Orgo bag
1/94
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
95 Terms
r
re
res
reserv
reserve
reserved
reserved
reserve
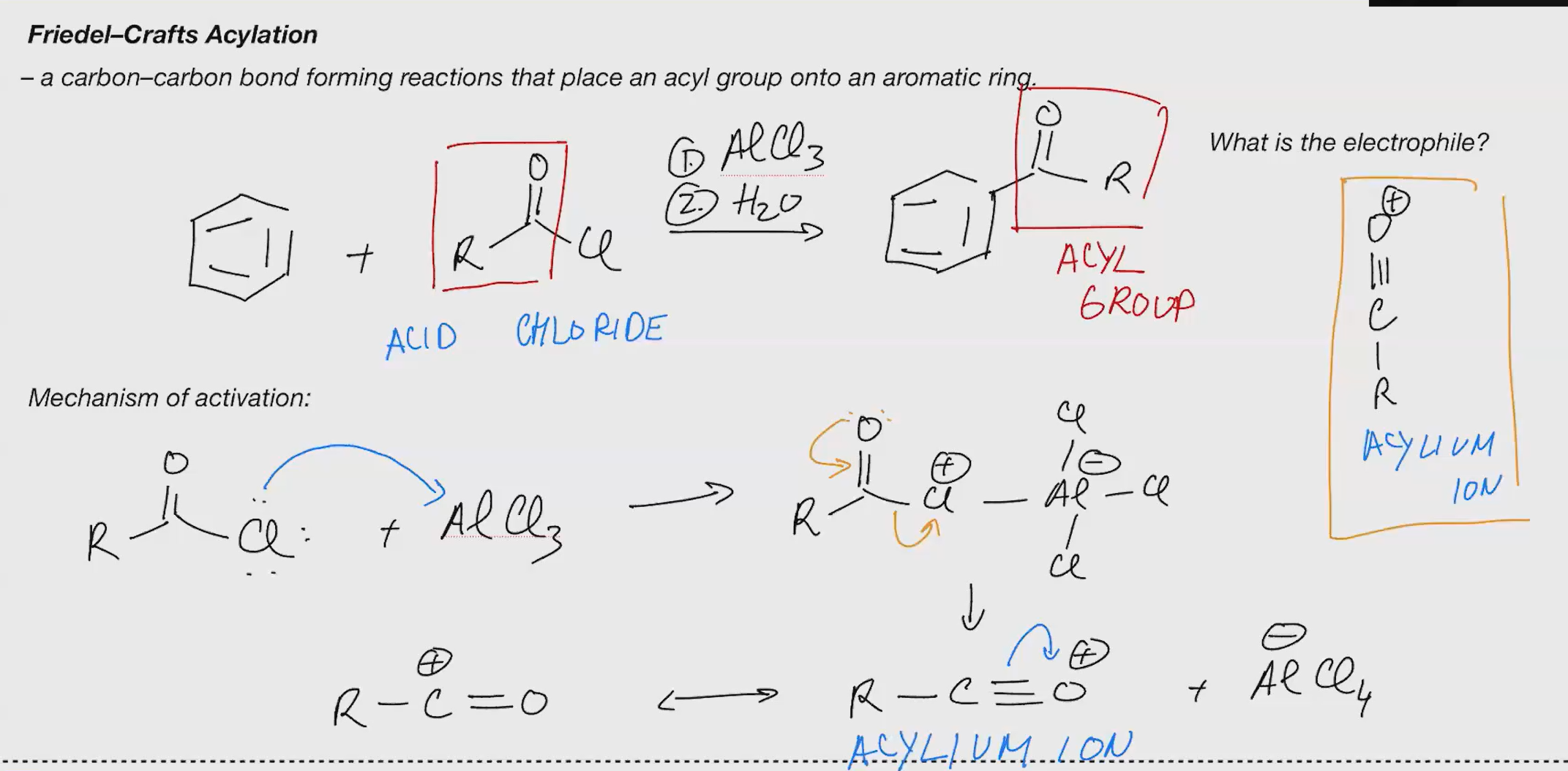
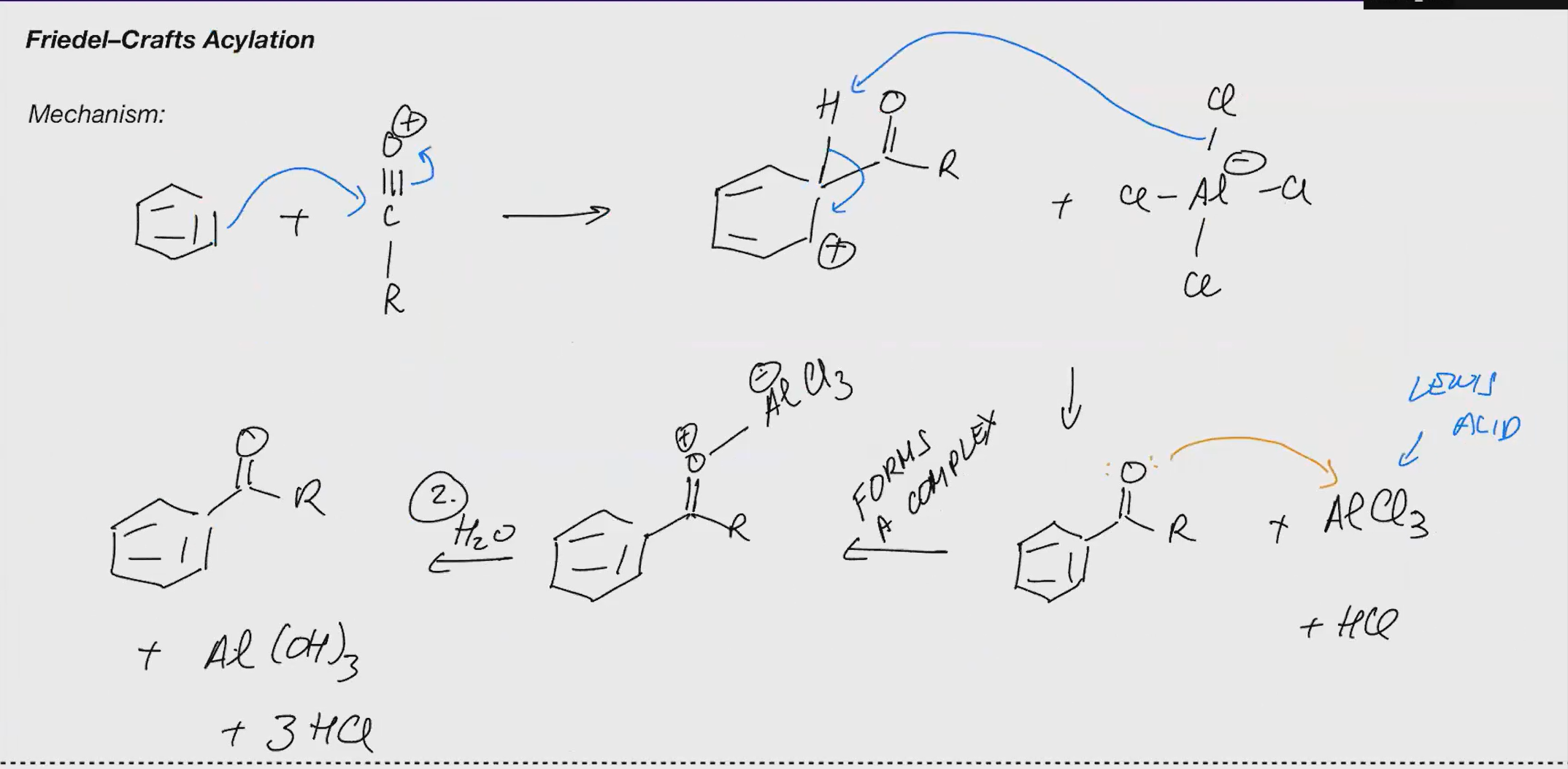
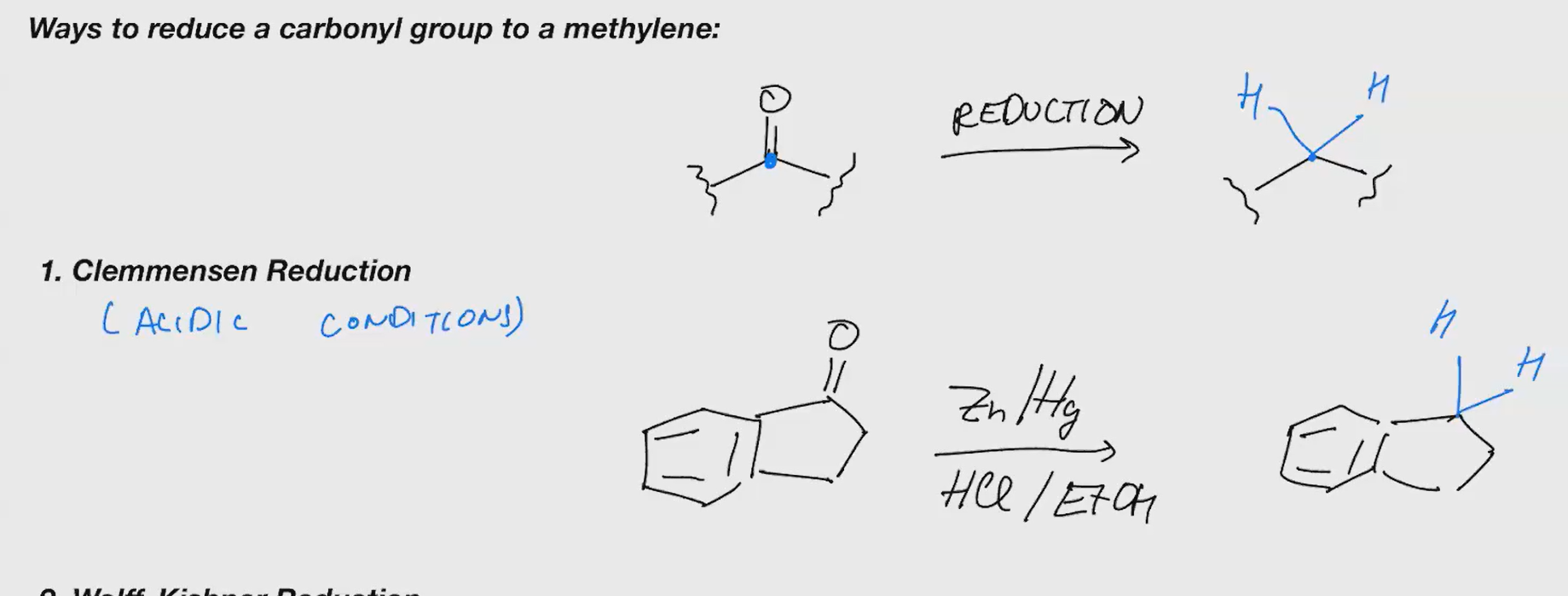
Friedel craft acylation (ACID CHLORIDE)
Reagents: 1. AlCl3 2. H2O
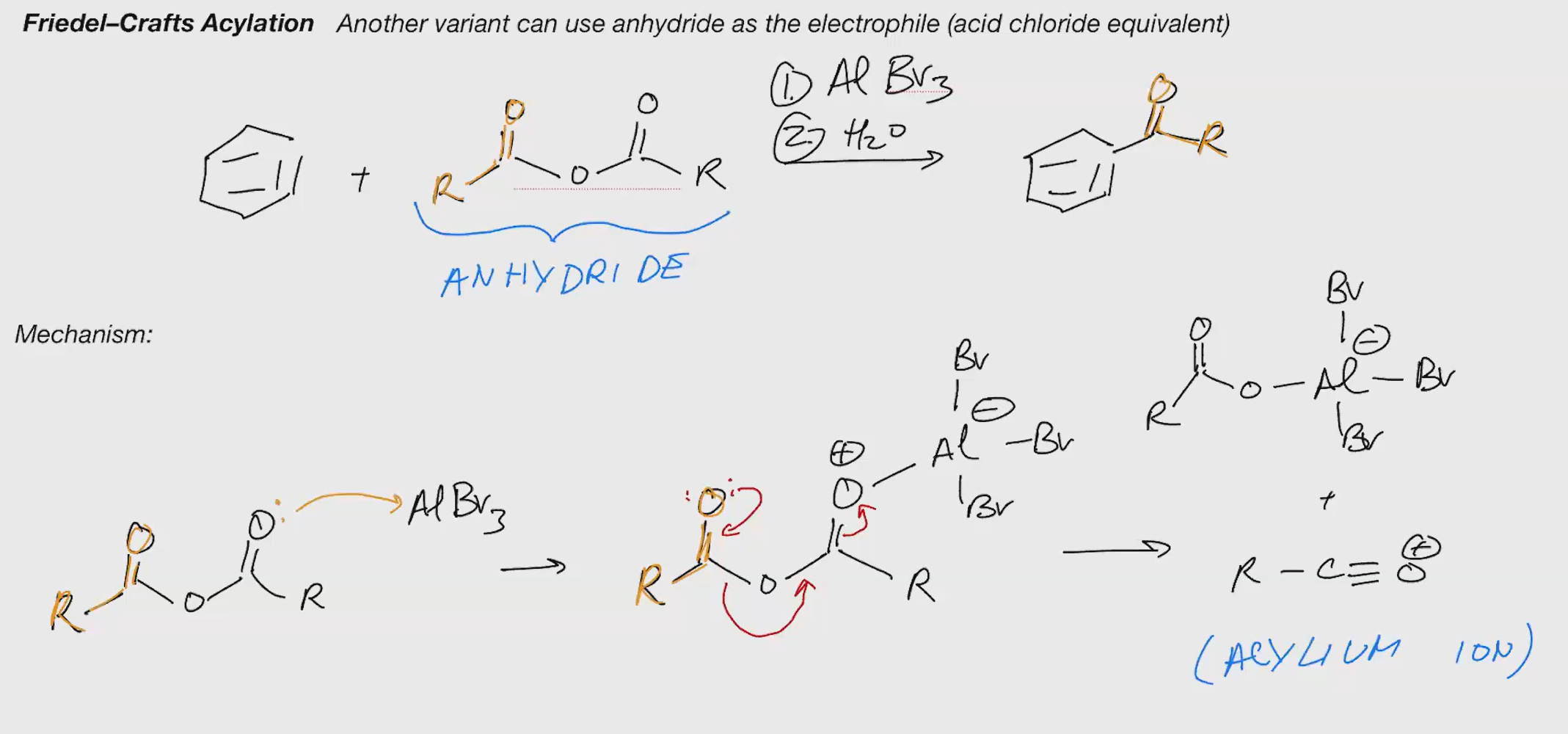
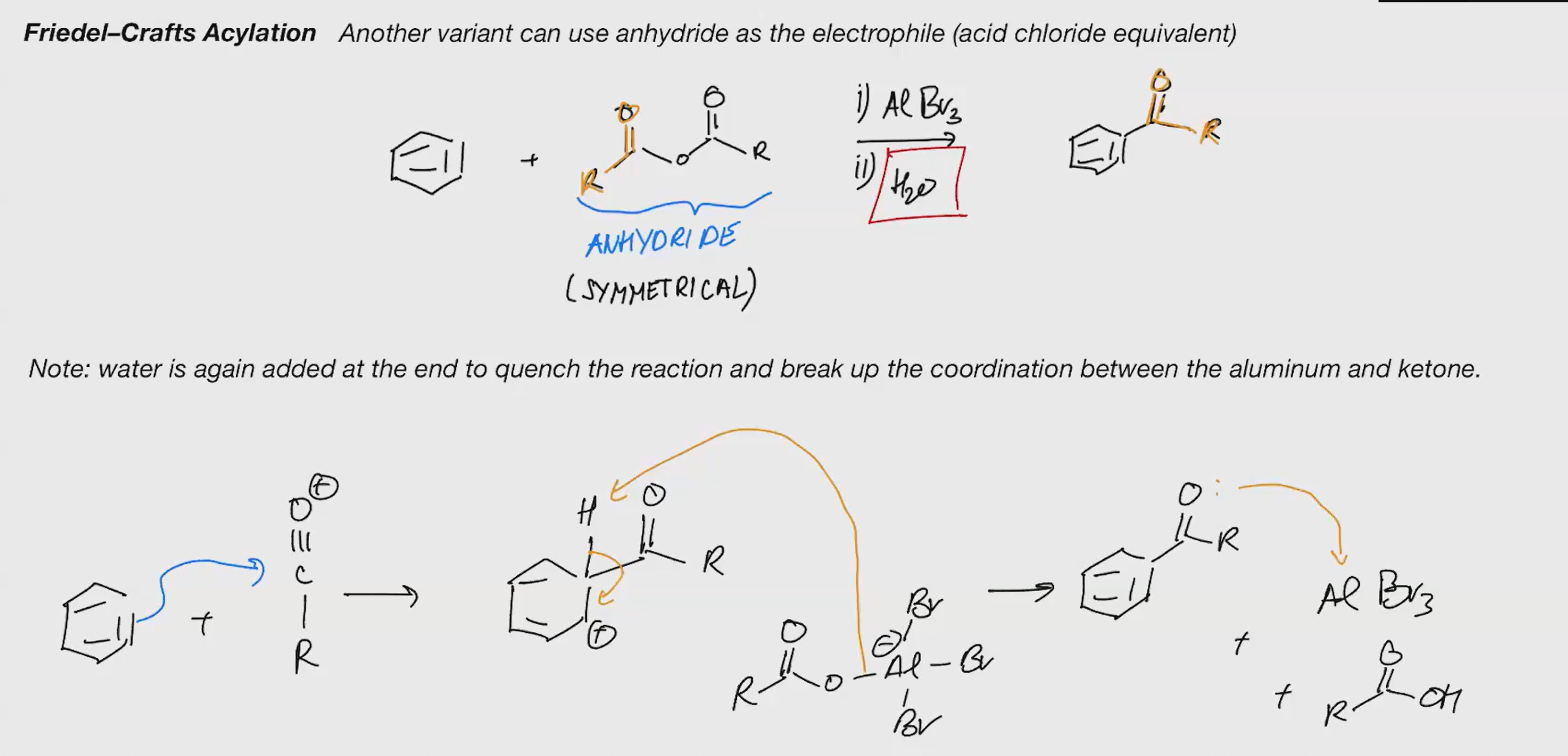
Friedel craft acylation (ACID CHLORIDE)
Reagents 1. AlBr3 2. H2O
Clemmensen reduction
Reduce a carbonyl group to a methylene
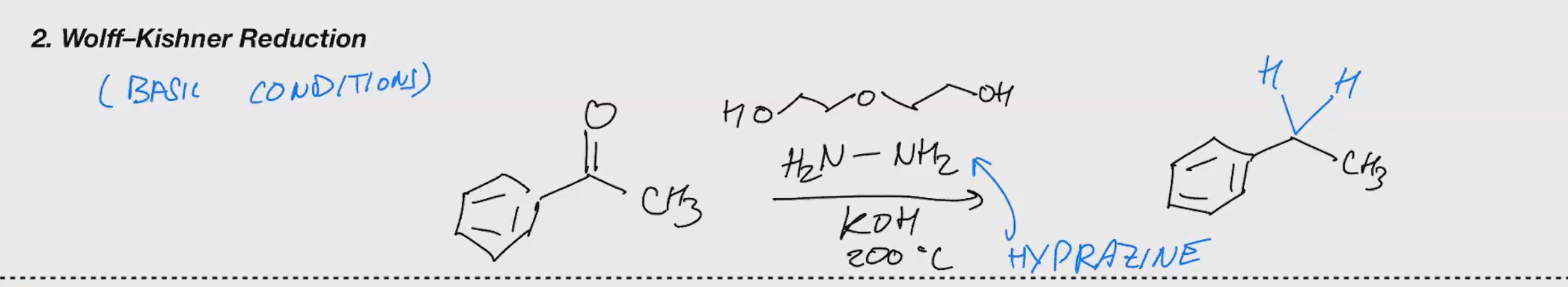
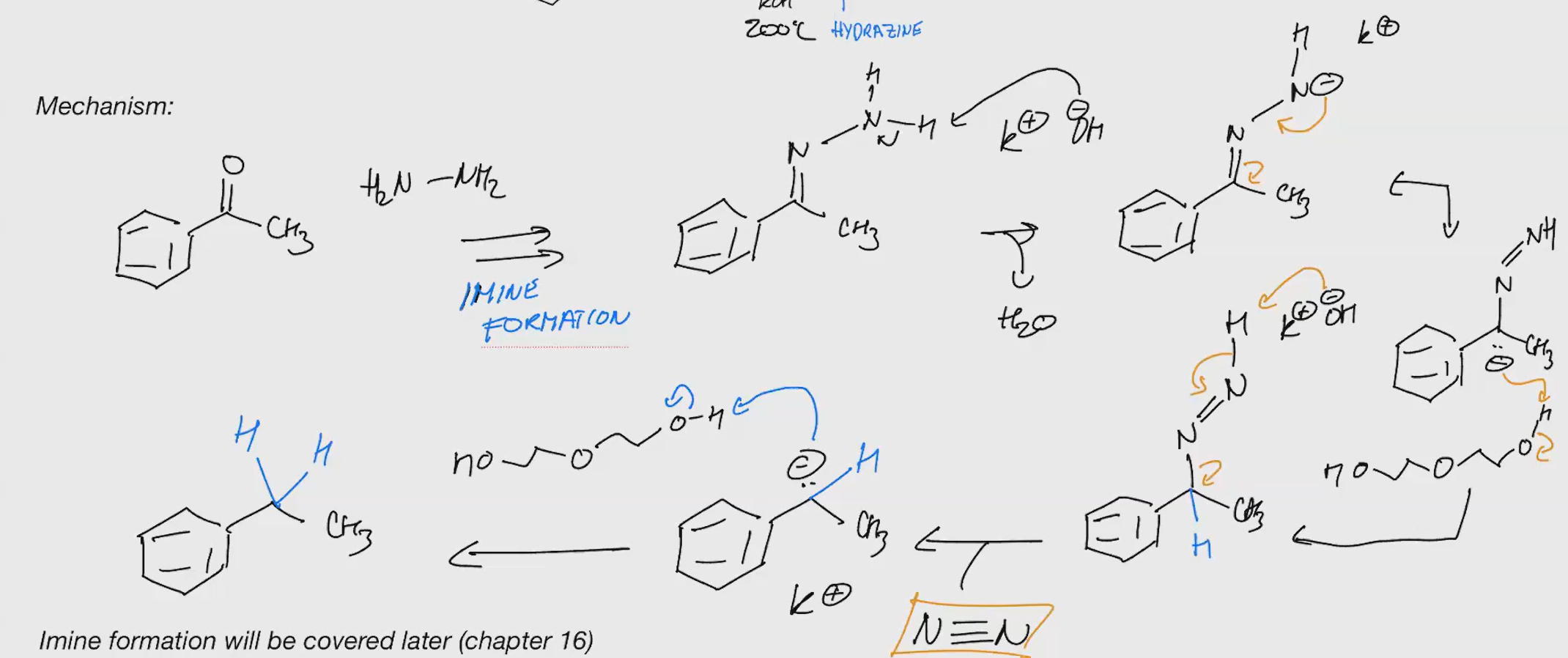
Wolff-Kishner reudction
Reduce a carbonyl group to a methylene
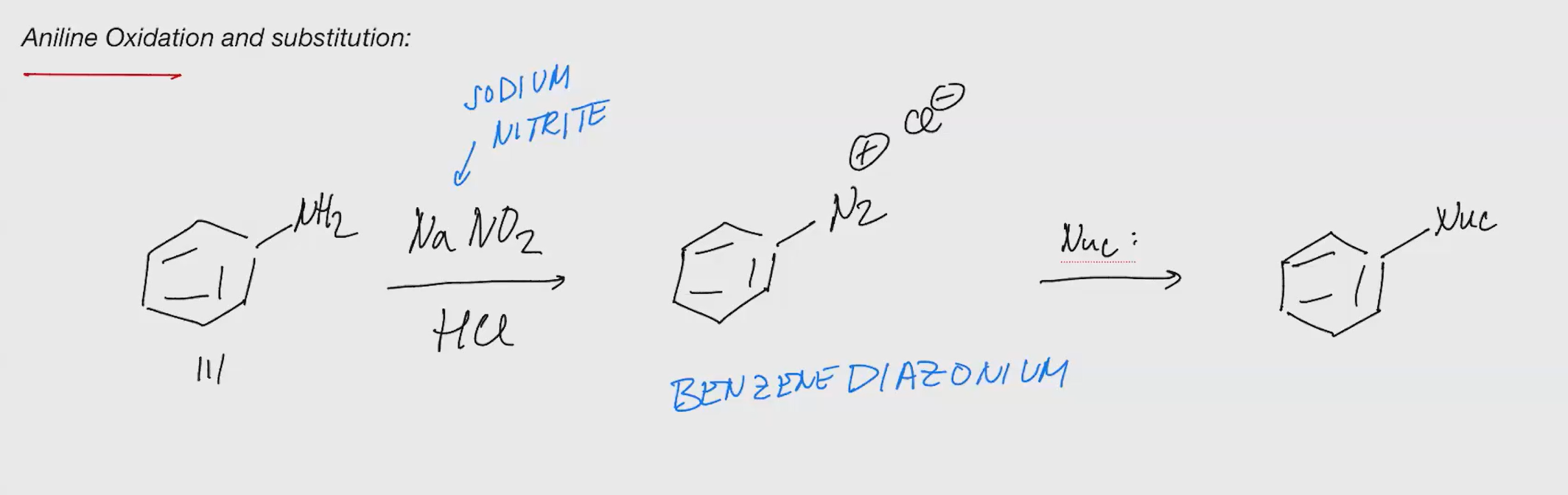
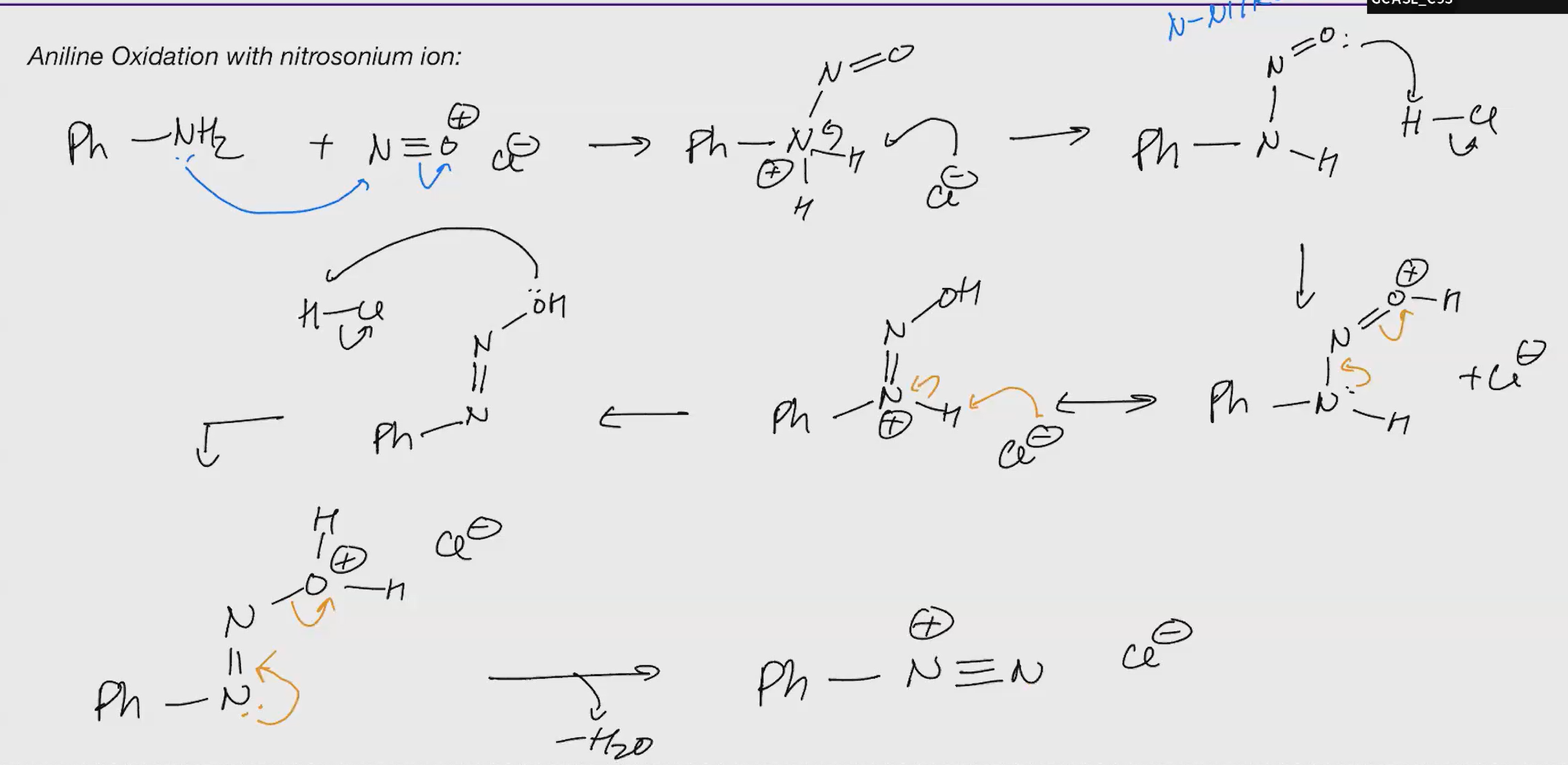
Aniline oxidation and susbstitution
NaNO2 Nuc
—> ——>
HCL
Nuc: Replaces the N2 After
CuCn
CuBr/CuCl
HBF4
H20
D3PO2
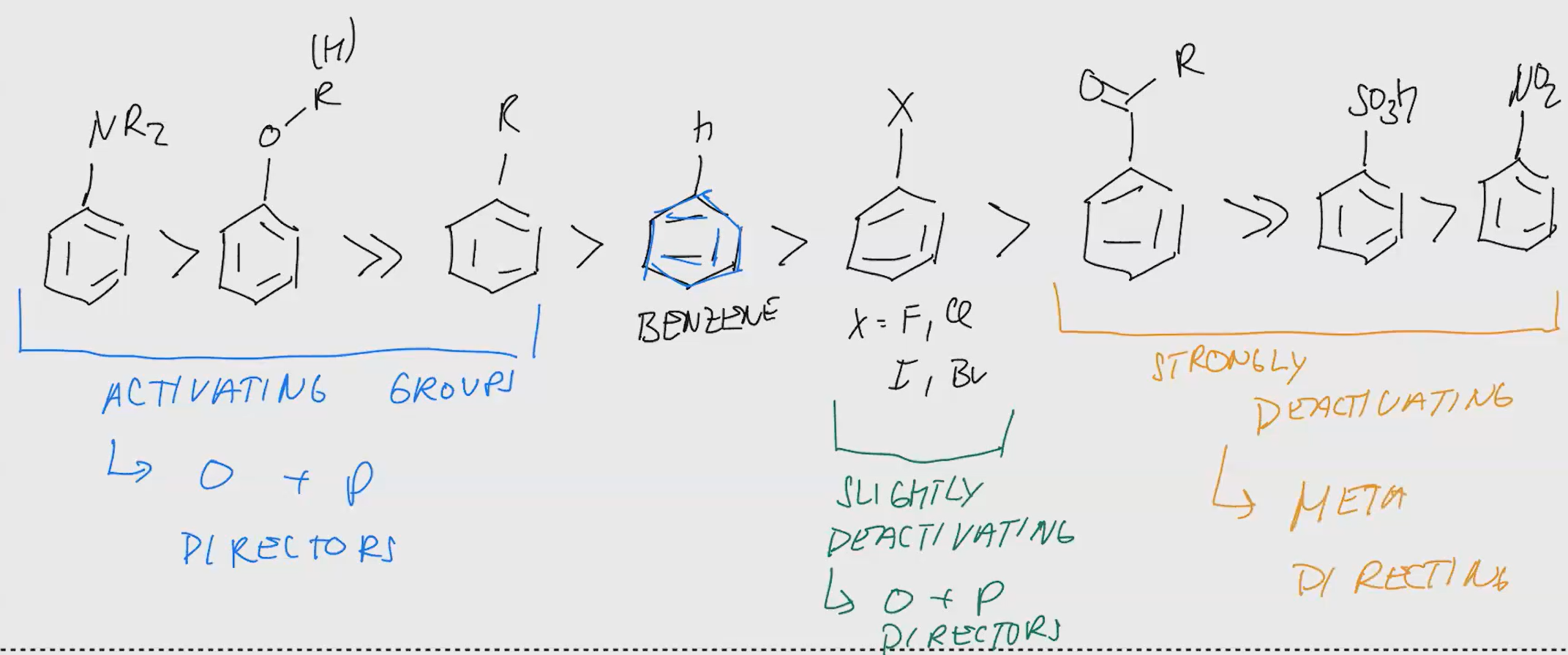
EAS Aromatic rules to know
Electron Withdrawing group - Deactivating - Meta Directing (Ortha and para have less electron density so they are less nucleophilic here)
Halogens - Slightly deactivating - Ortho & Para directing (they r electronegative and inductively withdraw electromagnetic density which makes aromatic system less nucleophilic
Sterics are important if electronic effects are similar & IF there are multiple substituents the more activating one controls the directions. (or more donating etc if they da same)
Nitro group (No2) prevents freideiyl crafts.
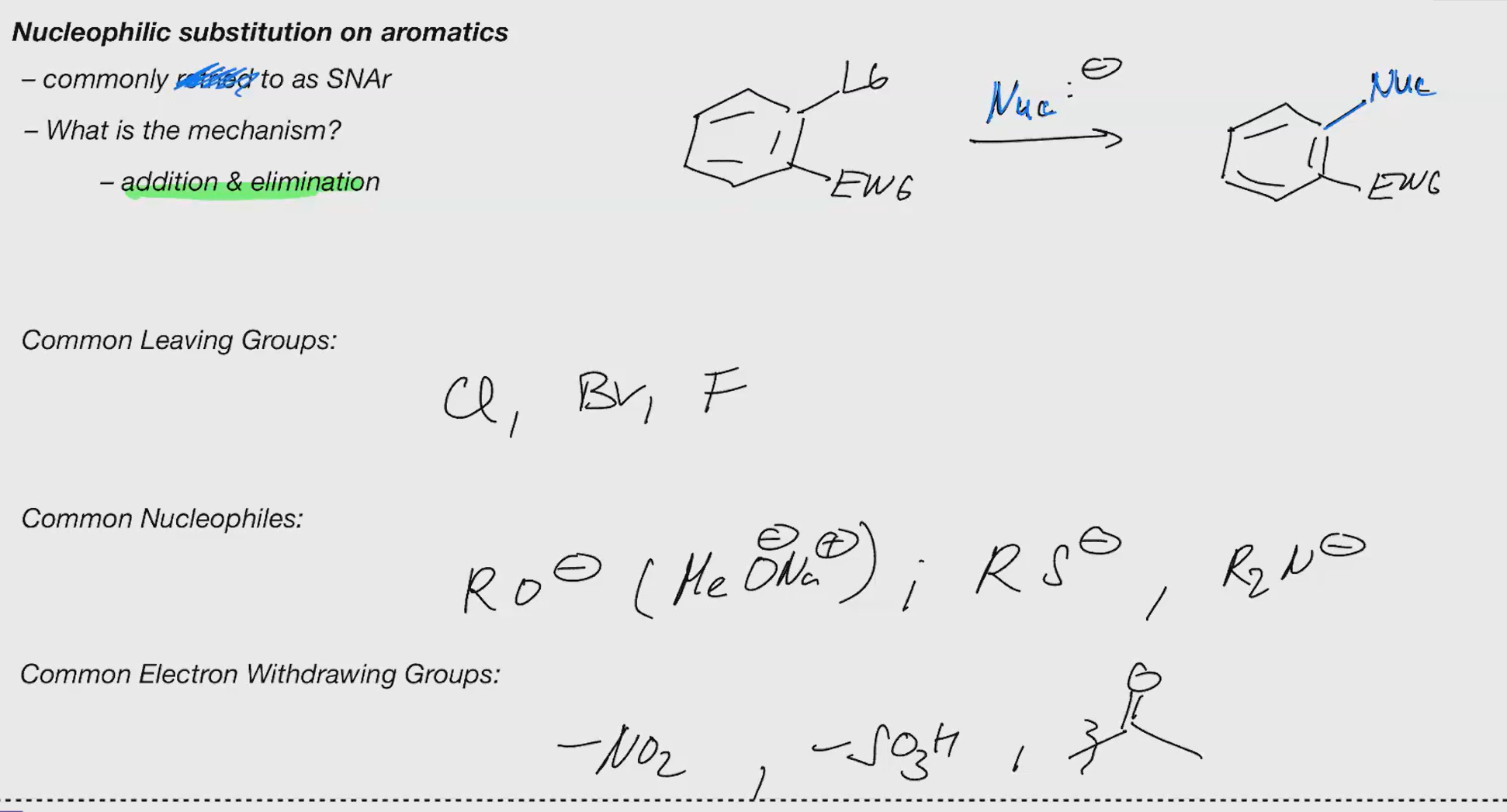
Nucleophilic aromatic substitution (SNAr)
Leaving Groups: Cl, Br, F
anion can be resonance stabilized 3 times
