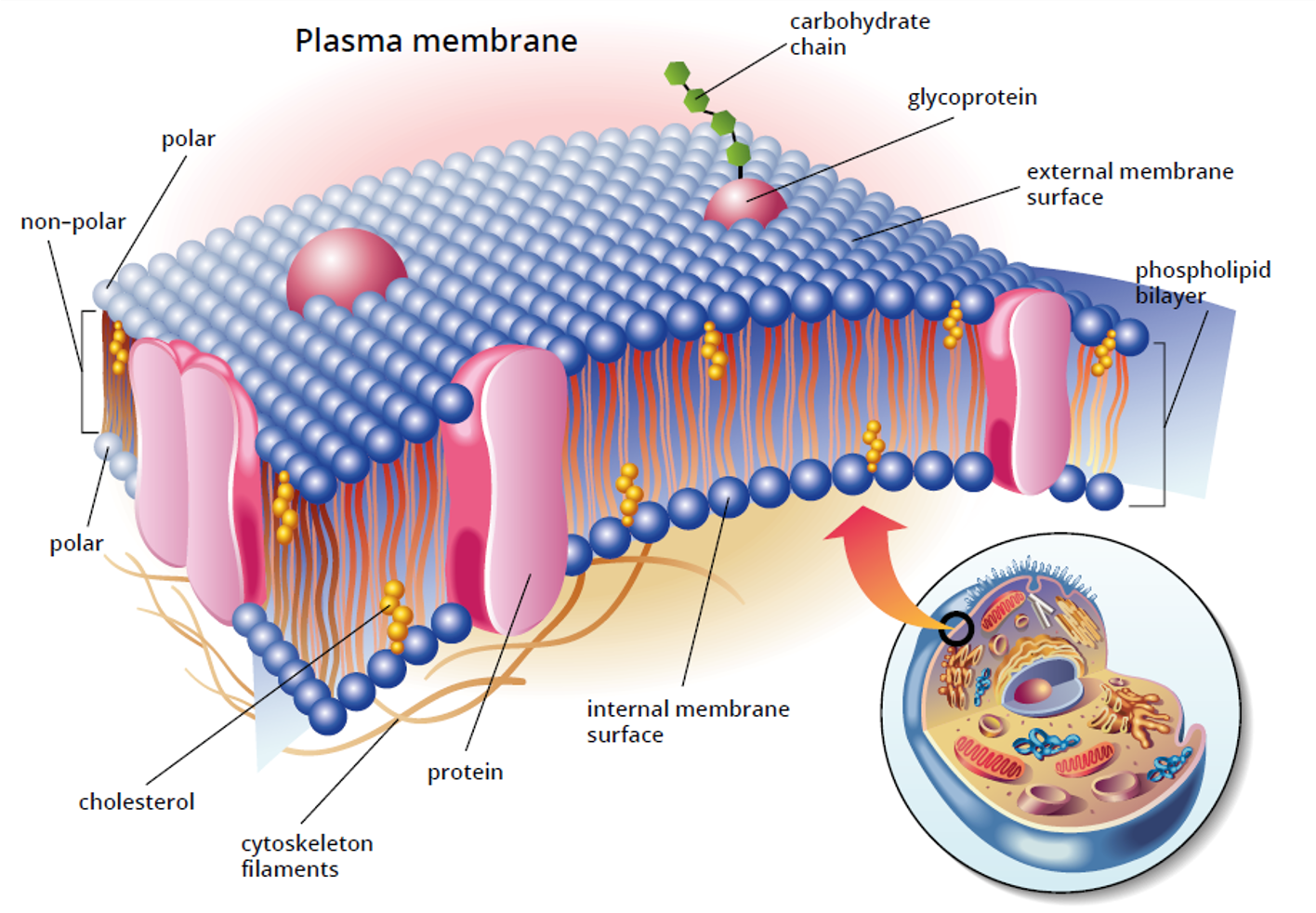
exporting proteins
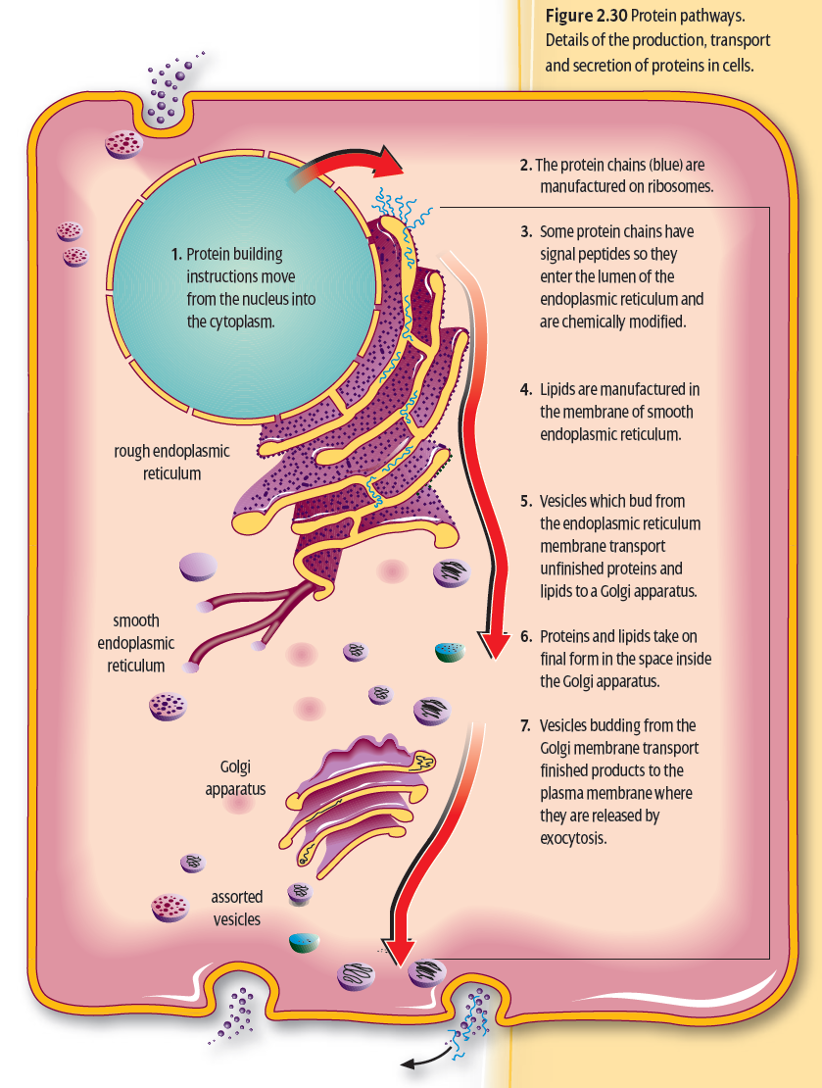
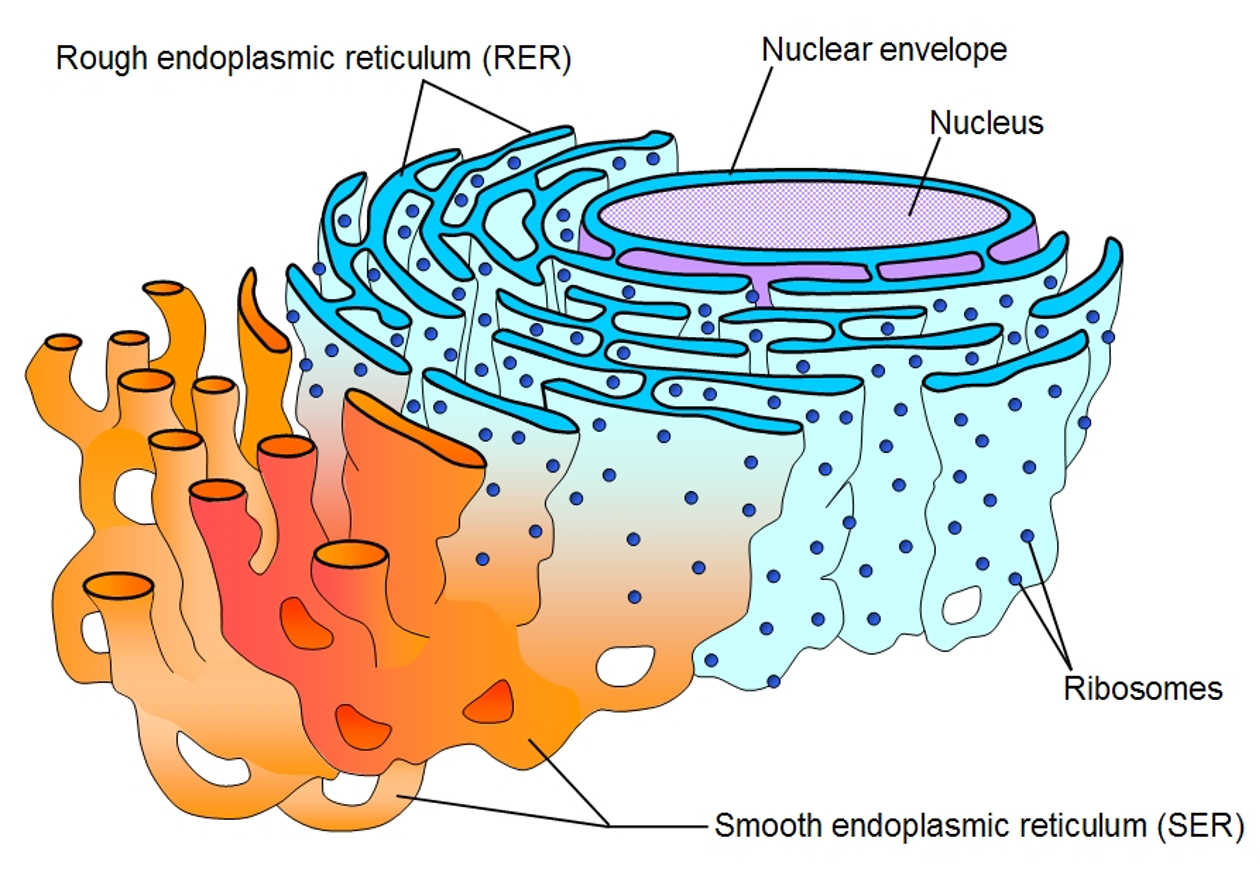