without EWG snar cant happen
Pyridine can bypass EWG CUZ OF THE N STABLIZING NEG CHARGE
(Heteroaromatics or EWGs for snar)
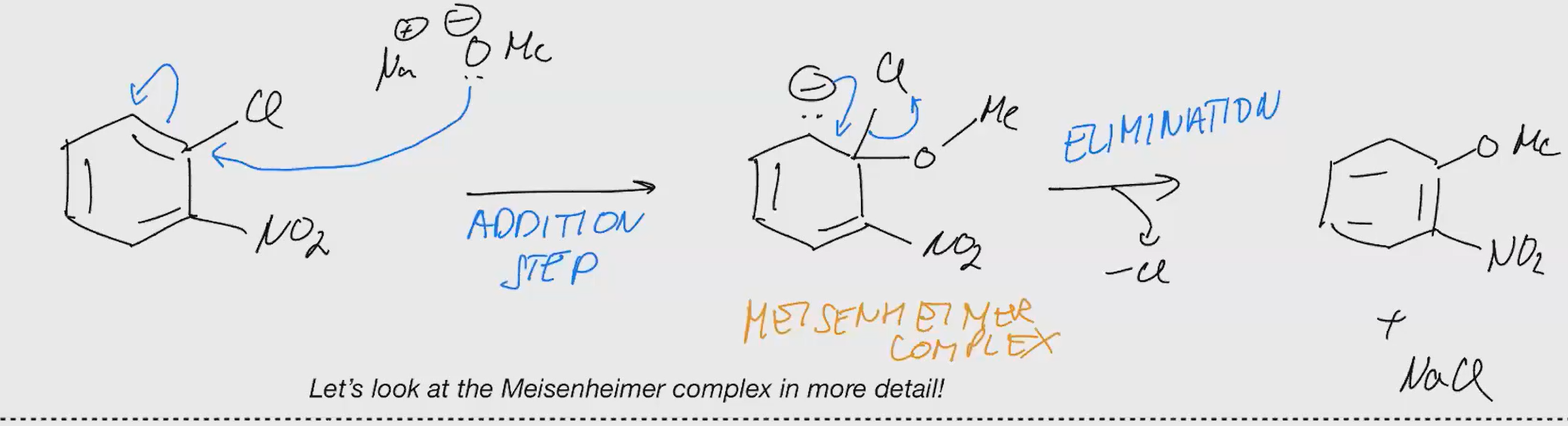
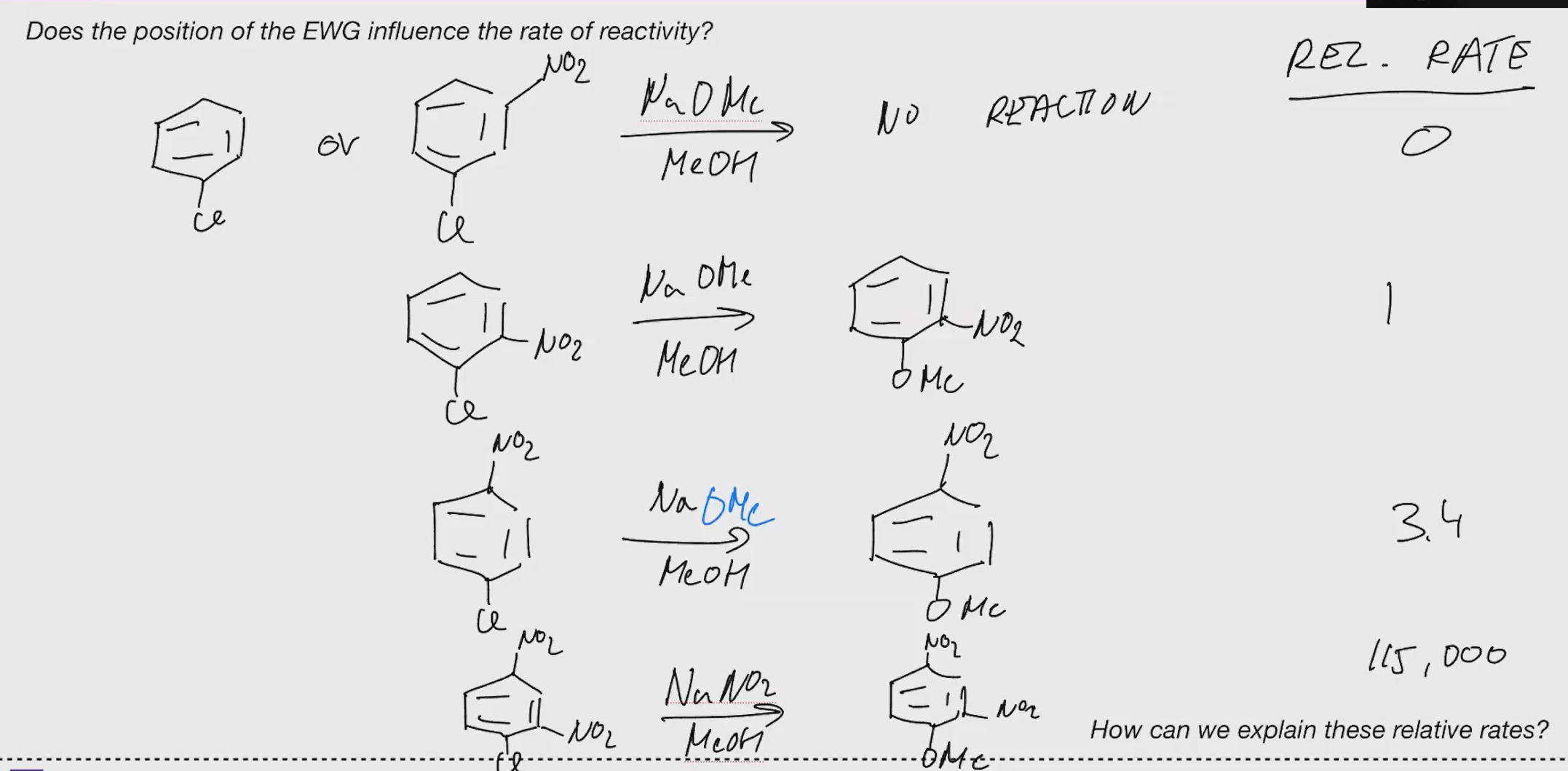
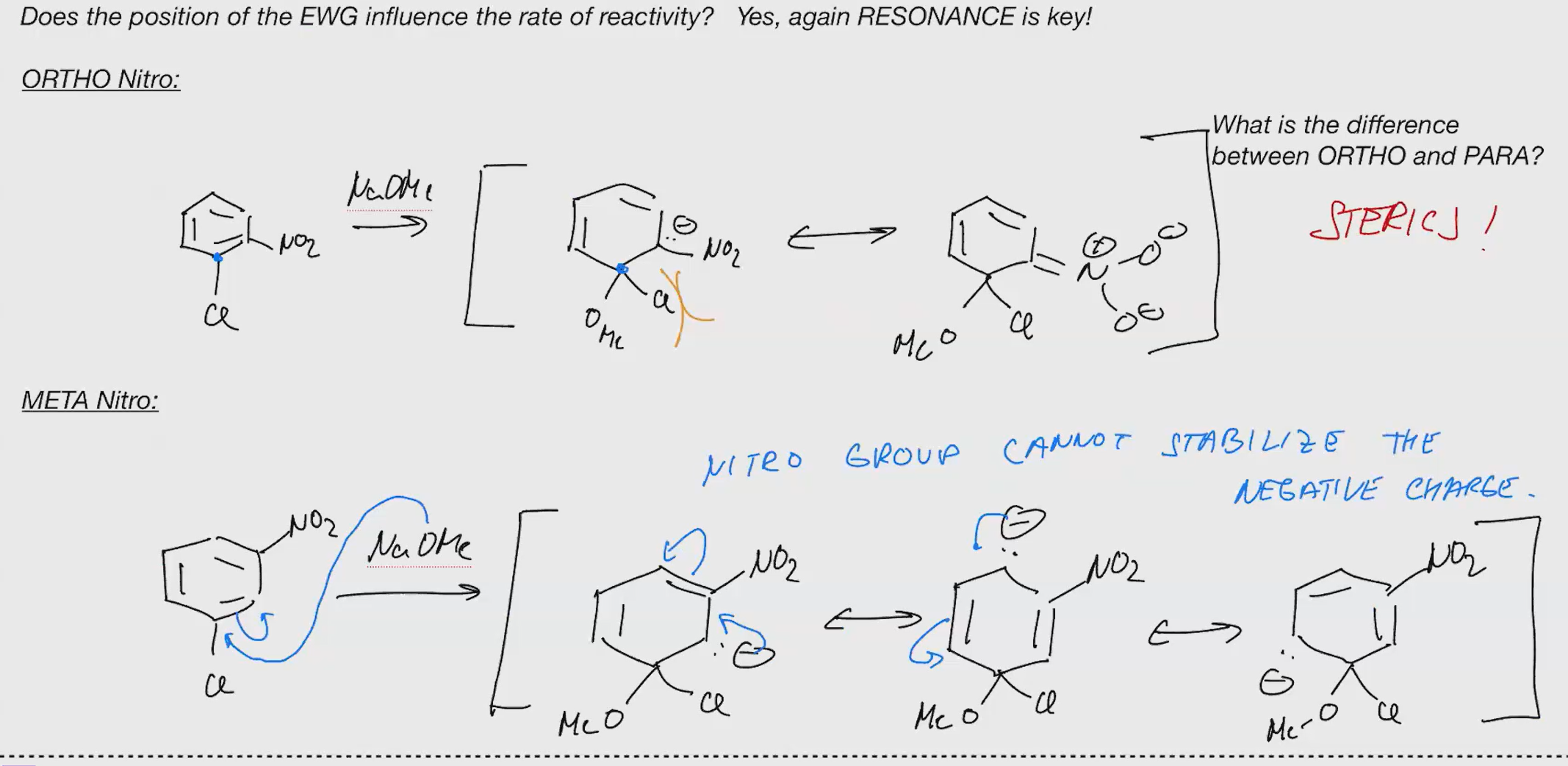
SNAR Rates
Explain
Electronics of heteroaromatocs
If the electron pair is part of aromatic it’s electron rich if not it’s electron poor
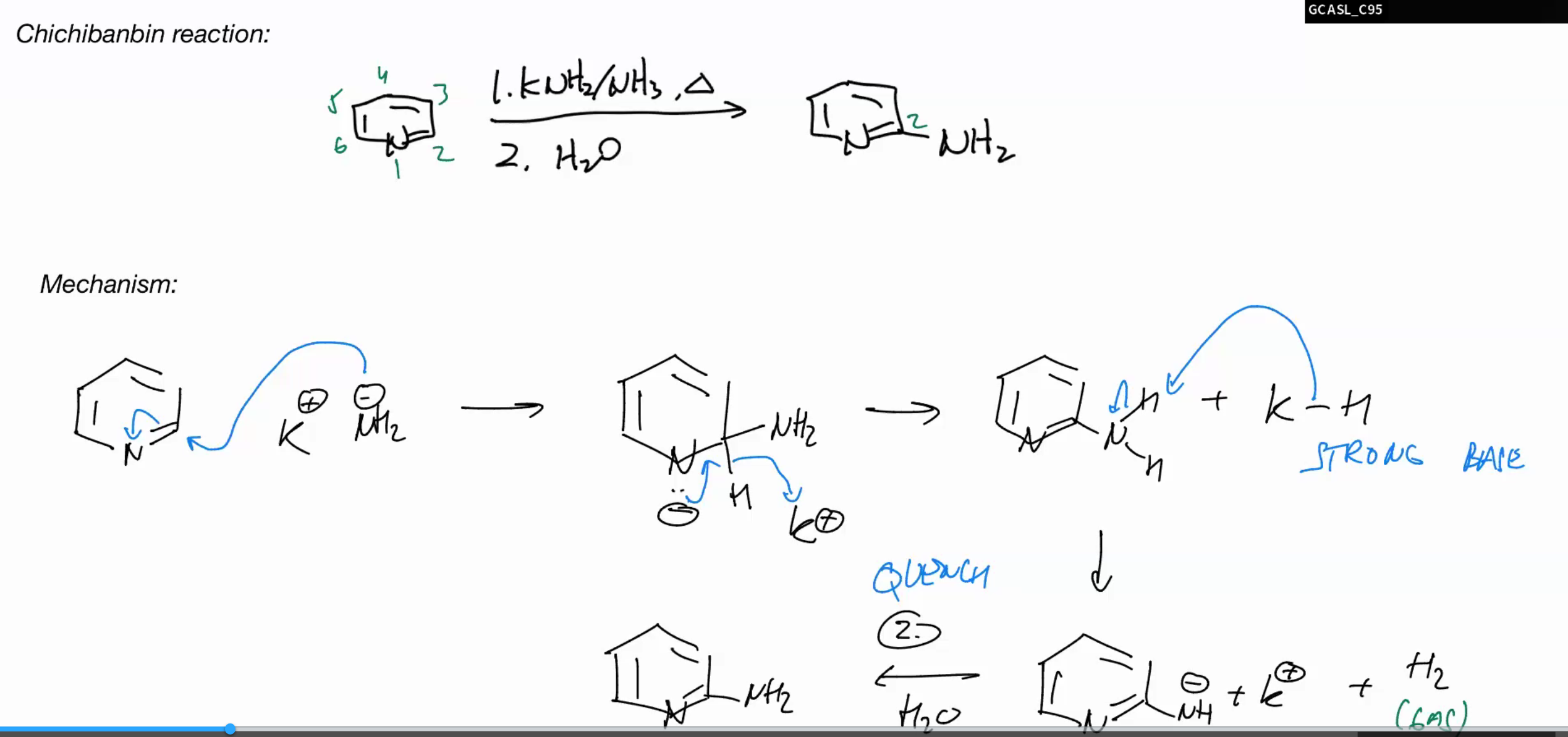
Chichibanbin reaction
KNH2/NH3
H2O
SINCE ITS A PYRIDINE AND ELECTRON OOR IT IS ACTIAVTED ENOUGH IN SNAR WHERE IT DOESNT NEED AN OFFICAL LEAVING GROUP
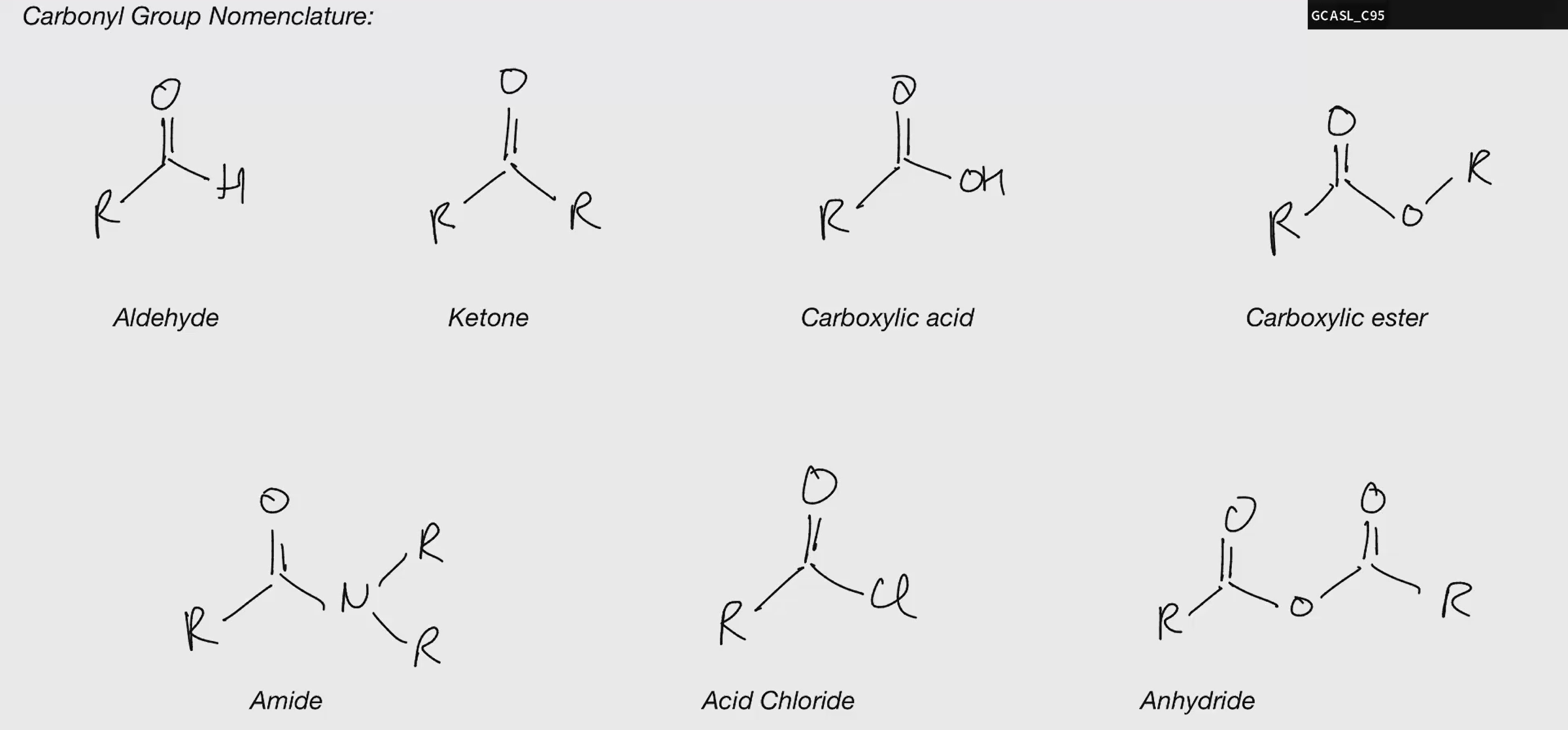
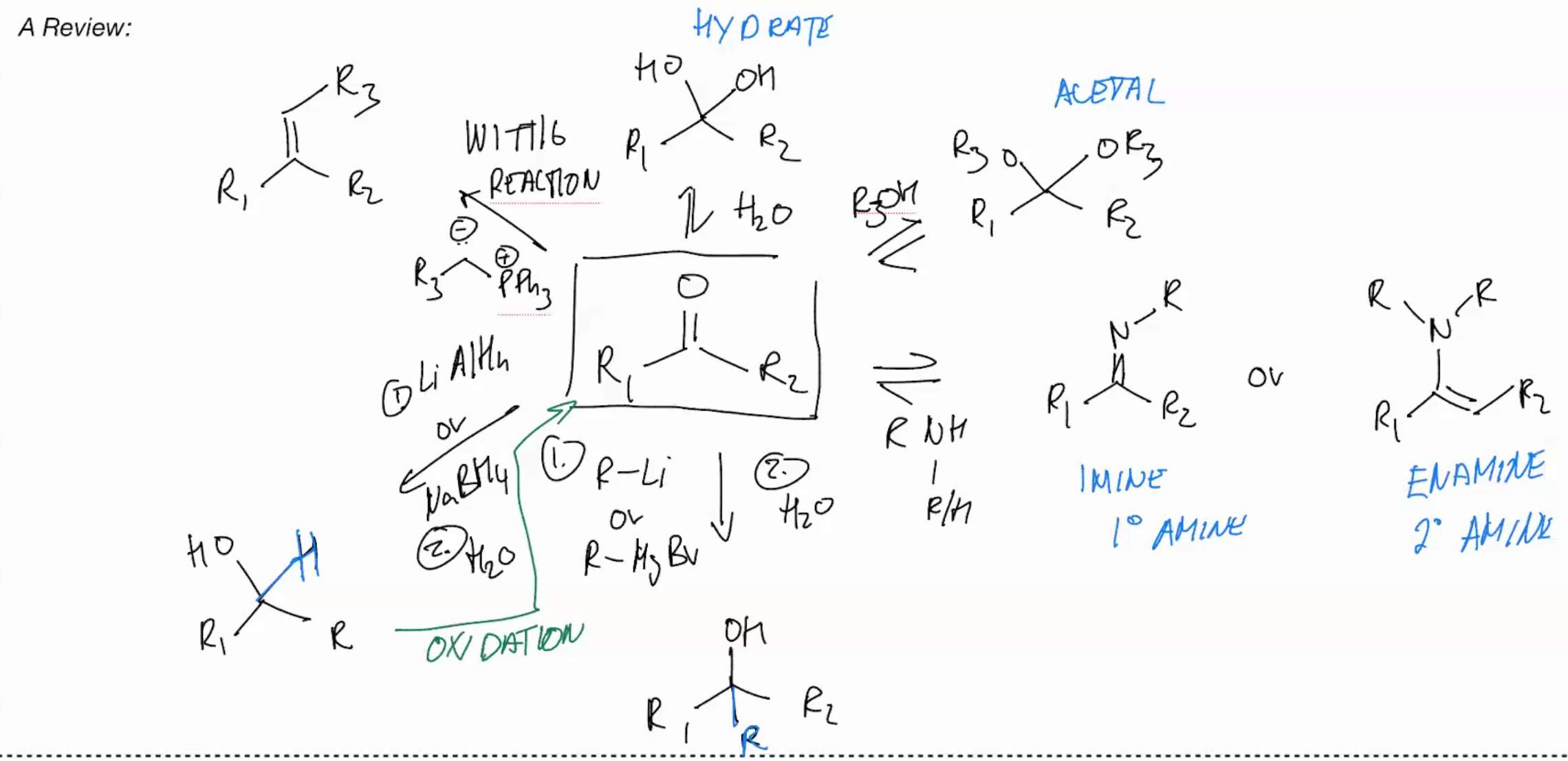
Carbonyl Nomenclature
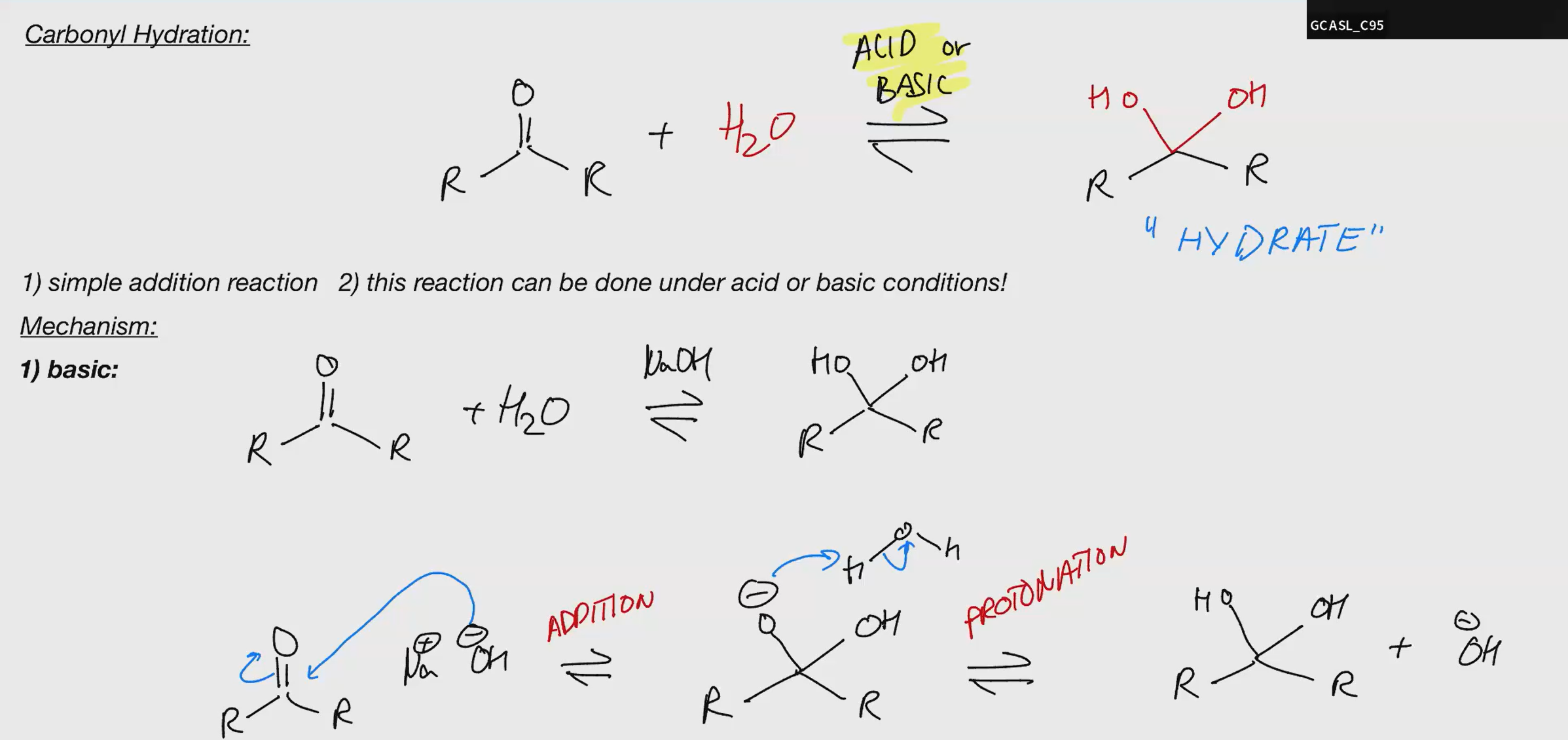
Carbonyl Hydration BASIC
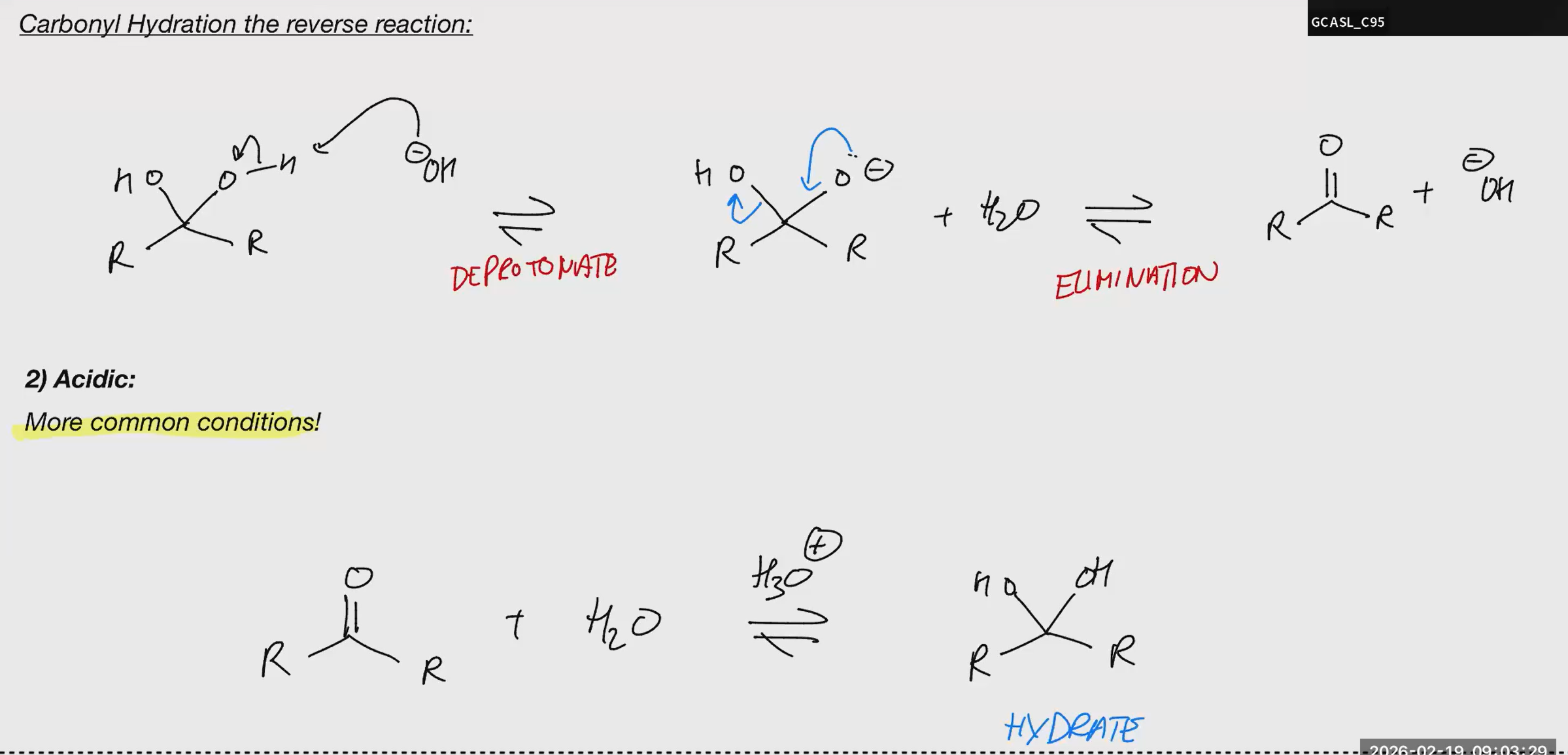
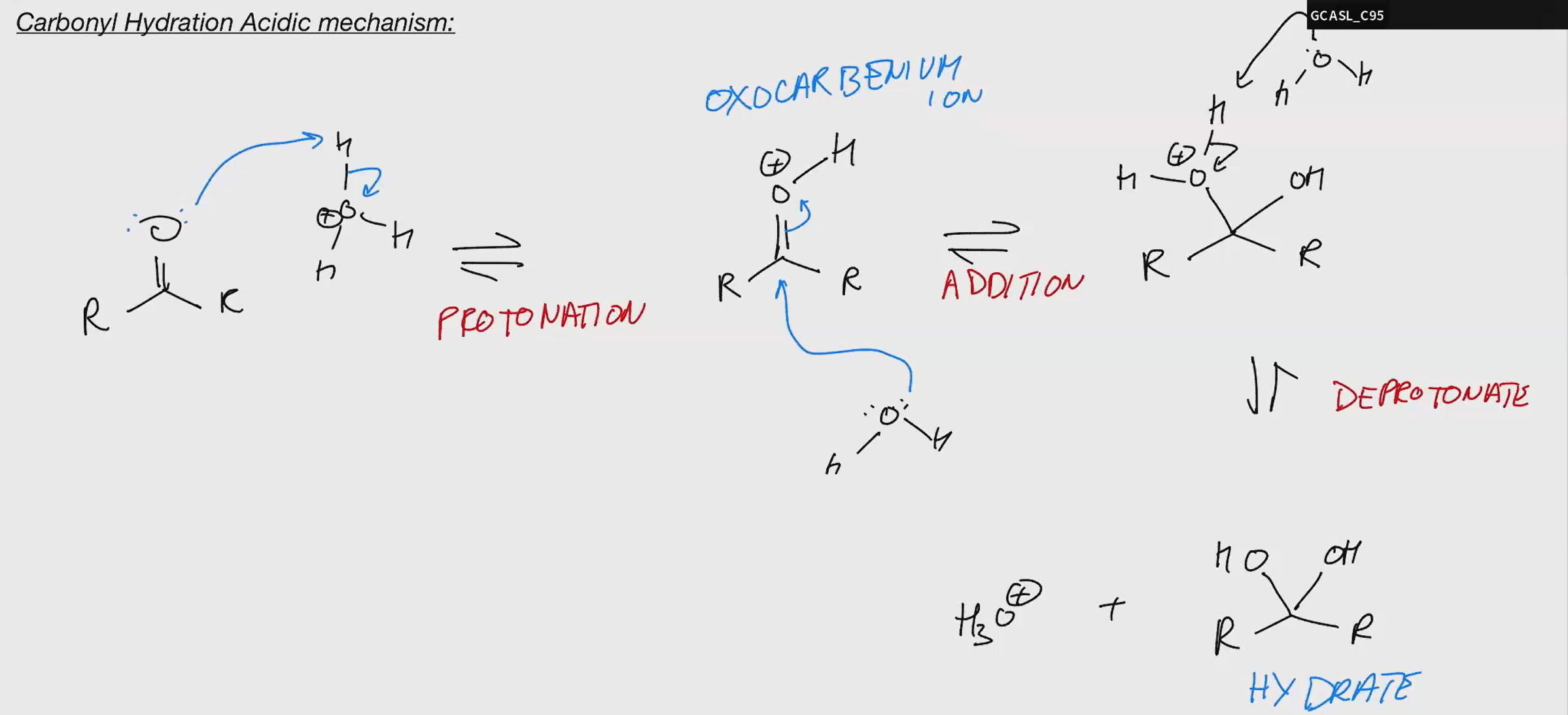
Carbonyl Hydration ACIDIC
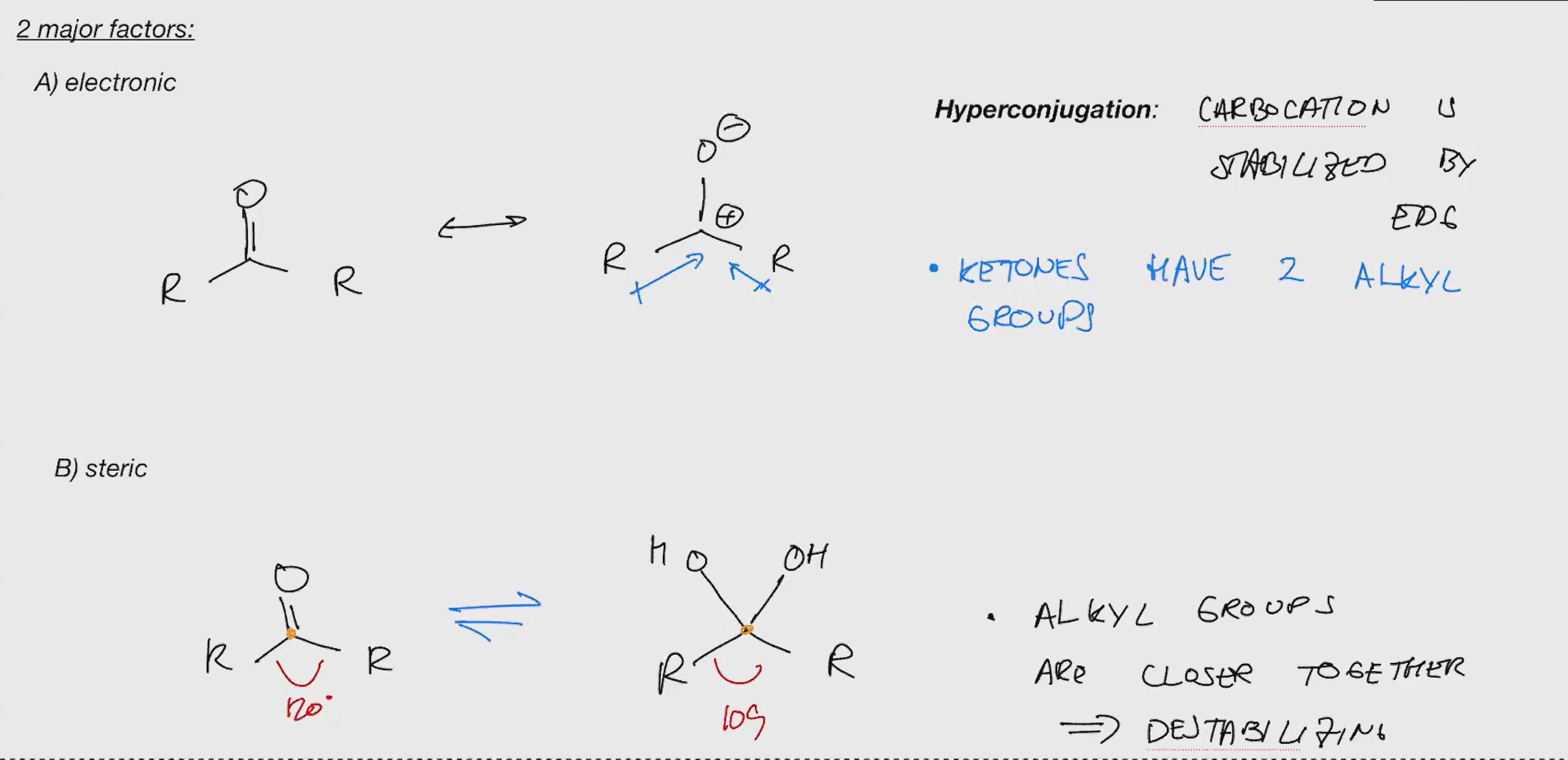
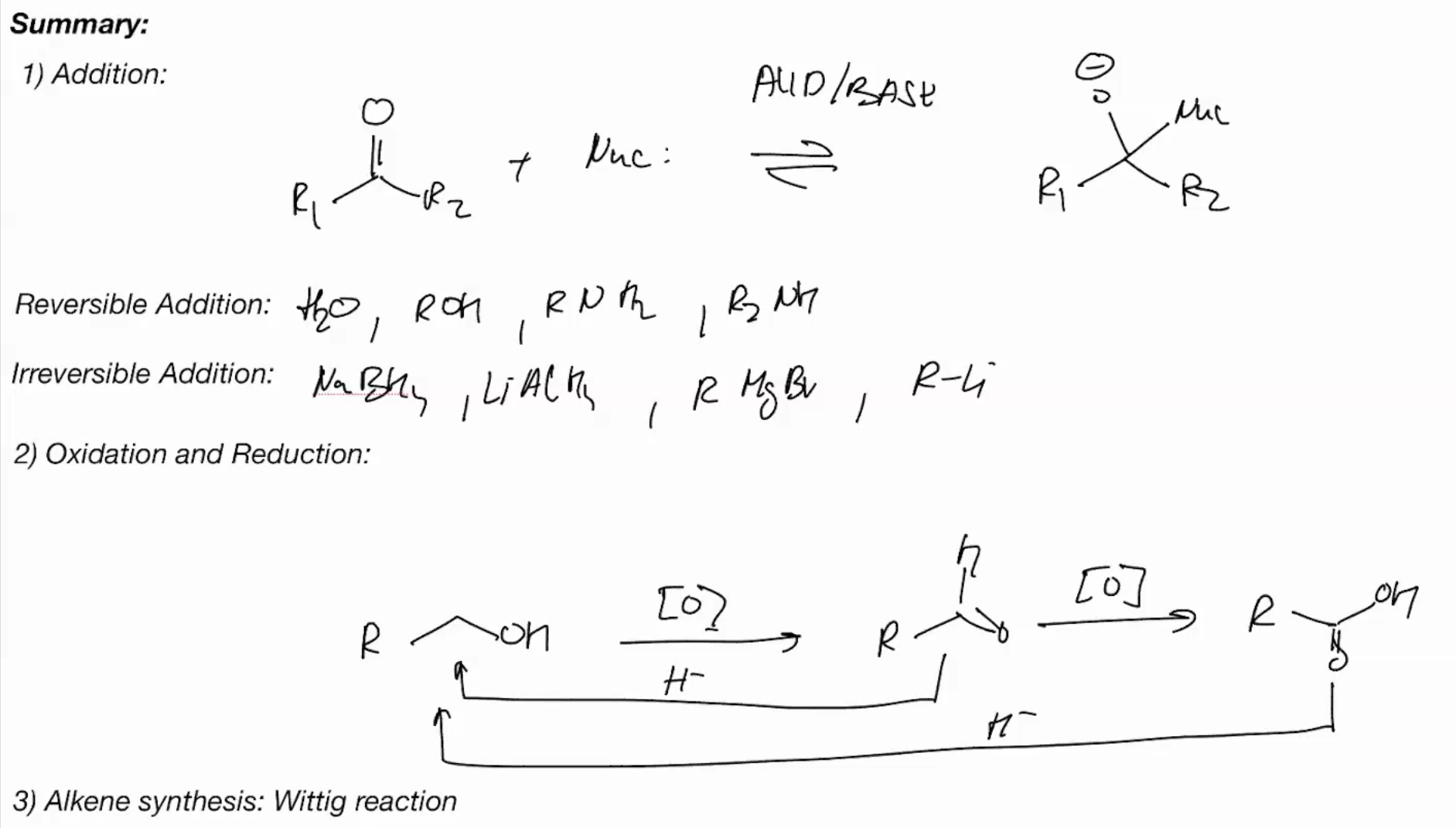
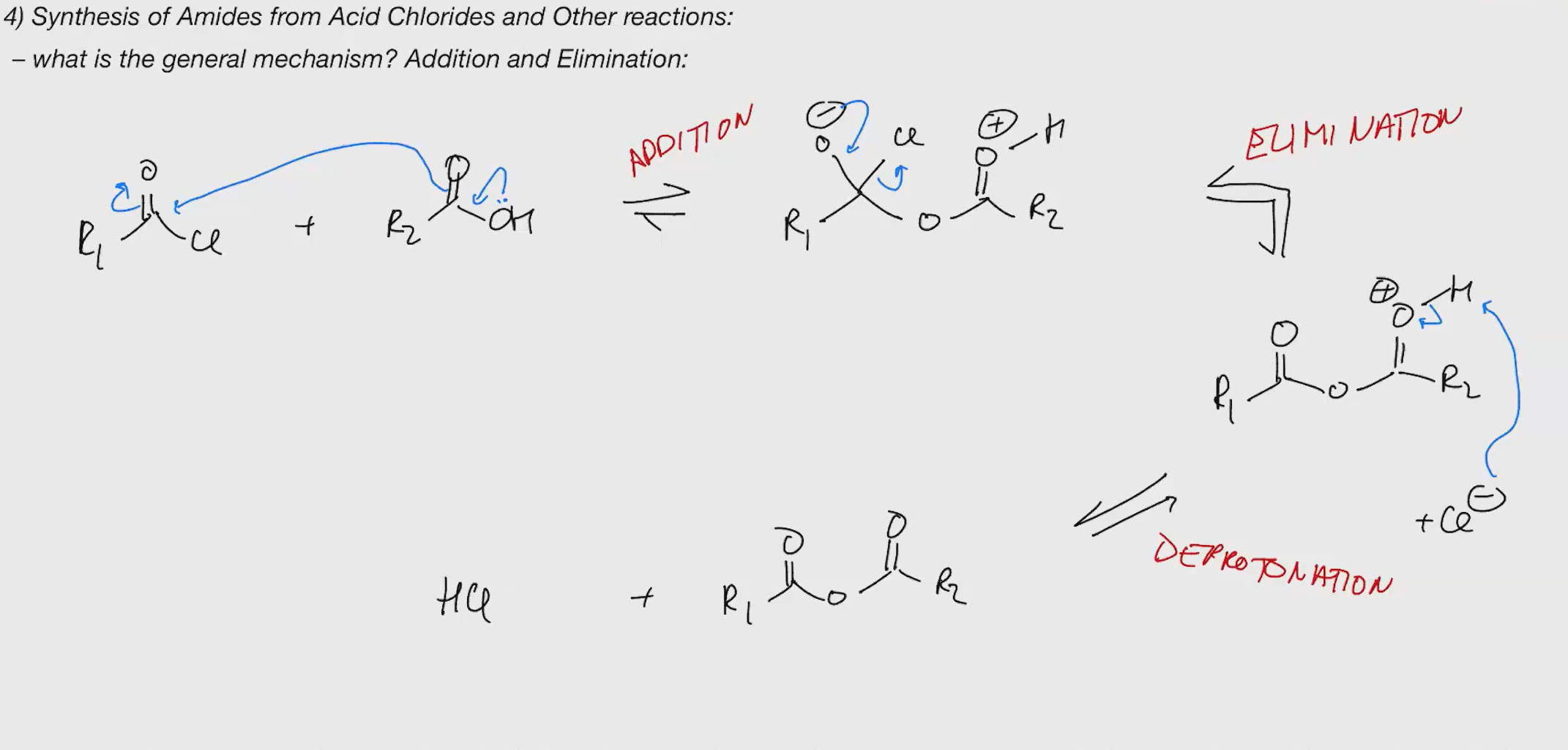
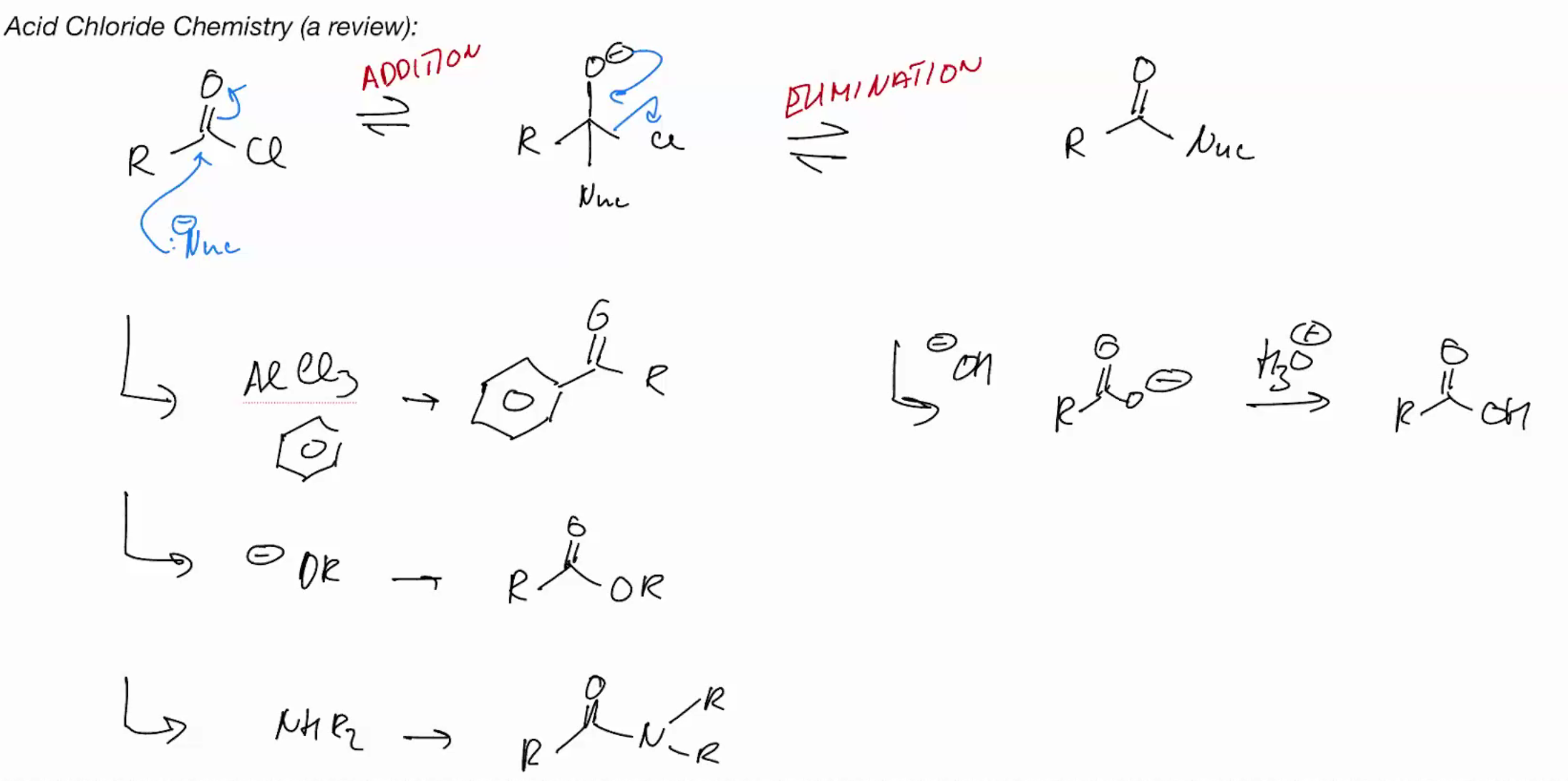
key reactions are addition and elimination
nucleophiles can be added directly or the addition can be accelerated by protonation first
Generally strong nucleophiles like OH directly, but weak ones like water need protonation
oxocarbenium are much more electronegative/reaction that regular carbonyls
no strong bases and acids
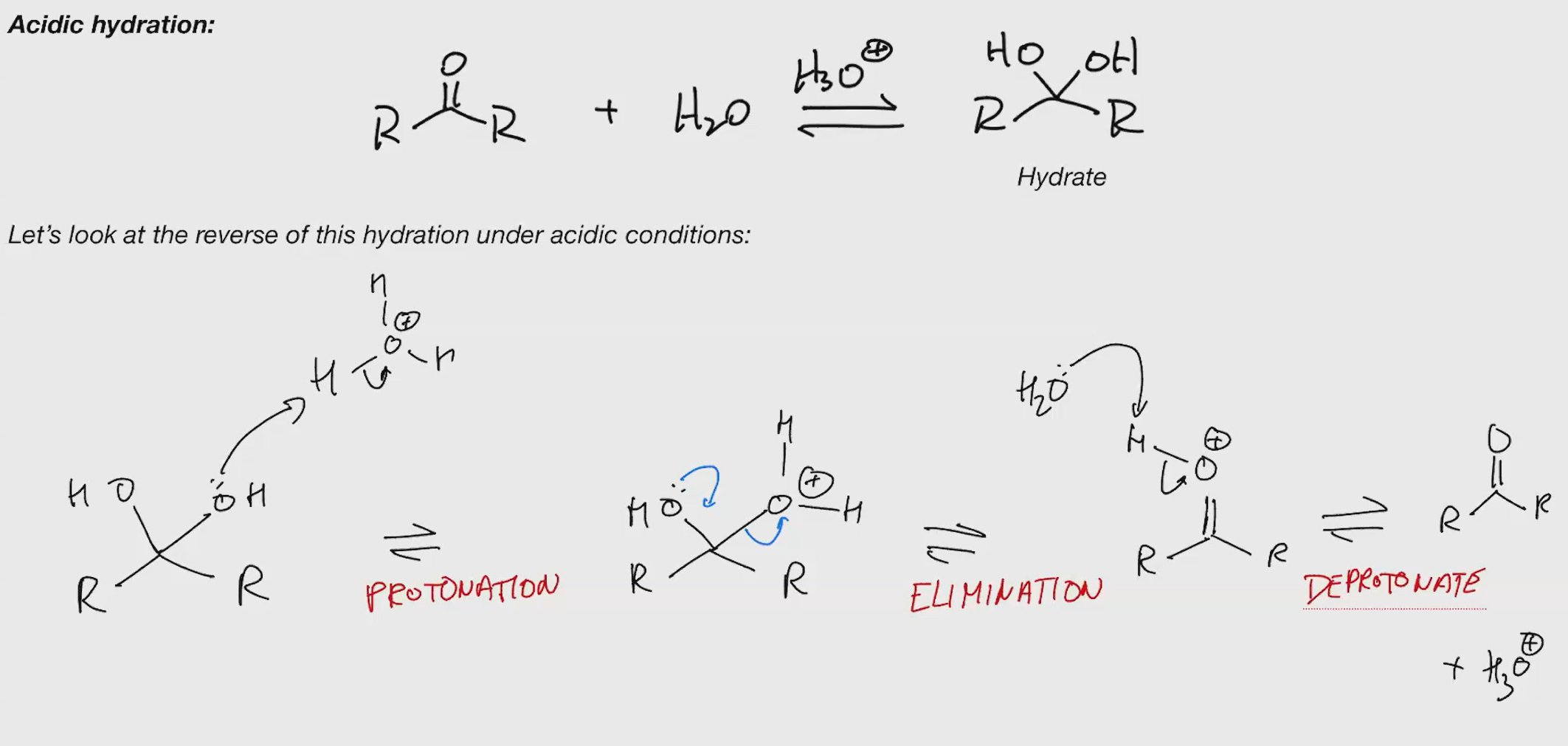
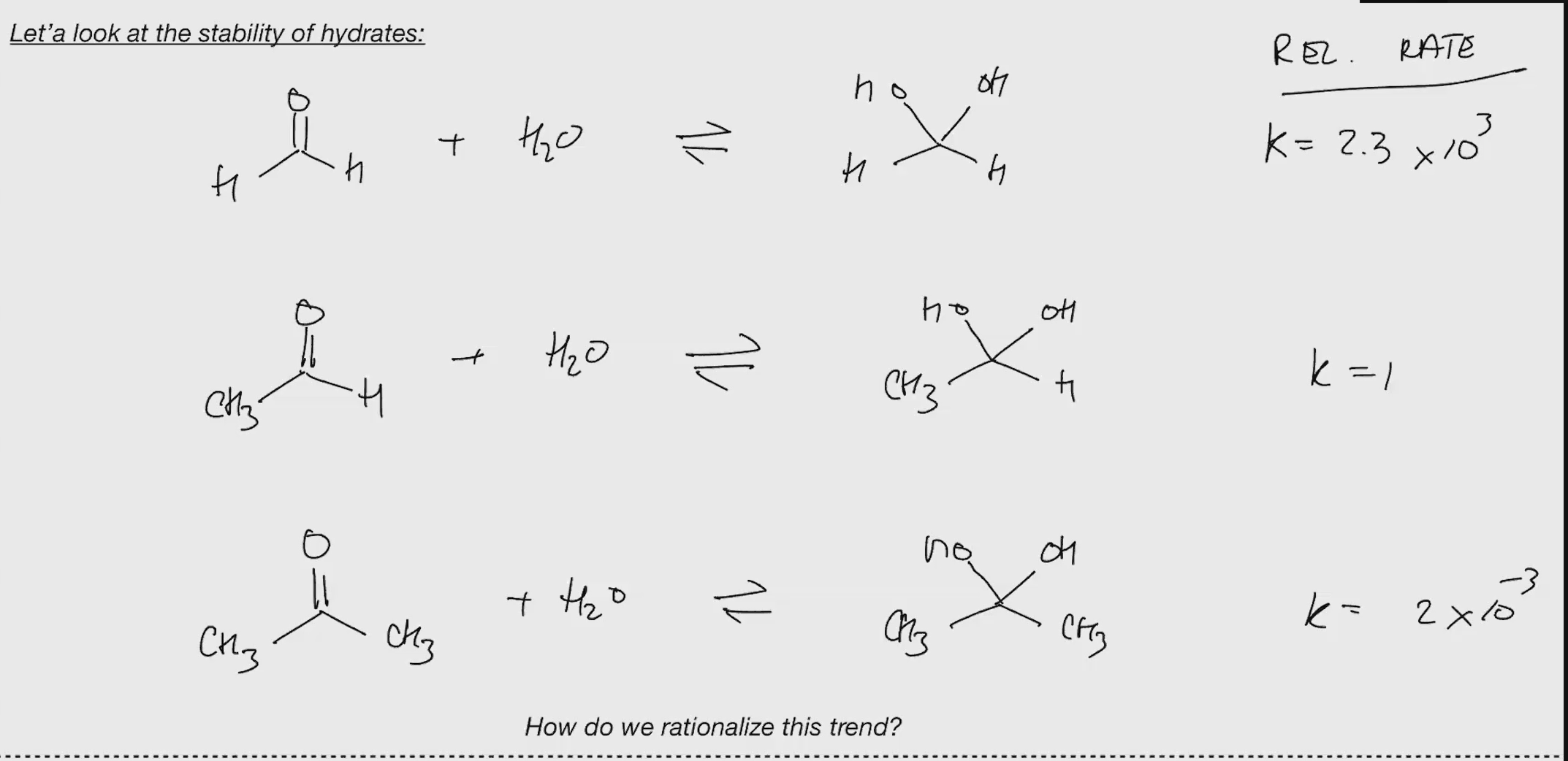
Stability of hydrates
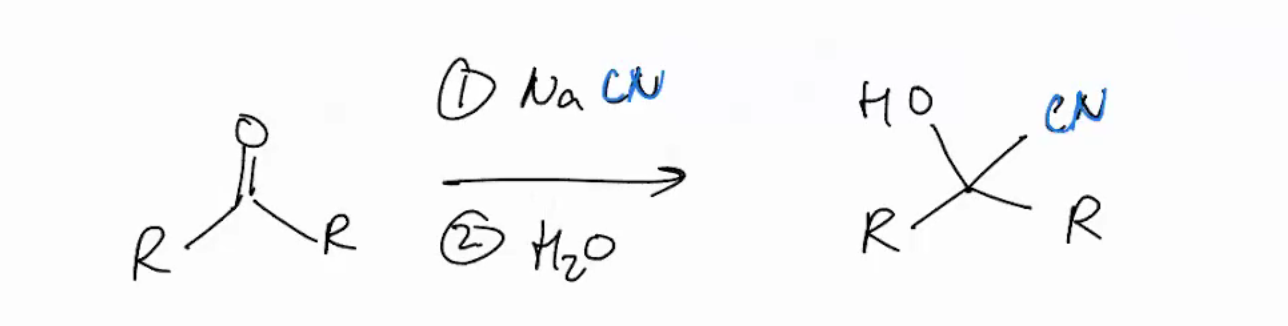
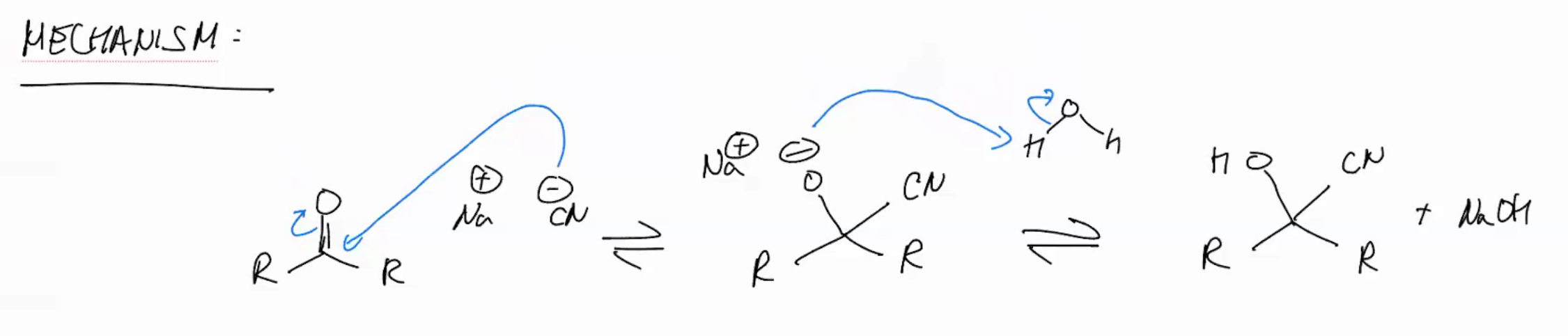
Cyanohydrin synthesis
NaCN
H2O
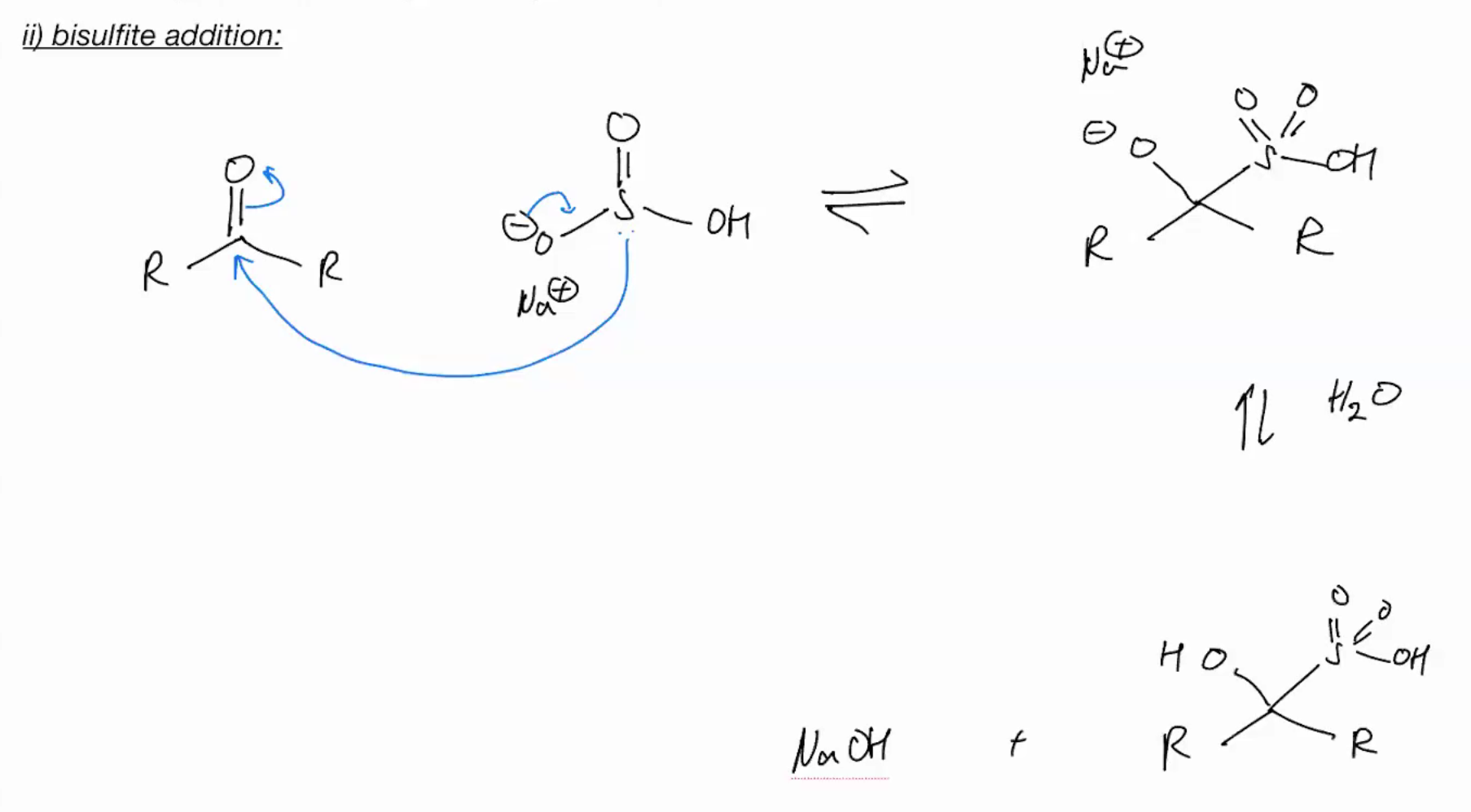
Bisulfite Addition
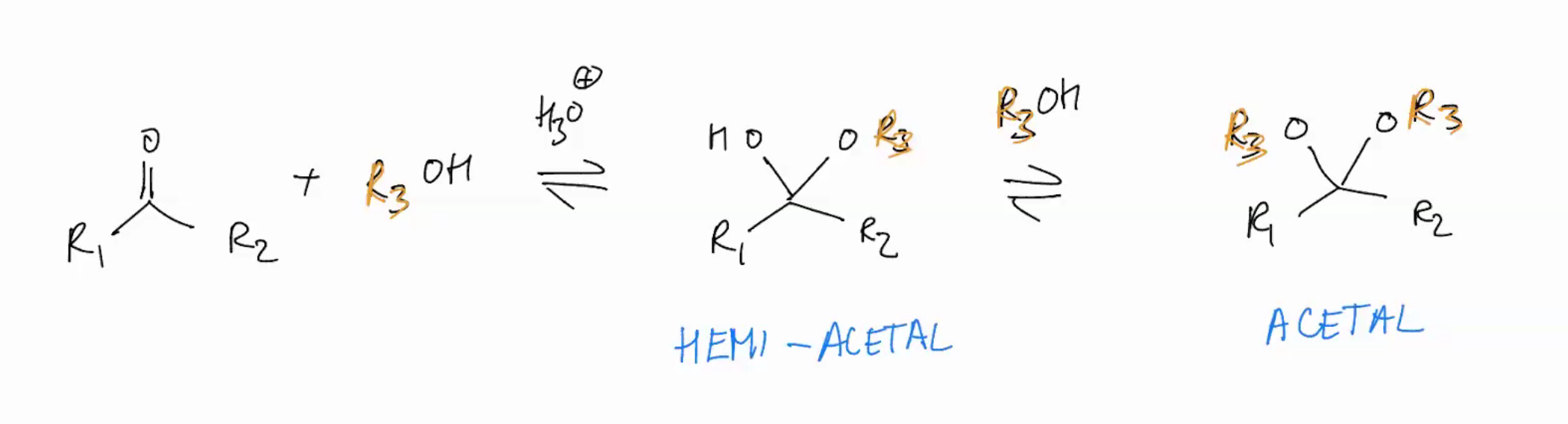
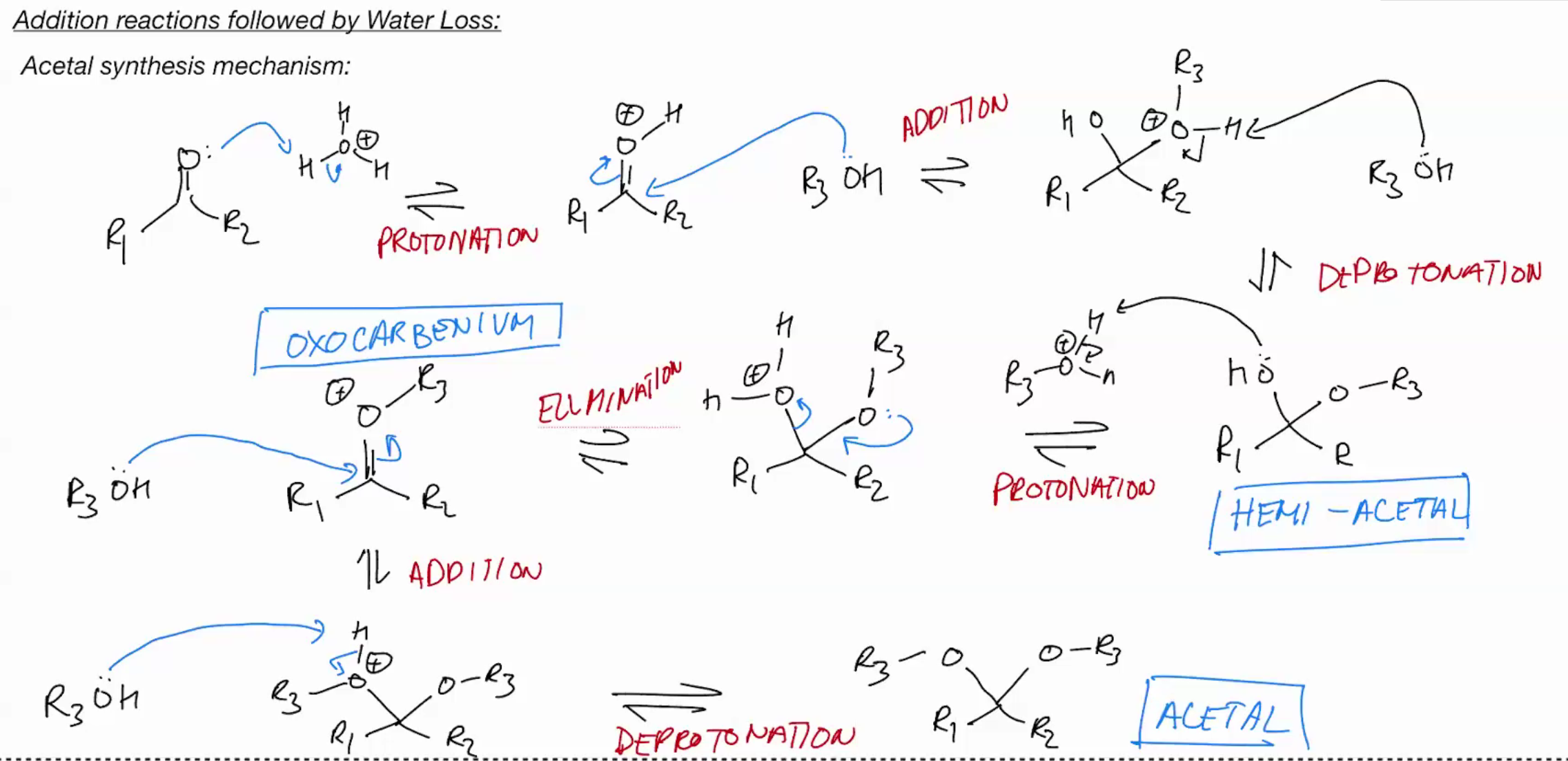
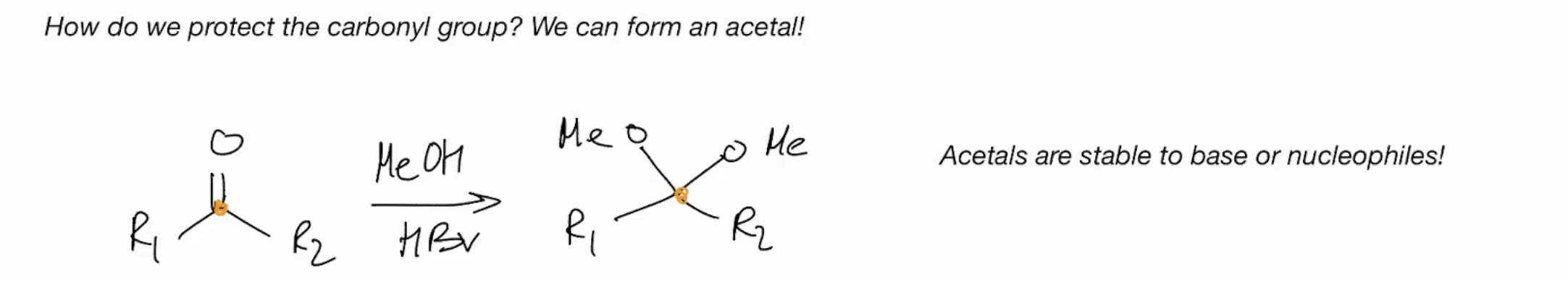
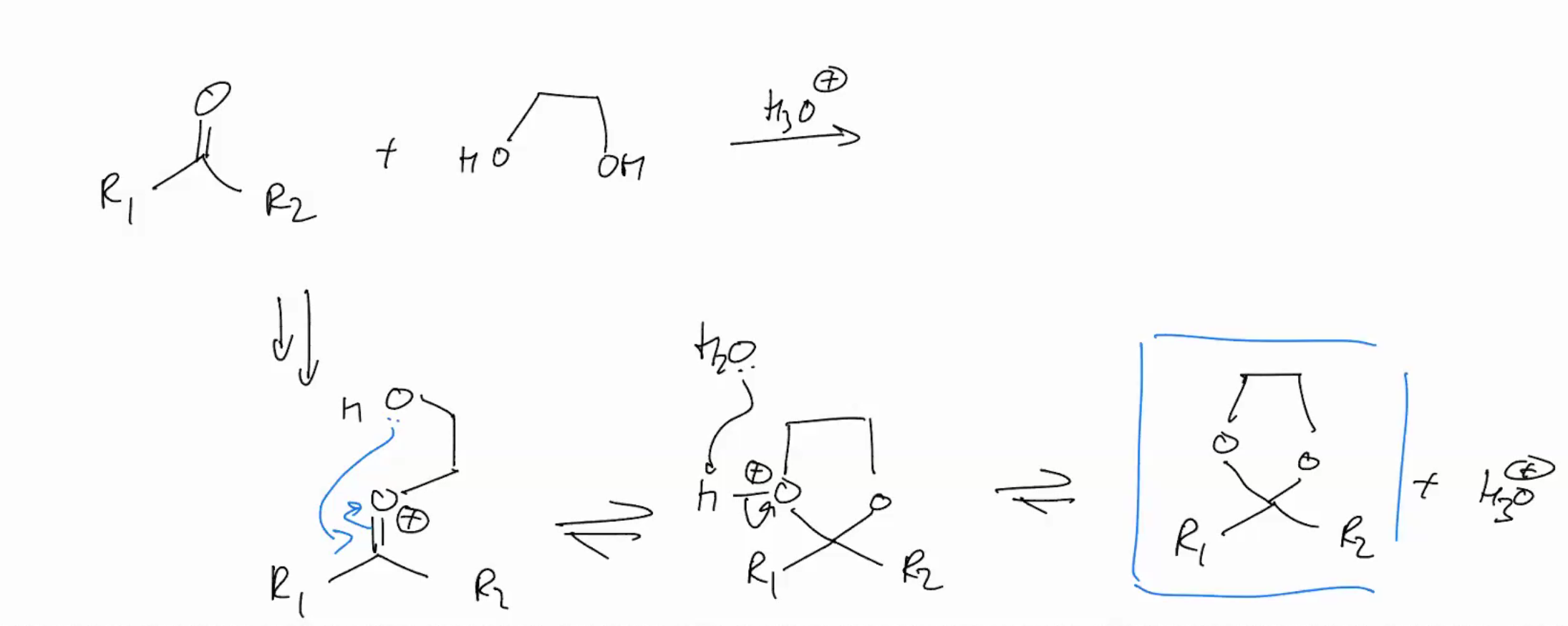
Synthesis of an ACETAL (ADDITION REACTIONS FOLLOWED BY WATER LOSS)
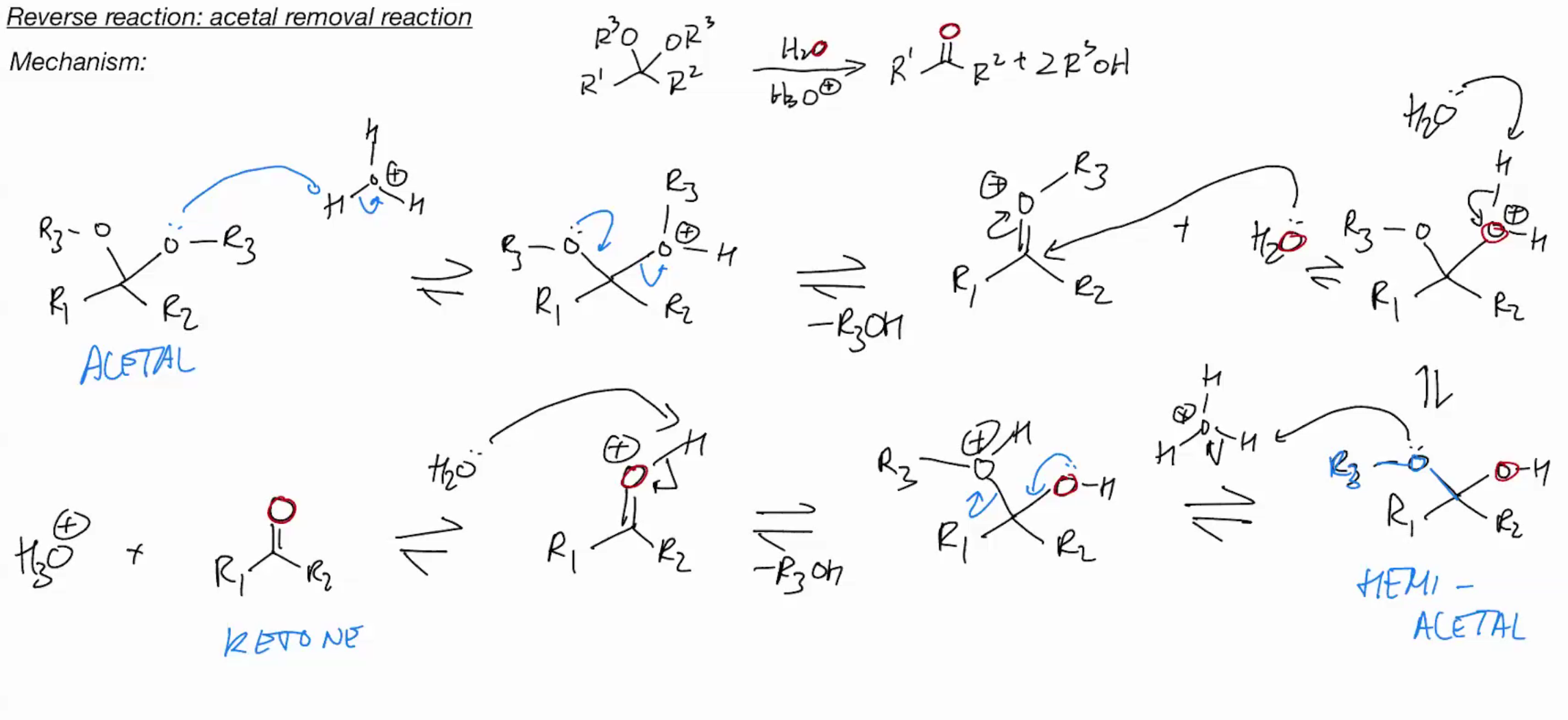
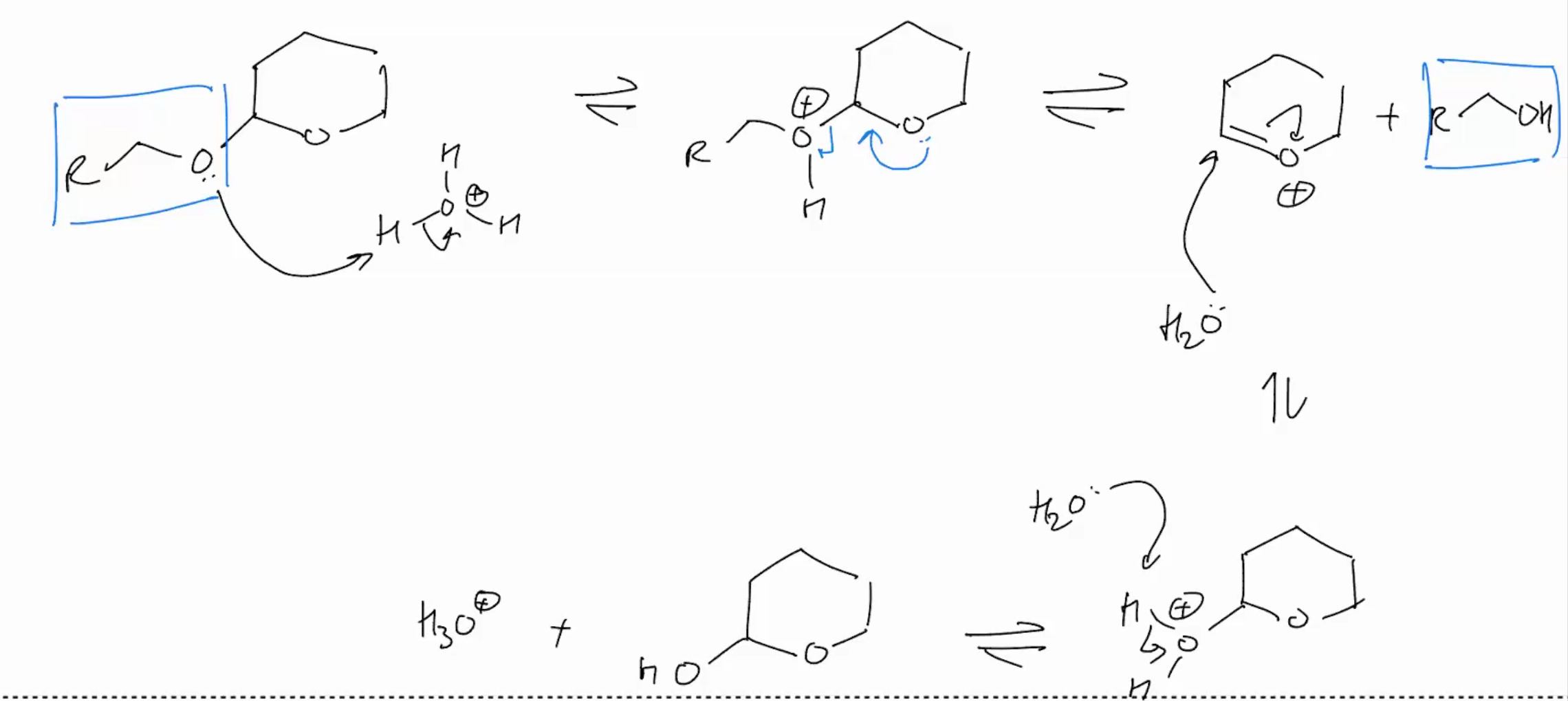
REVERSE Reaction: Acetal removal reaction
H2o
h3O
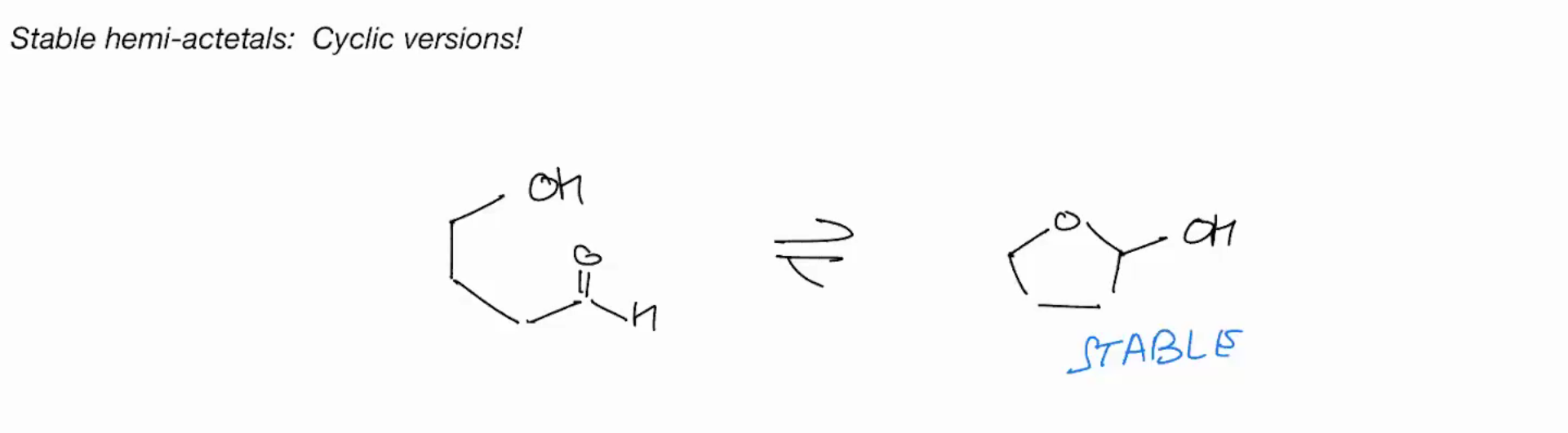
Stable hemi-acetals (CYCLIC VERSION!)
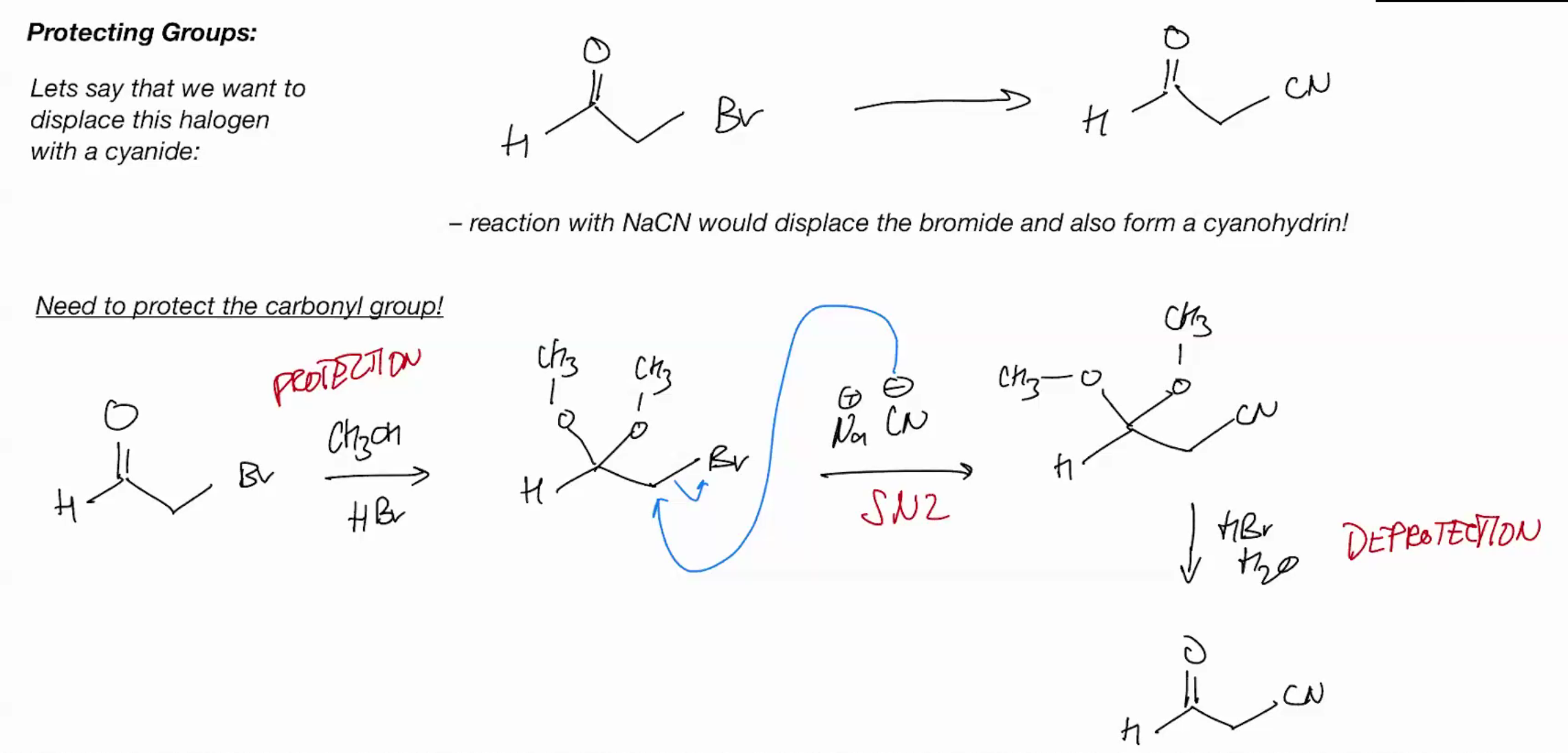
Why do i care about this… protection groups? for synthesis?
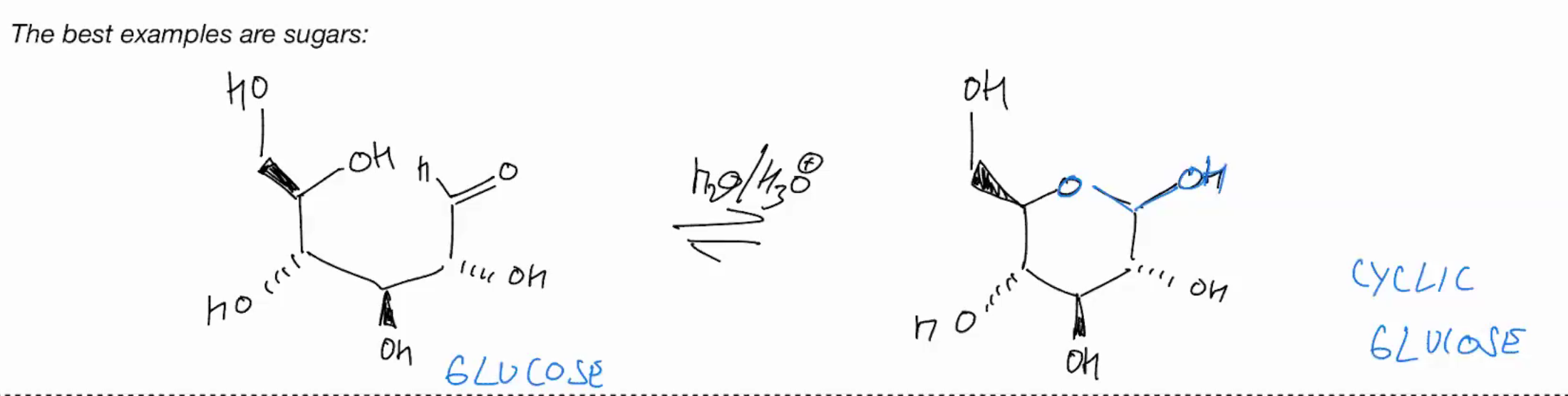
Protection Group (Protecting Carbonyl group by forming acetal)
Acetals are stable to base or nucleophiles.
Protection groups : Acetal
Formation of cyclic acetals is very favorable. Why the second step becomes intramolecular (cuz intra is hella faster)!
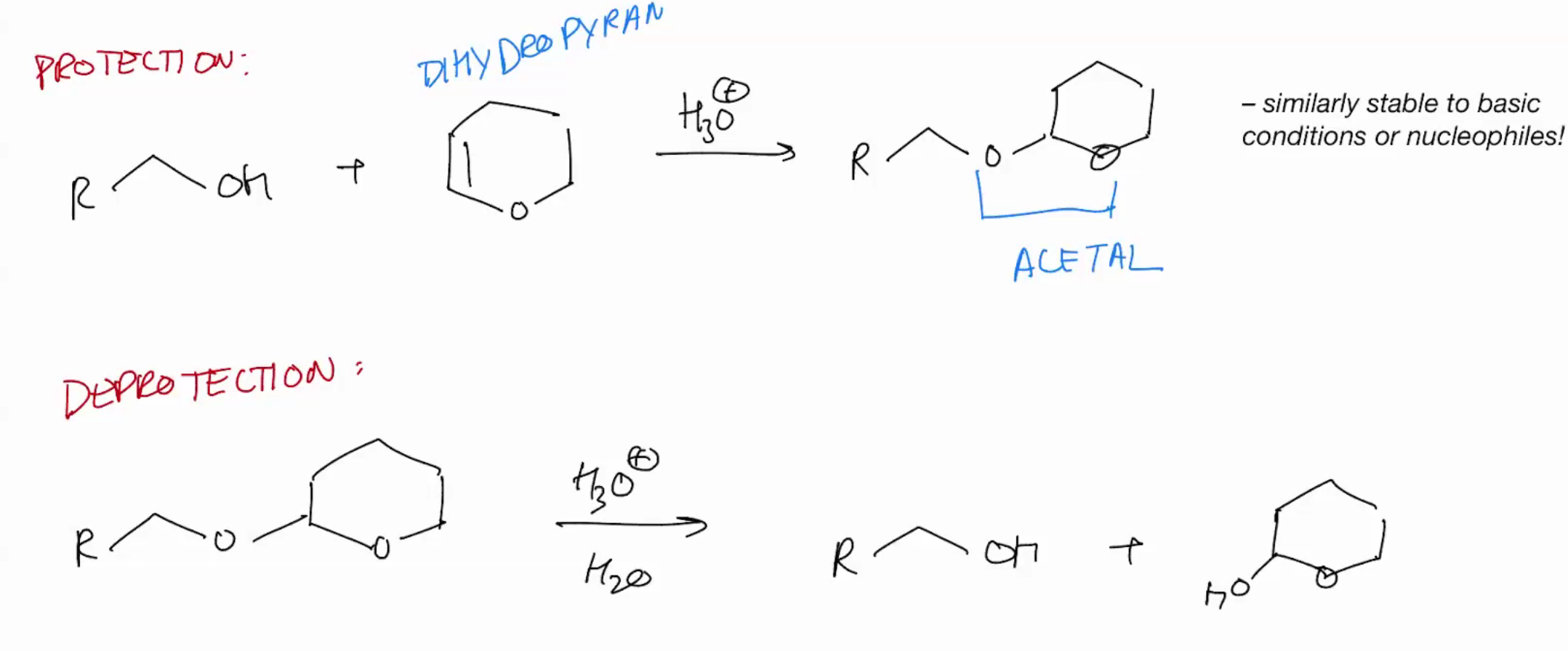
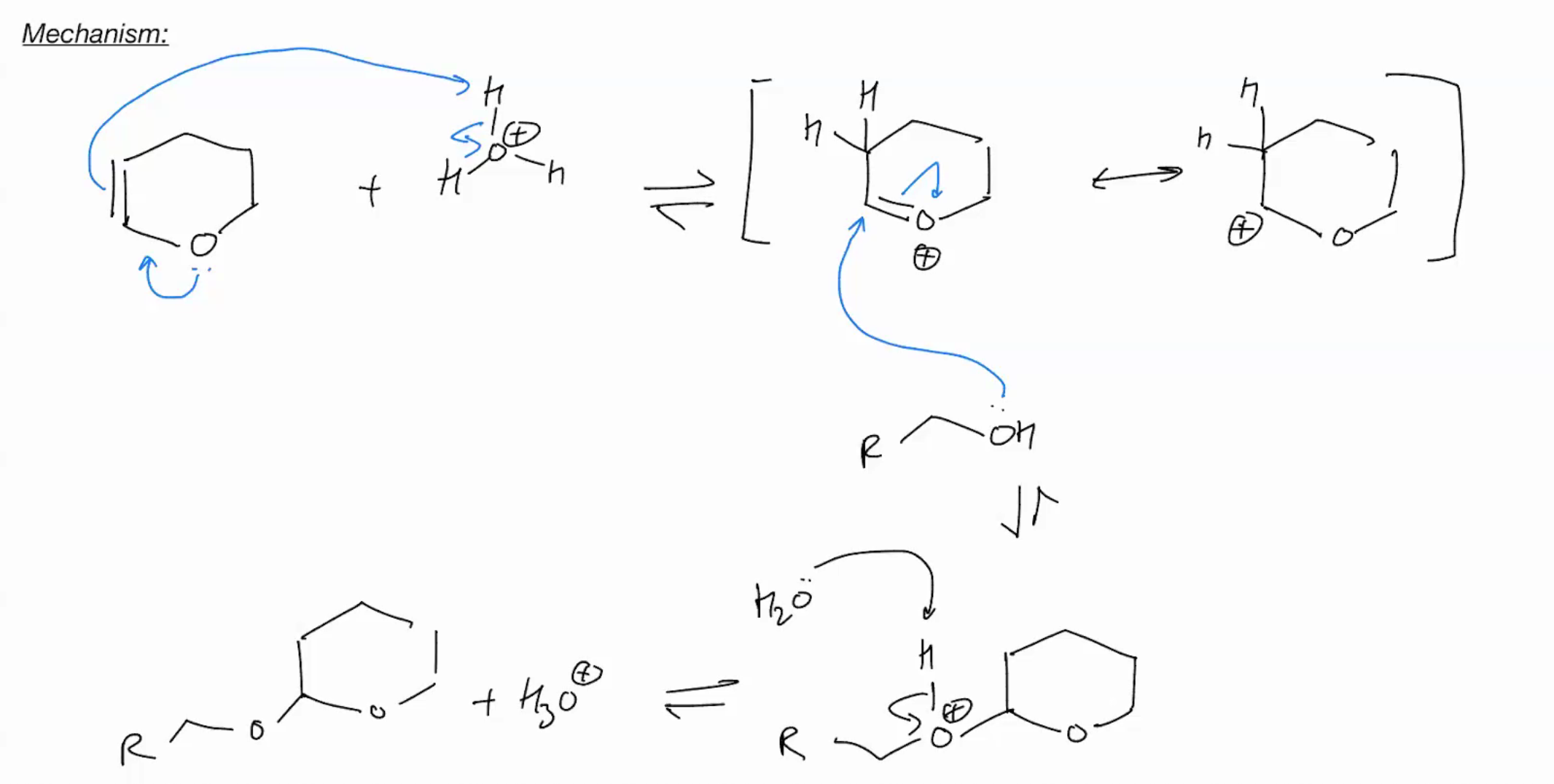
Protection group: Protect Alcohol as acetals
oxyvarbeneudn
Protection group: Protect Alcohol as acetals REMOVING IT
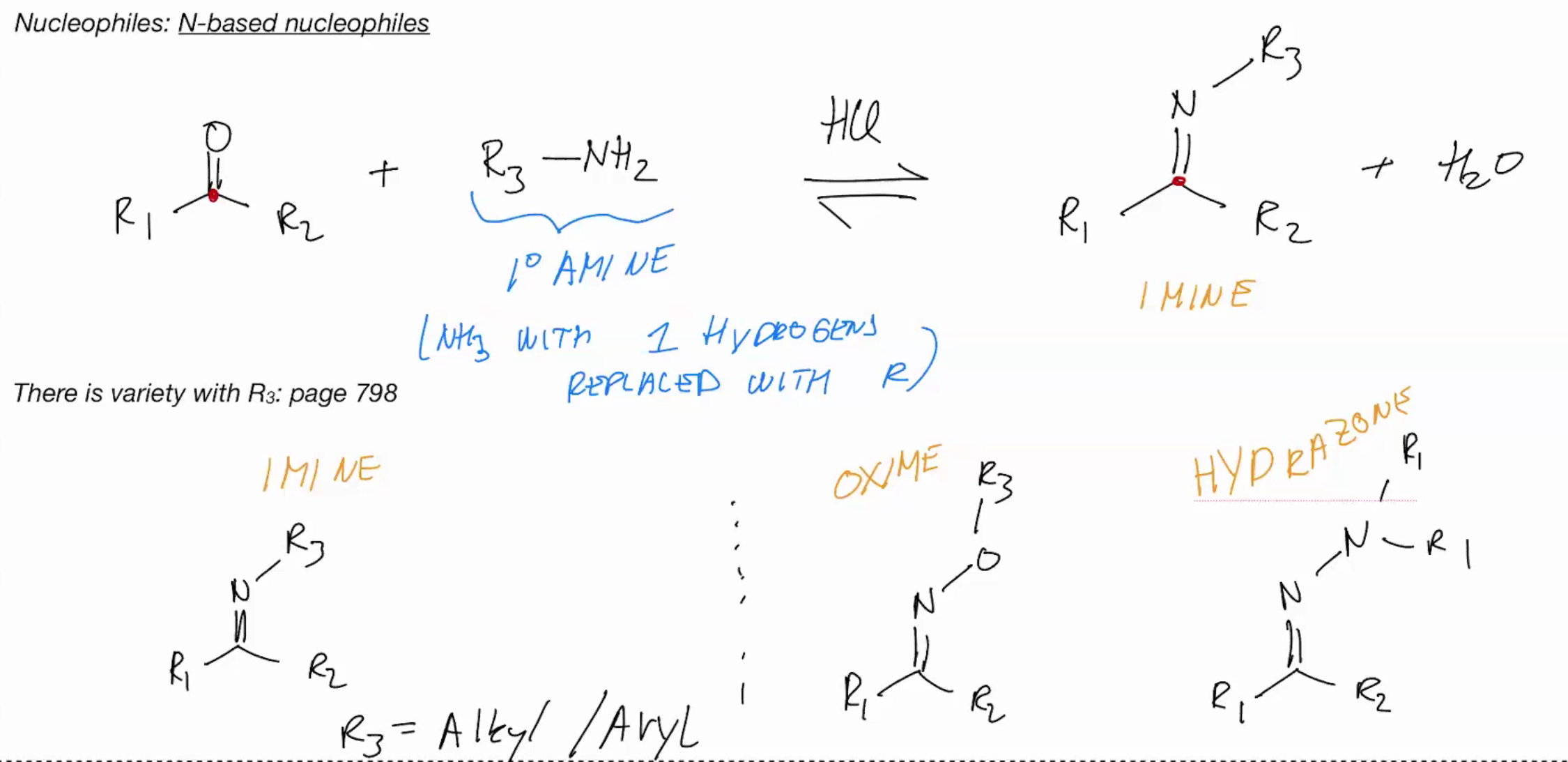
N-Based Nucleophiles pimrary amine
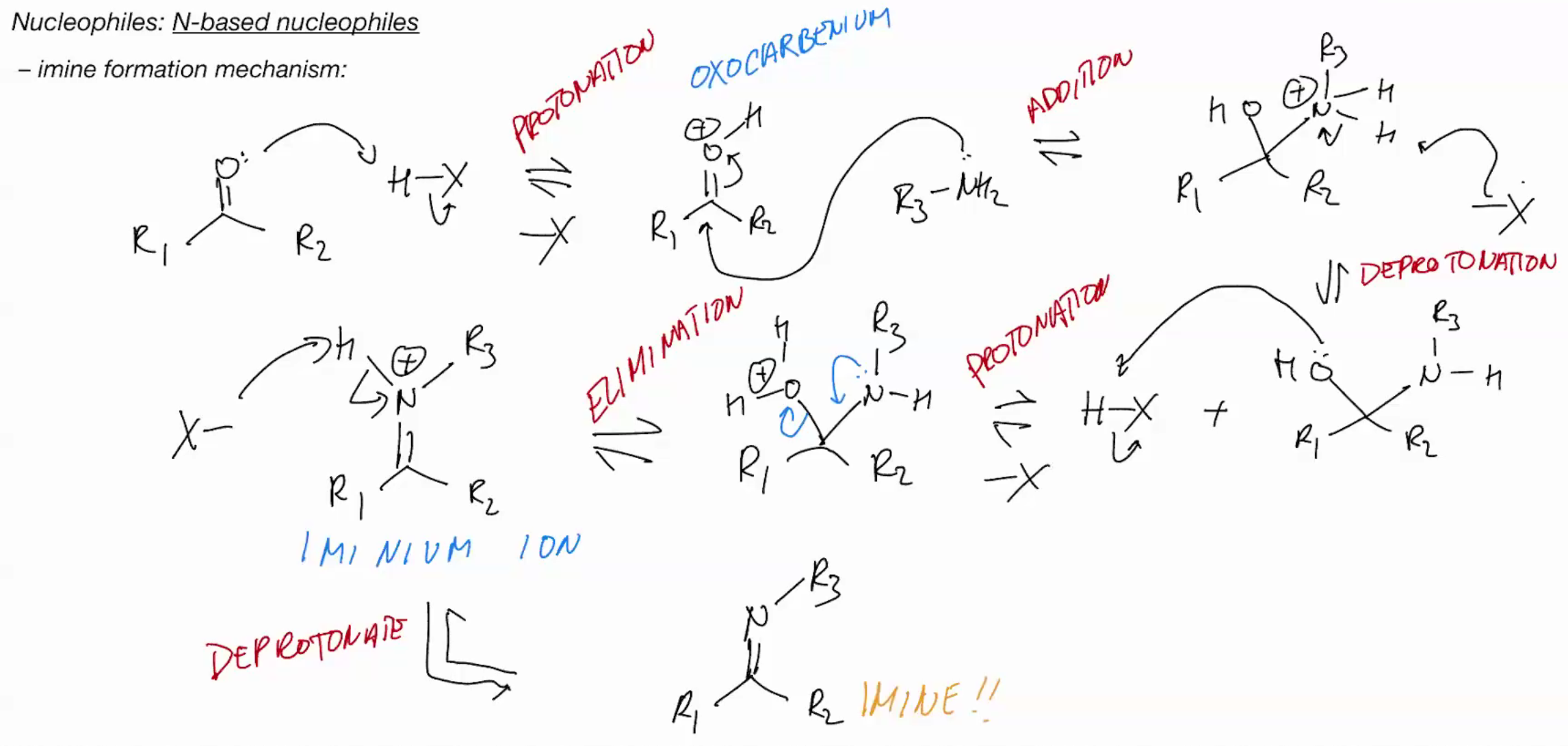
Imine formation PRIMARY AMINE
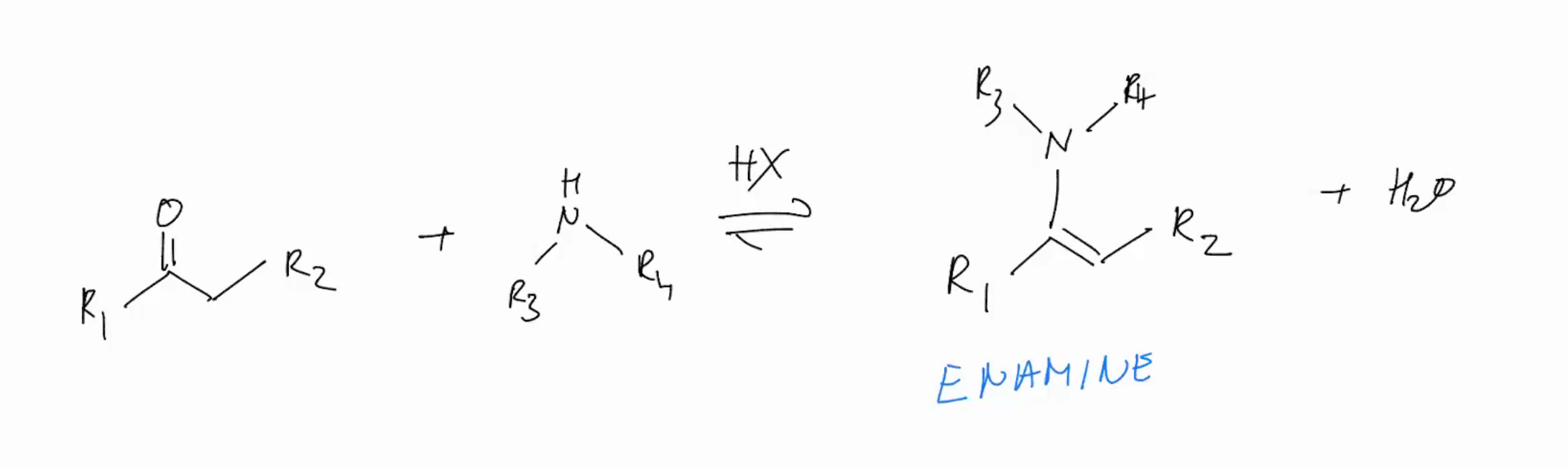
N-Based Nucleophiles (SECONDARY AMINE)
CANT FORM IMINE BUT WE OBSERVE FORMATION OF AN ENAMINE
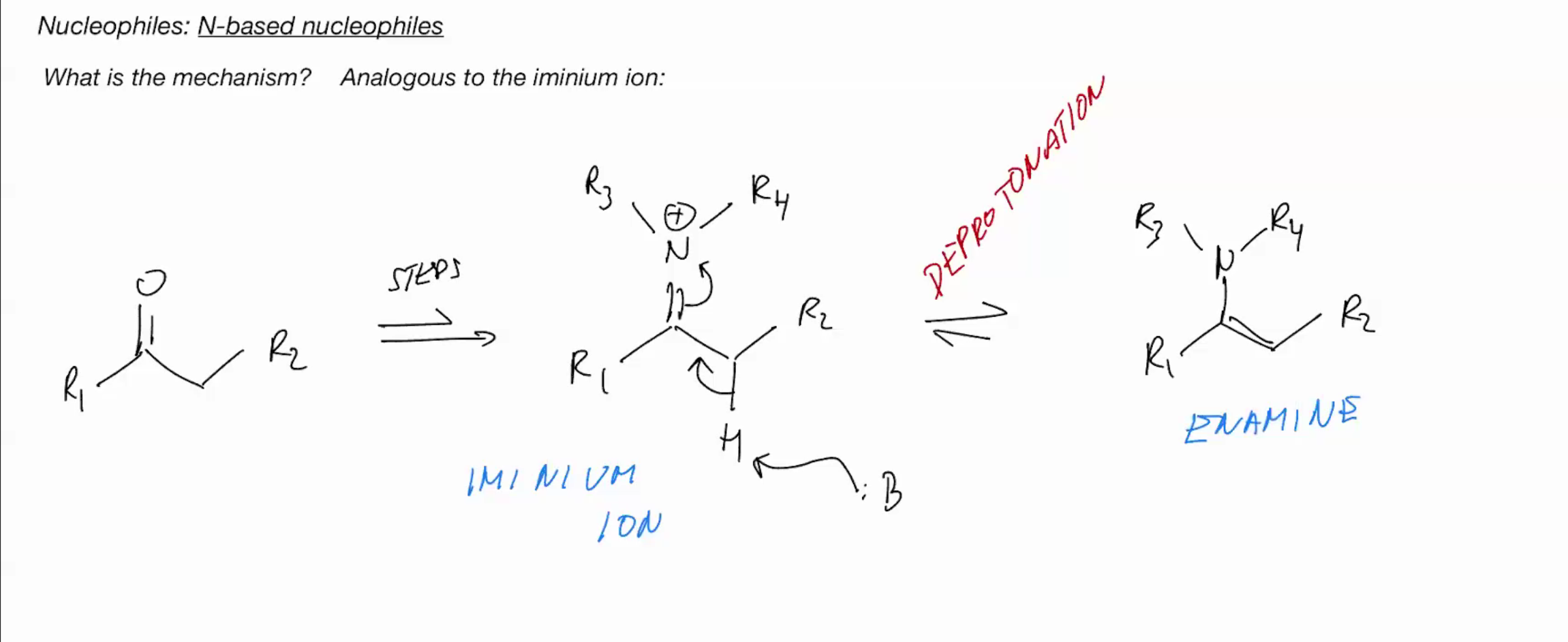
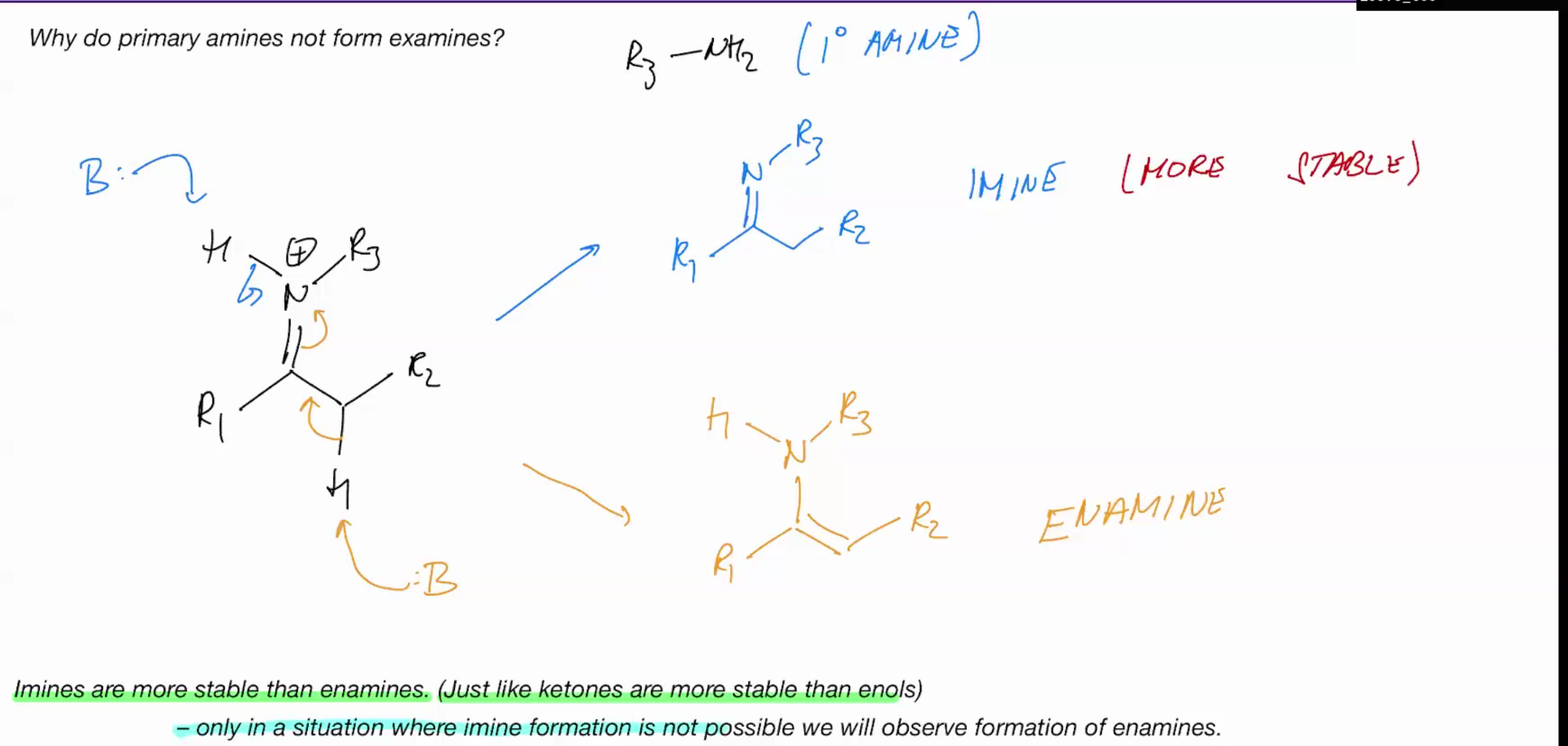
Why do primary amines NOT form enamines?
only in situations where imine formation is not possible we will observe formation of enamines
WE DONT HAVE AN ACIDIC NH WHICH IS WHY WE FORM A ENAMINE IN SCENARIOS.