Rough Endoplasmic Reticulum
series of folded membranes forming channels called cisternae).
Ribosomes attached.
Synthesis and process proteins by adding other molecules.
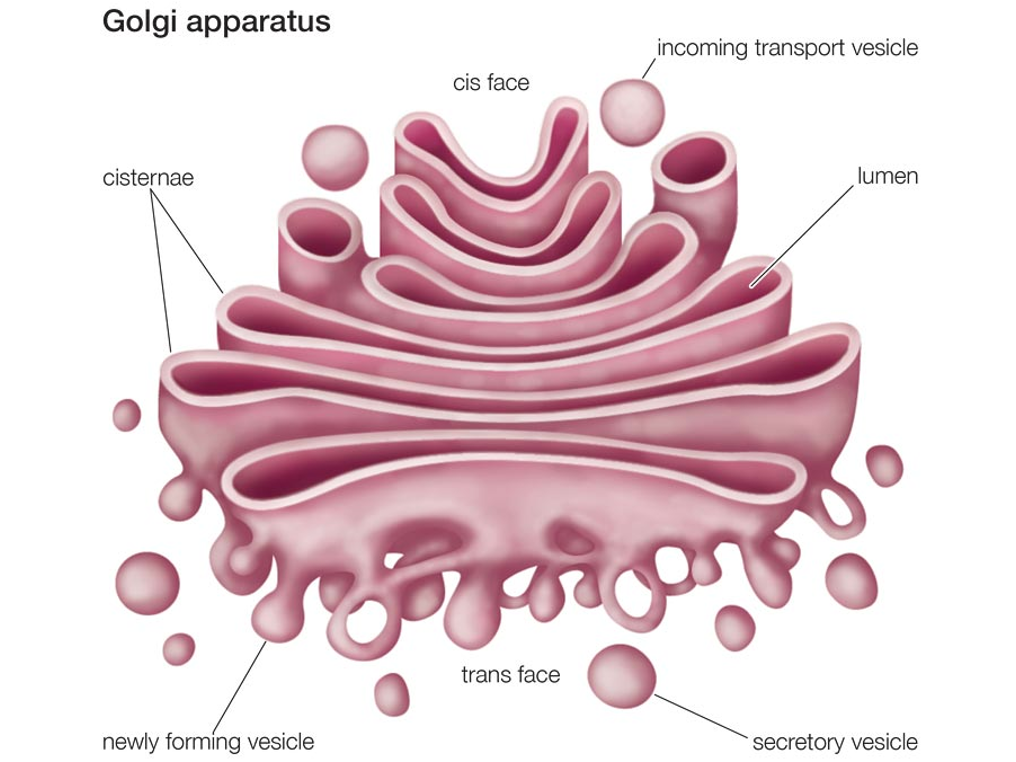
The Golgi Apparatus
Consists of several layers of membranes.