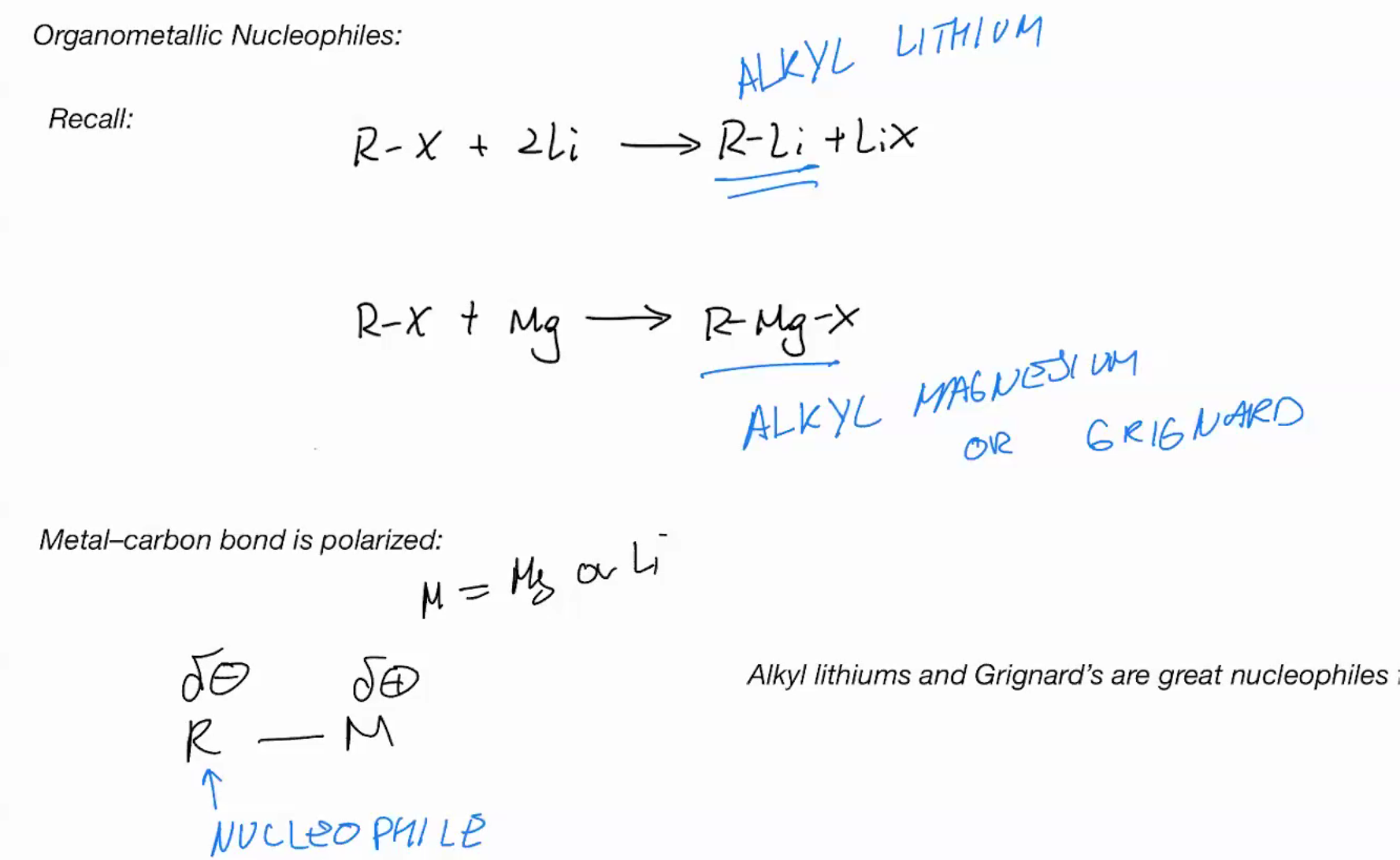
Organometallic nucleophiles
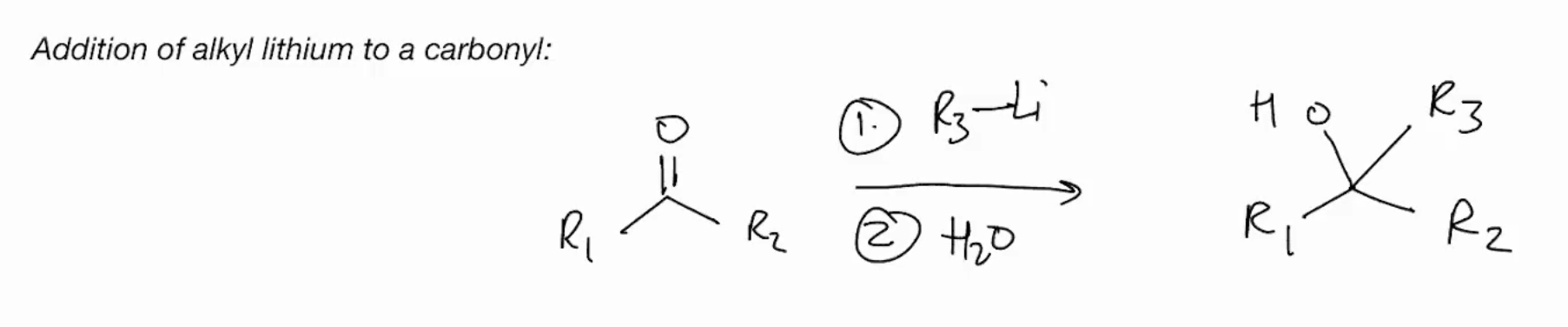
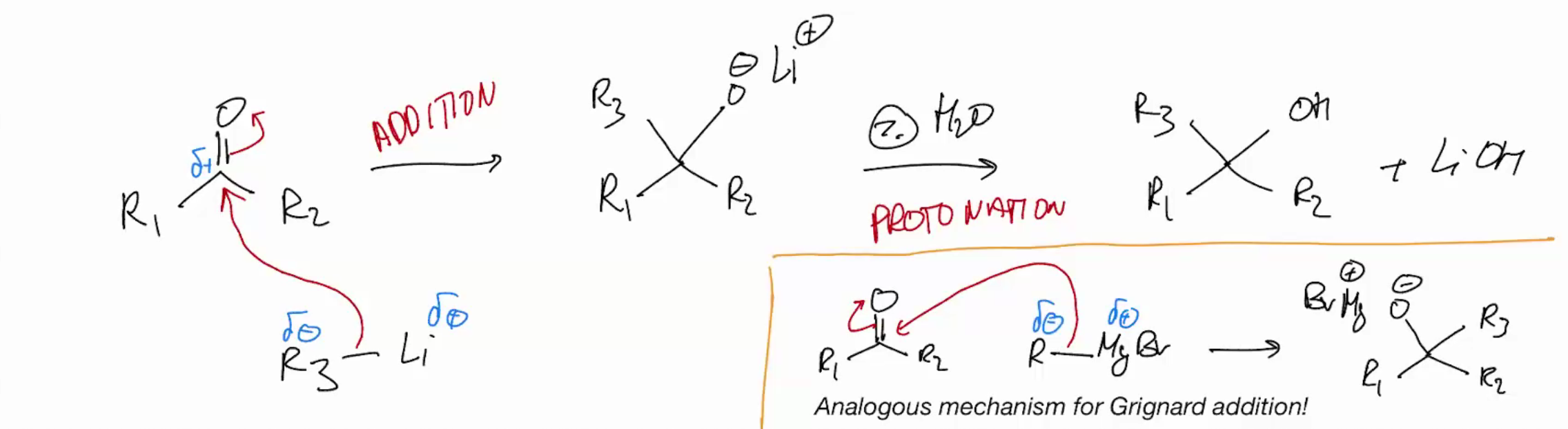
Addition of alkyl lithium to a carbonyl & Grignard addition
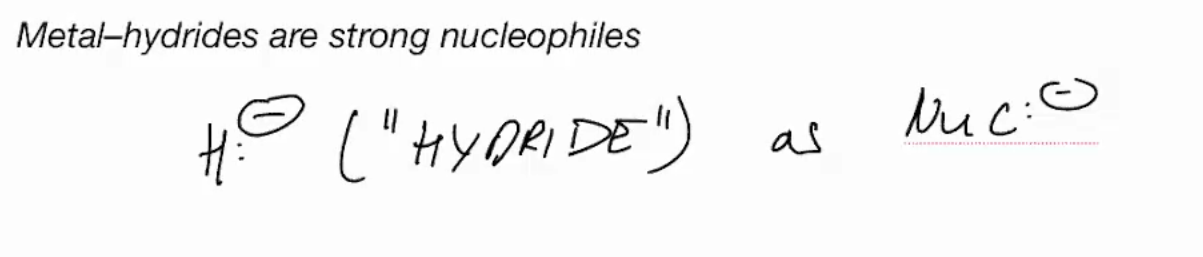
Metal hydrides are strong nucleophiles

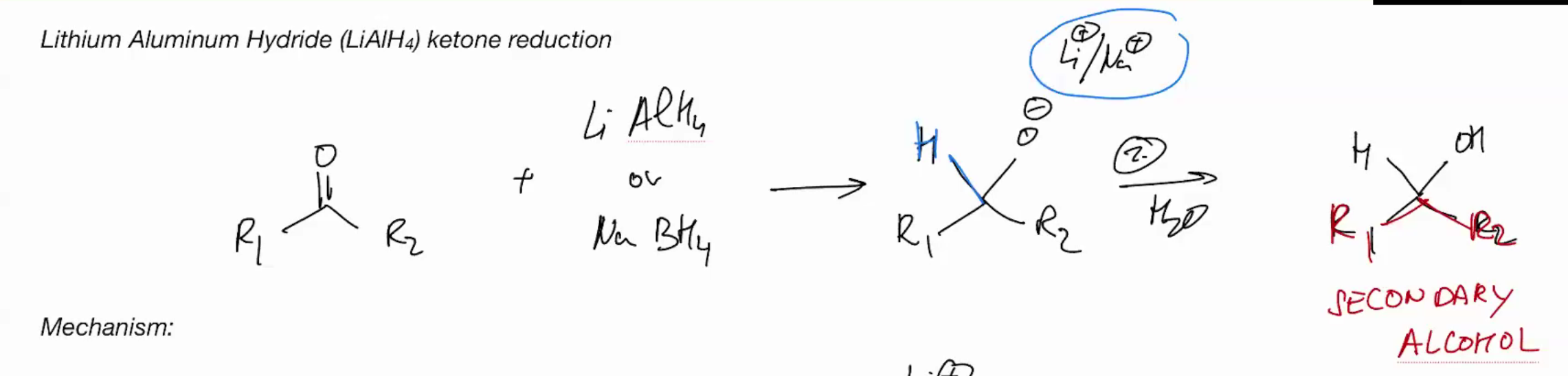
Lithium Aluminum Hydride (LiAIH4) Ketone Reduction
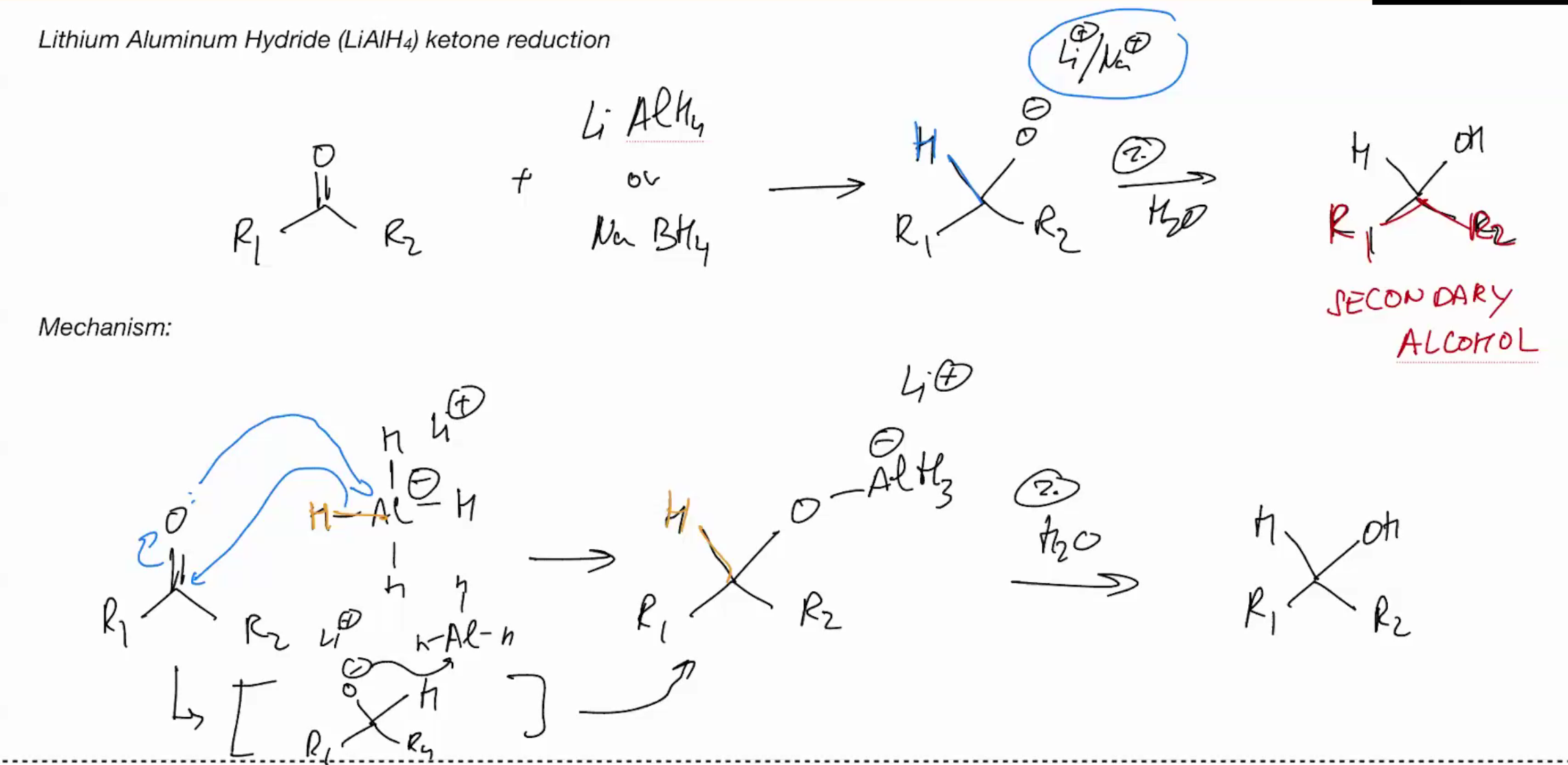
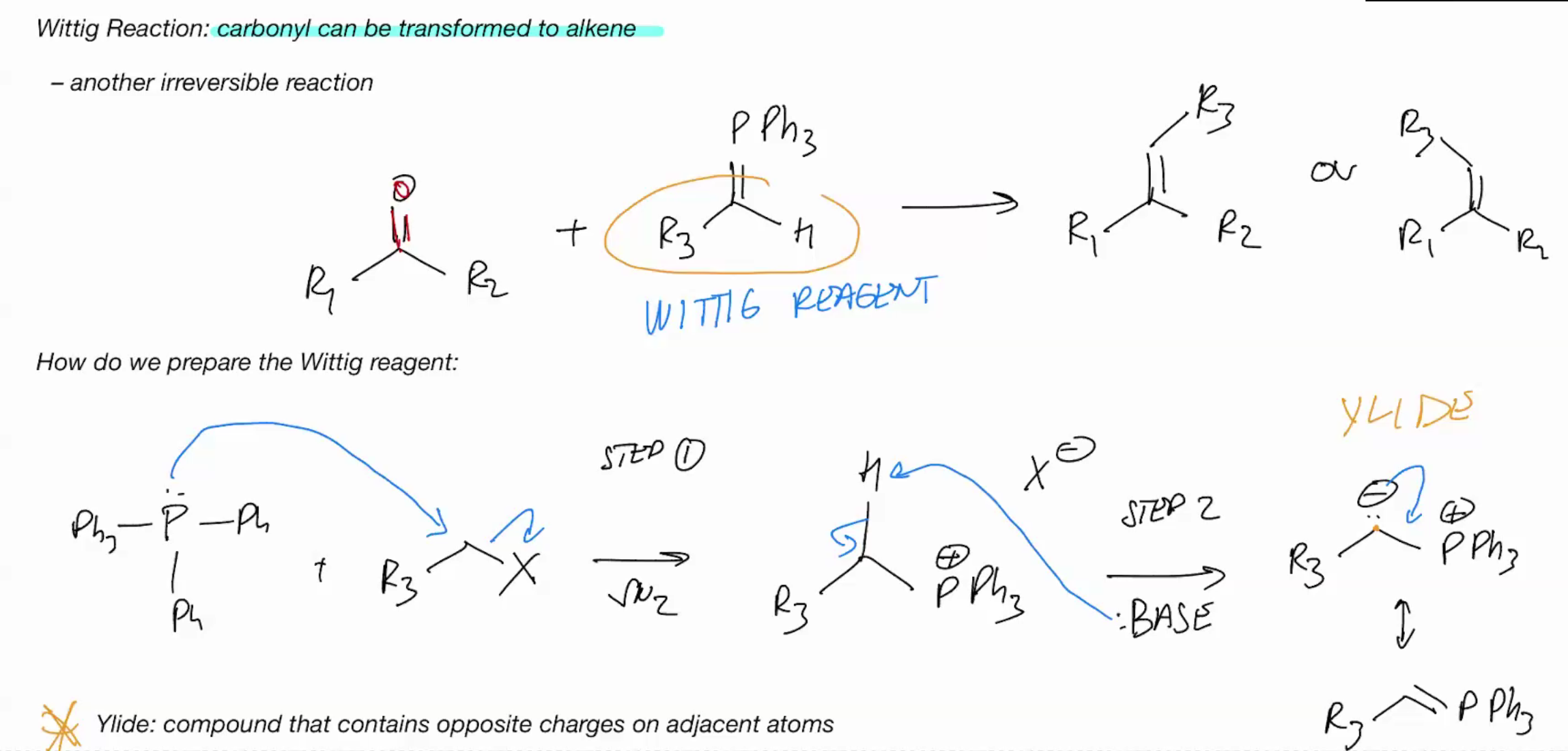
Wittig Reaction (IRREVERSIBLE RXN)
Make the reagent R3-PPh3
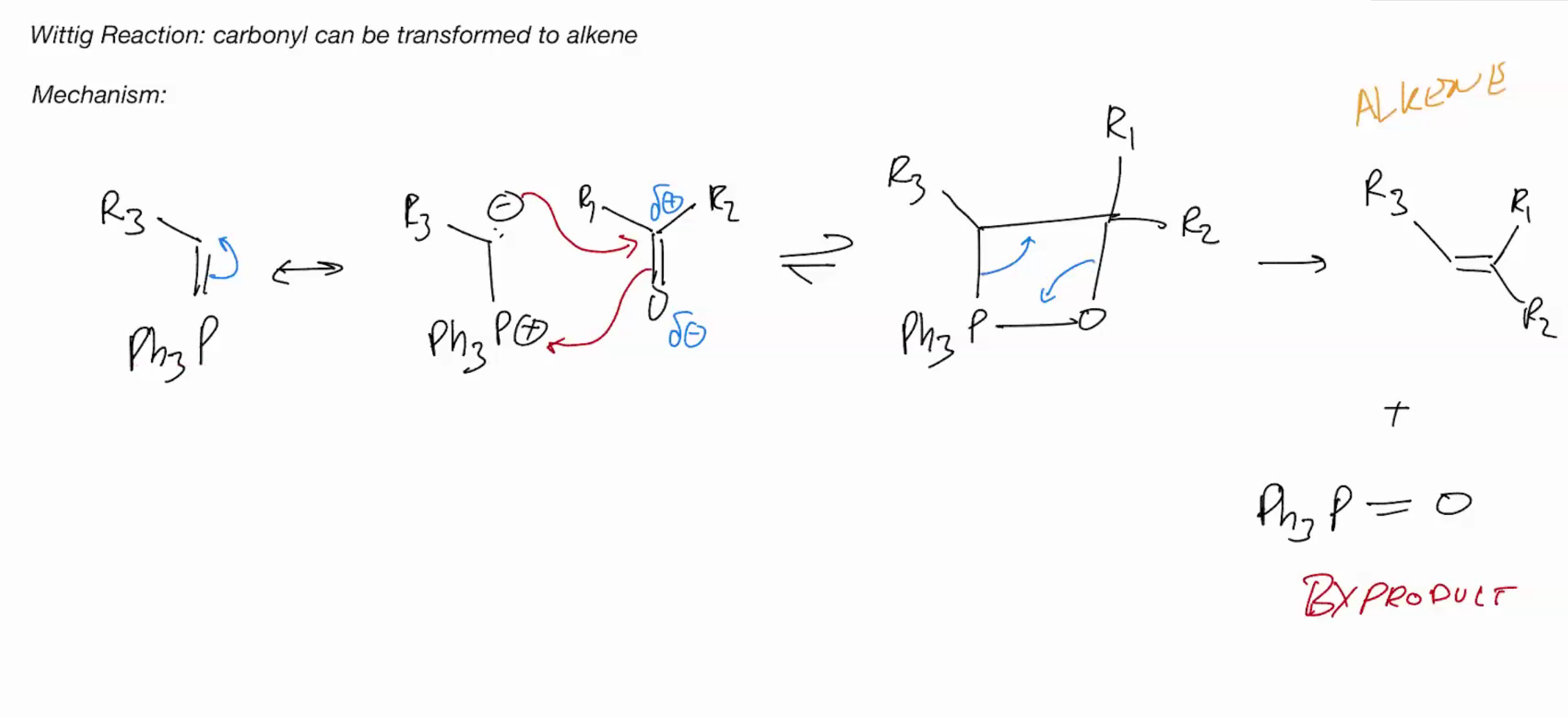
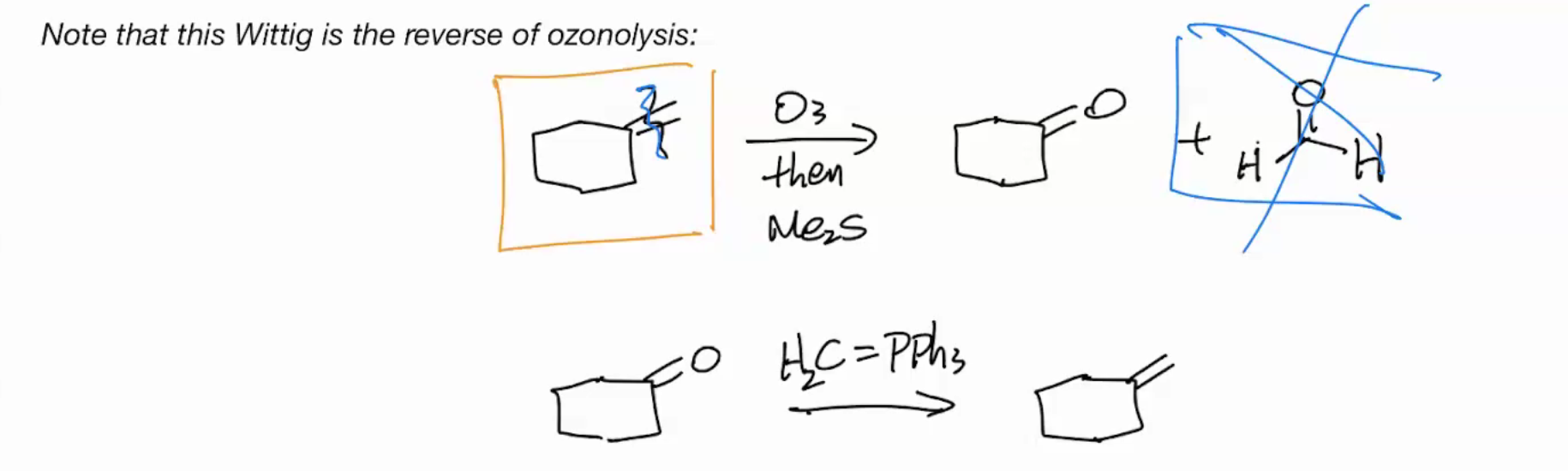
Wittig is the Reverse of Ozonolysis
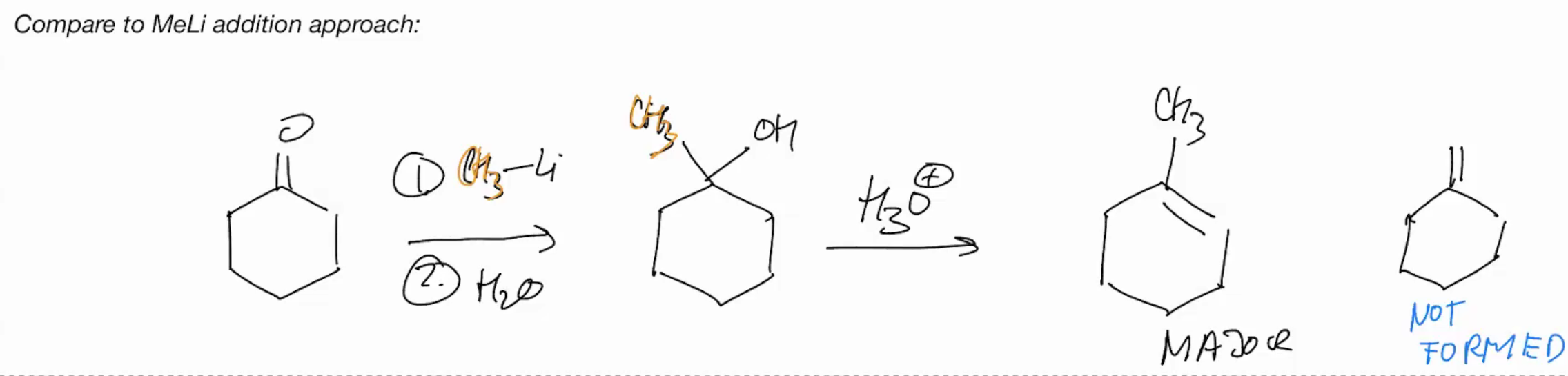
MeLi Addition approach ( basically ignore this u need to do a wittig rxn for the double bond there cuz itll be inside)
Synthesis of carbonyl overview
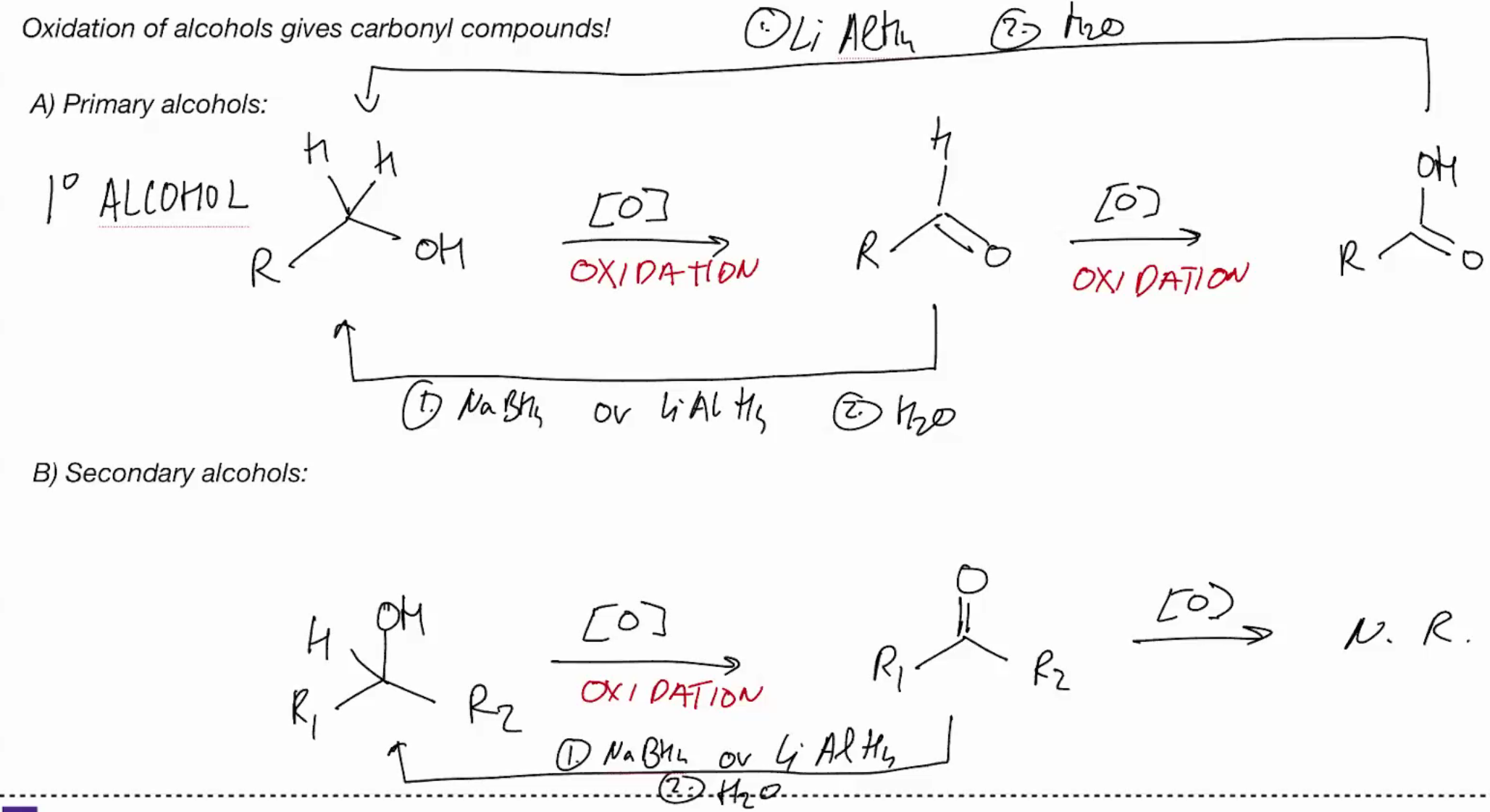
Oxidation of alcohols gives carbonyl compounds (tert acholhols have no rxn cuz no hyrogen to remove)
(tert acholhols have no rxn cuz no hyrogen to remove)
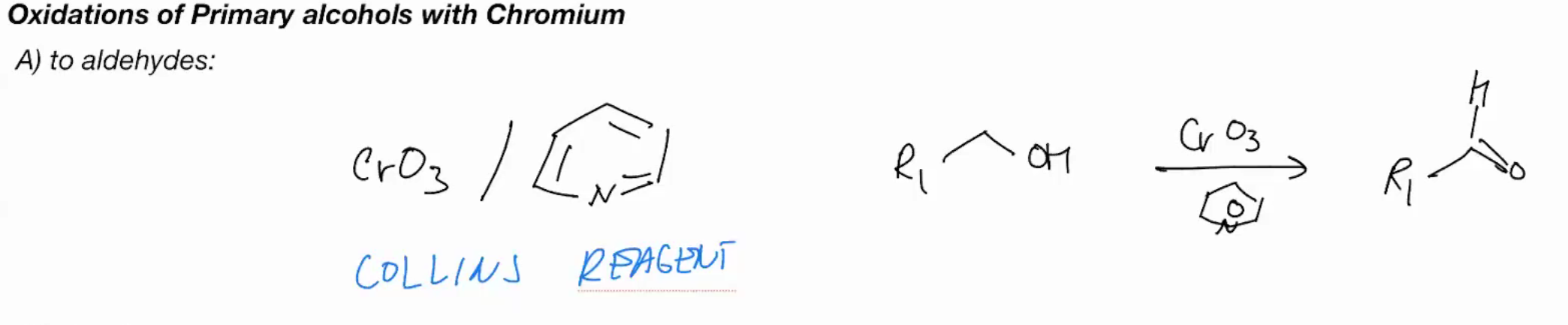
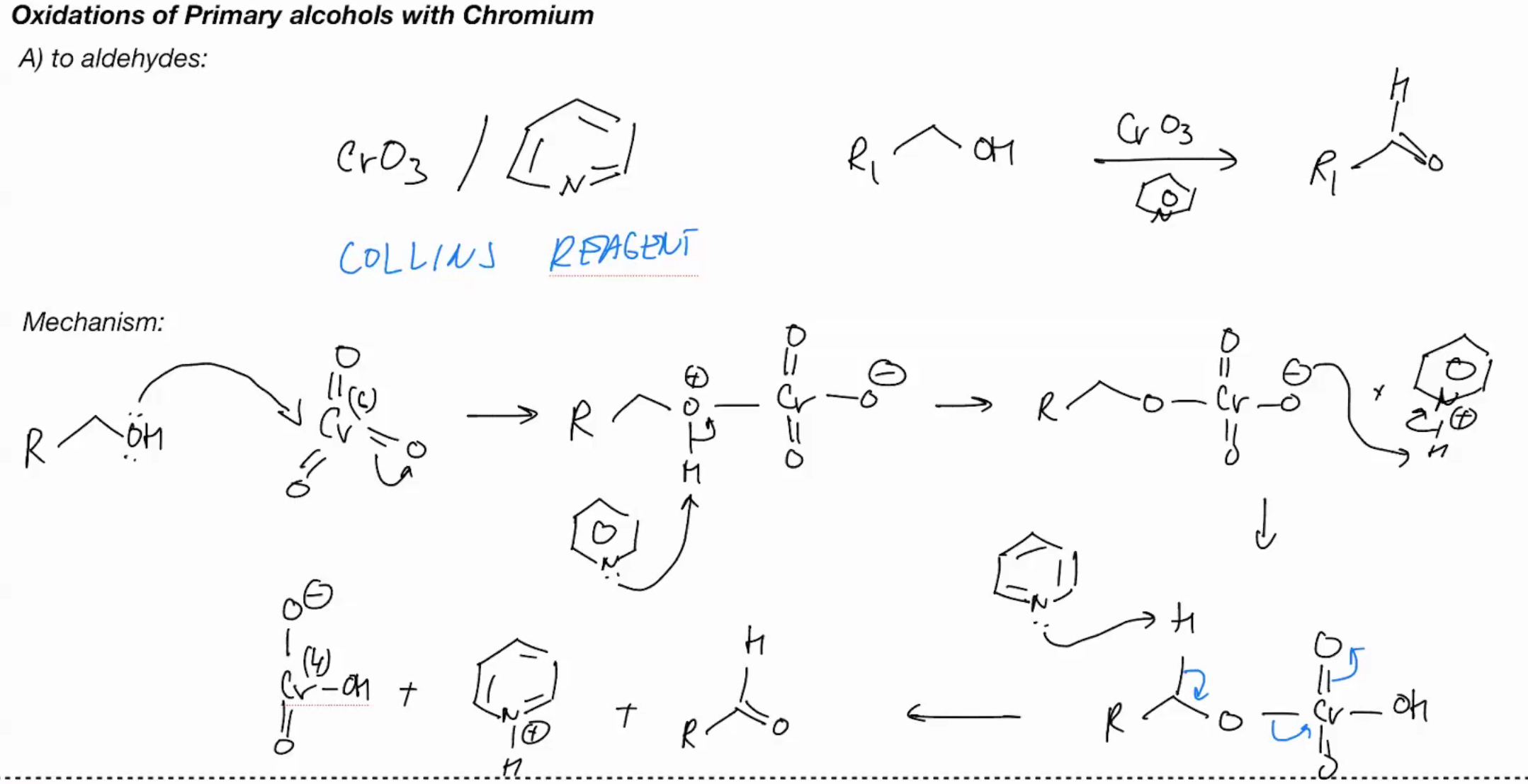
Oxidations of primary OH with Chromium (COLLINS REAGENT)
Cr03/ aromatic benzene with N - Collins reagent
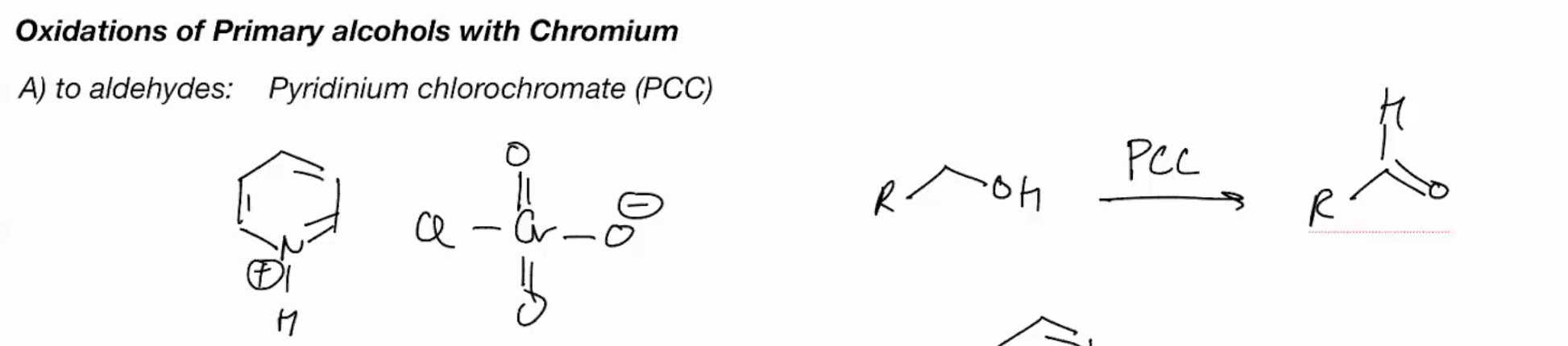
Oxidations of primary OH with Chromium (PYRIDINIUM CHLOROCHROMATE) - PPC
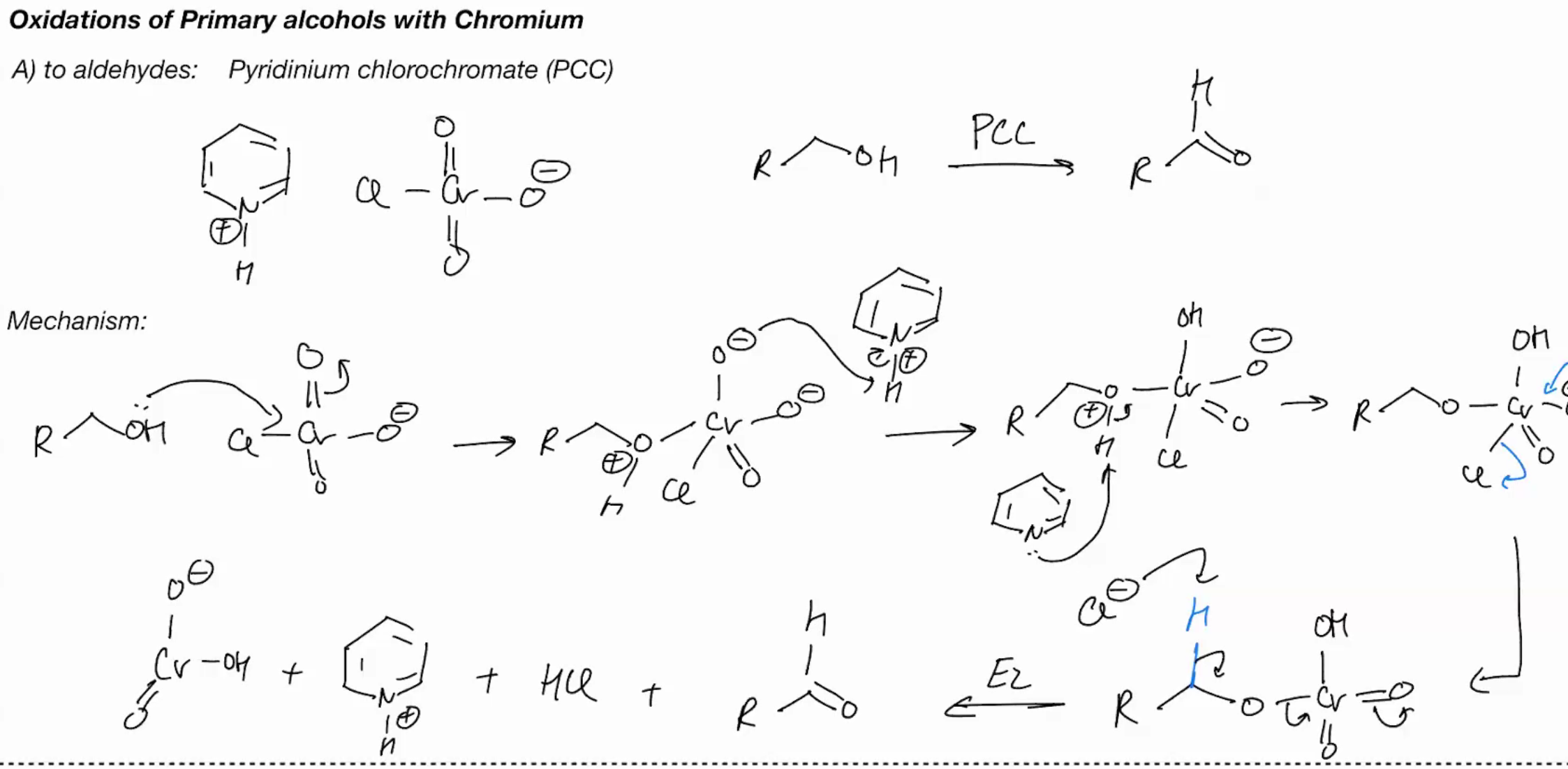
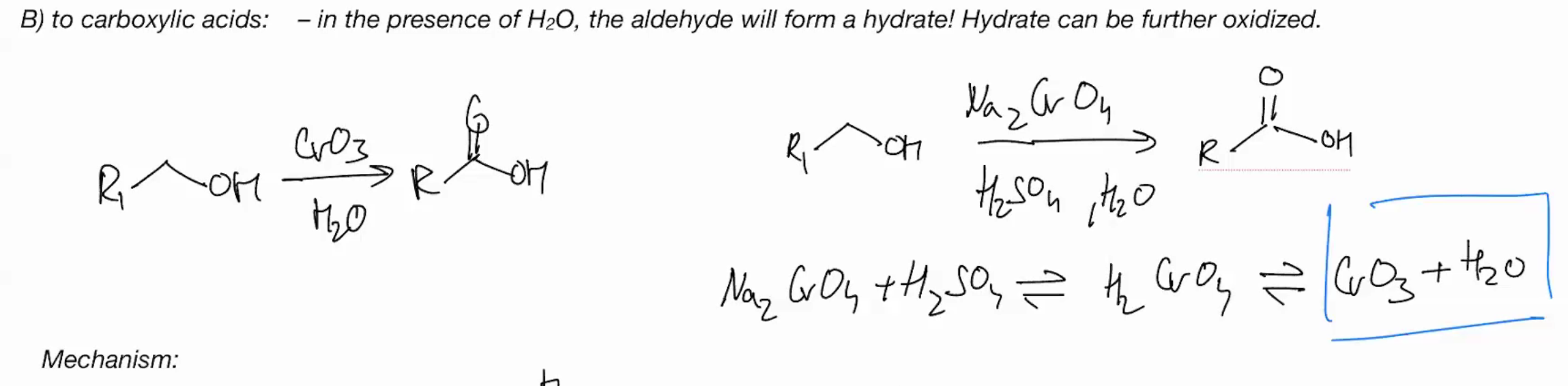
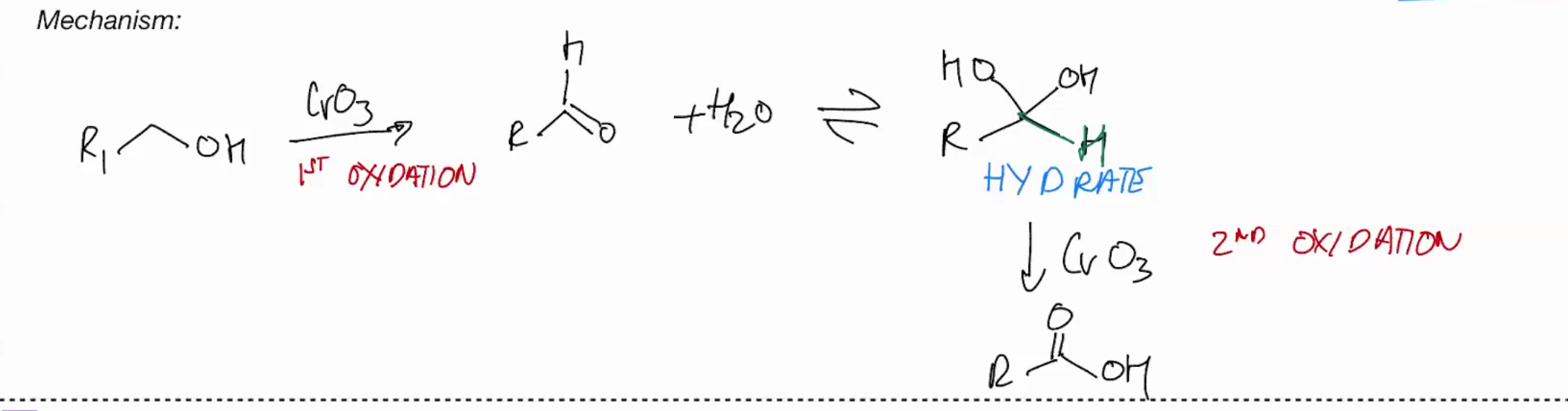
Oxidations of primary OH with Chromium - OH into a carboxylic acid
CrO3 + H2O
use water as the pyridine , deprotonate w that
Secondary OH provide ketones! CAUSE THEY CANT OVEROXIDIZE
misc
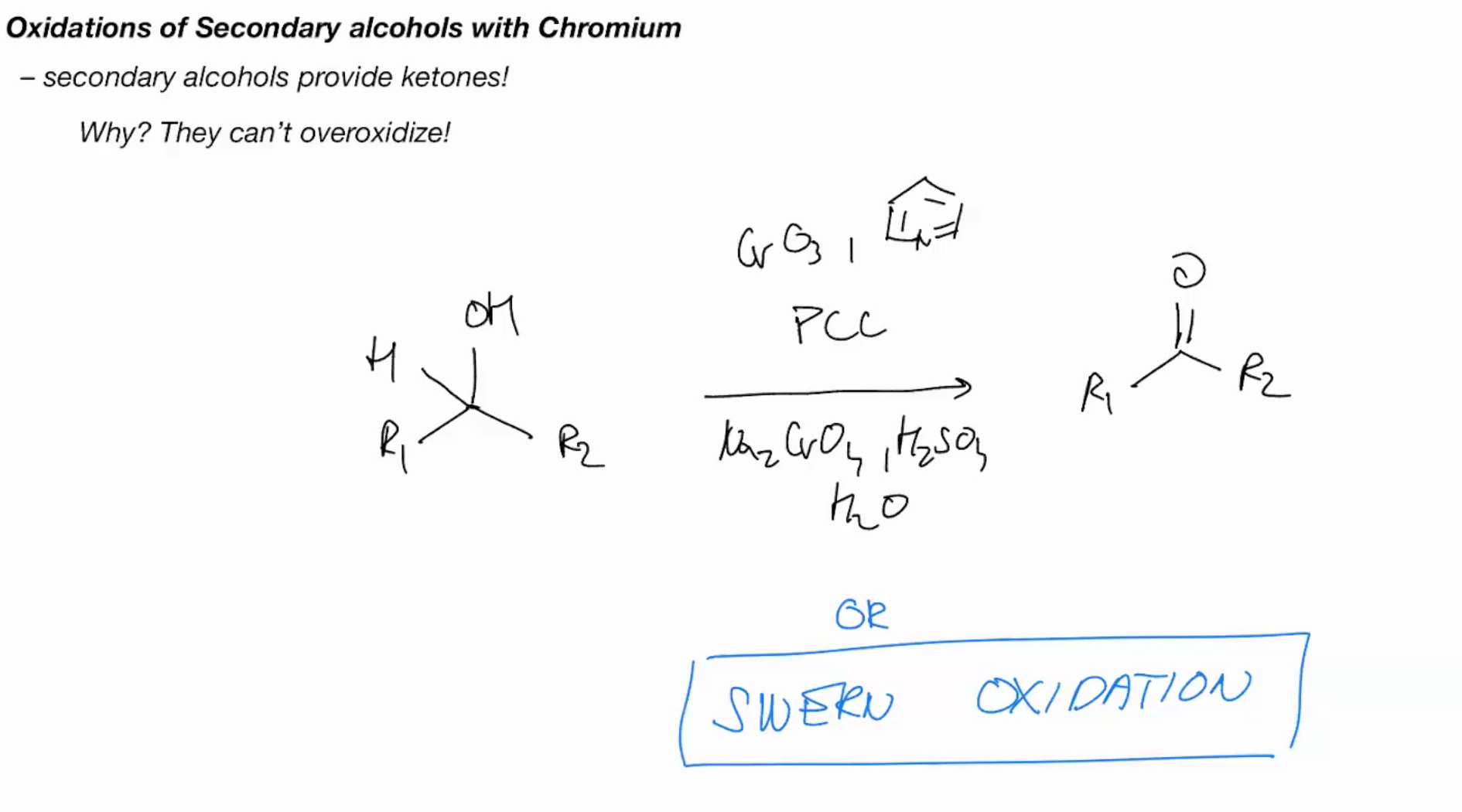
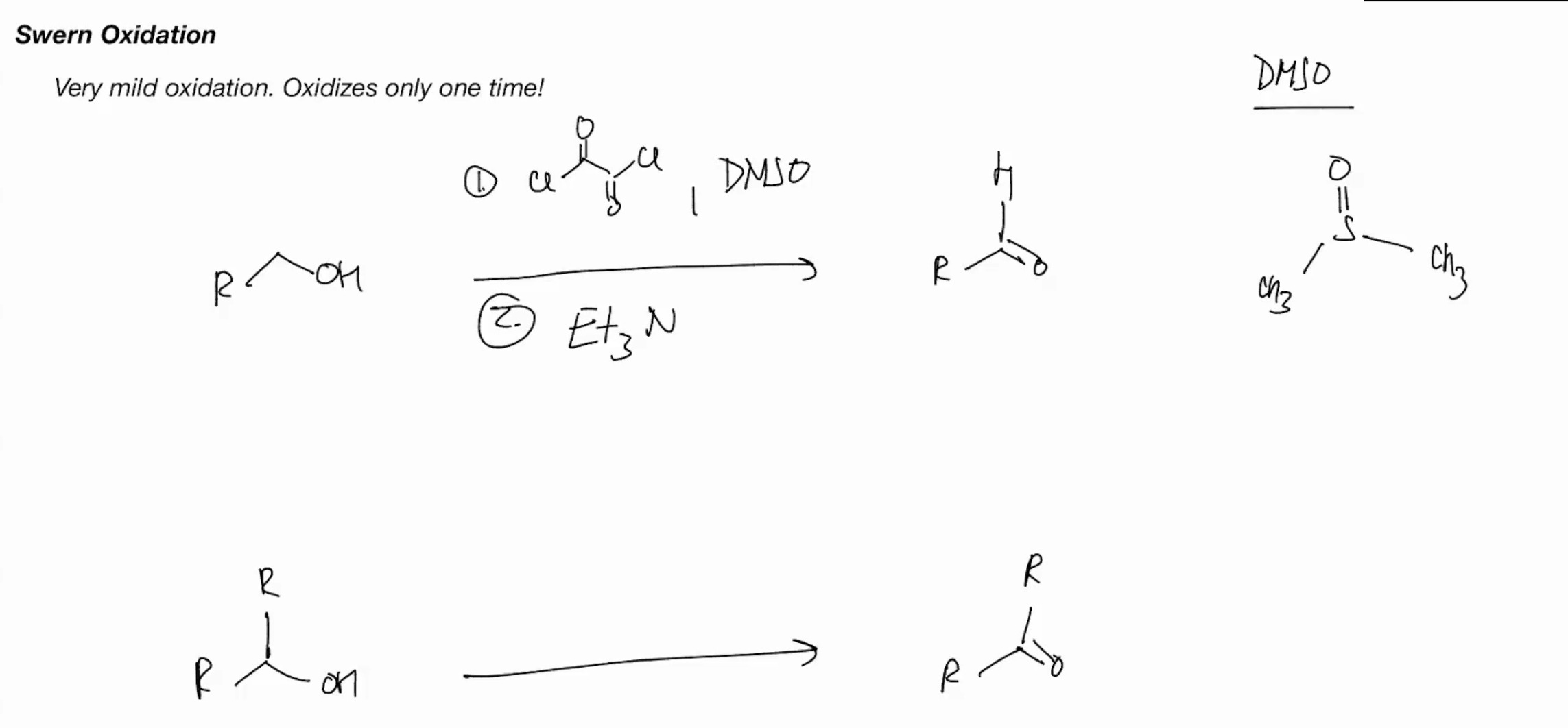
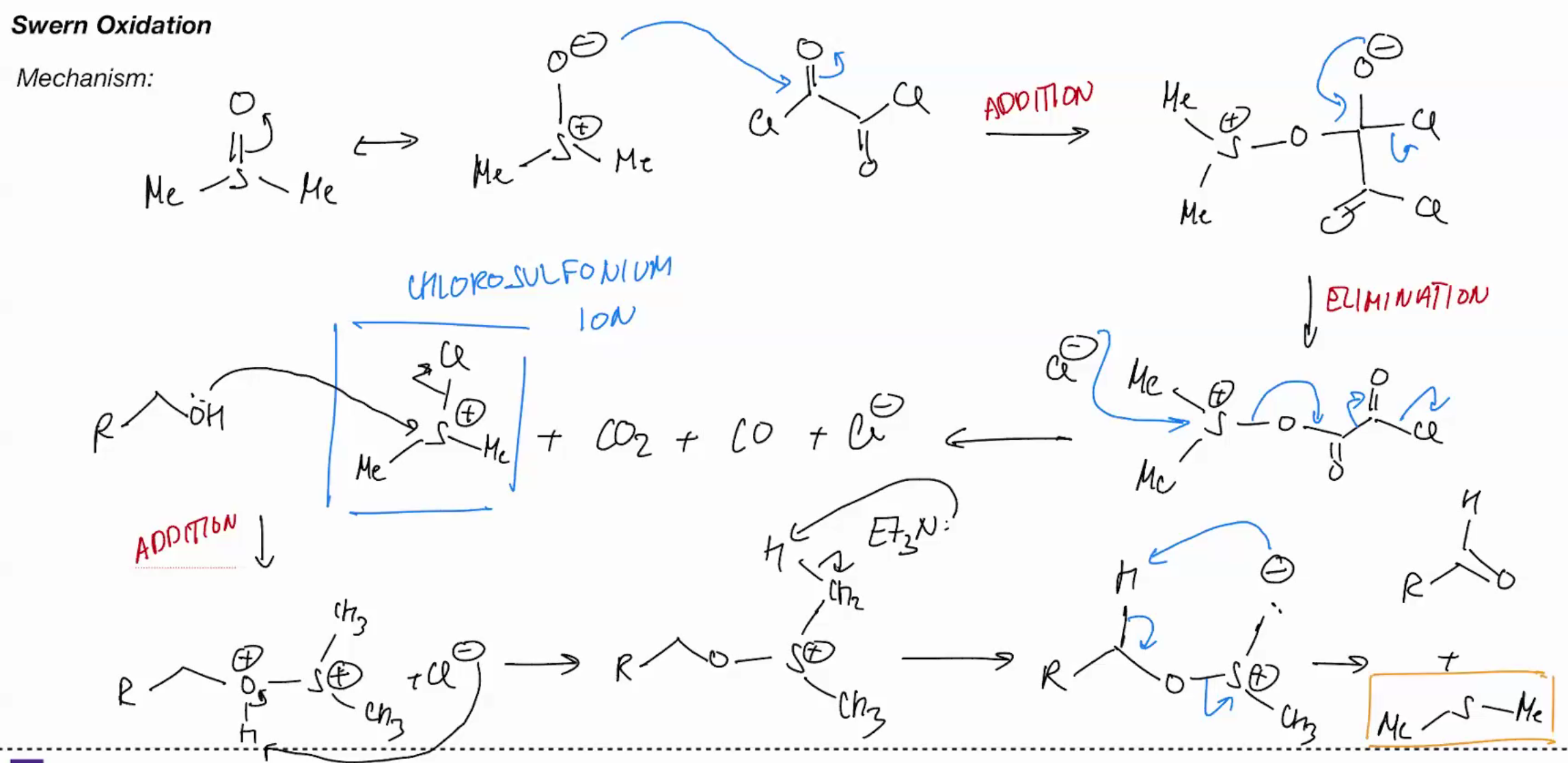
Swern Oxidation (only oxidizes one time)
1brick , dmso
etn3
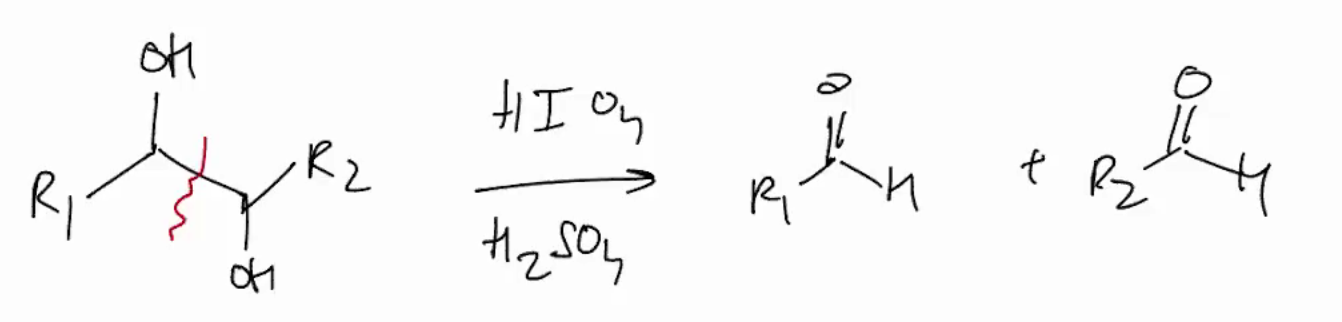
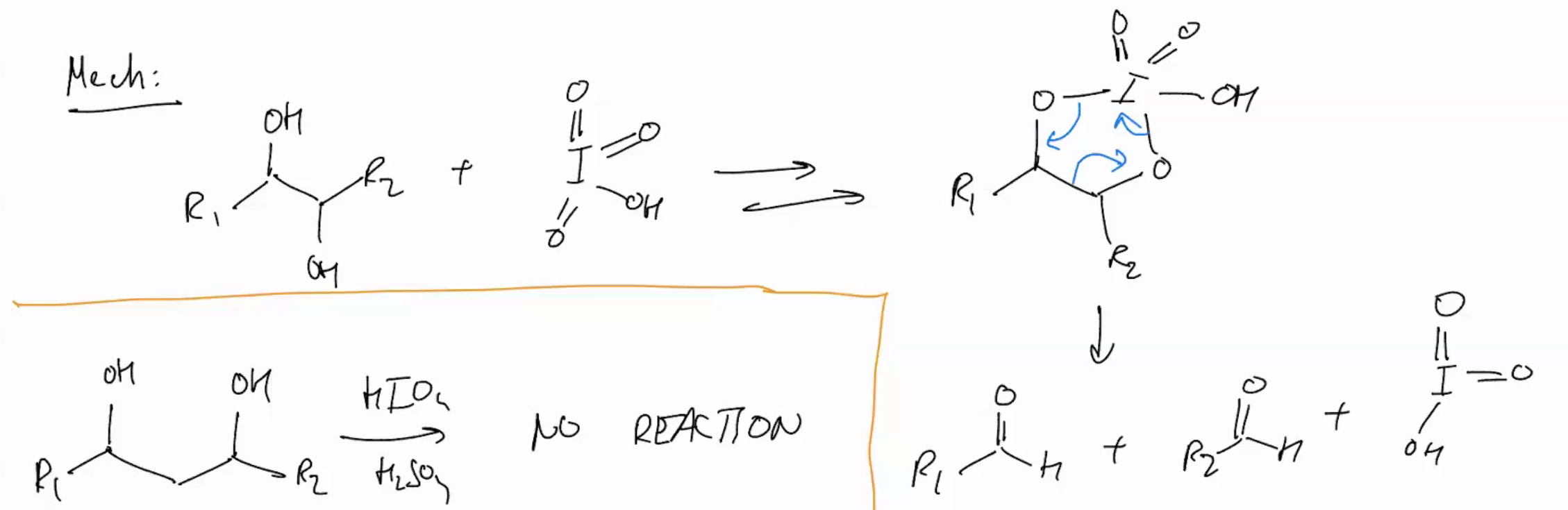
Oxidation of Diols (oxidative cleavage)
not on exam yay
HiO4
H2sO4
only works an adjacent OH’s
RECAP OMEGA!