Proteins are transported in transition vesicles and are sent to the Golgi apparatus from the ER.
Further processing and modification of proteins.
The Golgi complex packs the proteins into secretory vesicles and are stored in the cytosol and then excreted via exocytosis (except lysosomes).
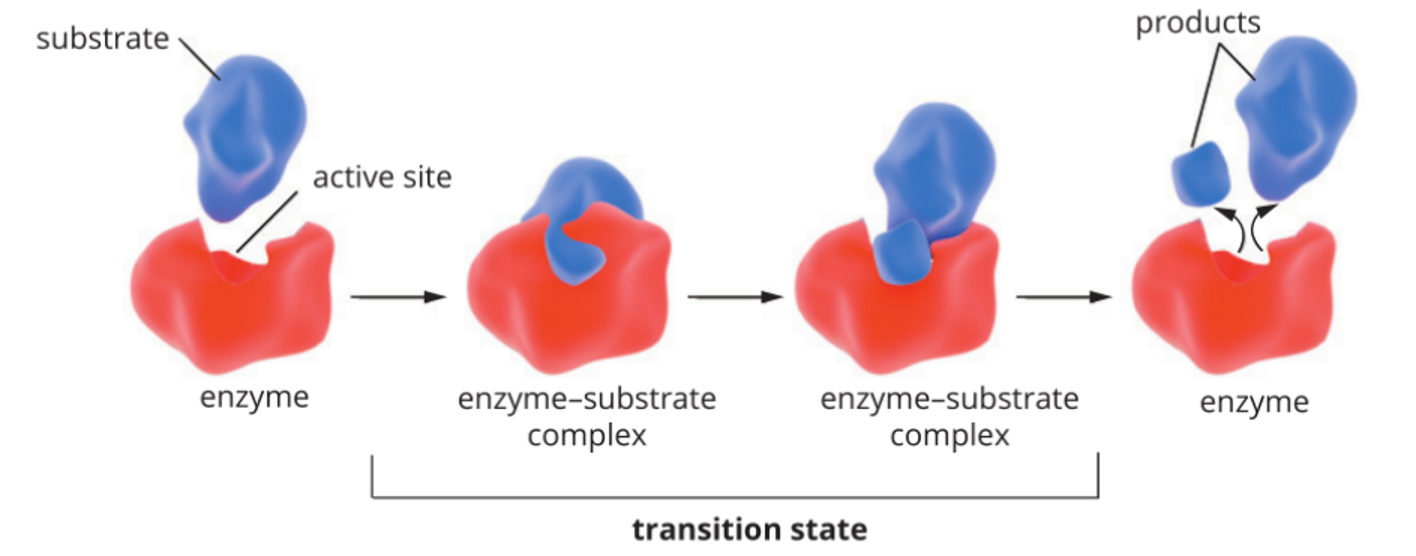
enzymes
Globular proteins with tertiary or quaternary structure, role is to increase the rate of (catalyse) biochemical reactions. end in ase. reusable.
How do Enzymes Work?
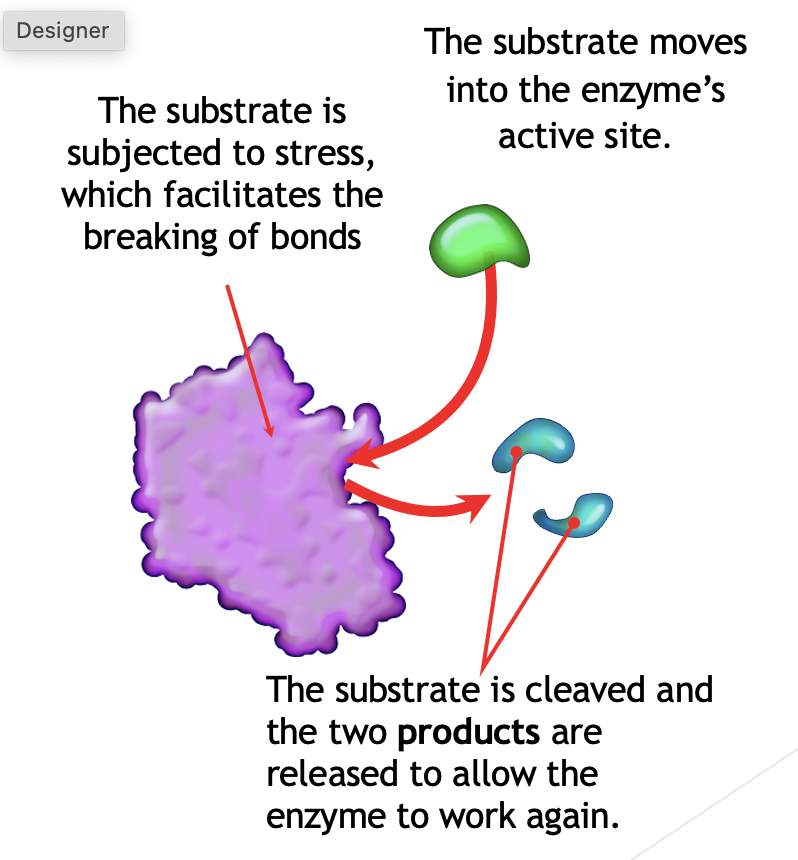
Enzymes work on molecules - substrates by binding to them. Specific structure to match a specific substrates. Either building molecules or breaking down large molecules into smaller ones. Specific region called the active site where the substrate binds and where catalysis occurs. (enzyme-substrate complex is formed).
Lipase + Fat à Fatty acids + Glycerol
Lipase is the enzyme.
Fat is the substrate.
Fatty acids and glycerol are the products.
active site: flexible and capable of changing its shape in order to conform to the shape of substrate and achieve a tighter fit. (conformational change).
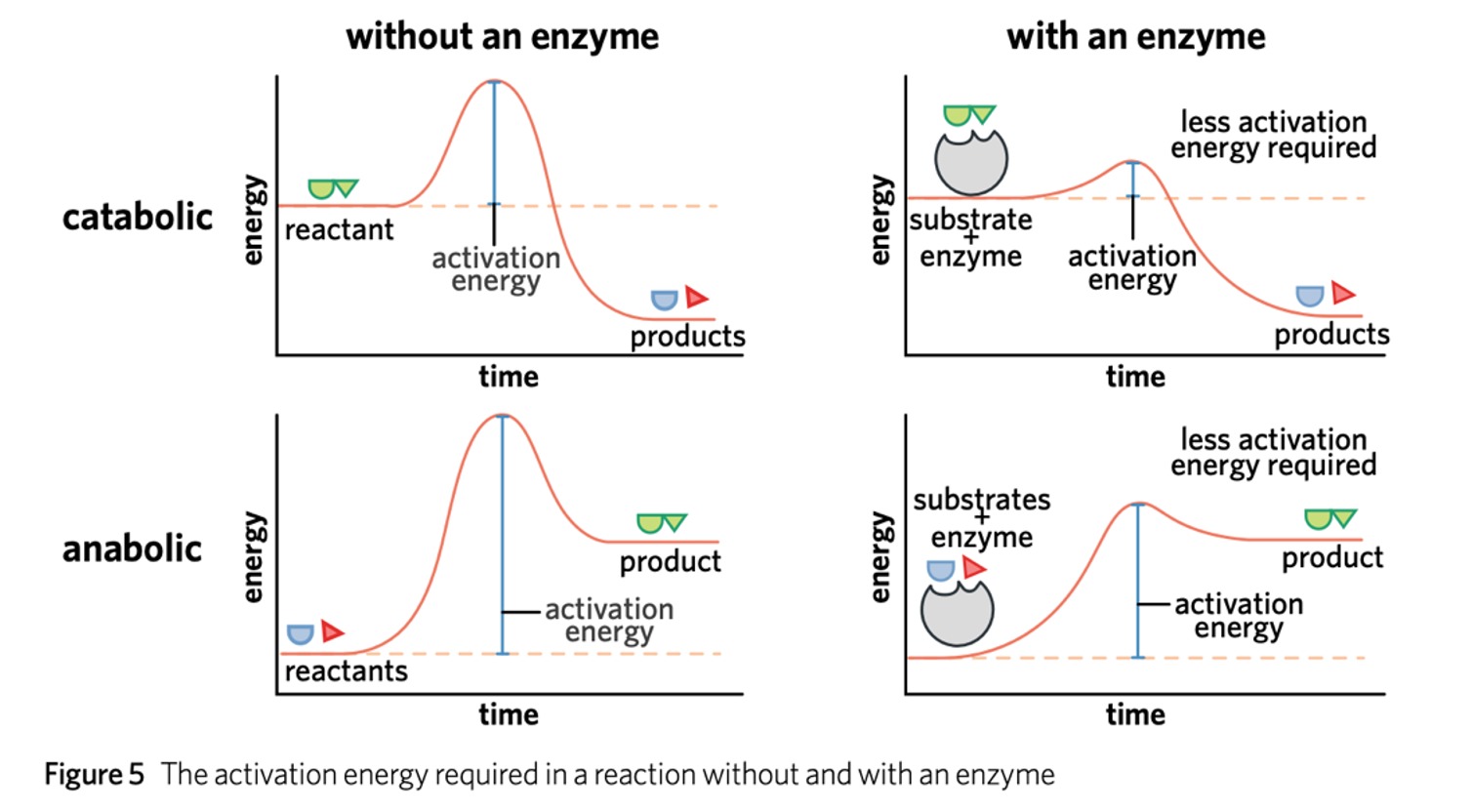
Activation Energy
energy required to get a reaction started.