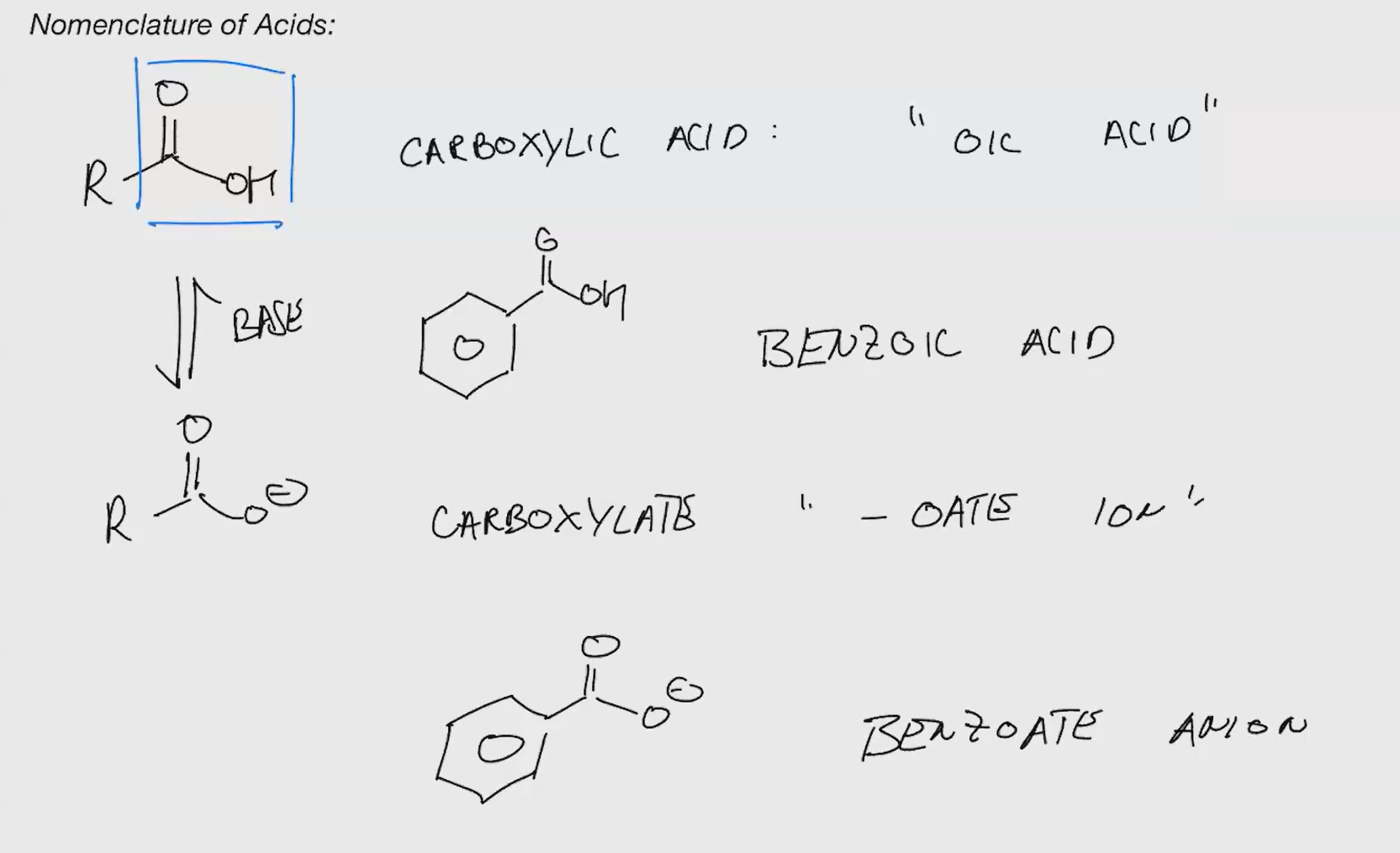
Nomenclature of acids
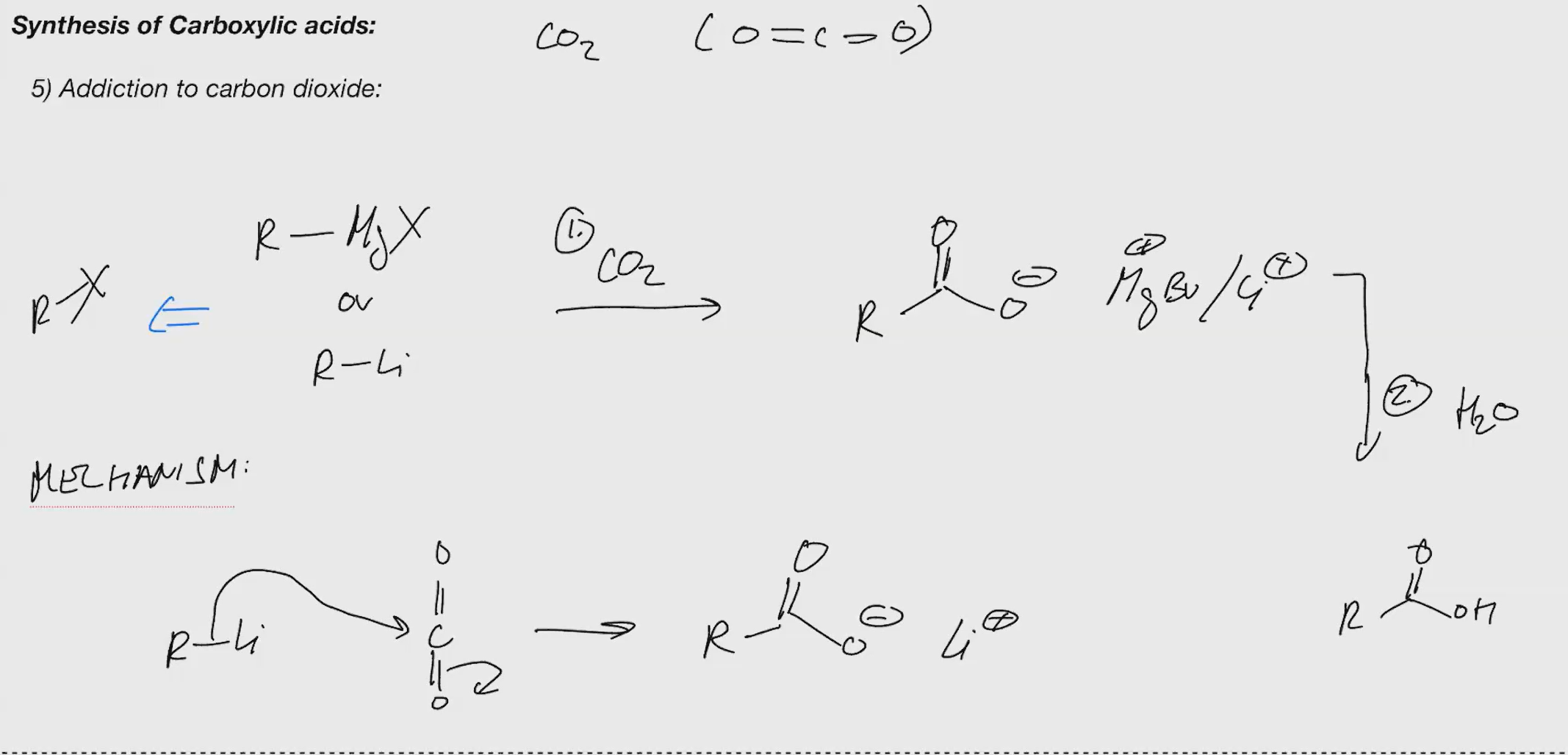
Synthesis of carboxylic acid
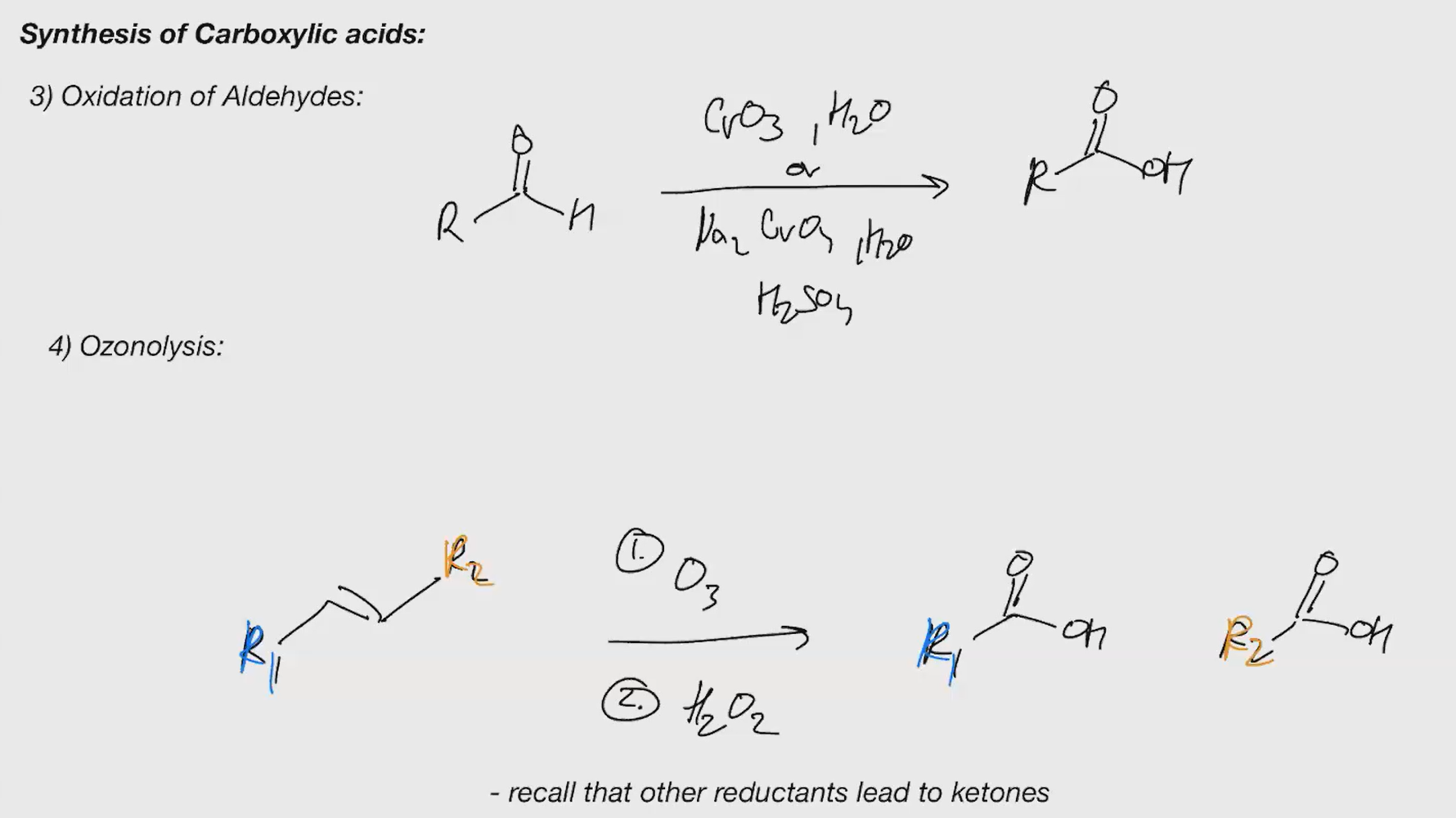
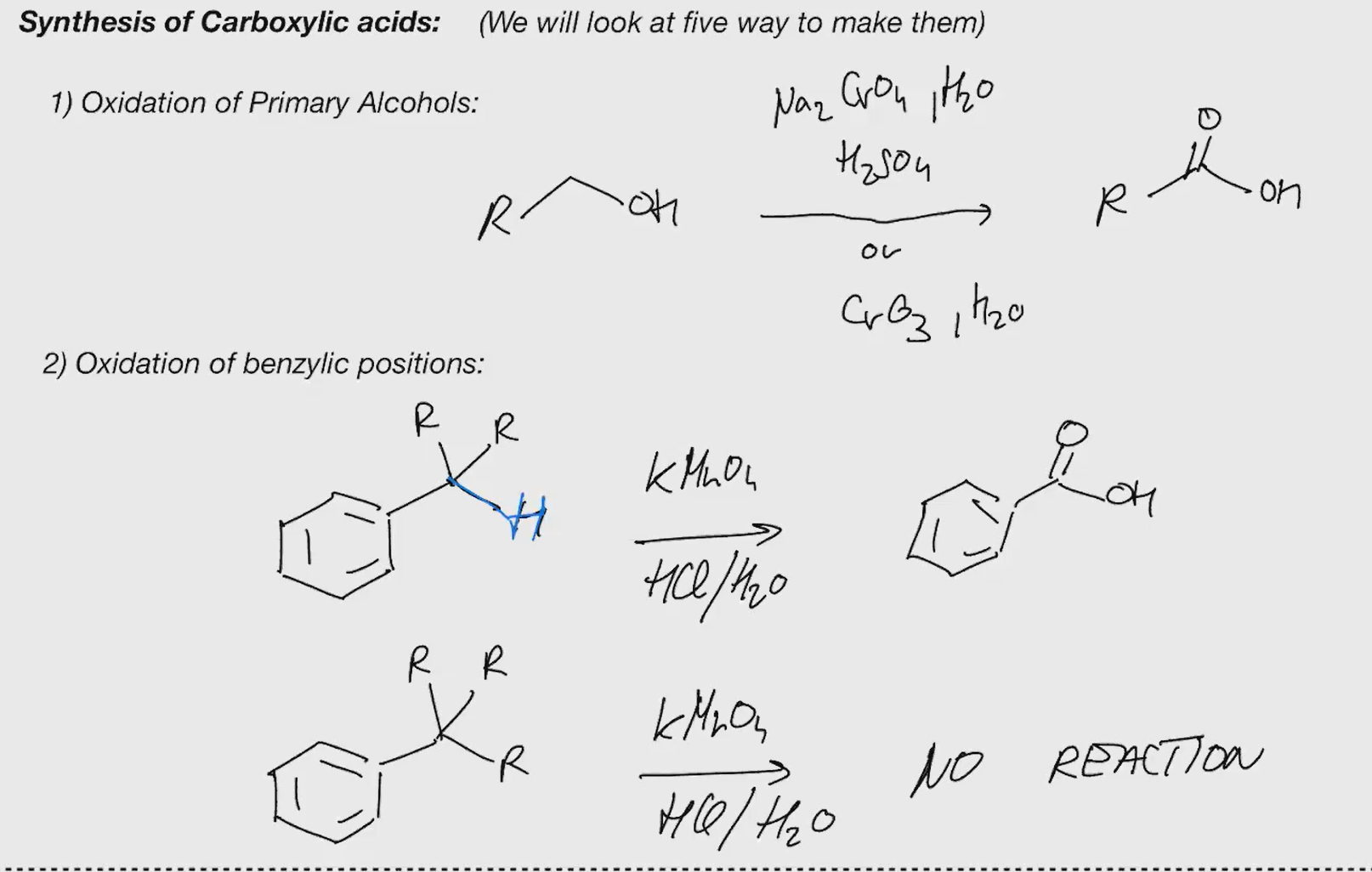
syntehsis of carboxylic acid

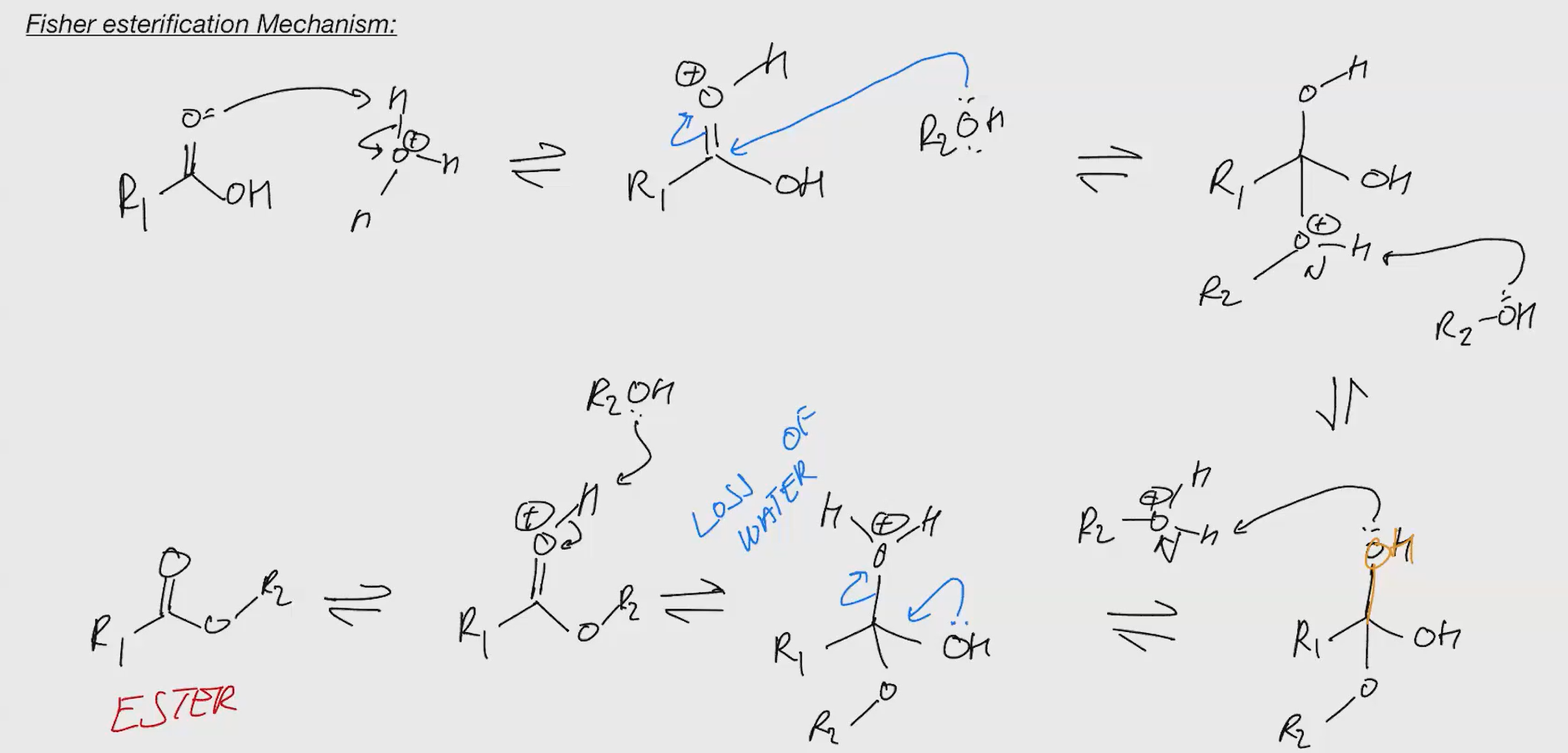
Fisher esterification
Remember under acidic condition u cant kick out bases. that why we protonate
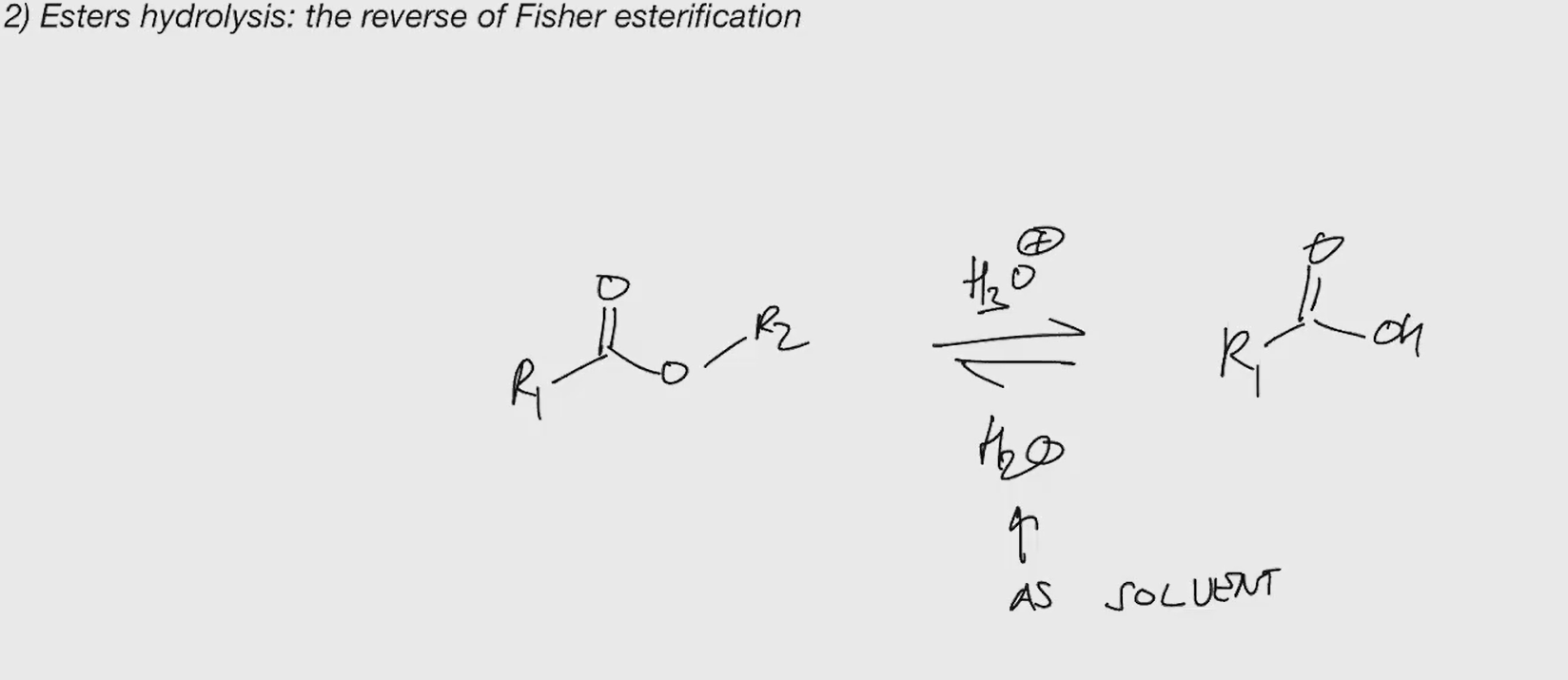
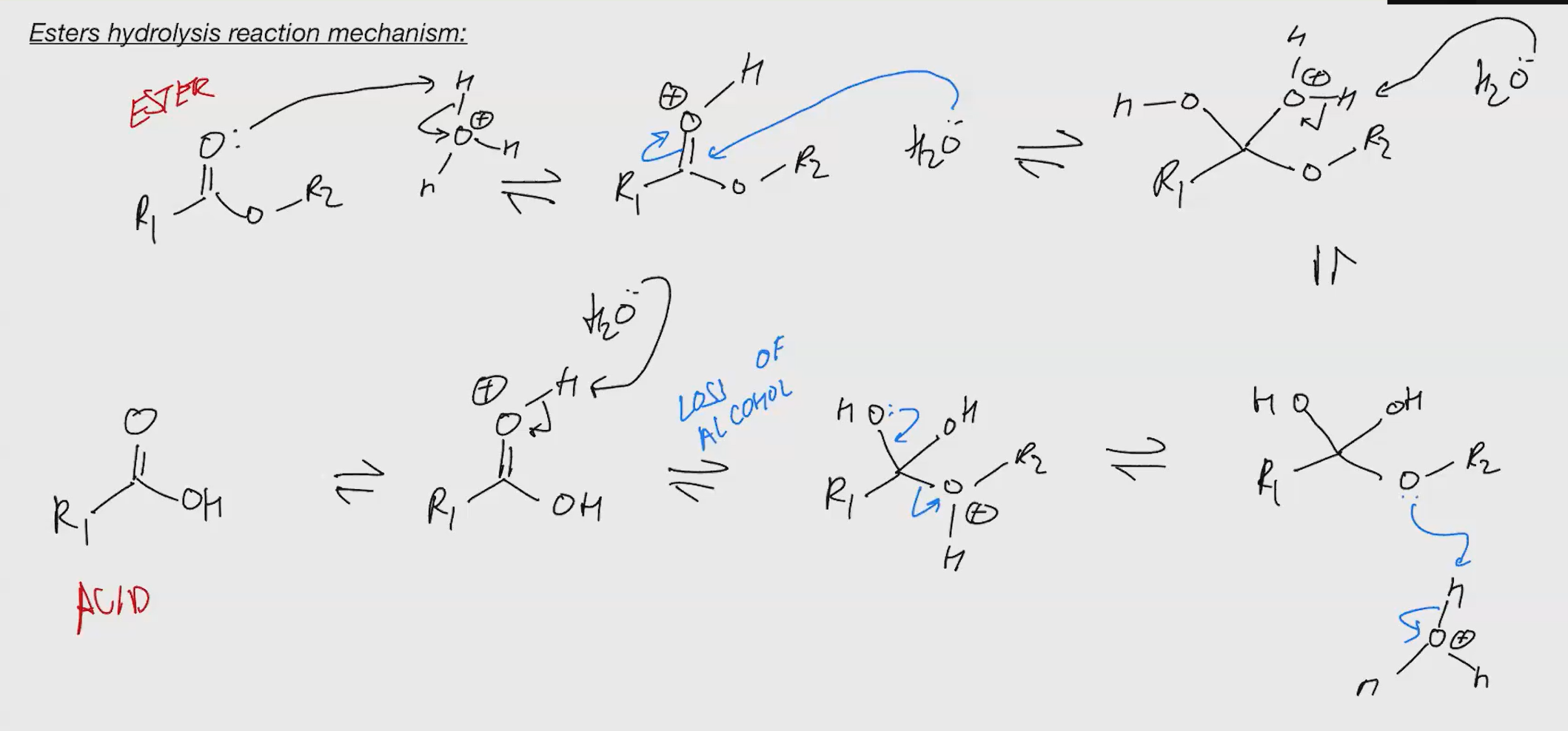
Ester hydrolysis
we kick out OH?
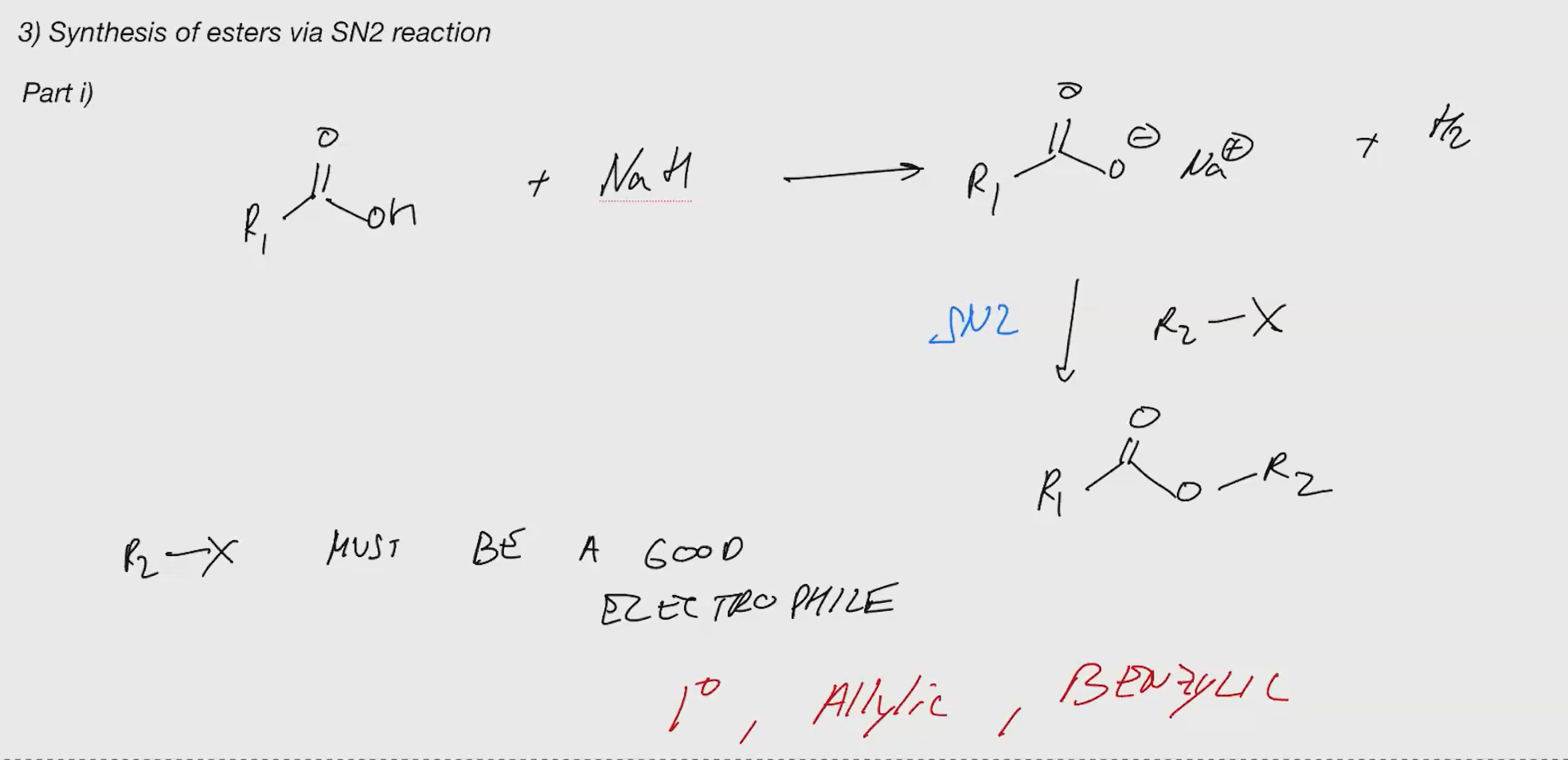
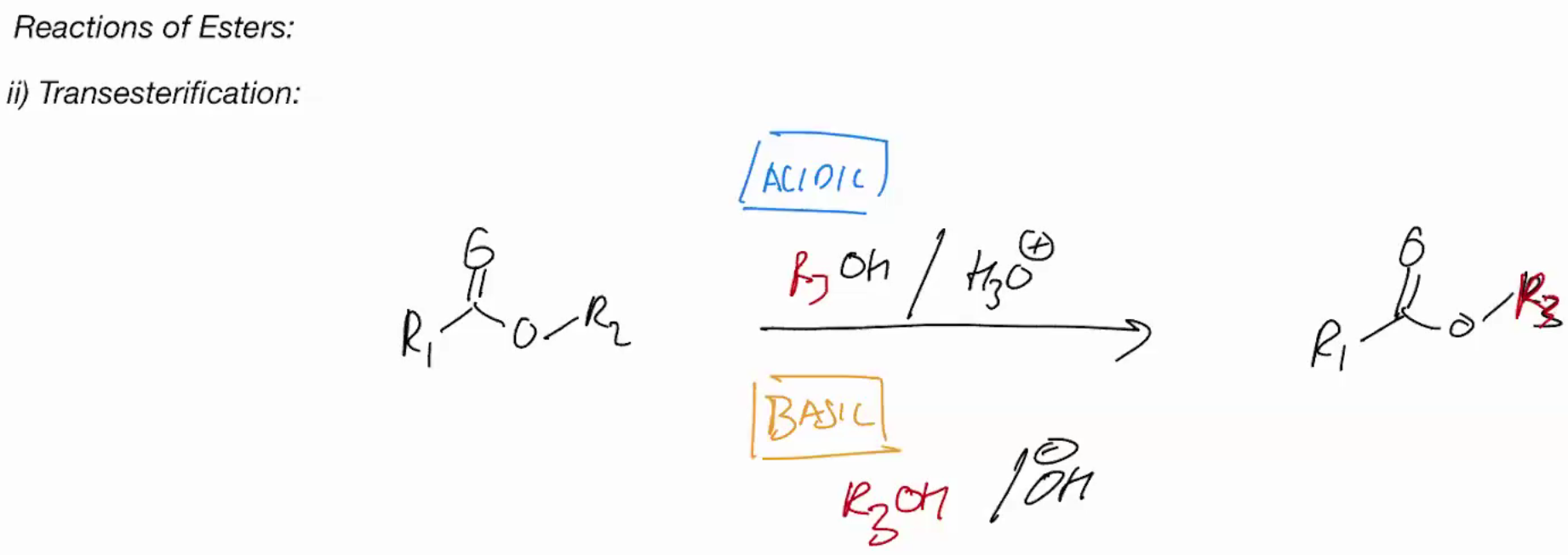
synthesis of esters via SN2
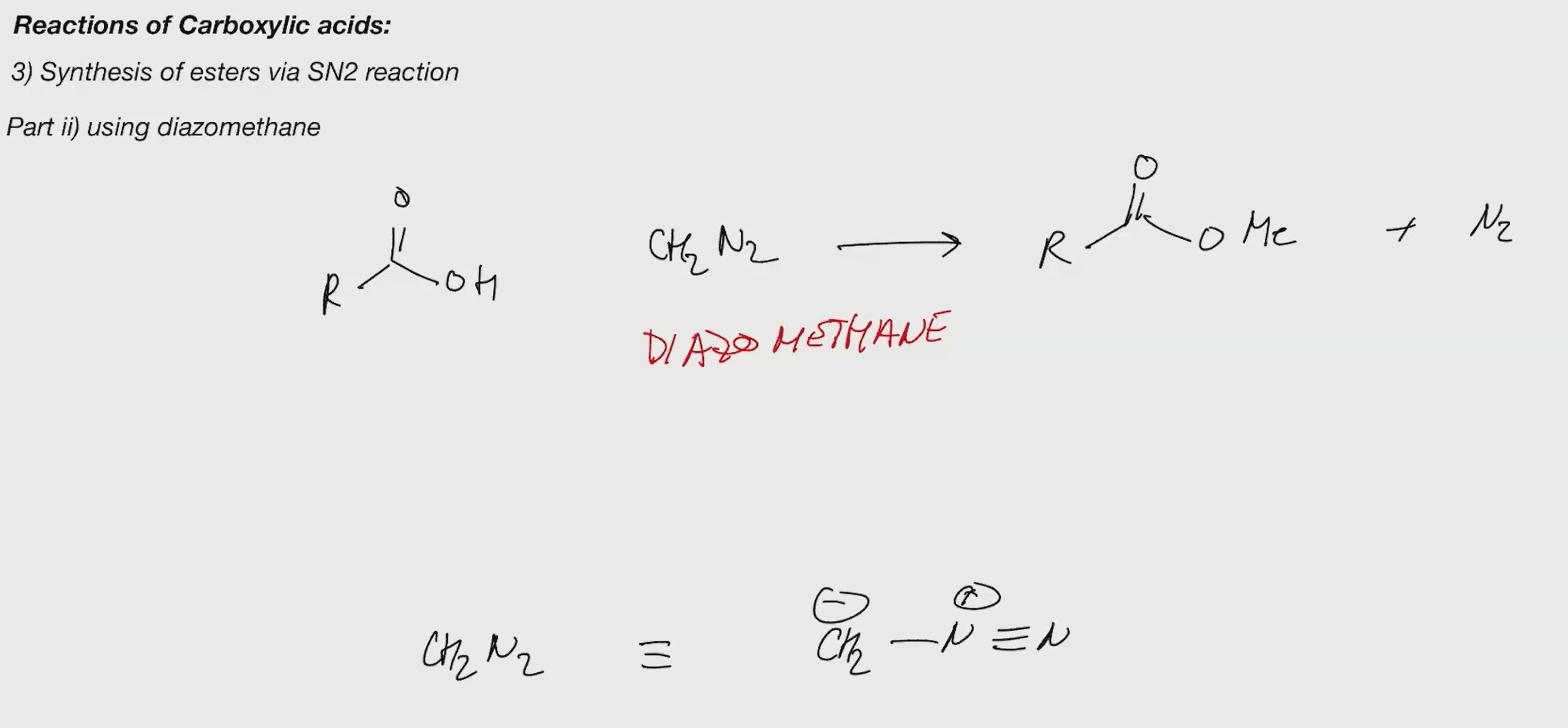
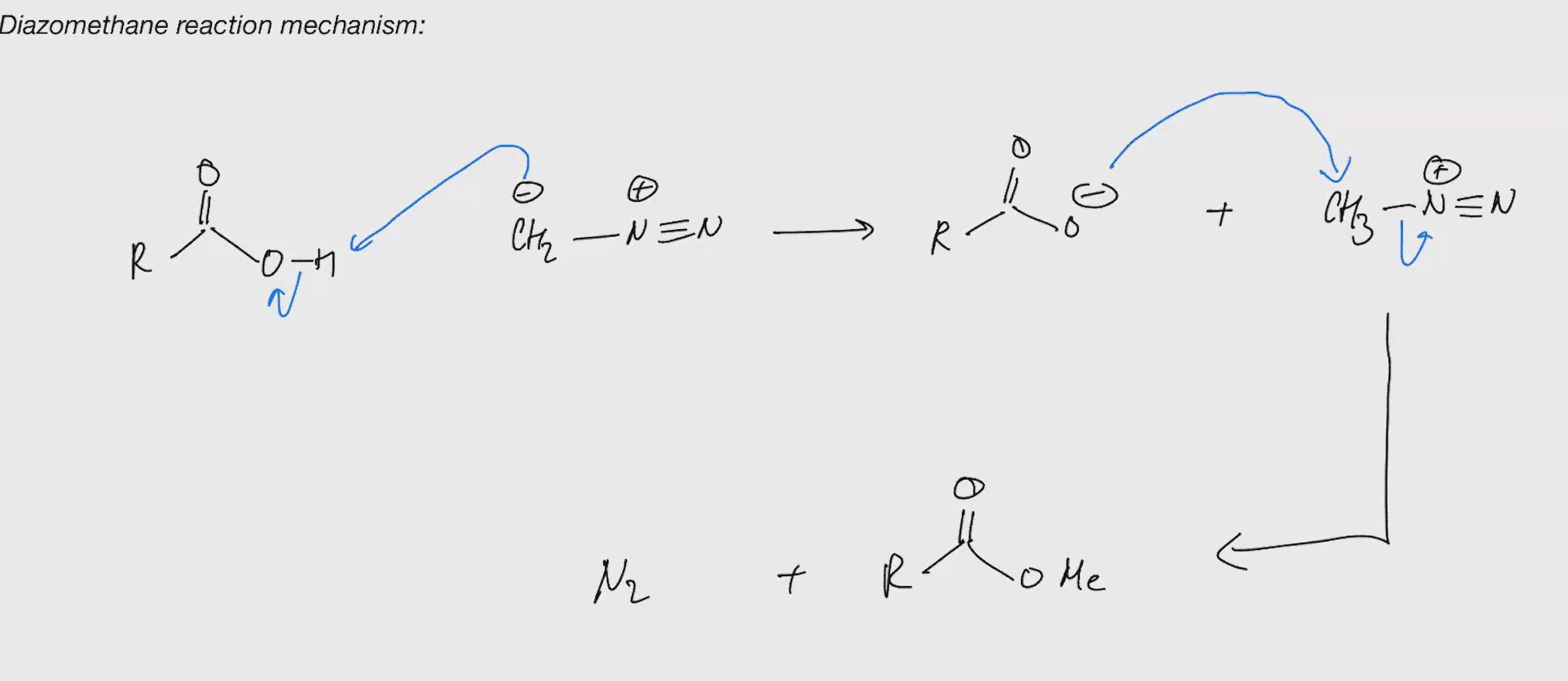
Part II using methane (sn2 ester synthesis)
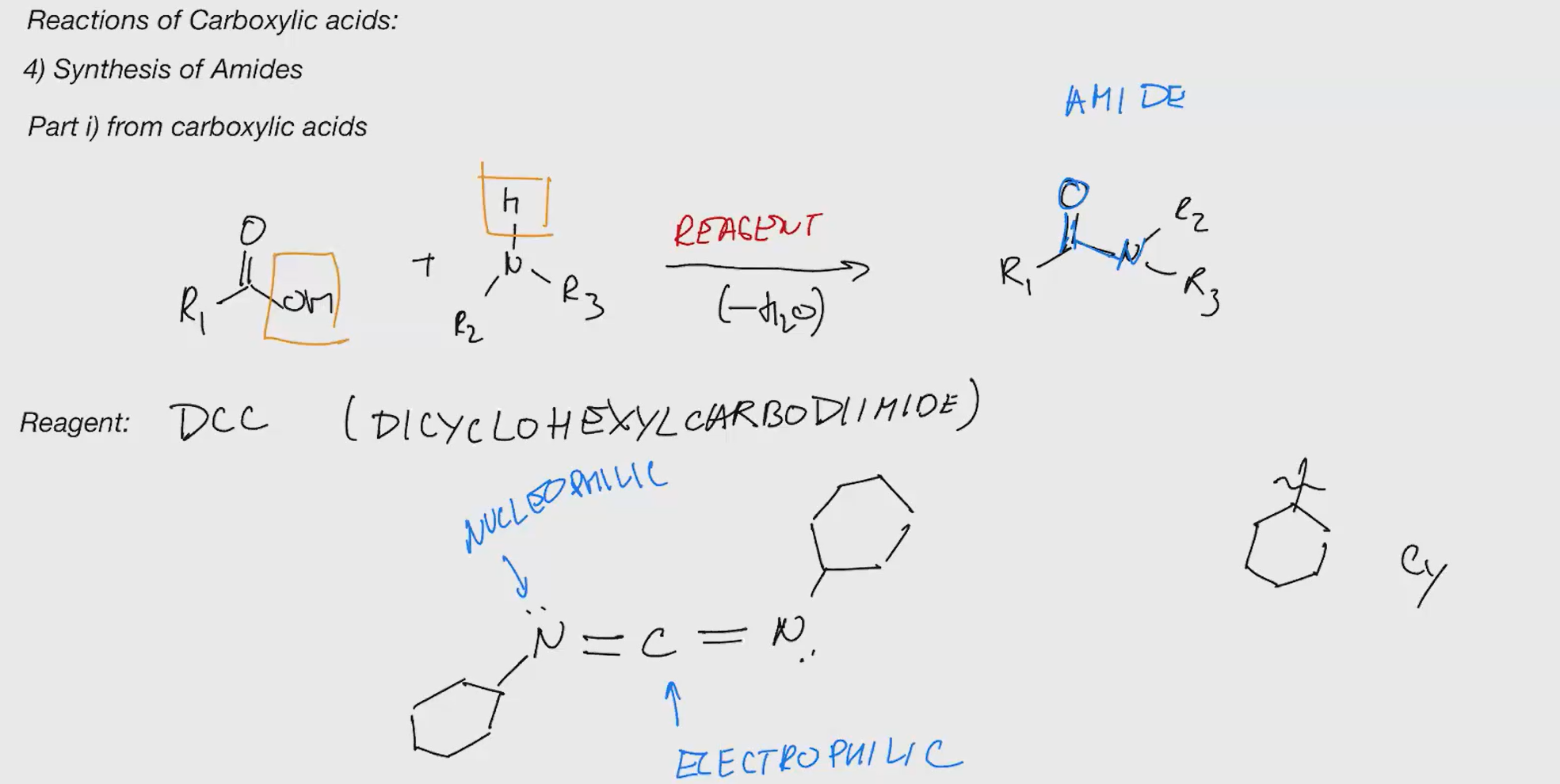
Synthesis of amides
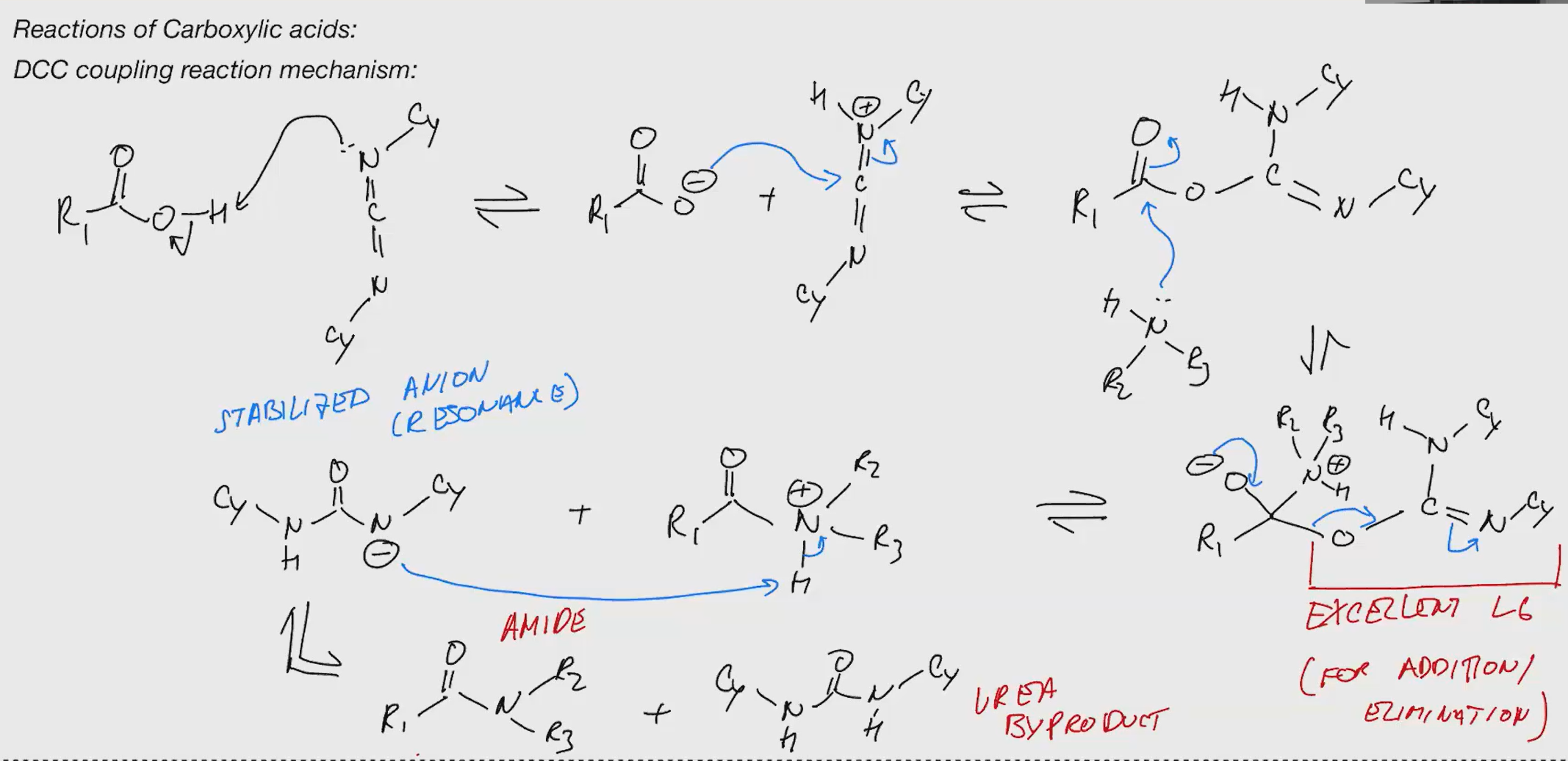
DCC
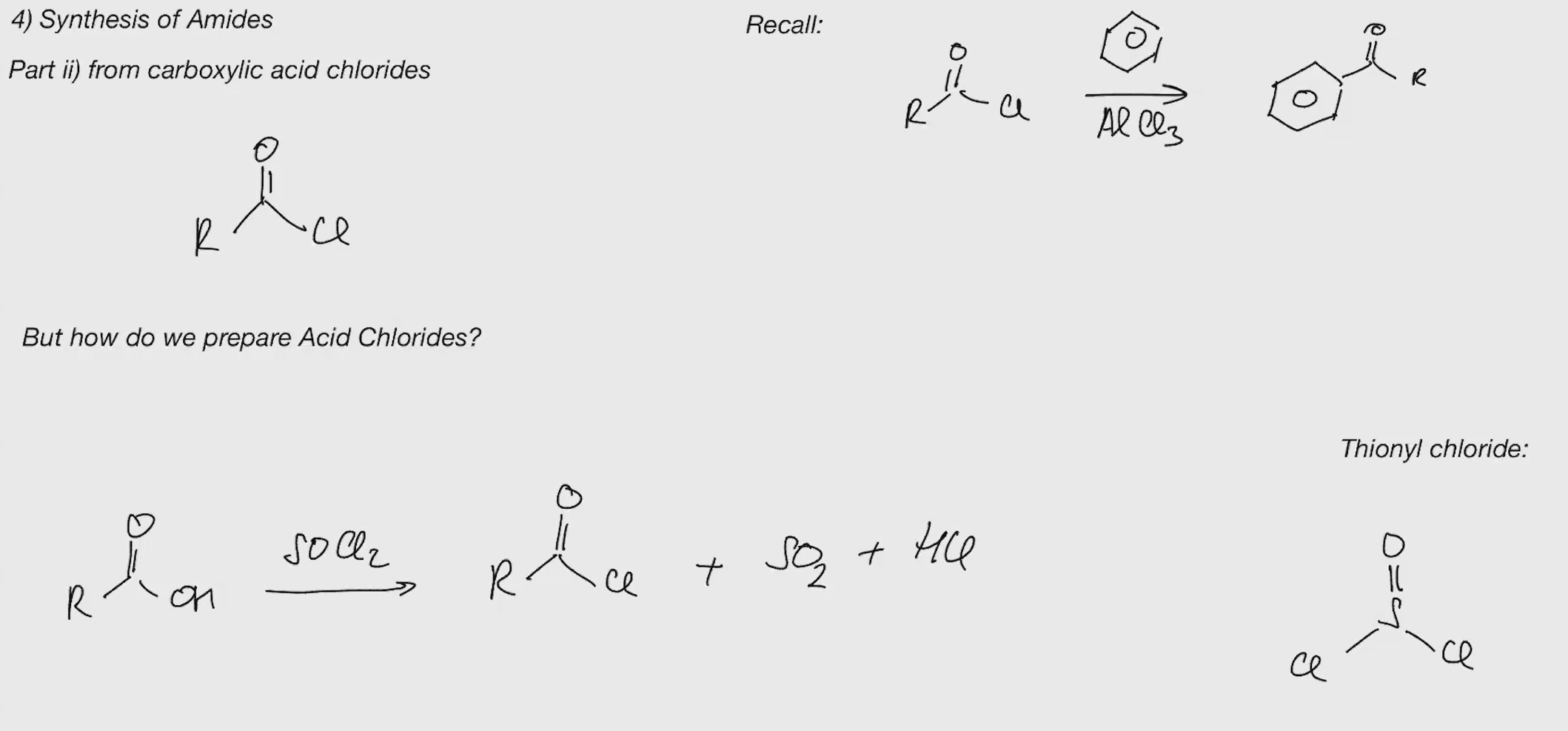
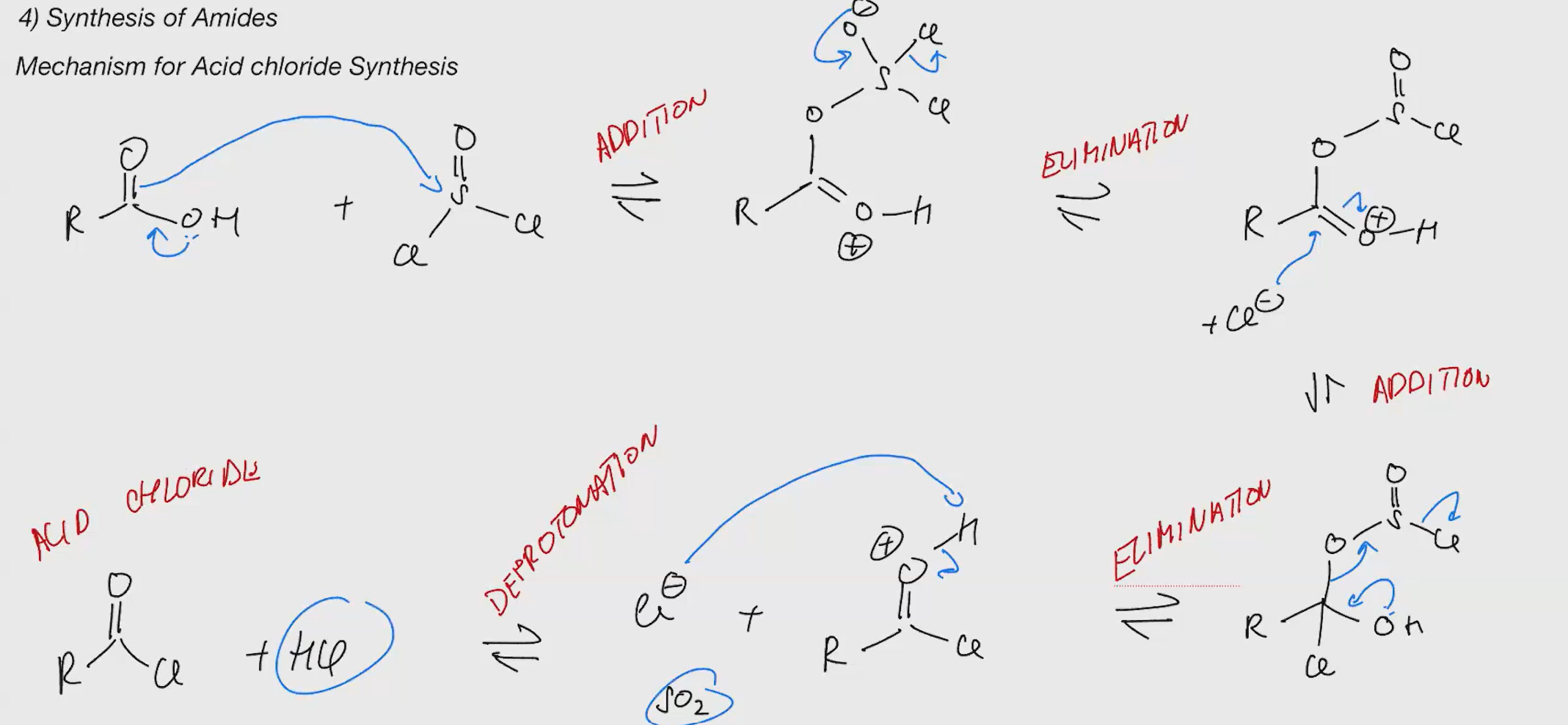
Acid chloride synthesis
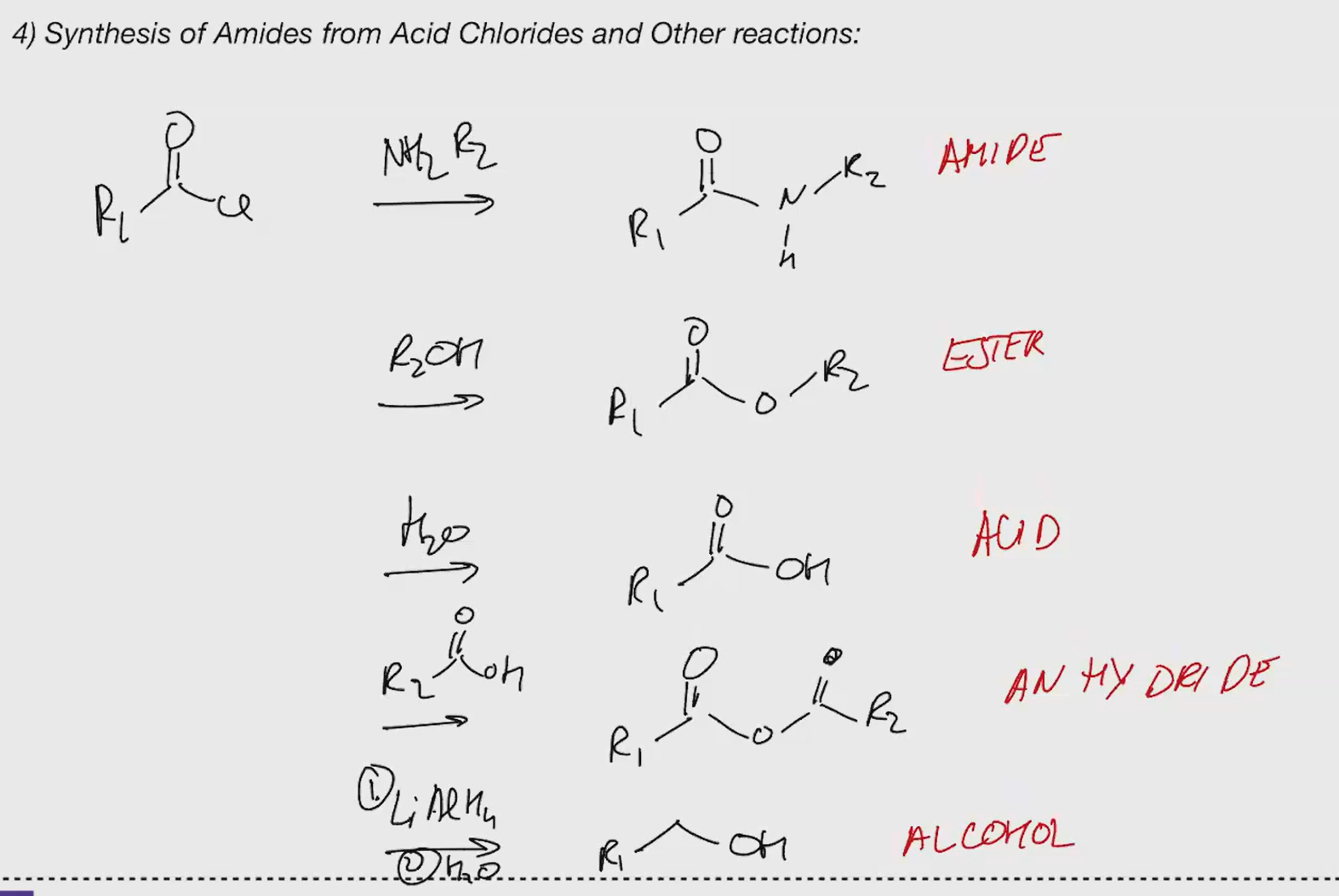
nswiwb
anhydride makeing
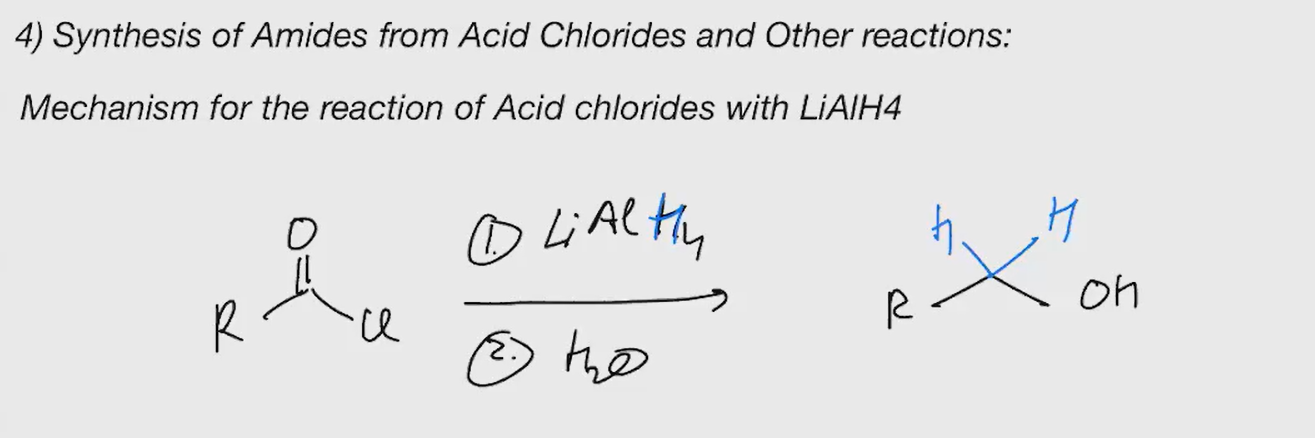
LiAlh4 acid chloride
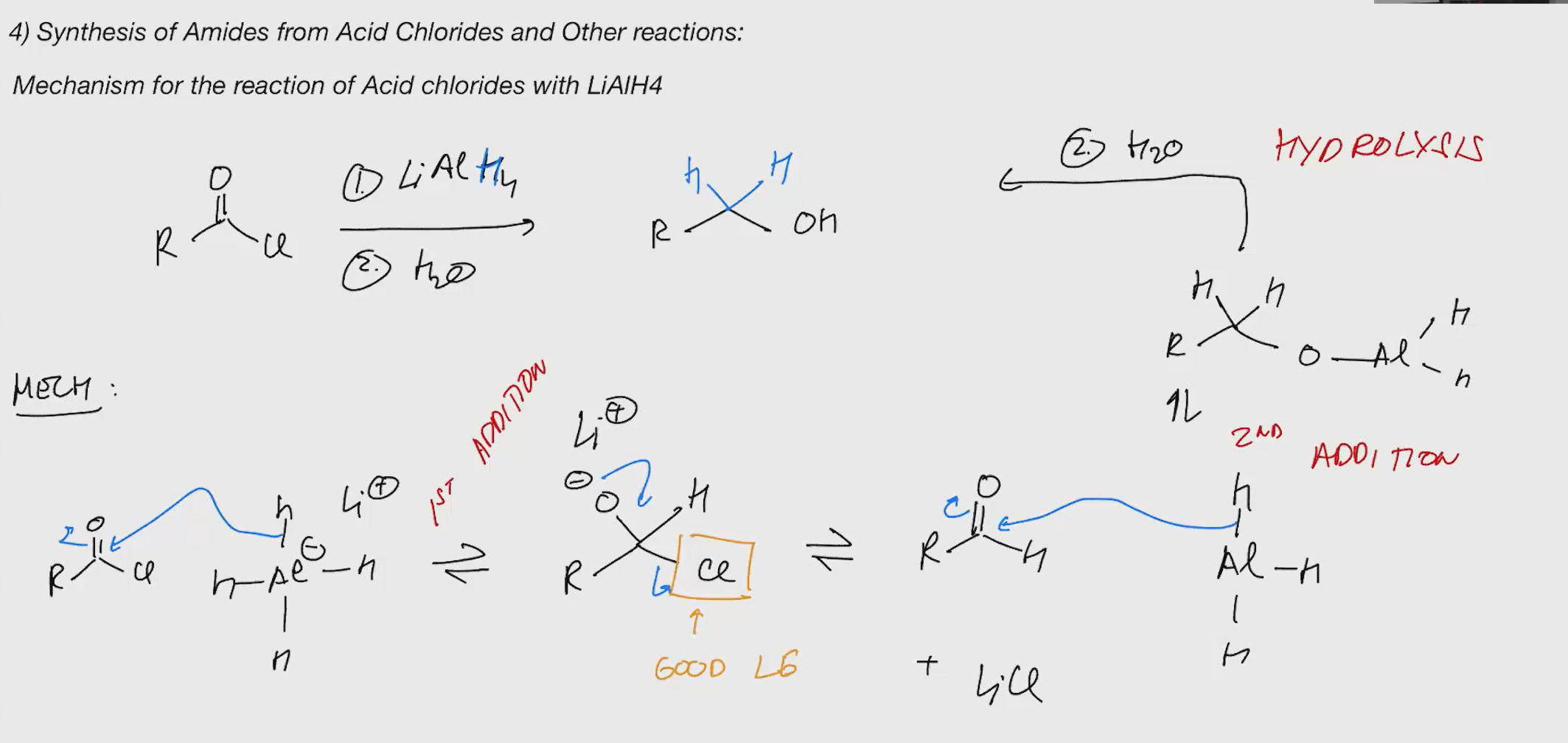