Catabolic Reactions
Breakdown of larger molecules into smaller components, with the release energy (called exergonic reactions).
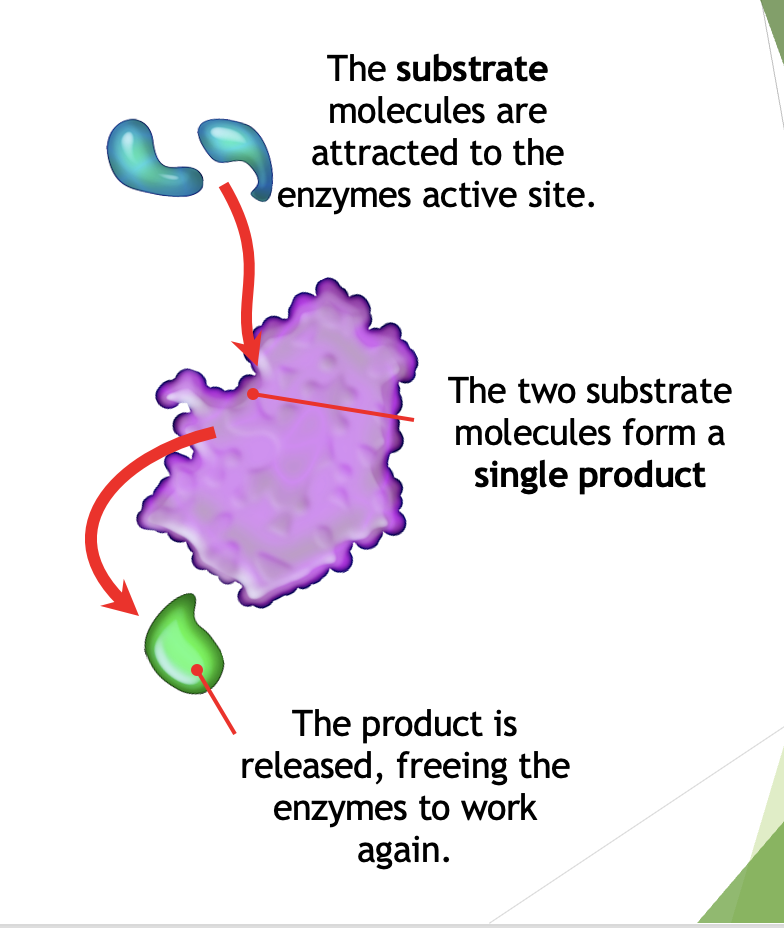
Anabolic Reactions
smaller molecules are joined to form larger ones, an input of energy is required (called endergonic reactions).