bad
selectivity
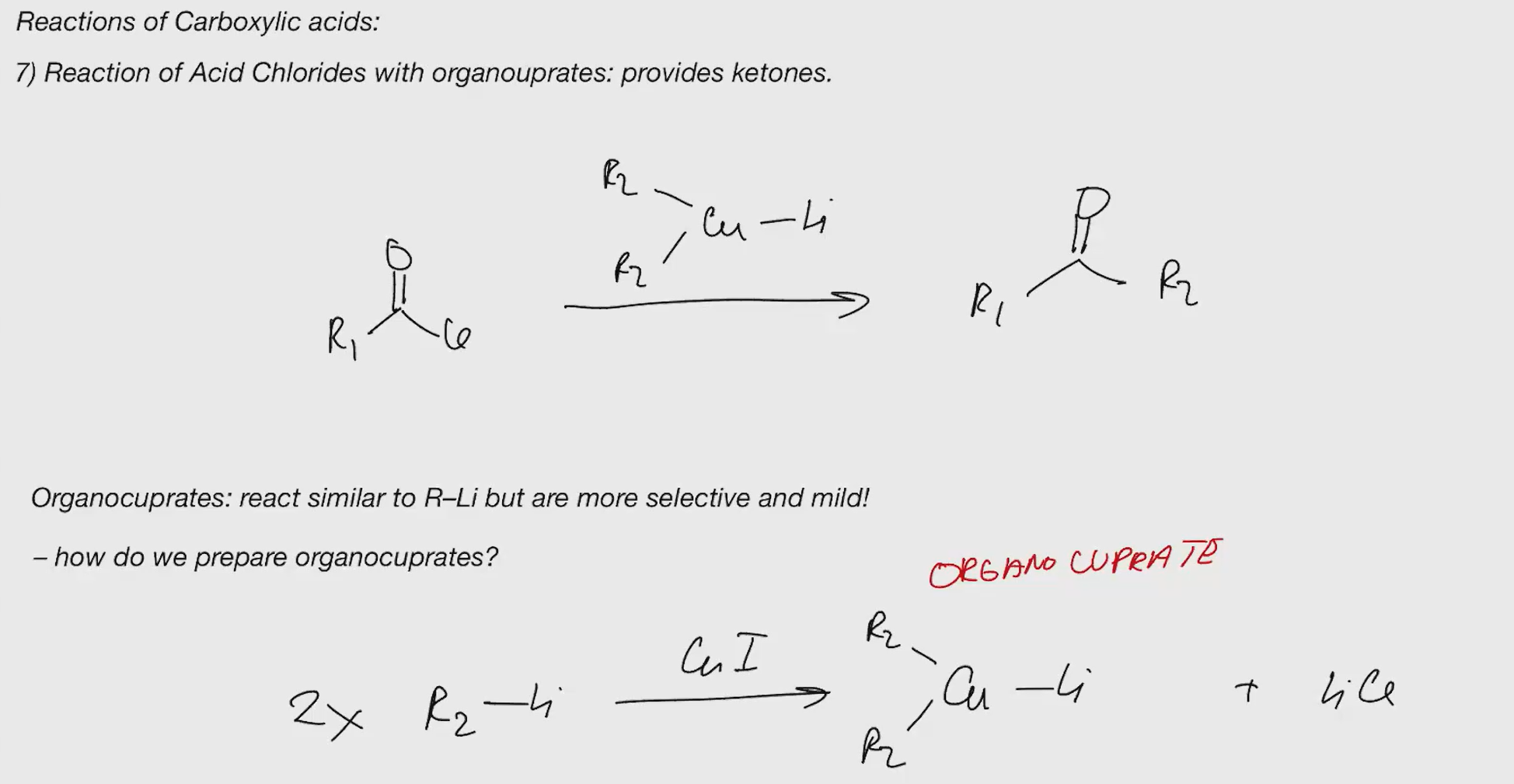

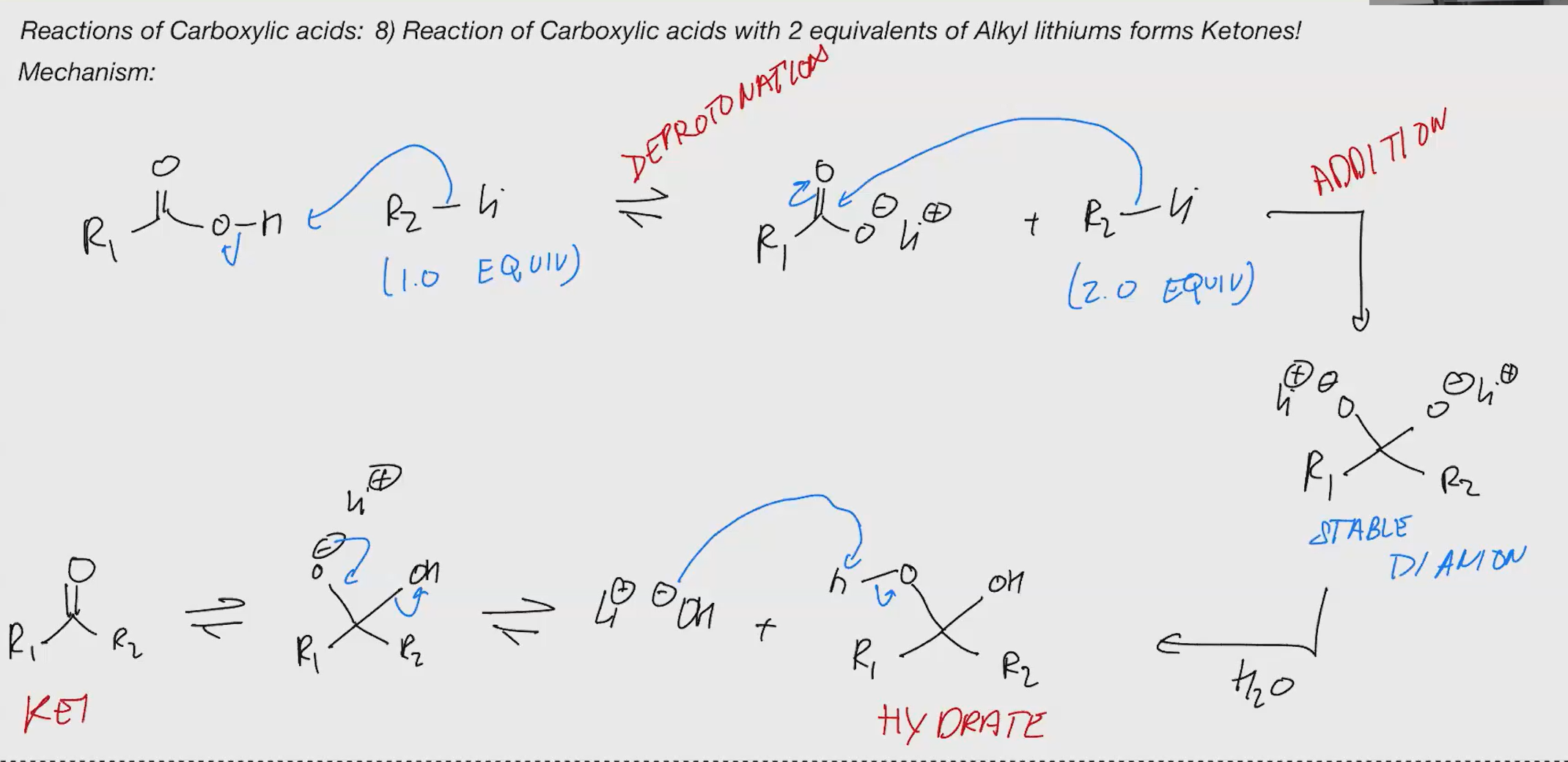
carboxlylic acid to ketone
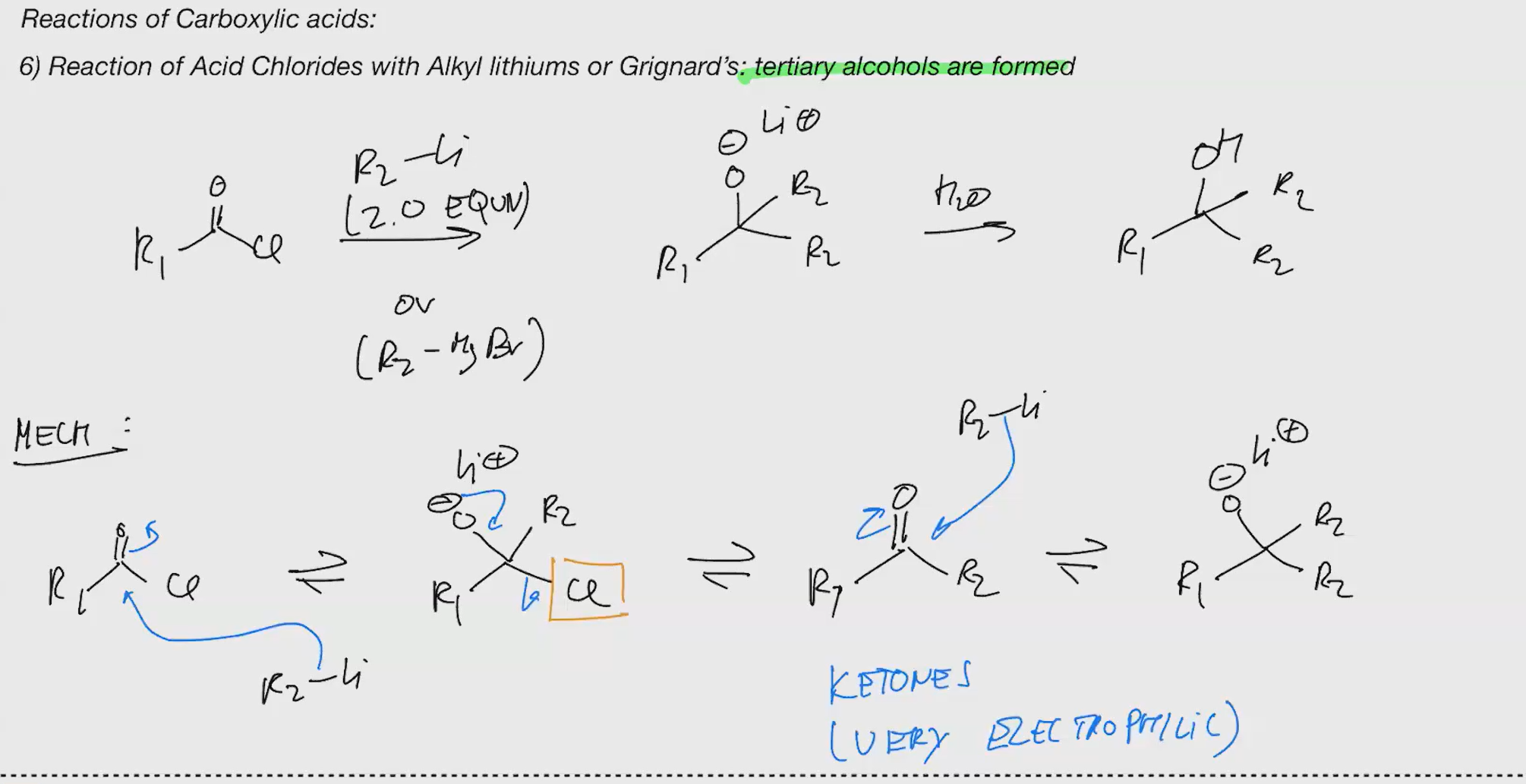
grinard wont work, its negatsivey charged species thats y so u need lith
a random OH spawns somehow
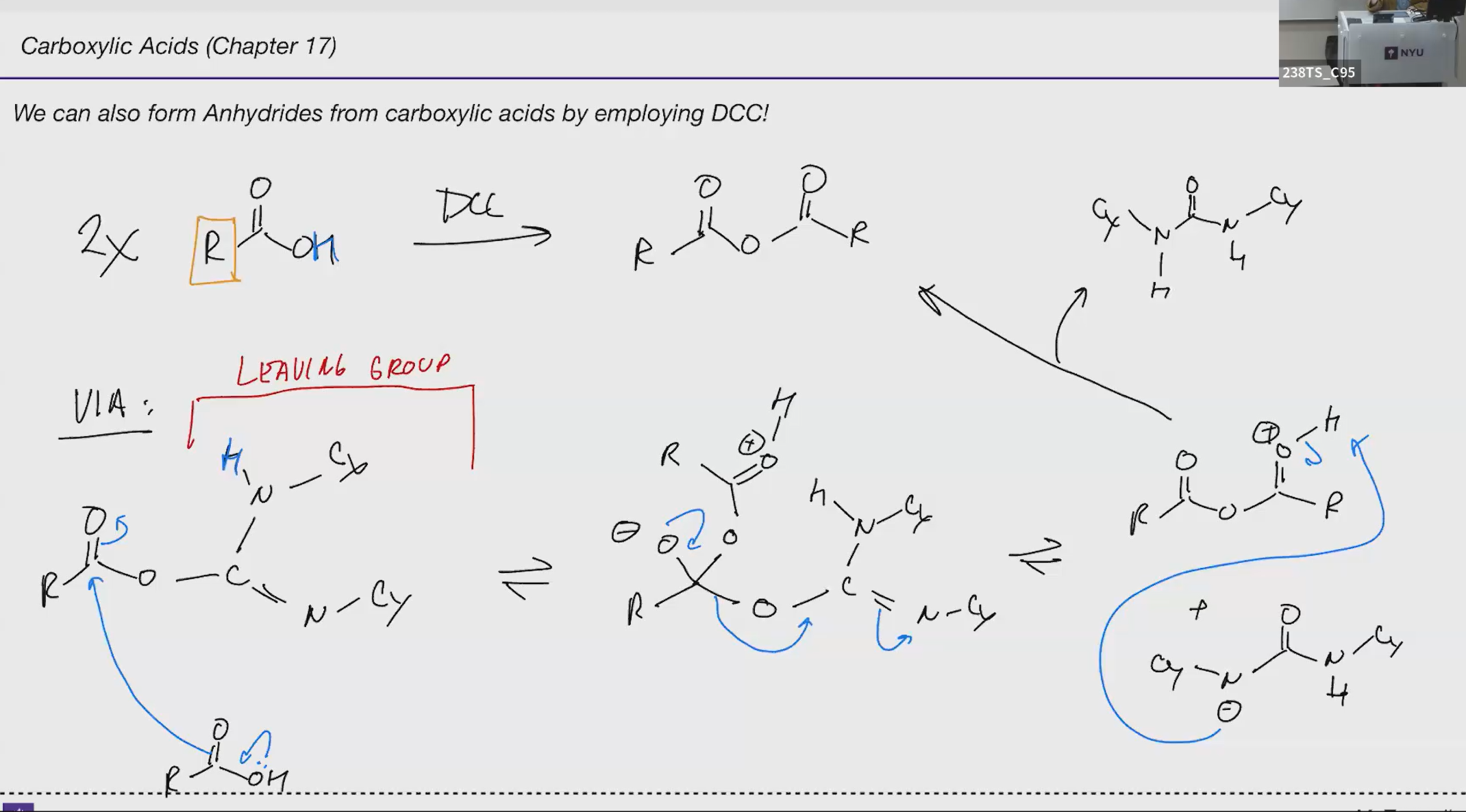
Anhydride DDC
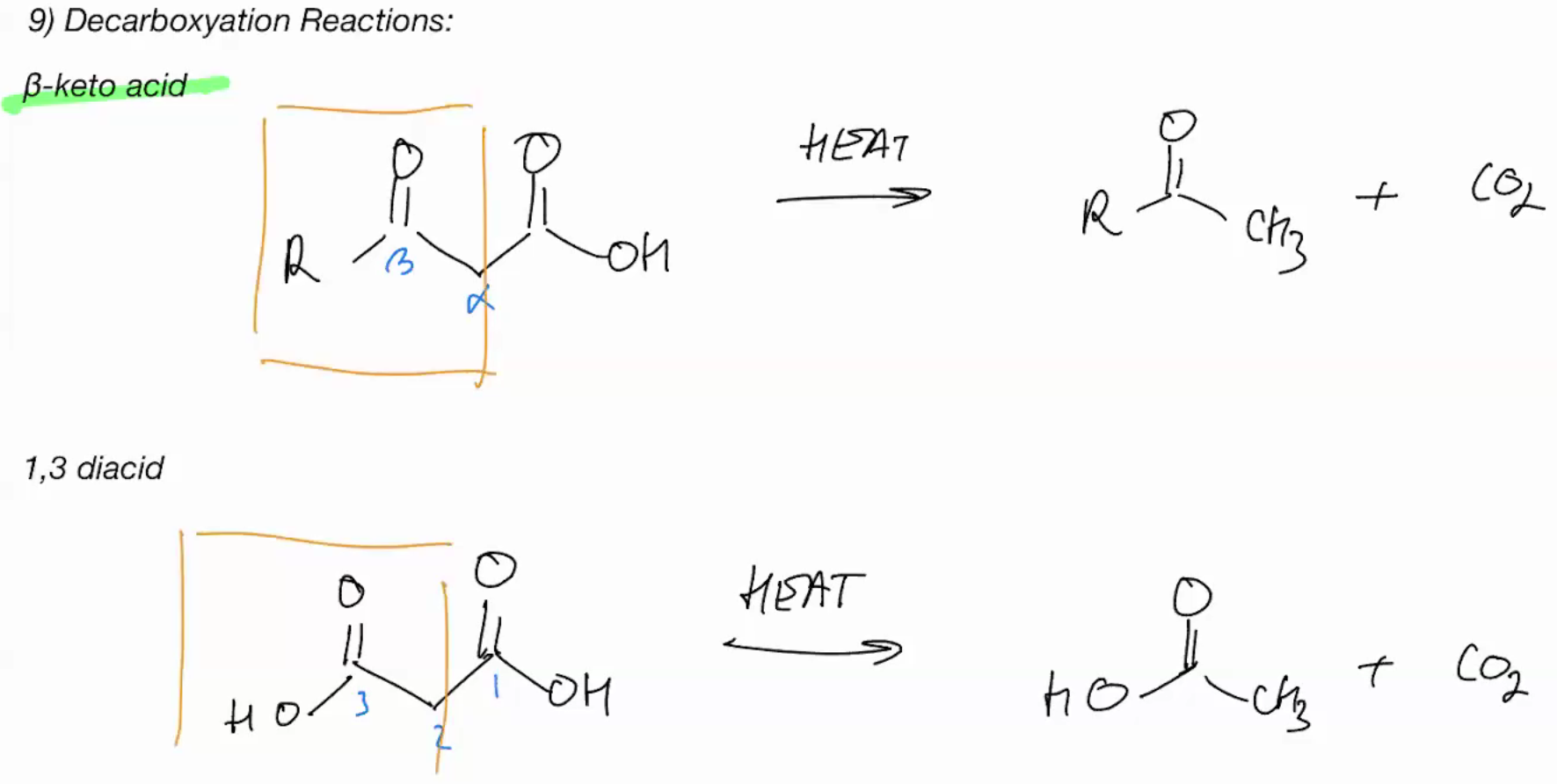
beta keto acid & 1,3 diacid (DECARBOXYLATION)
Use heat we retain left and remove right for co2 and rotate ur bond
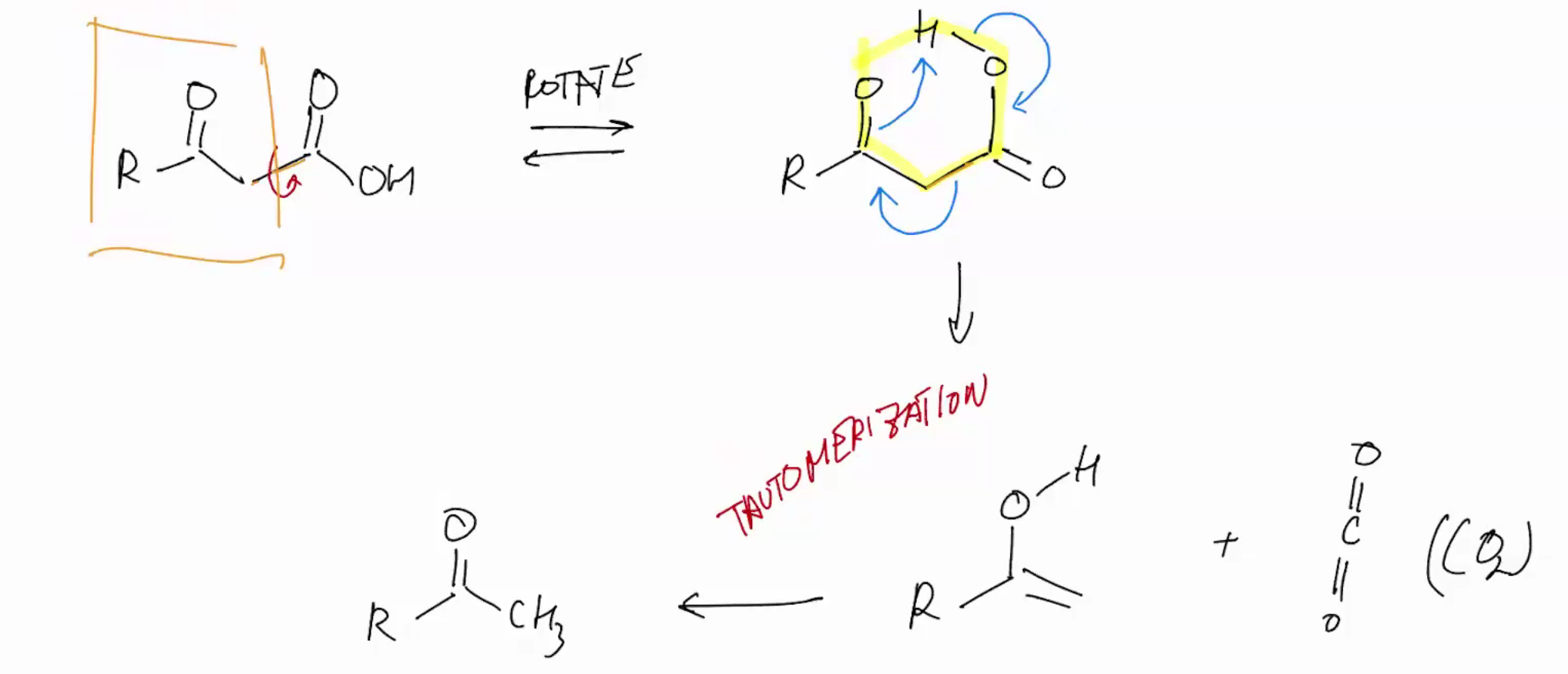
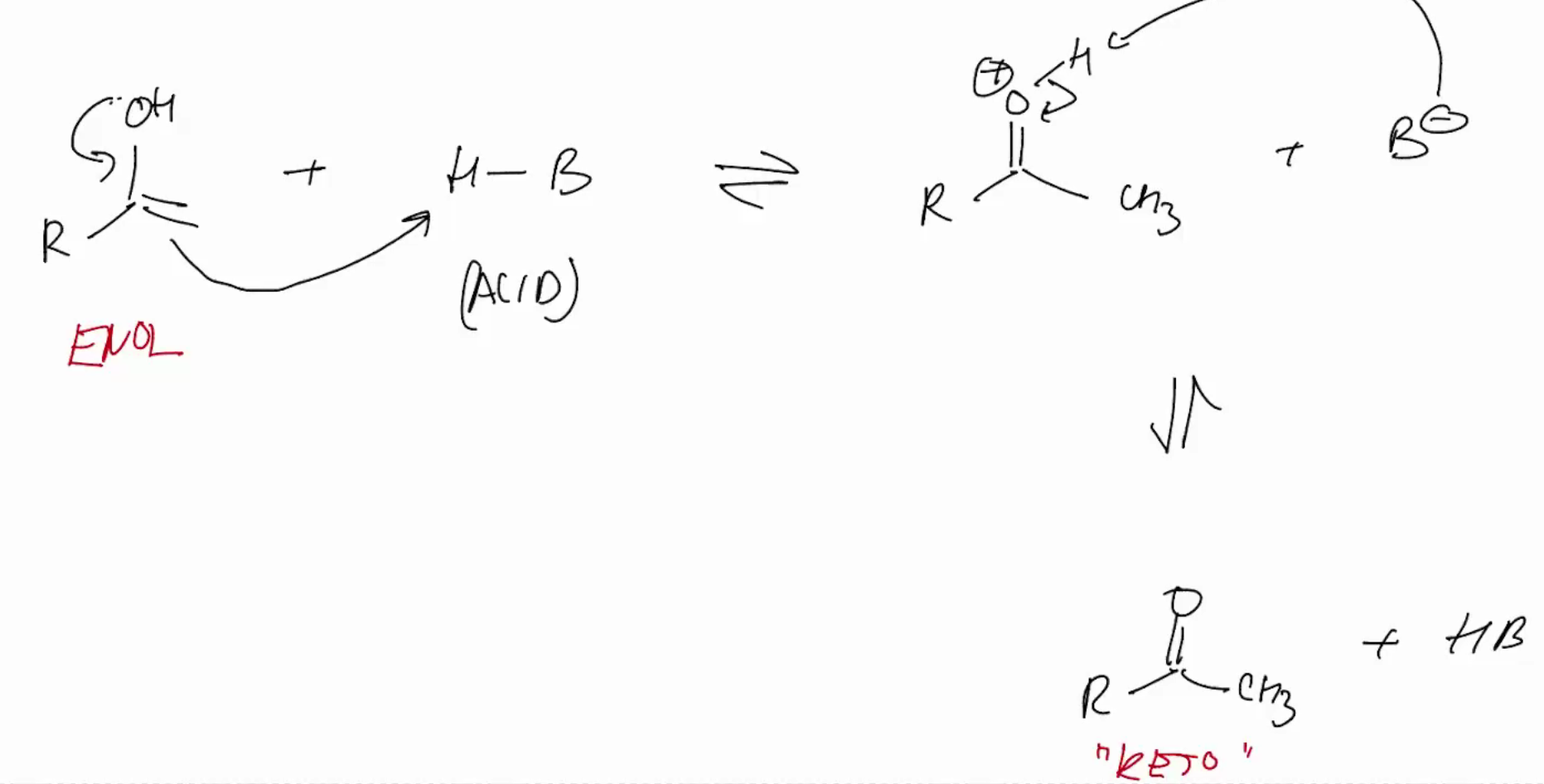
Keto/Enol tautomerization
nomenclature
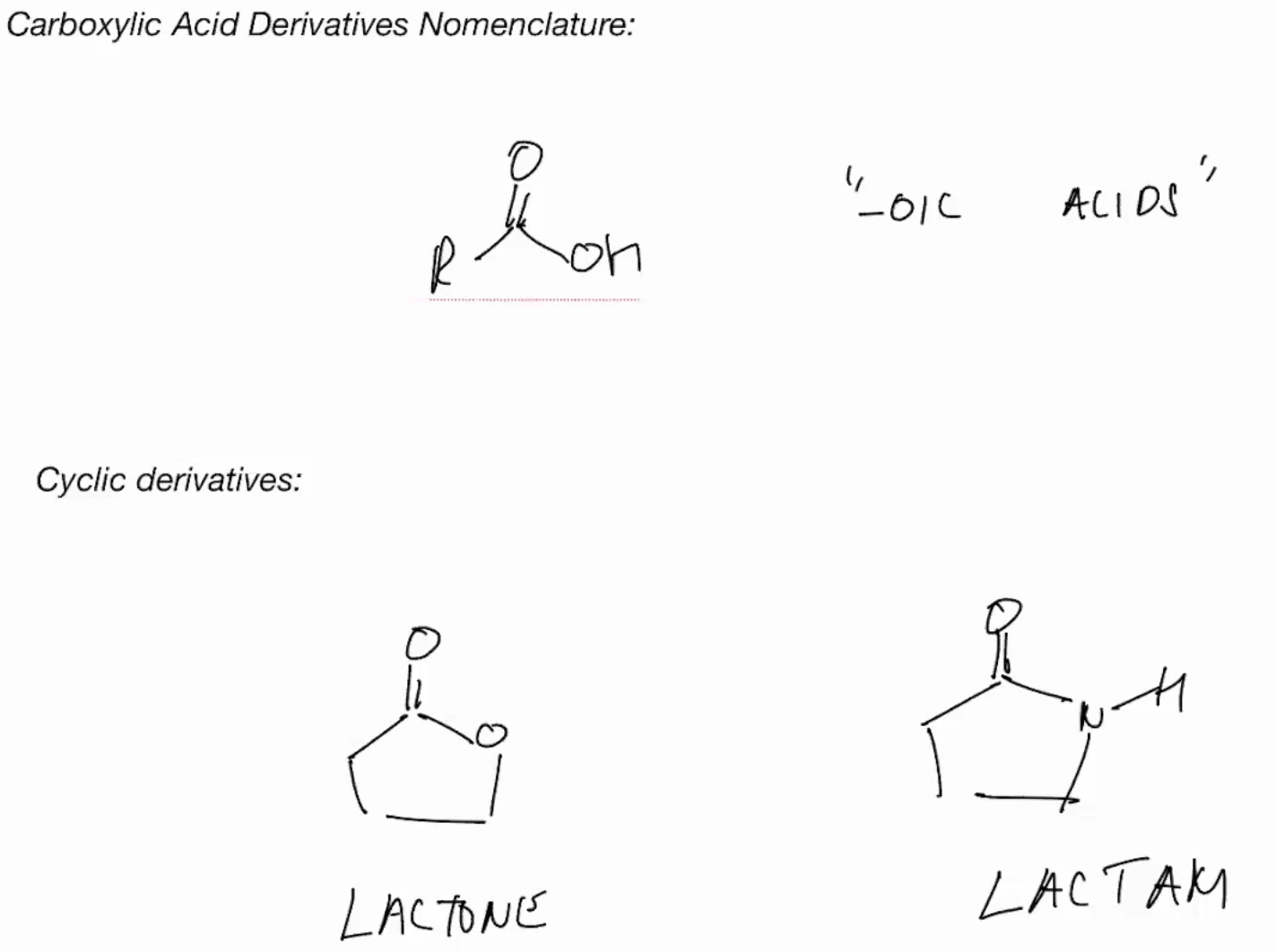
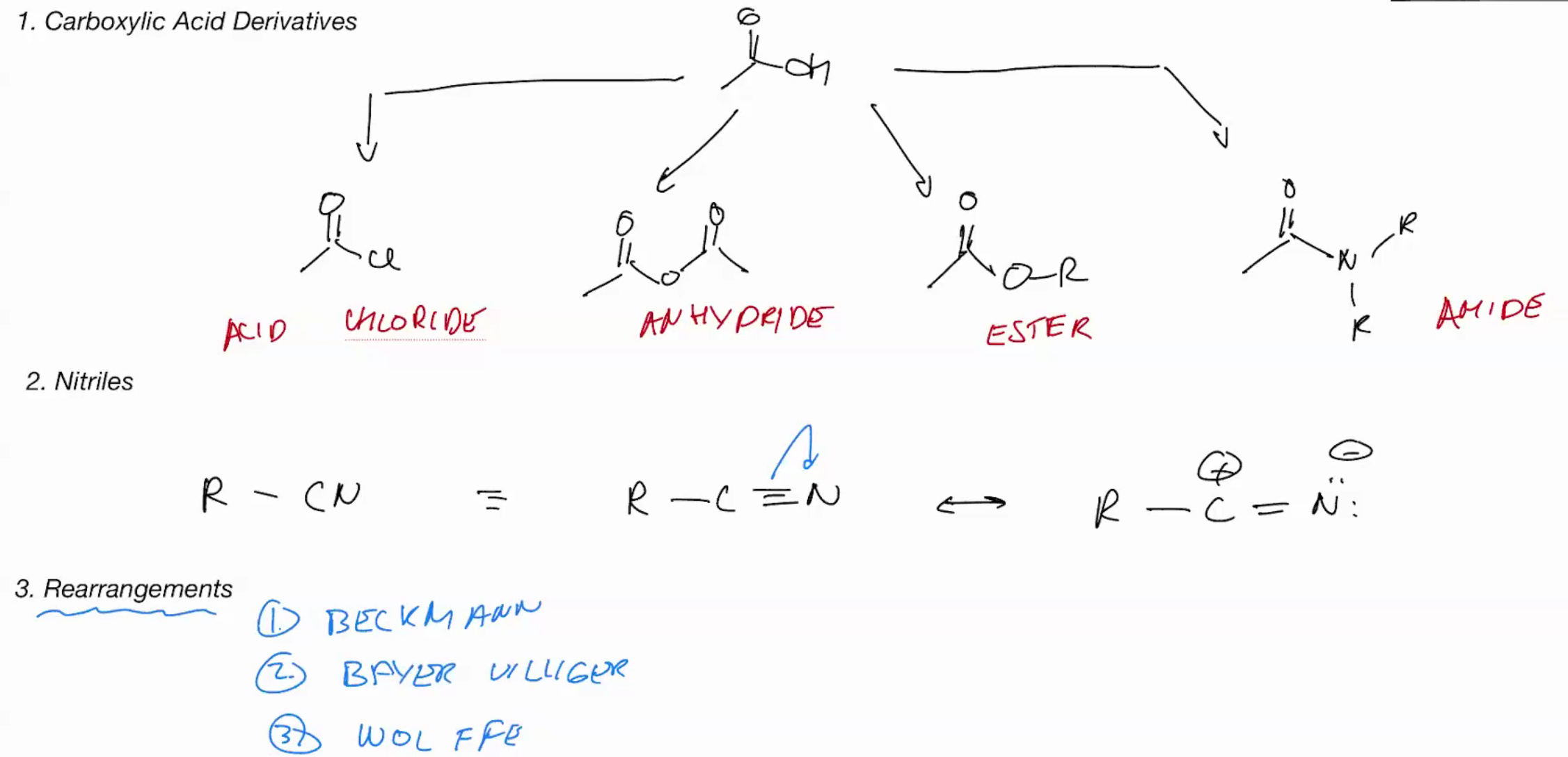
Carboxylic acid derivatives
Acid chloride review
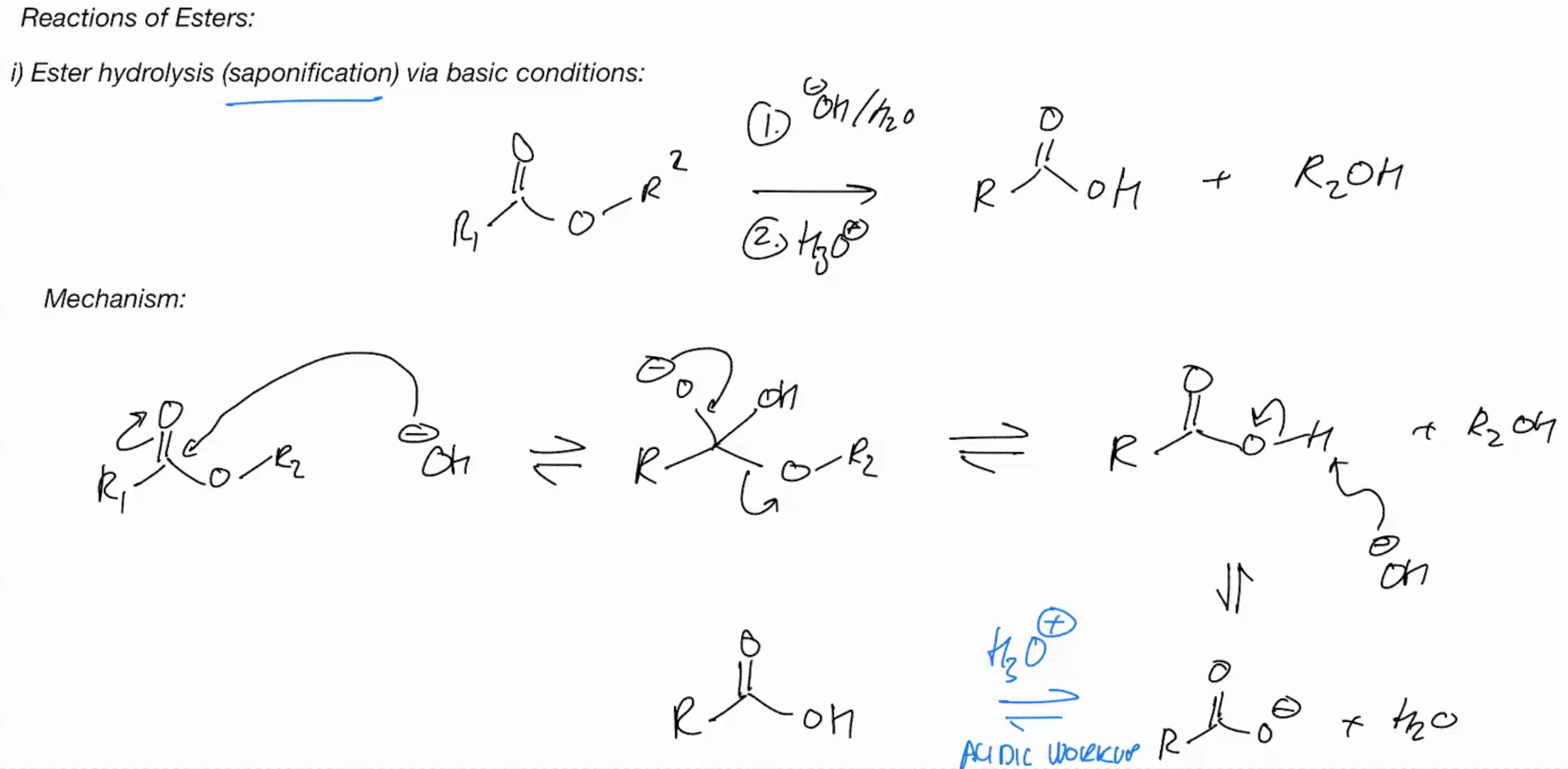
Ester hydrolysis (saponification) BASIC CONDITIONS
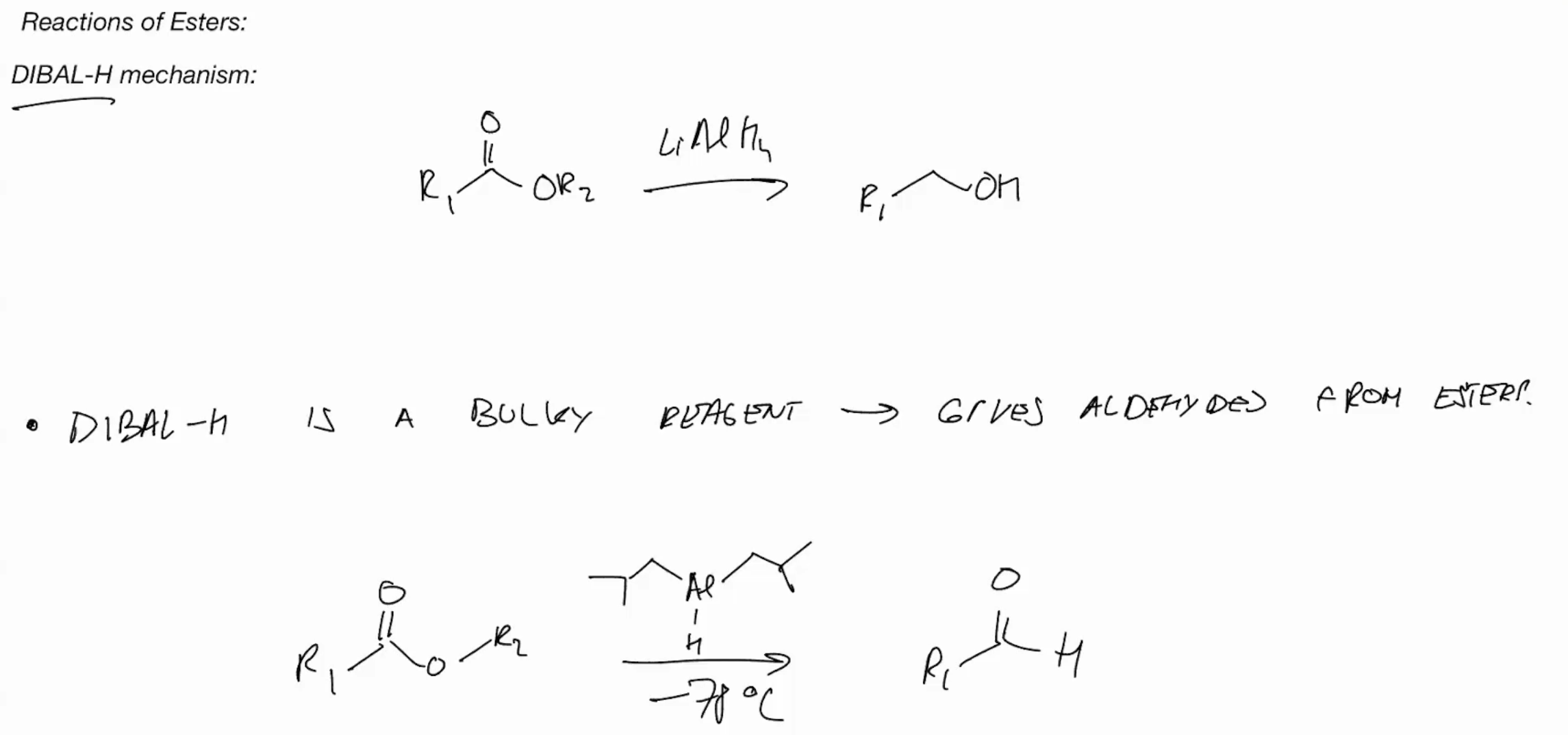
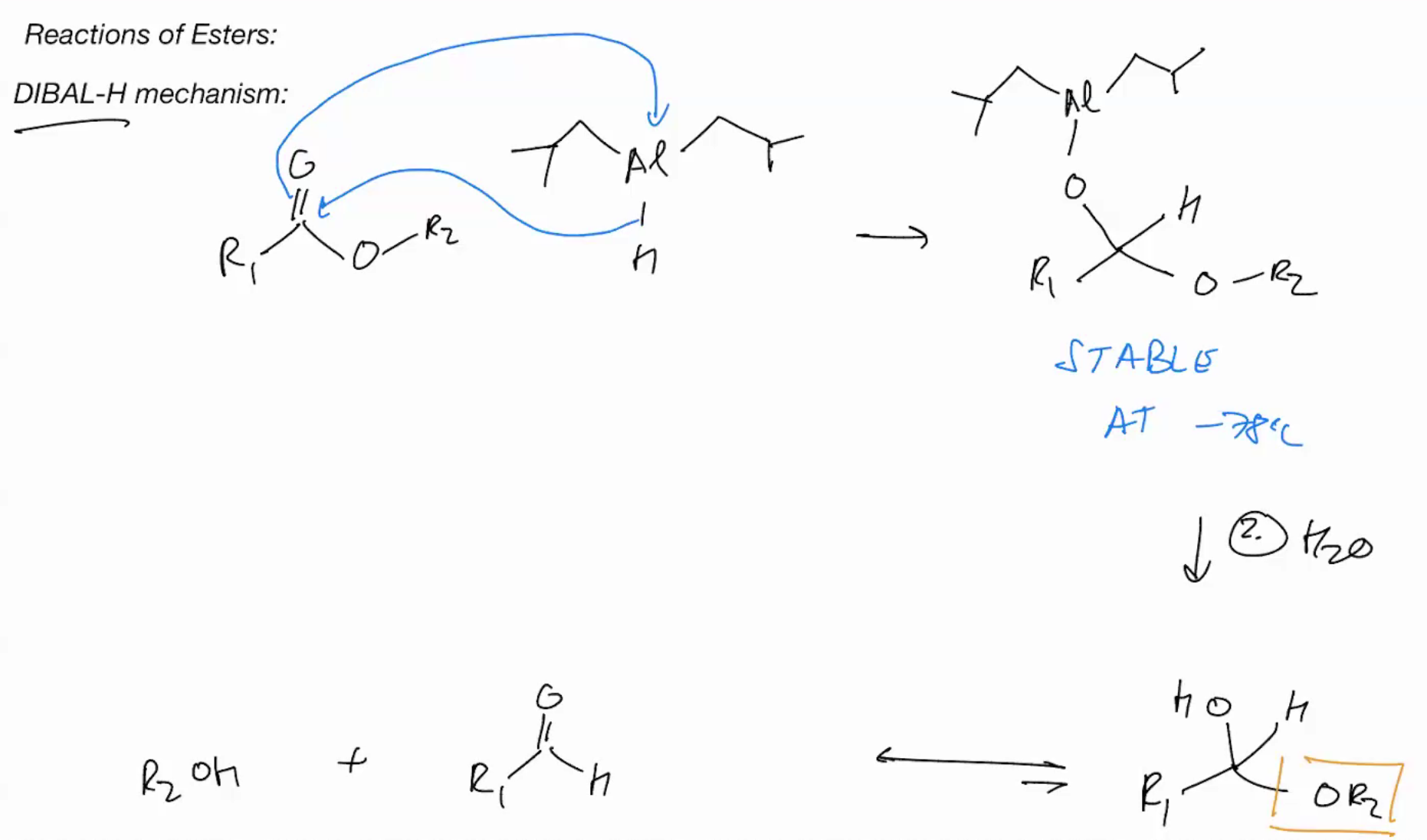
DIBALH (GIVES ALDEHYES FROM ESTERS)
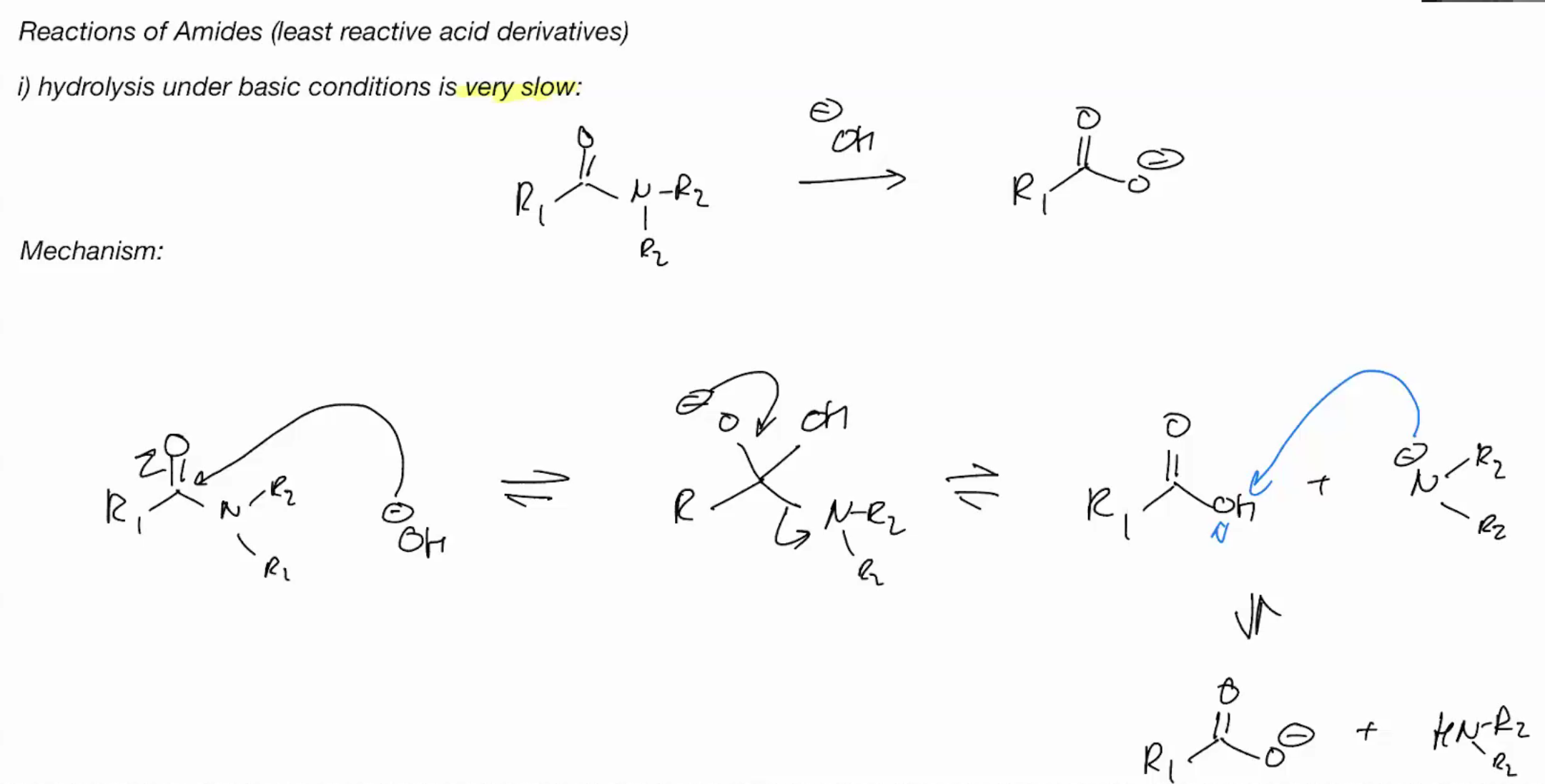
Hydrolysis of amines under BASIC CONDITIONS (SLOWW ASF)
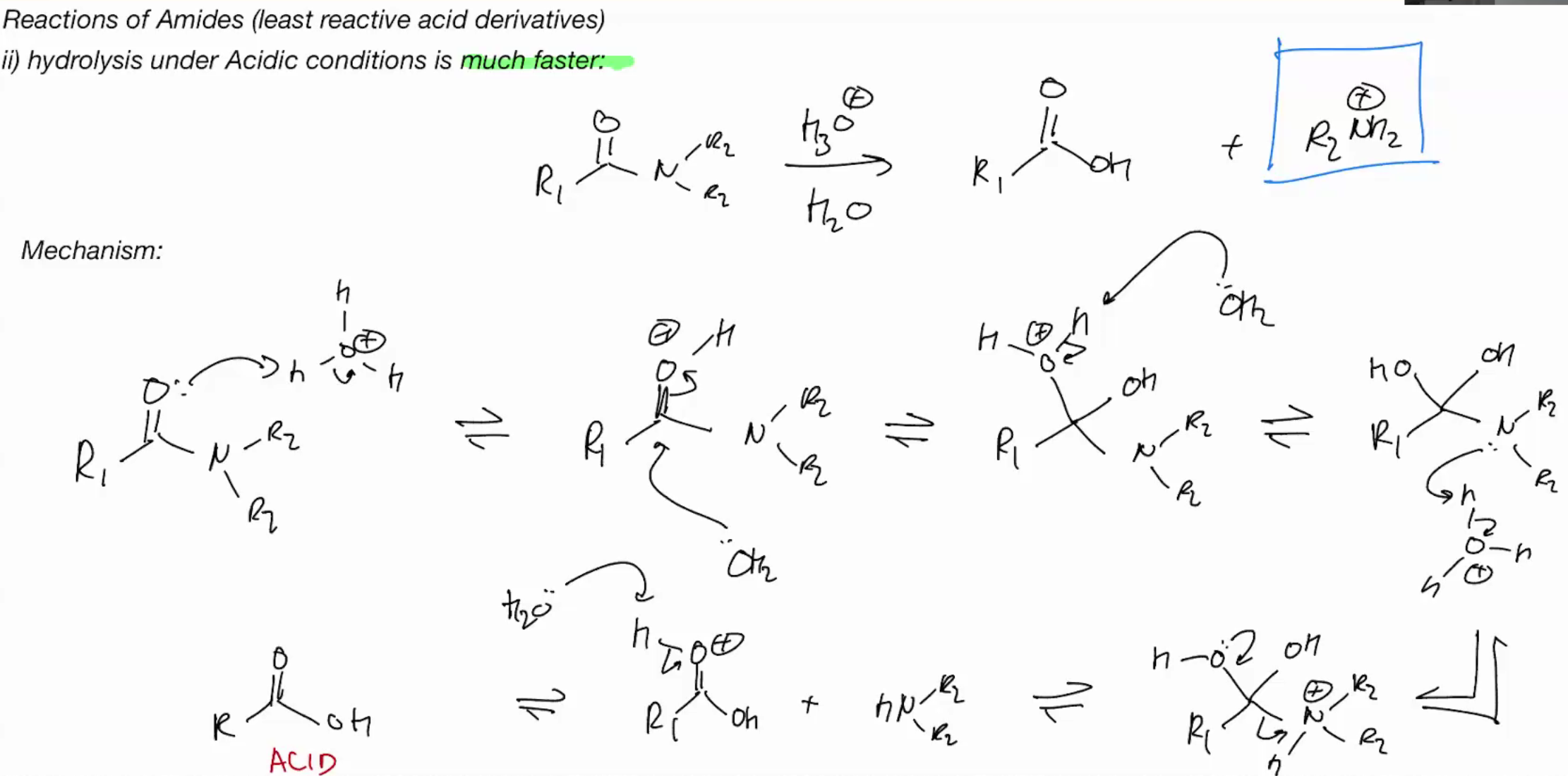
Hydrolysis of amines under ACID CONDITIONS (FAST CUZ MAKING IT BETTER ELETROPHILE OR SUMSHI)
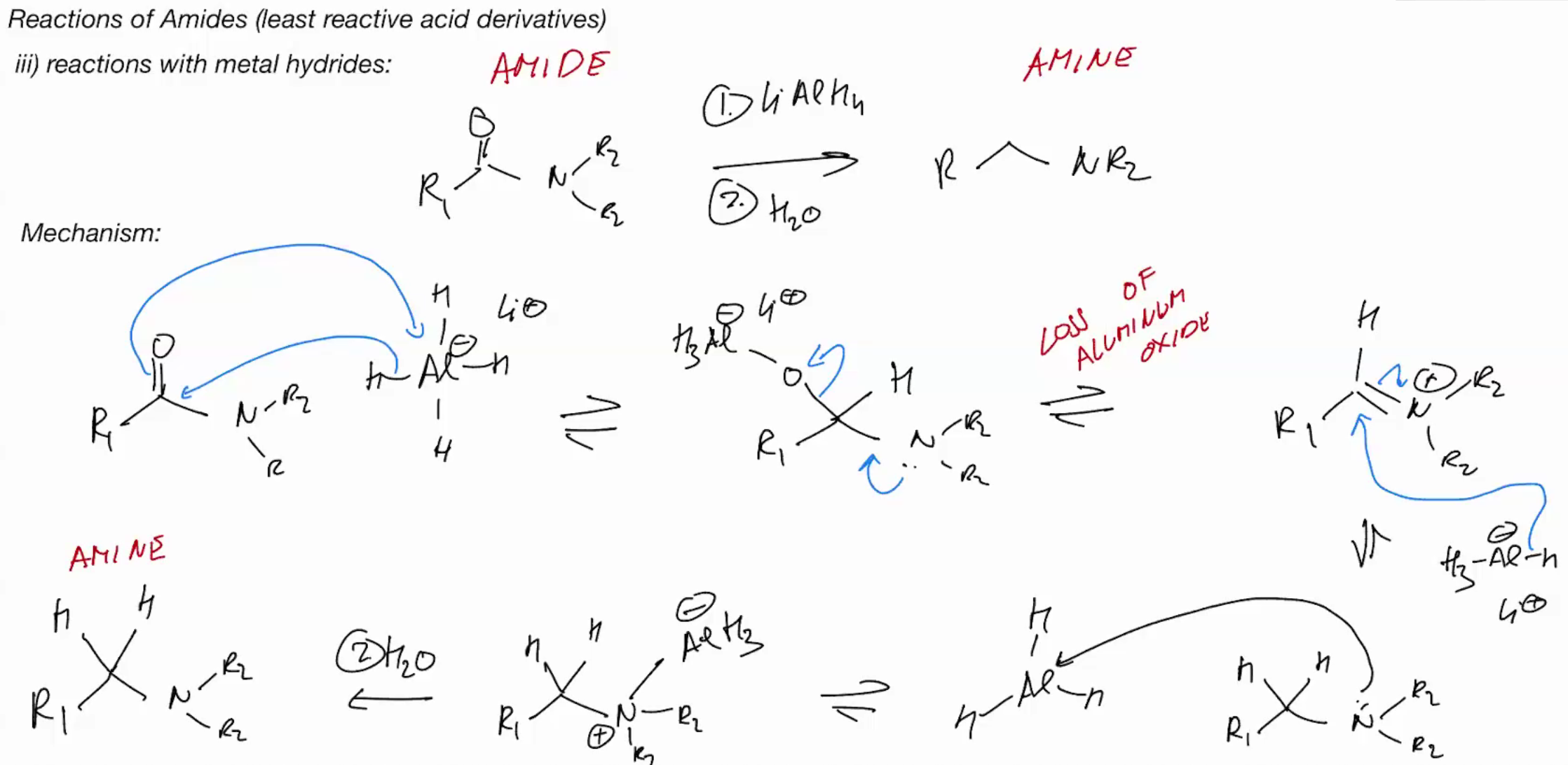
Amide TO AMINE with metal hydride (RMBR THE QUENCHHH)
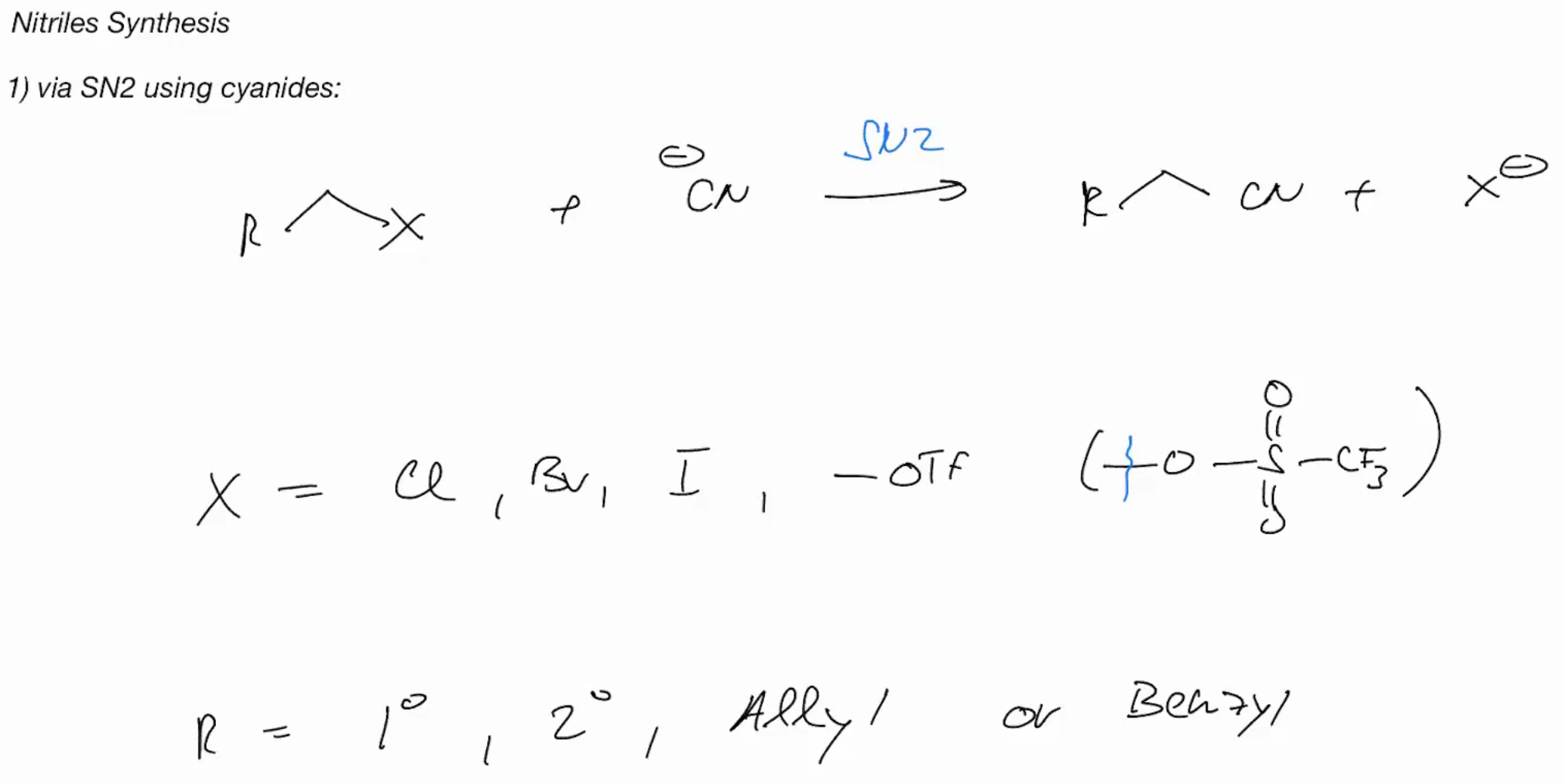
Reaction Of Nitriles (SYNTHESIS)
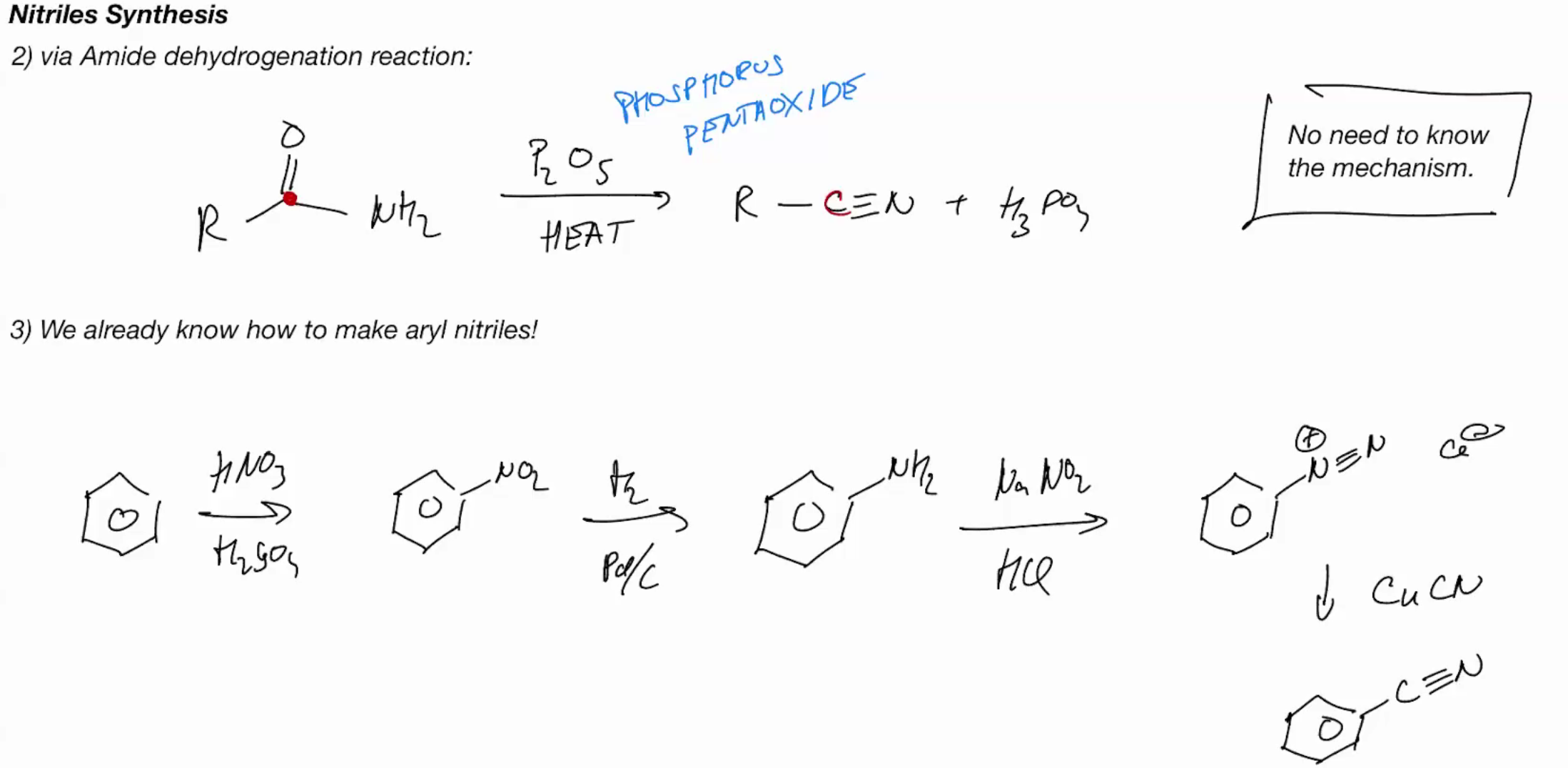
Nitrile Synthesis (amide dehydrogentation)
Phosphorous pentaoxide (P2O5) + heat —→ h3po3 + R - CN Triplebond
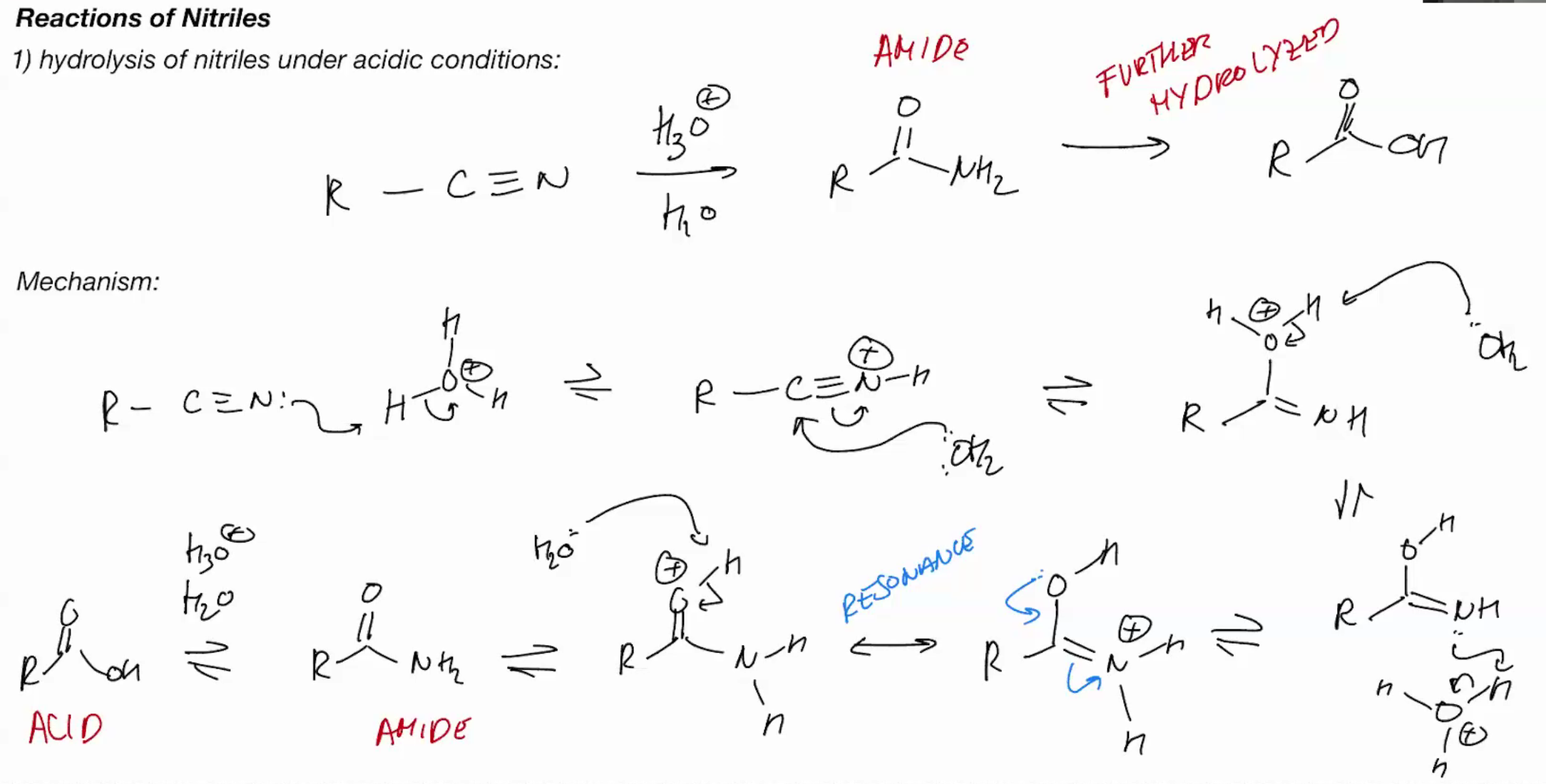
Hydrolysis of nitriles (ACIDIC CONDITIONS) (SKILS AMIDE GOES TO CARBOXLYLLIC ACID)
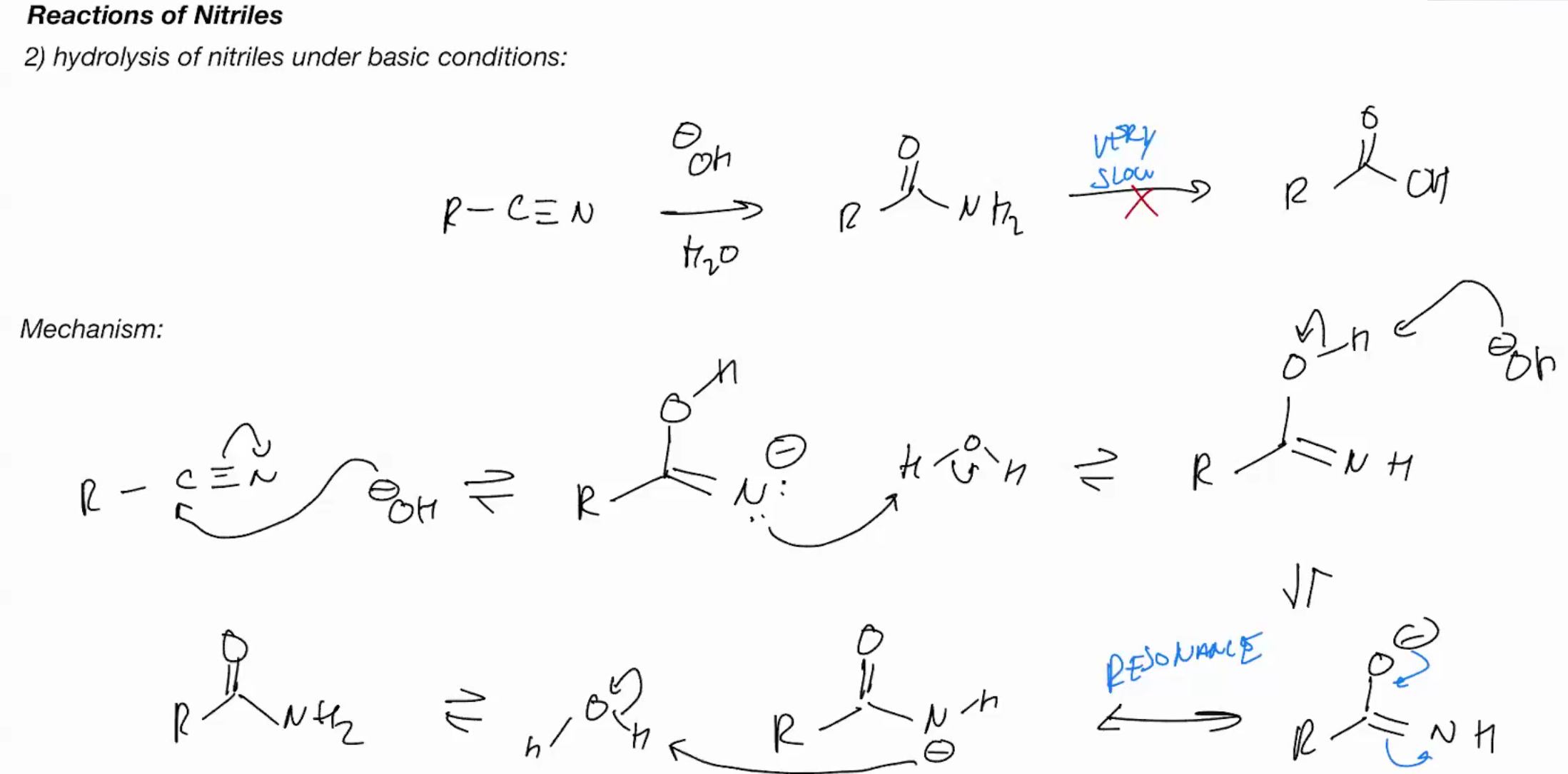
Hydrolysis of nitriles (BASIC CONDITIONS) (STOPS AT AMIDE)

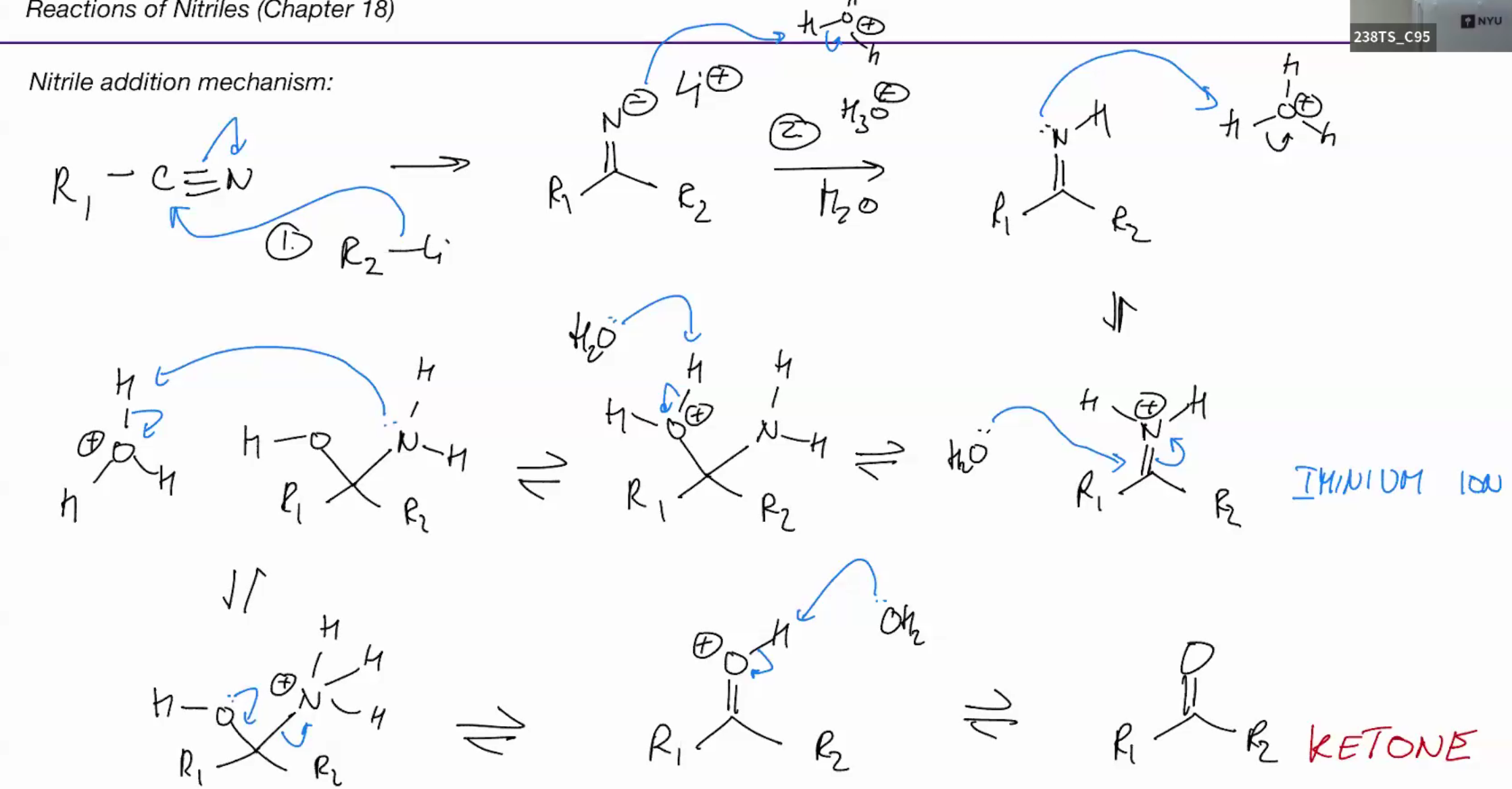
Nitriles with alyk lith grinards make ketone
mechanism
Reagaents - r-li and h3o h20
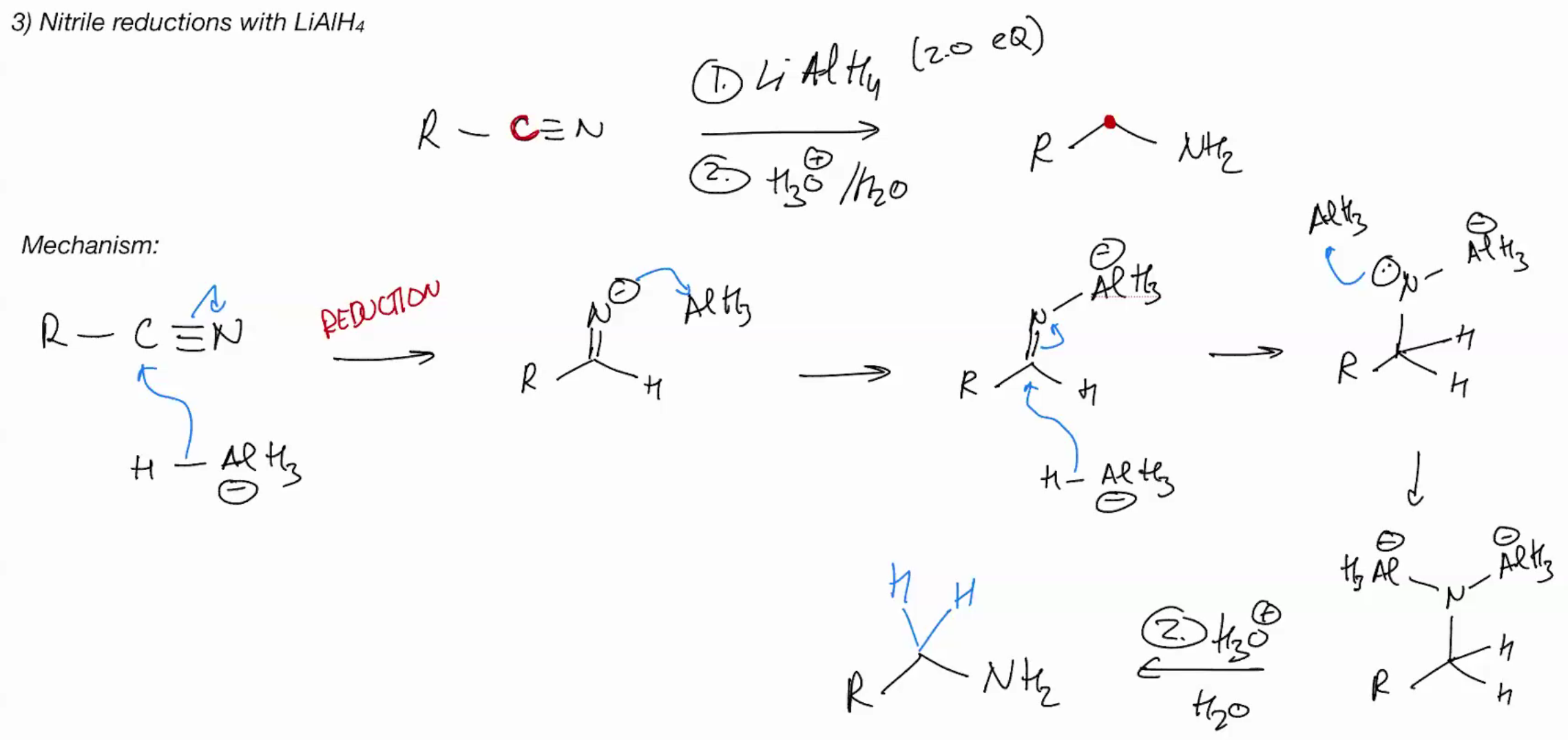
Nitrile reduction to a primary amine
Reagent LiAlH4 + ACID
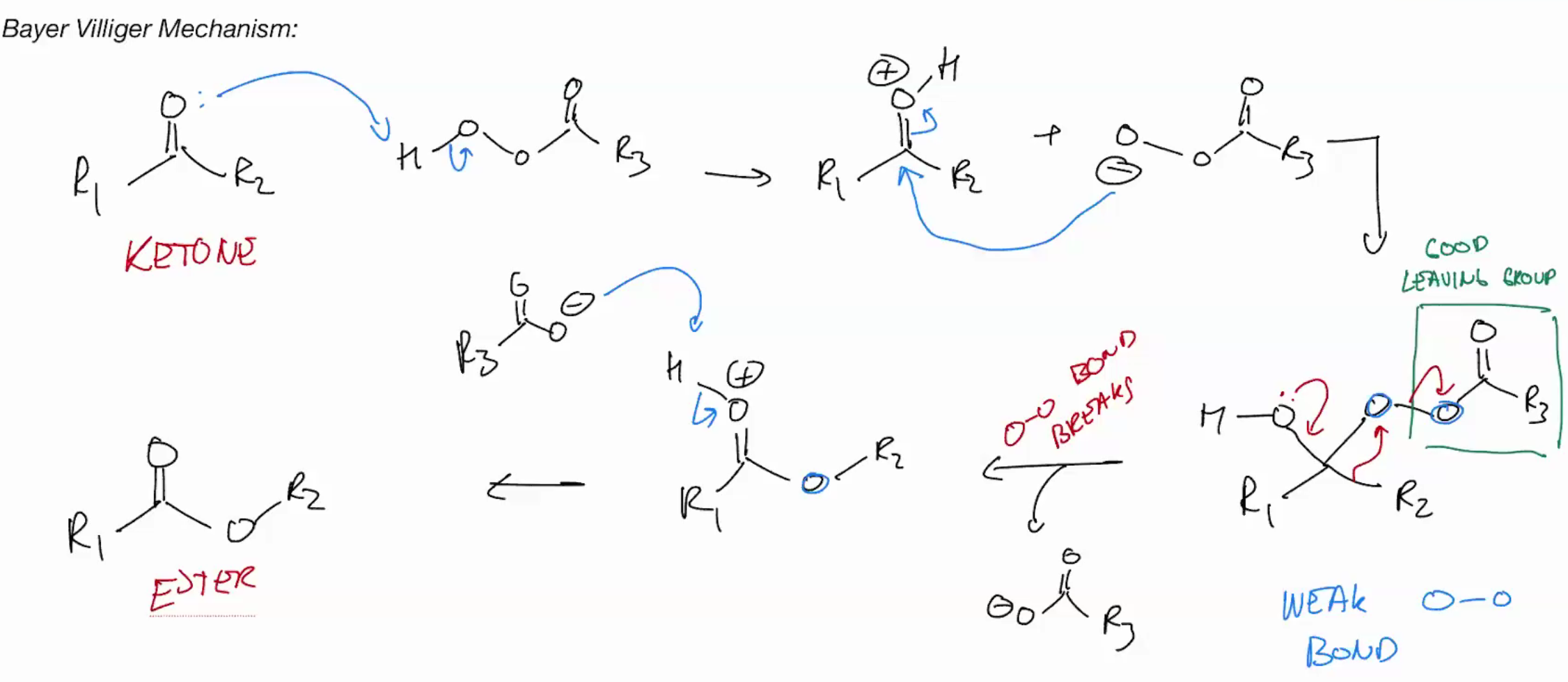
Bayer Villiger (KETONE TO ESTER) rearrange (return ketone to oxycarbenium)
Use Peroxy acid to react + stff
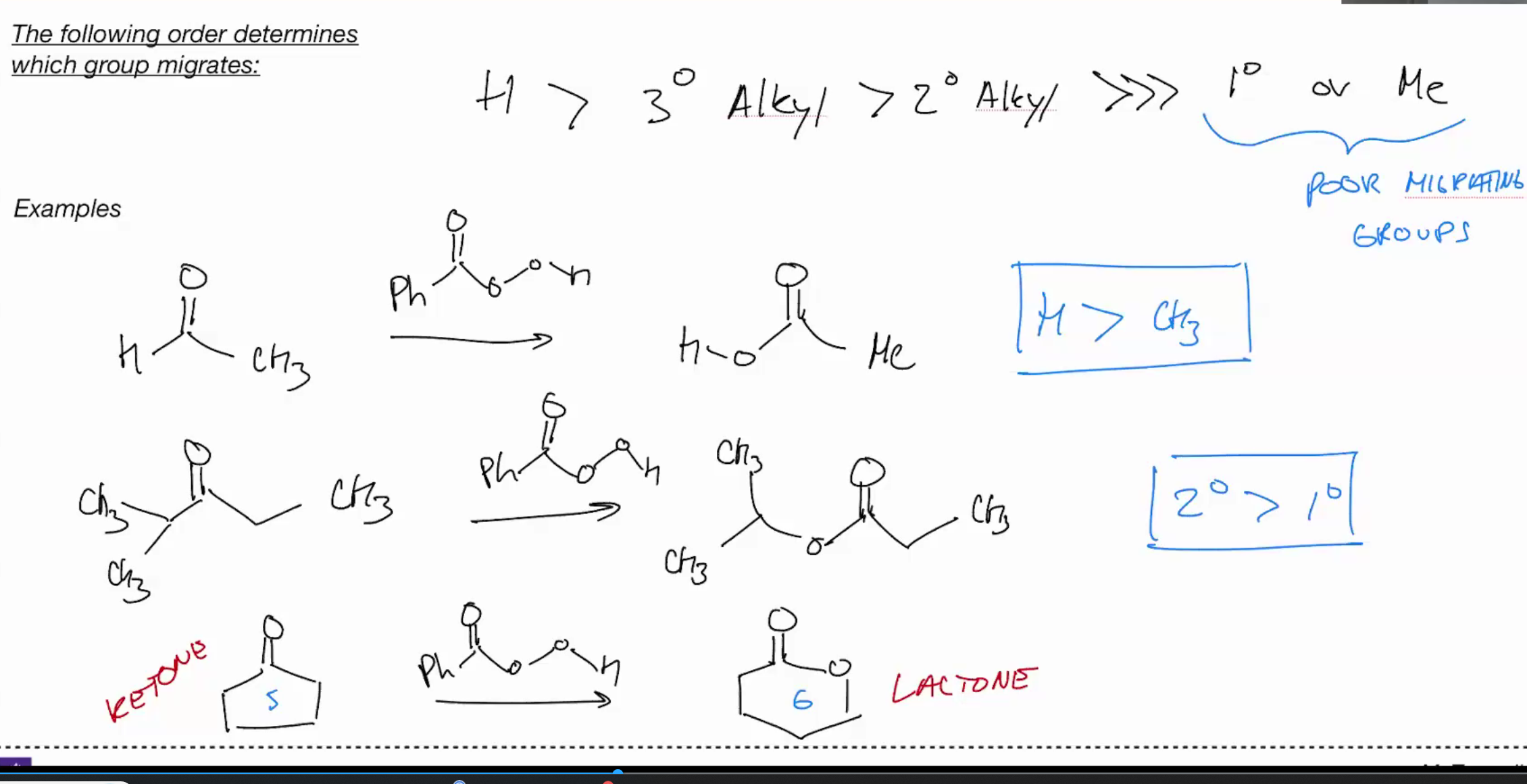
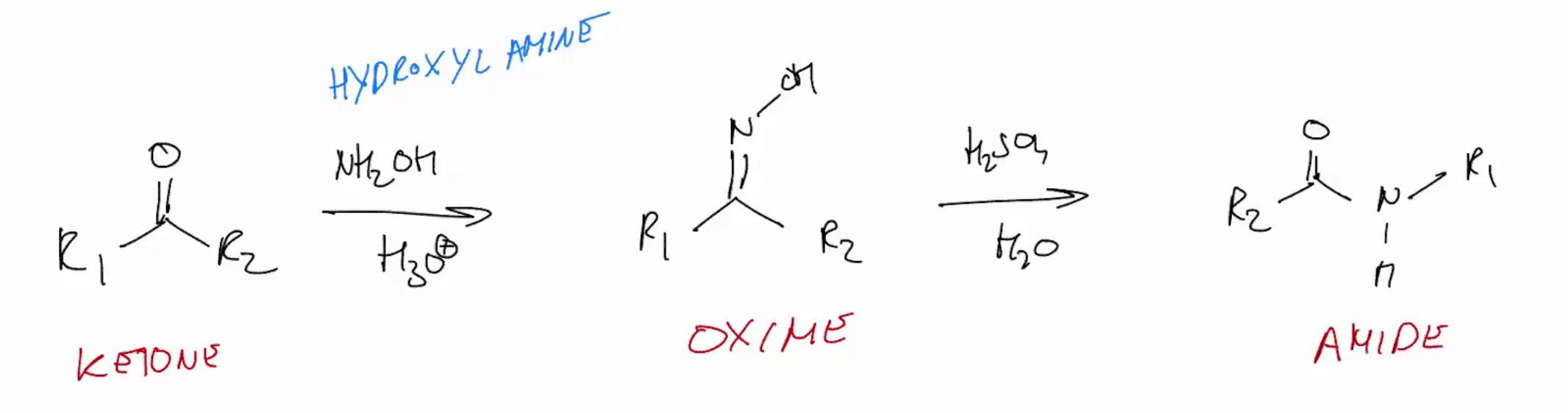
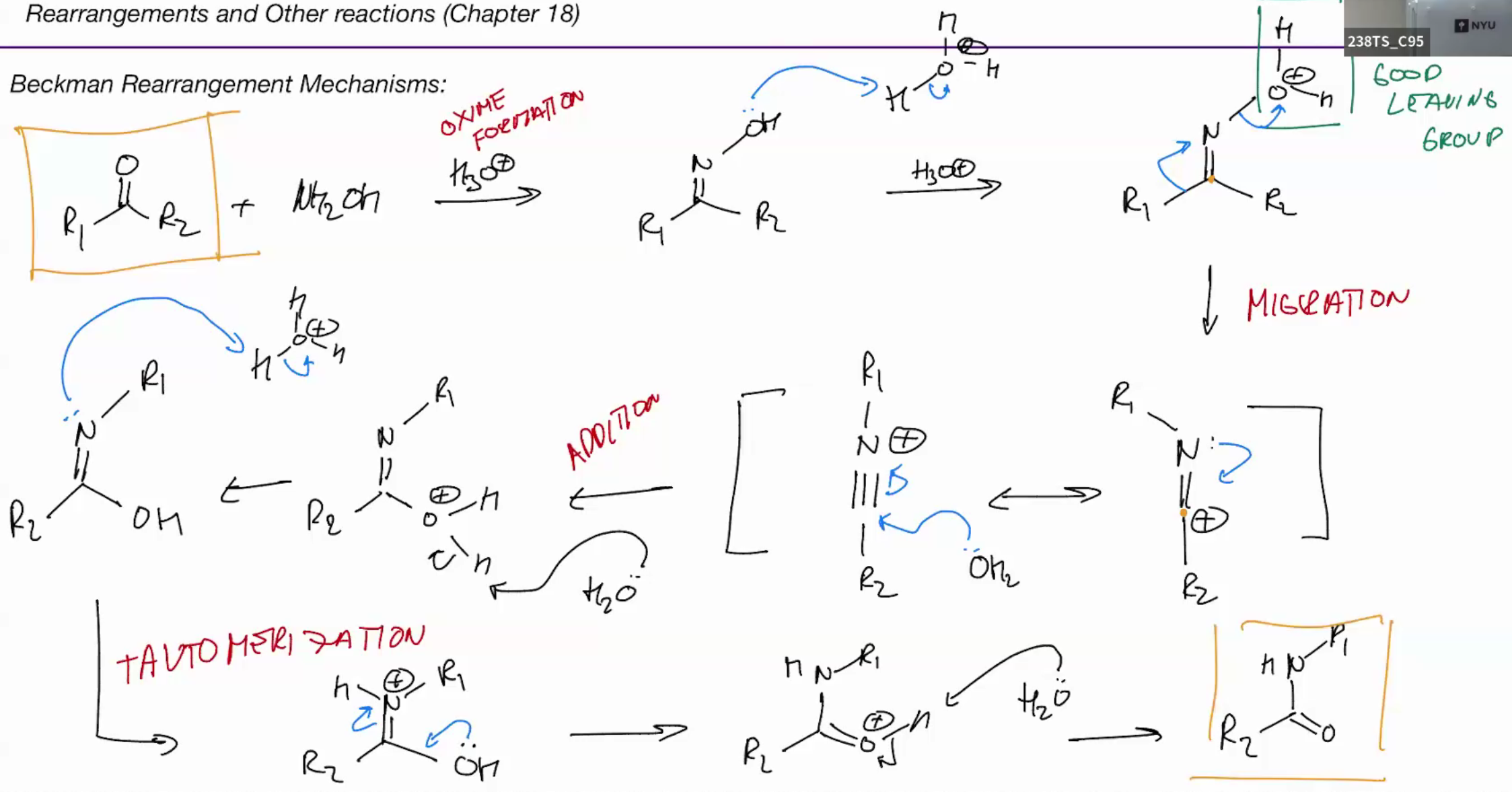