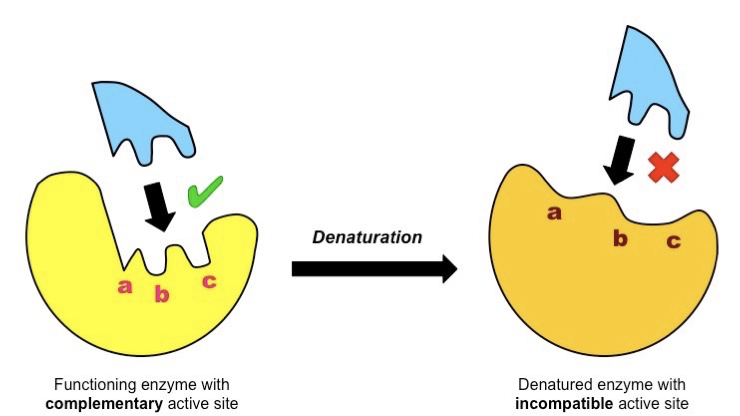
Denaturing Enzymes
The secondary and/or tertiary structure of the protein is lost – the amino acid chains unravel.
This destroys the shape of the active site.
can no longer act as a catalyst.
-heat, extreme, ph, chemcials
irreversible
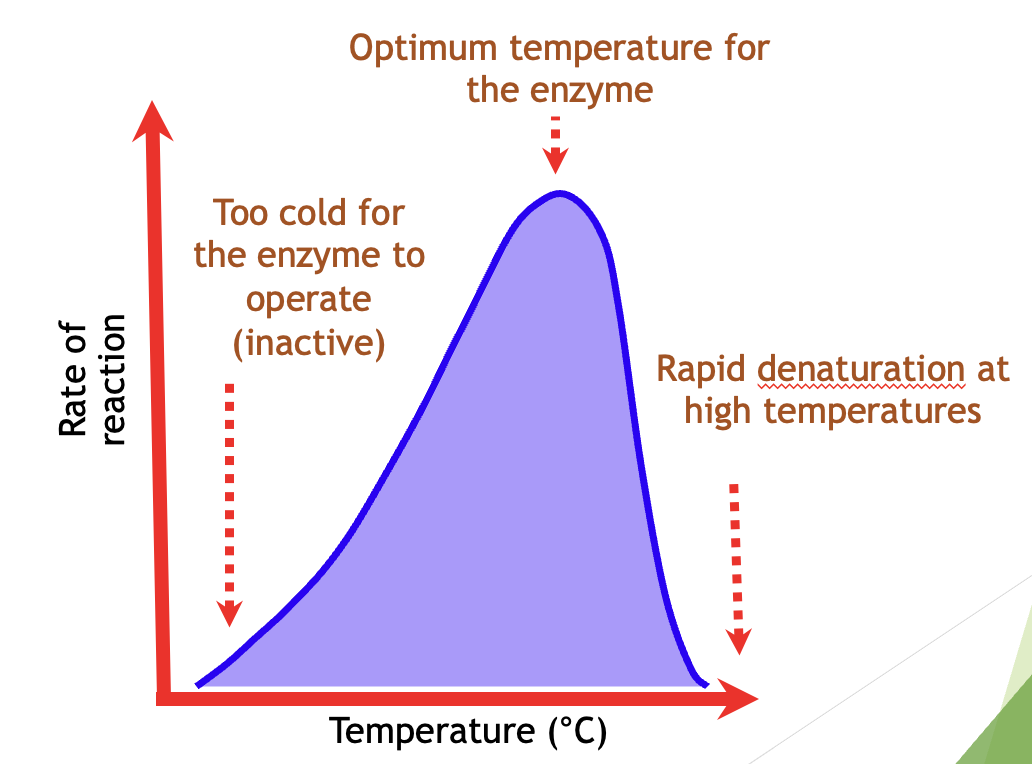
Effect of Temperature
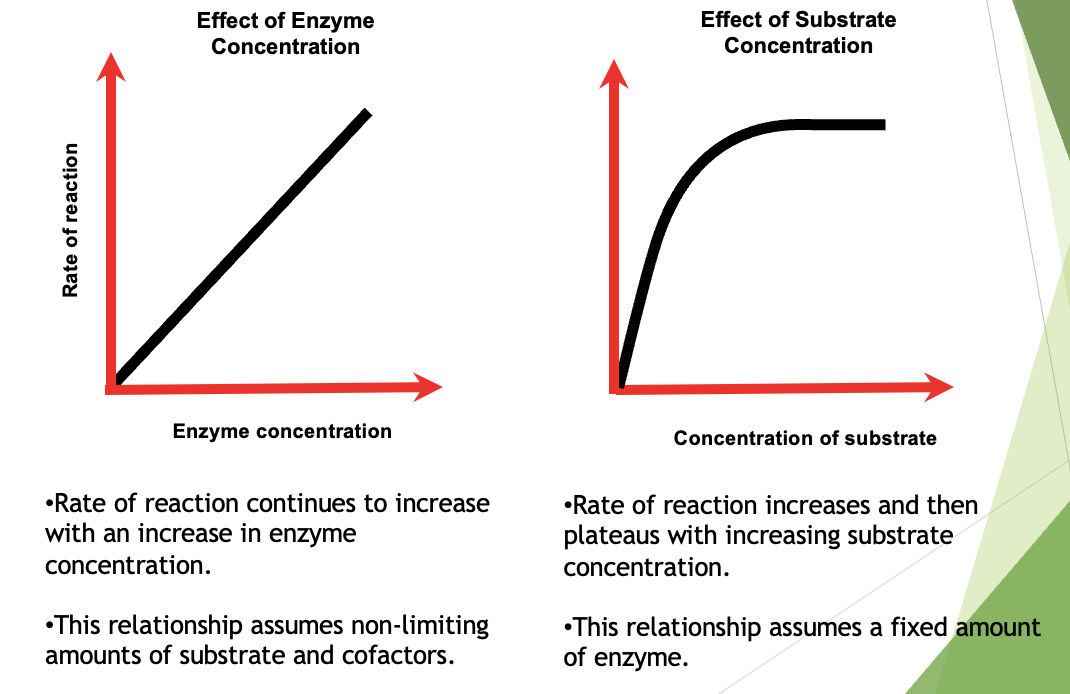
Effect of Enzyme Concentration
uAs long as there is an excess of substrate molecules, there will be a proportional increase in the rate of reaction as the enzyme concentration increases.
uIf there is not an excess of substrate molecules, then eventually the rate reaches a maximum speed.
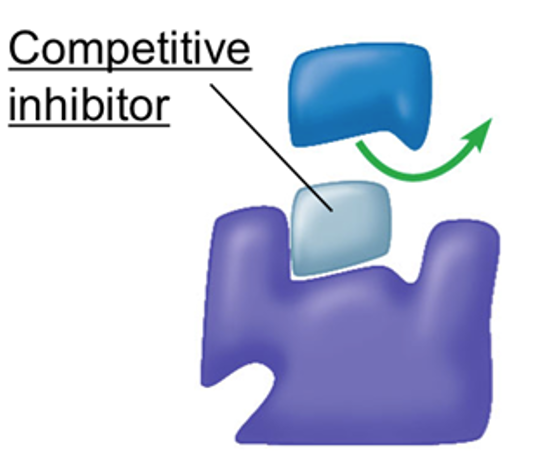
Competitive Inhibition (Reversible)
A competitive inhibitor is a molecule that competes with the substrate for binding to the enzyme’s active site.
As its shape is complementary to the active site, it binds temporarily to the active site, preventing an enzyme-substrate complex from forming.
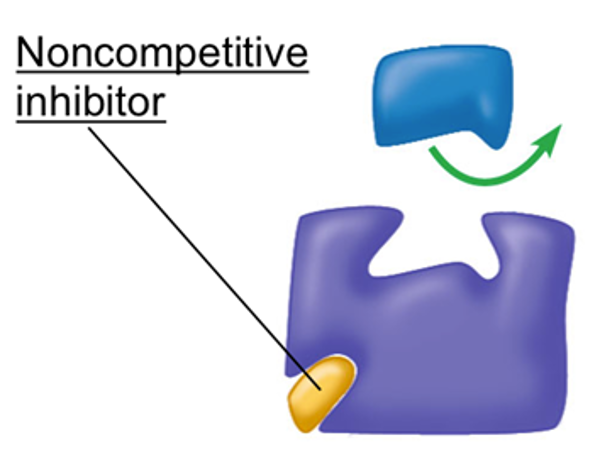
Non-Competitive Inhibition (Reversible)
The inhibitor molecule binds with the enzyme in another part of the enzyme (allosteric site), not at the active site.
This binding alters the shape of the enzyme, and therefore it’s active site.
The enzyme cannot successfully form an enzyme-substrate complex.
Nucleic Acids
organic macromolecules that store and transmit inherited characteristics of organisms (hereditary information).
They encode instructions for the synthesis of proteins.
They are linear polymers made of repeated subunit monomers called nucleotides.
Two types – DNA and RNA.