Beckman rearrangement (amide from ketones via oximes)
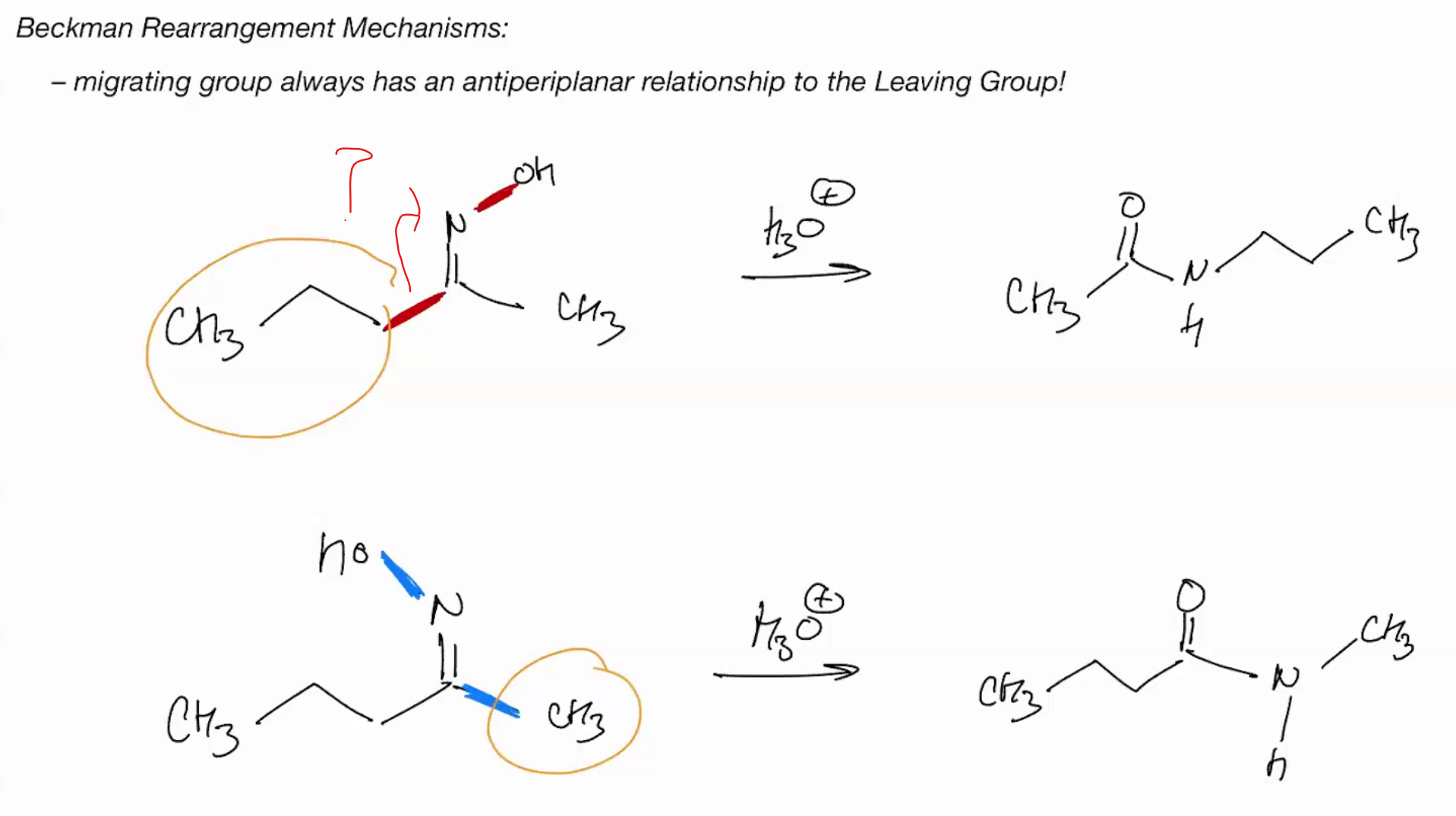
For becker migrating group always has anti pariplanar to leaving group
ketocarbene are prepared from diazo ketones
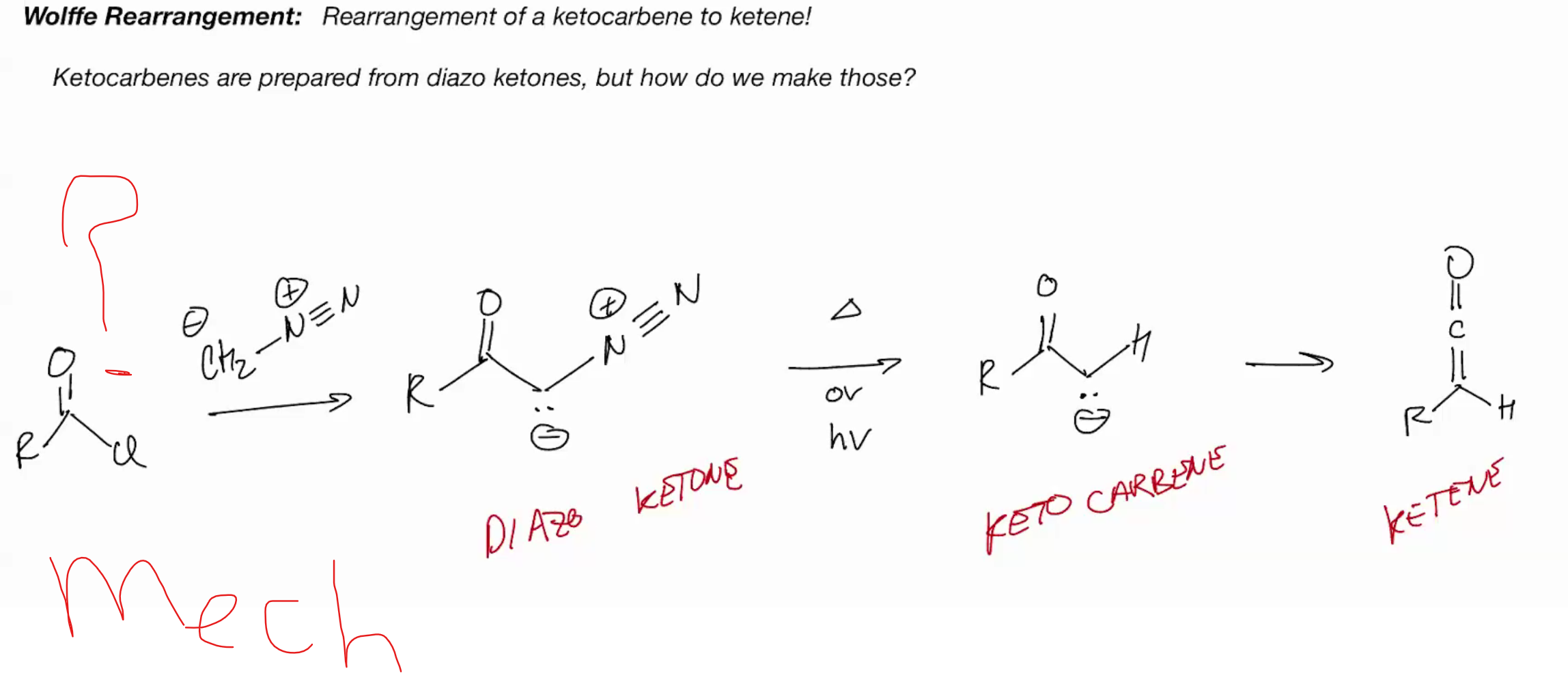
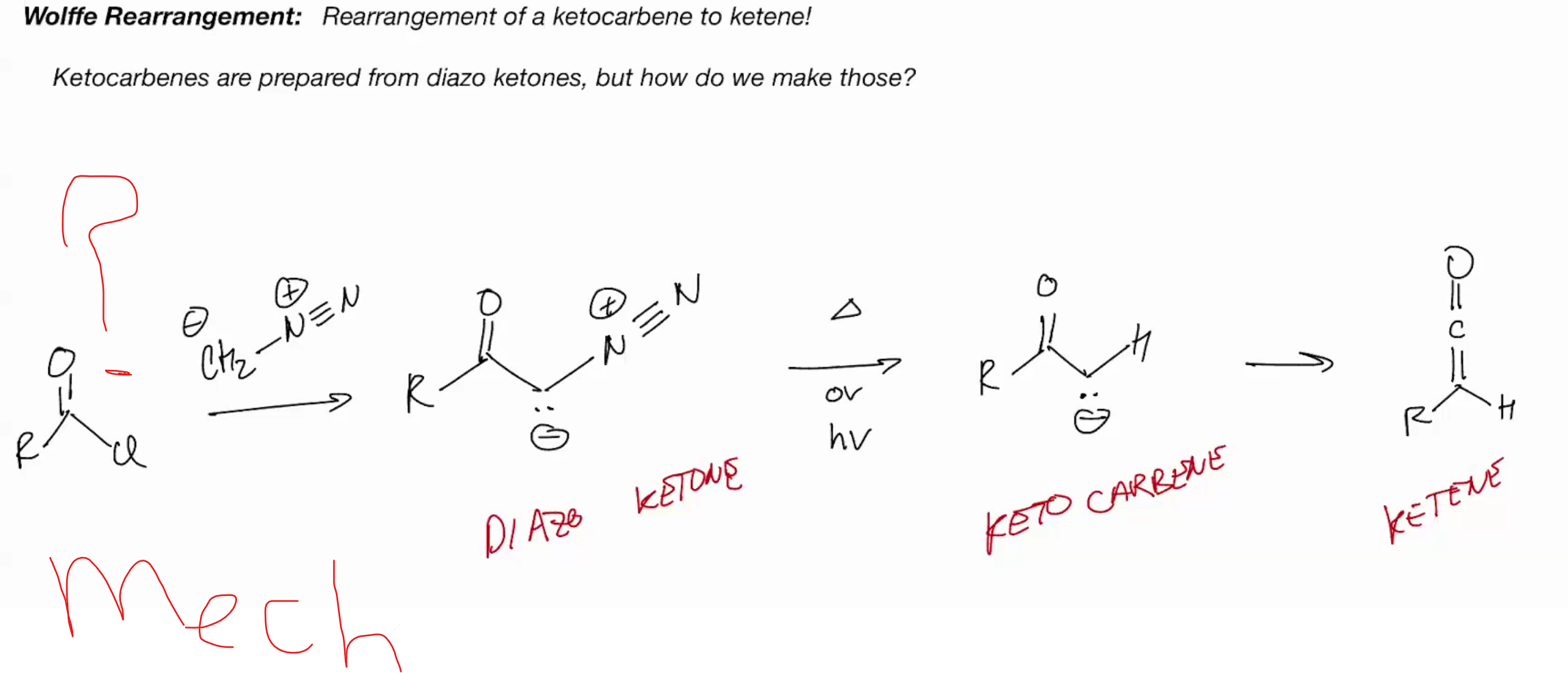
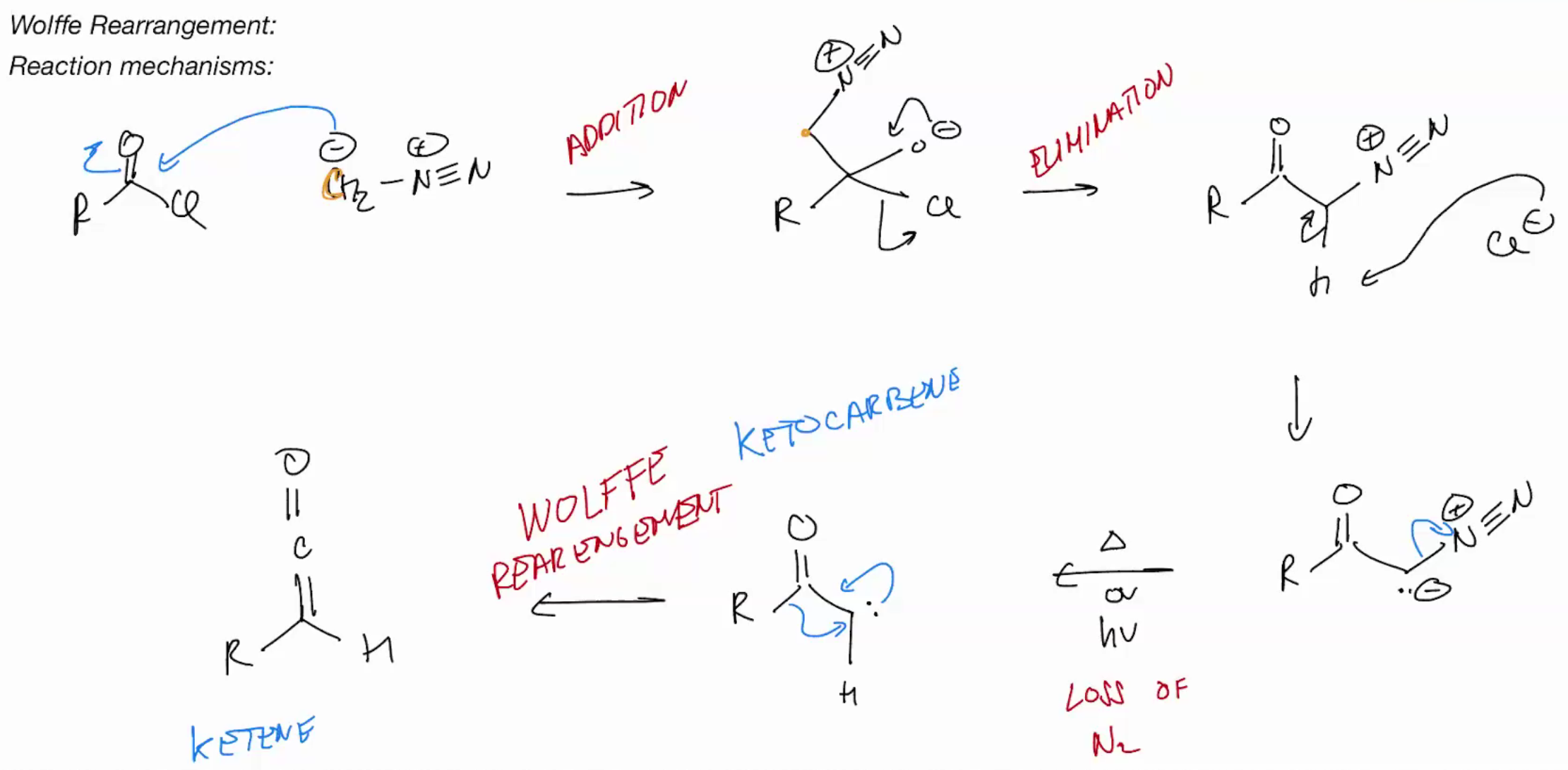
Wolffe Rearrangement (ketocarbene to ketene)
diazomethane + acid chloride = diazoketone + light + heat = keto carbene one step into the elctrophilic product
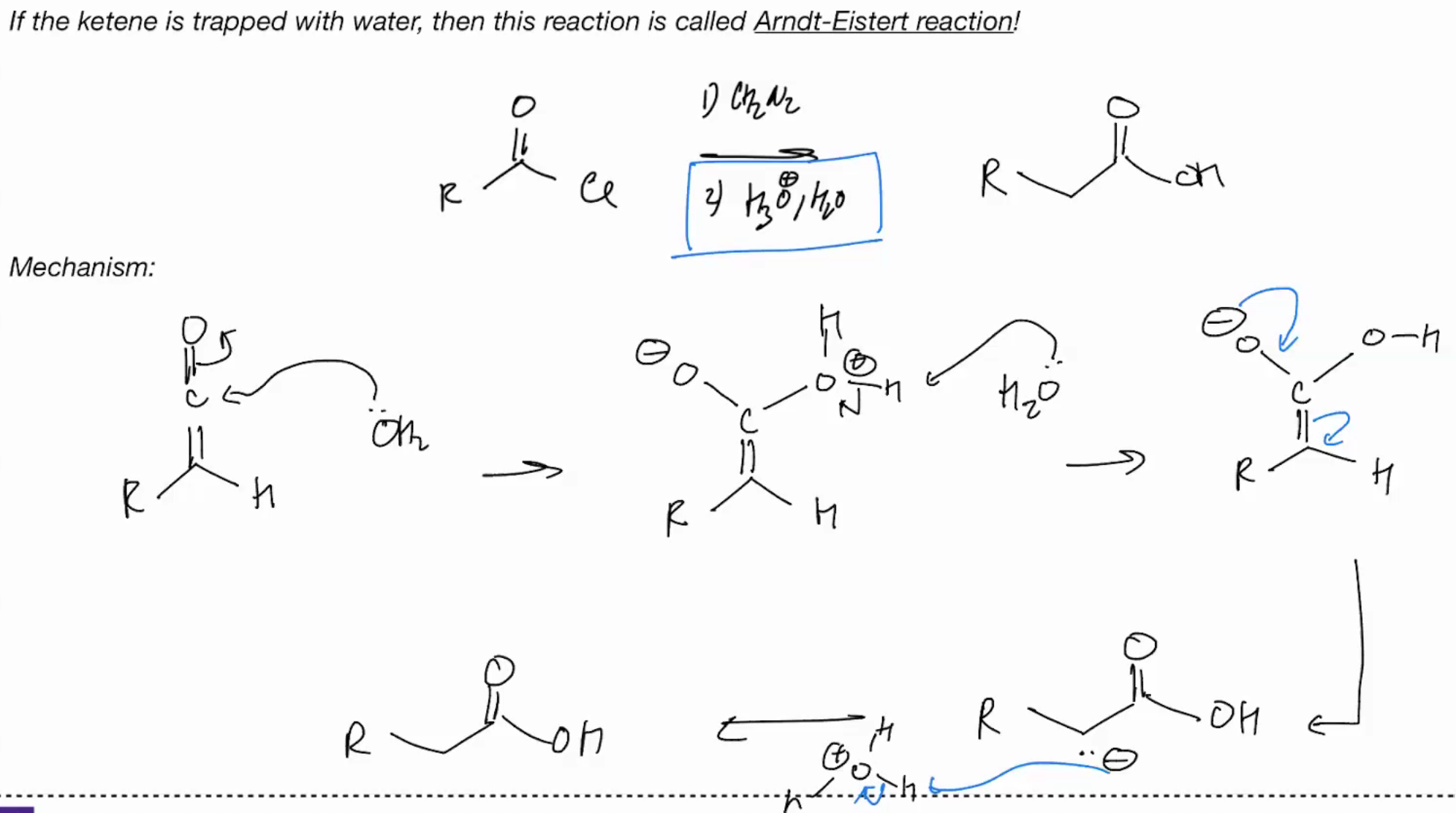
Arndt Eistert reaction
h30/h20
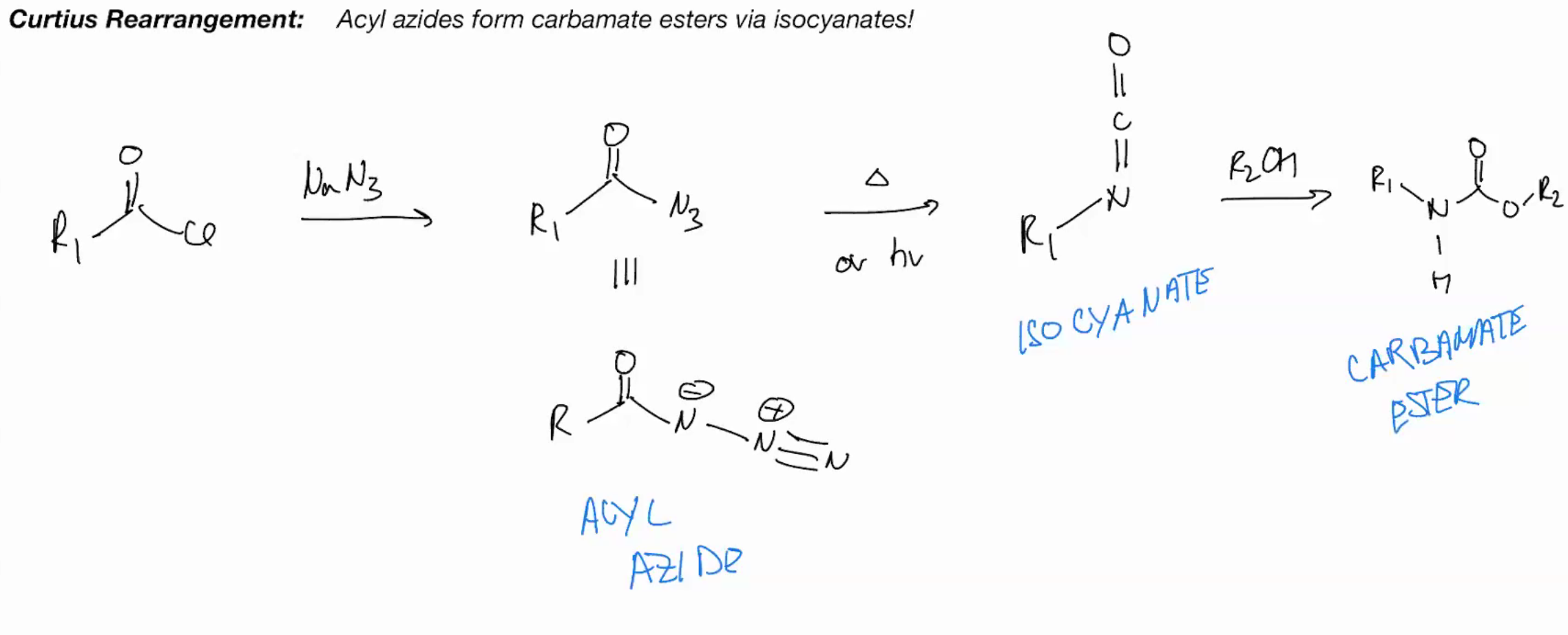
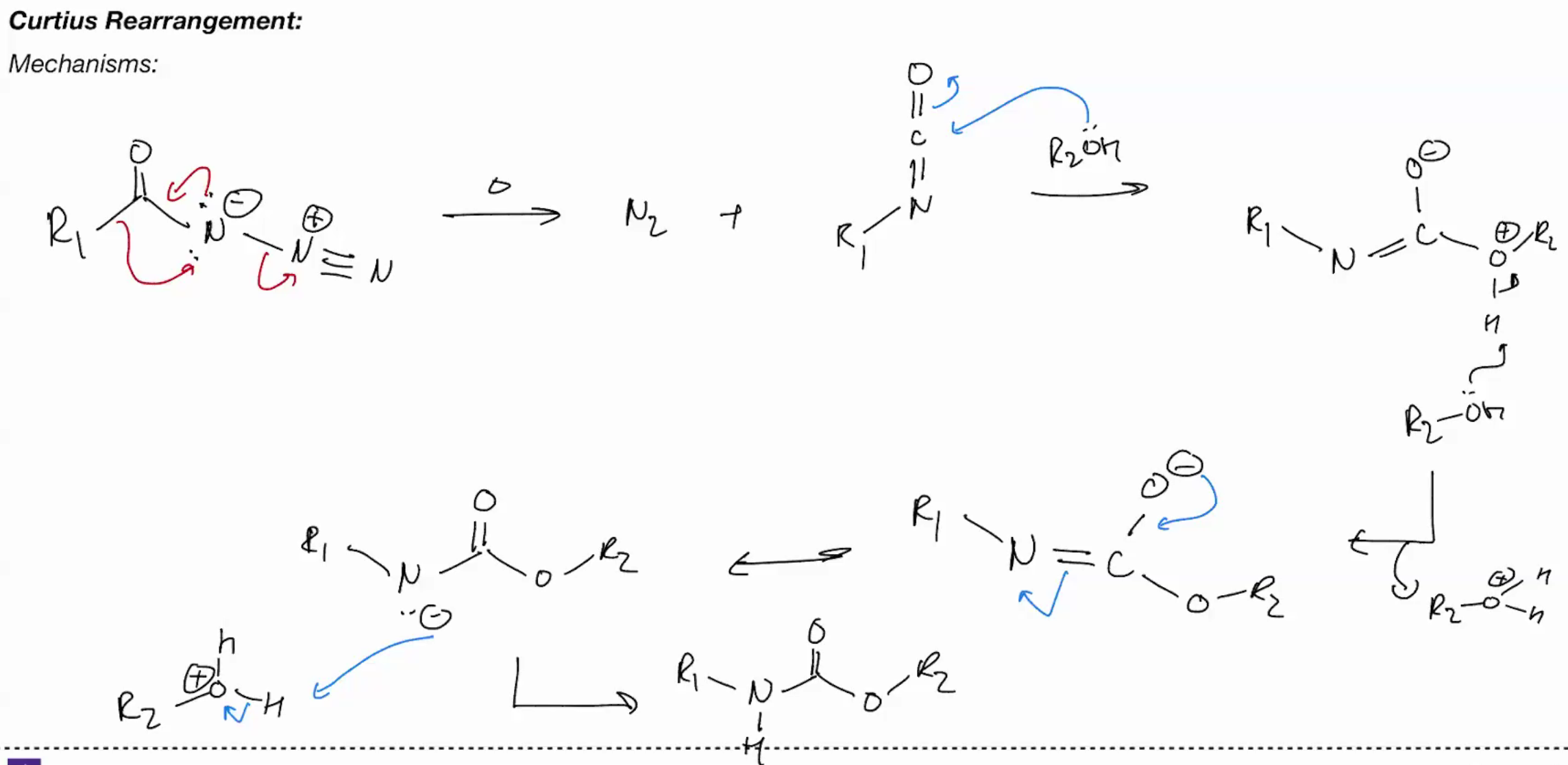
Curtius (acyl azides from carbomate esters via isocyanates)
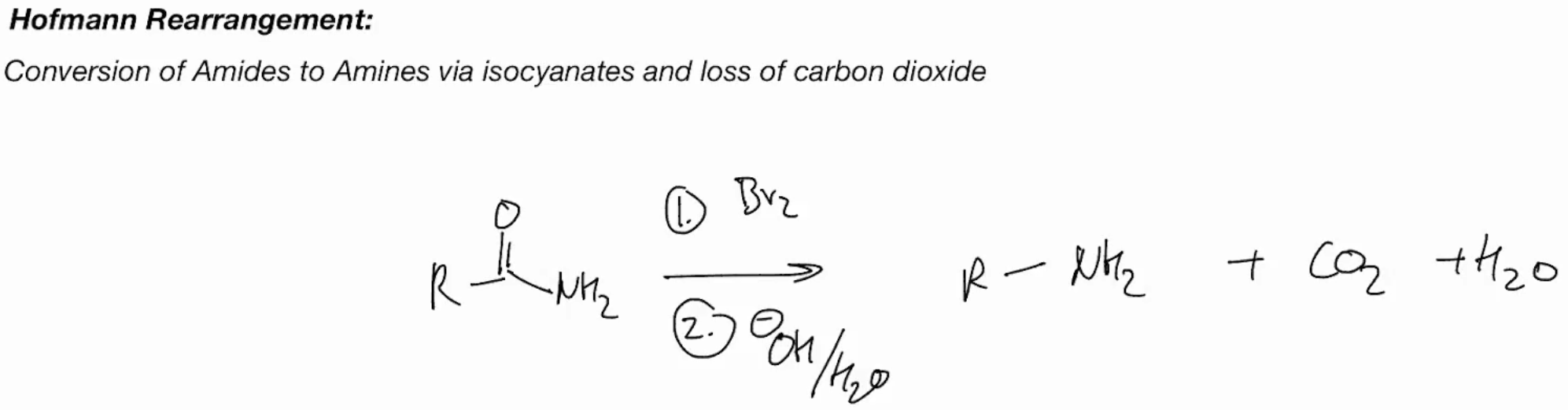
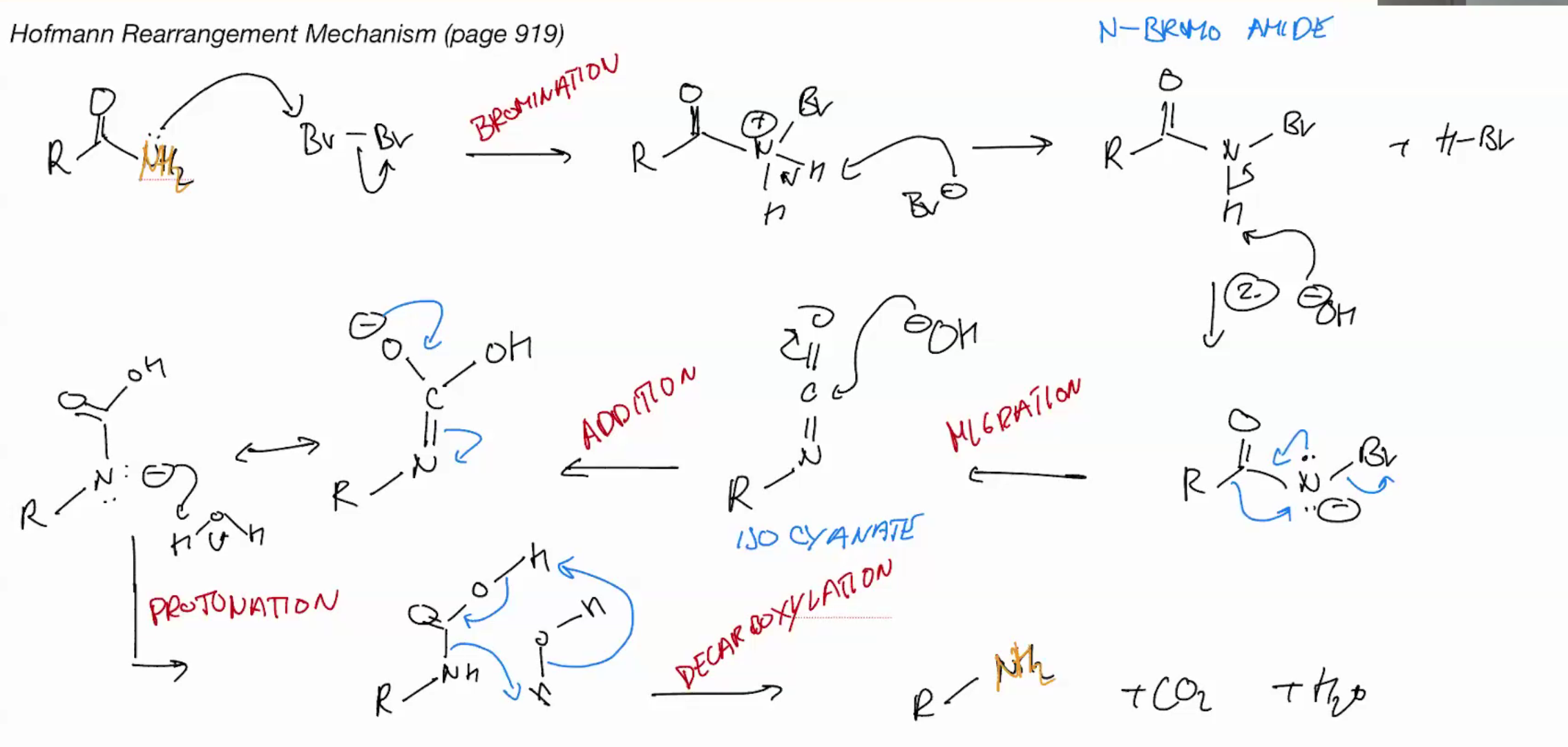
Hoffman rearrangement (AMIDES TO AMINE)
stops as isocyanate cuz basic
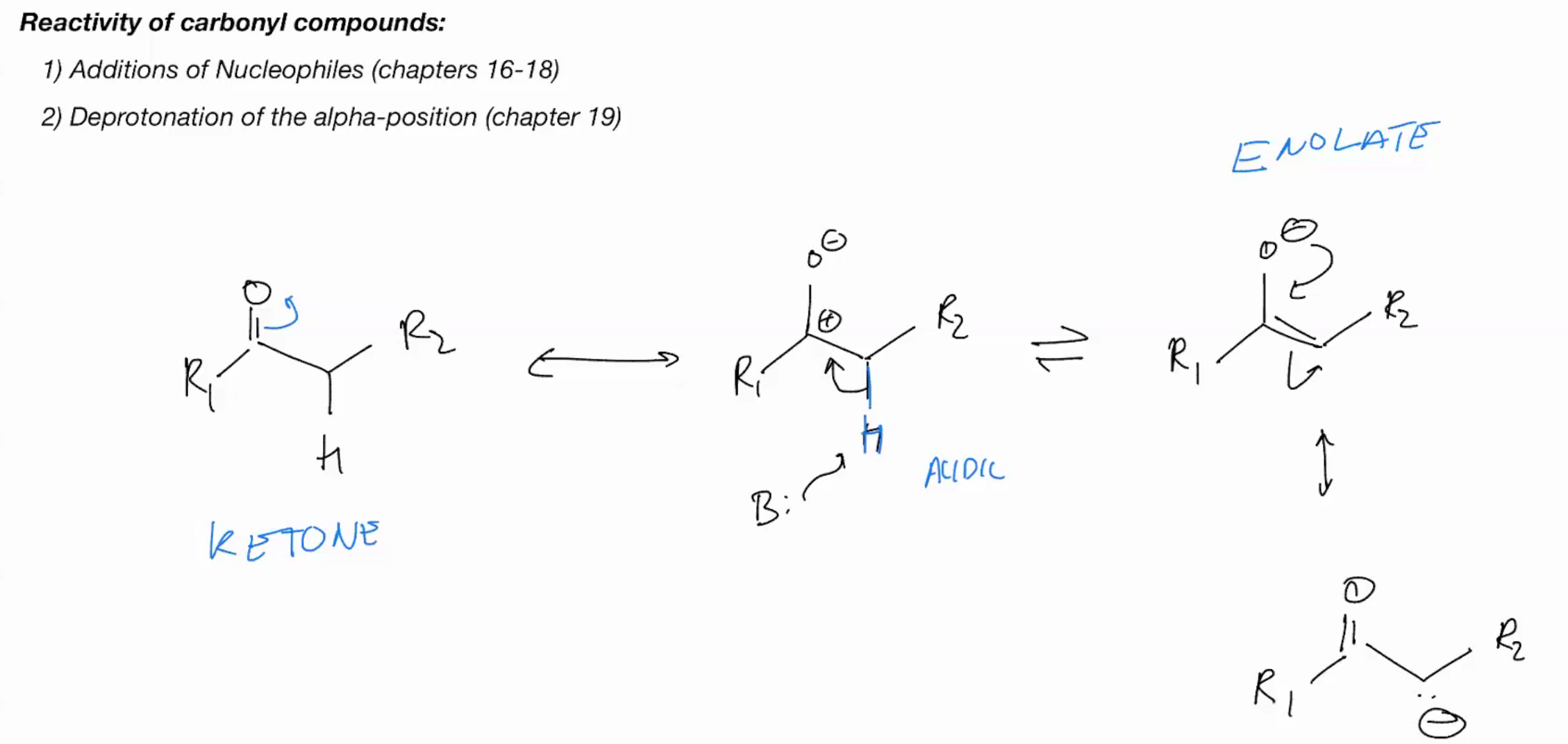
reactivity of carbonyl compounds
CAN AHPPEN UNDER ACIDIC OR BASIC CONDITIONS (KETONE NOMENCLATURE)
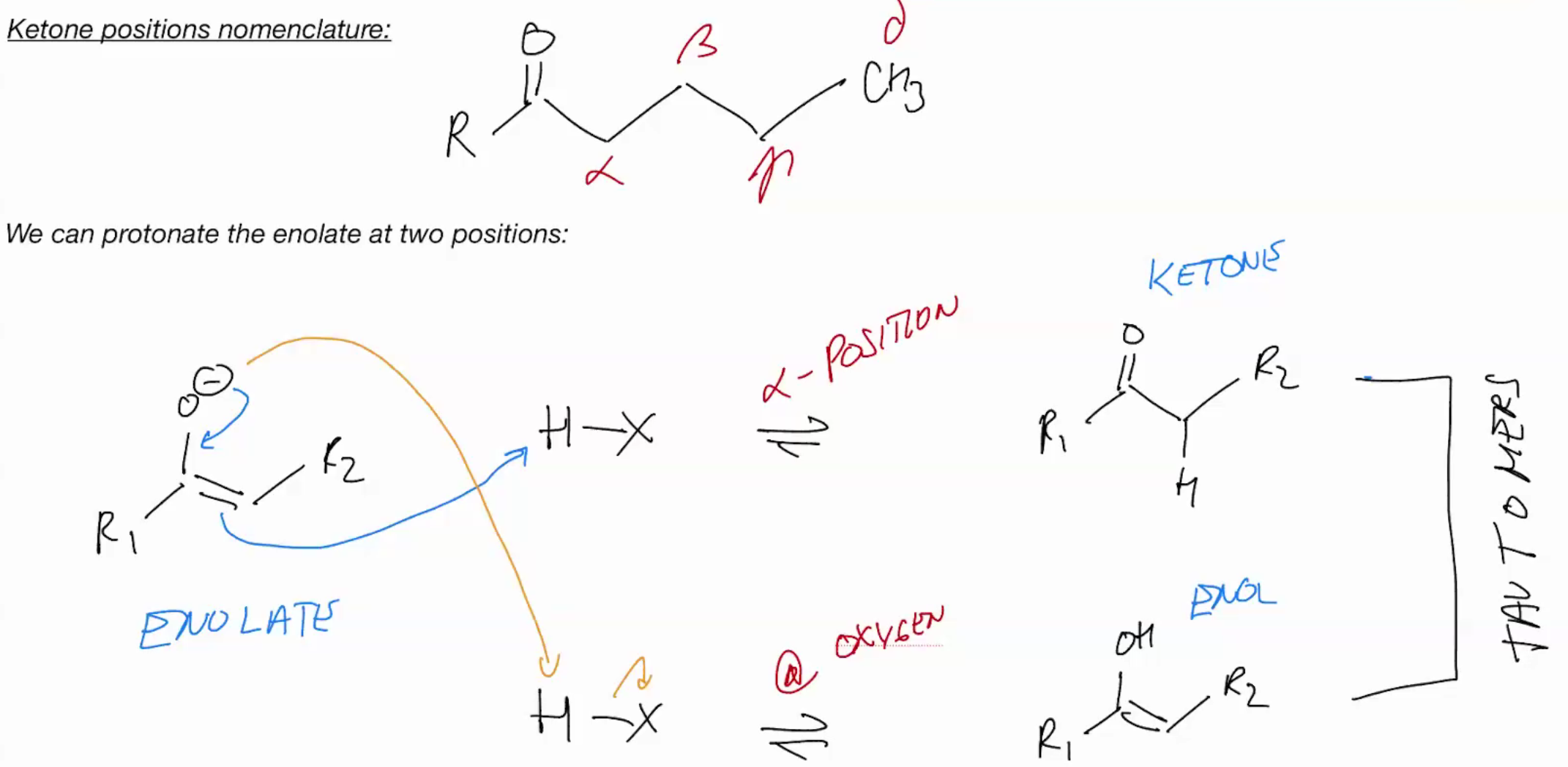
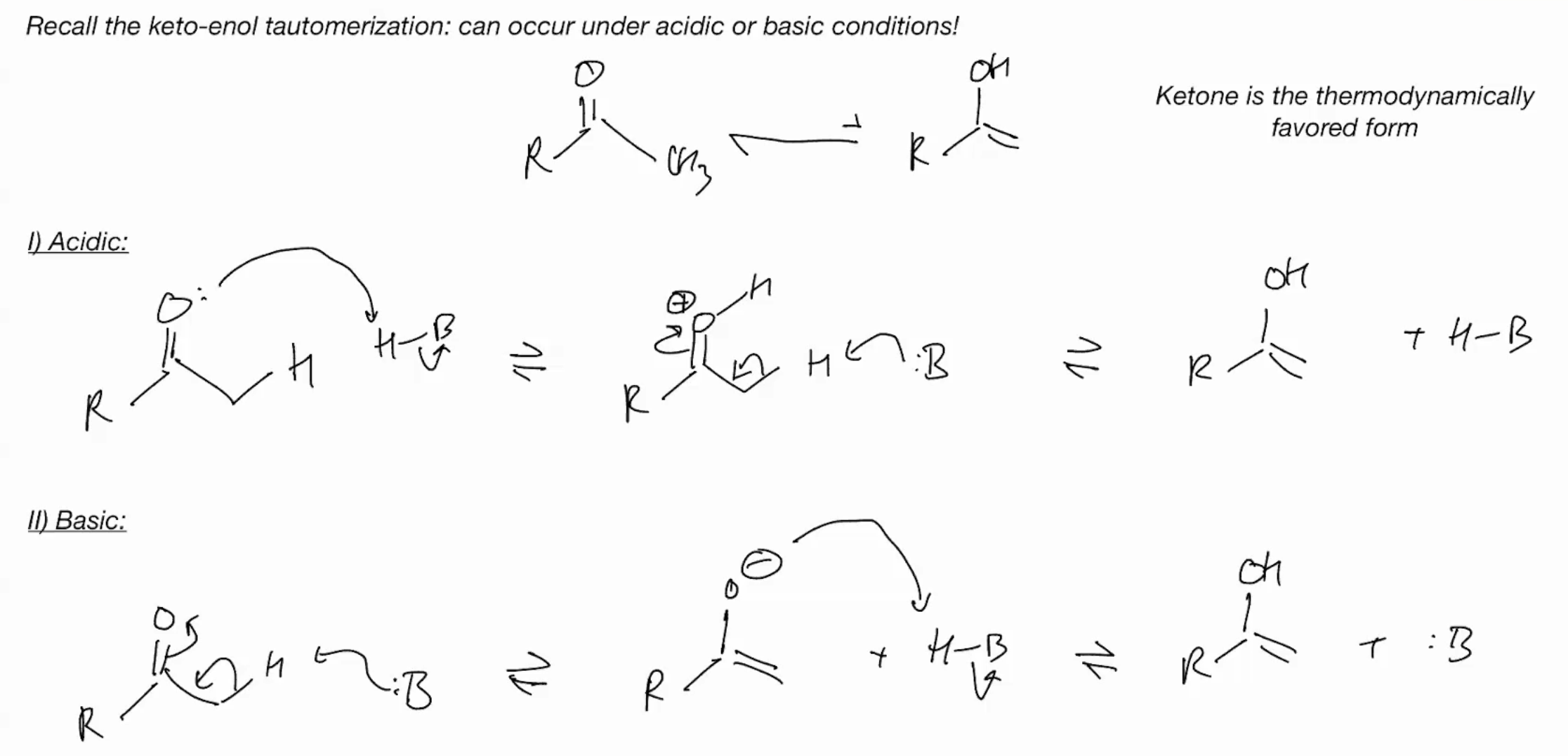
keto-enol tautermizrrization
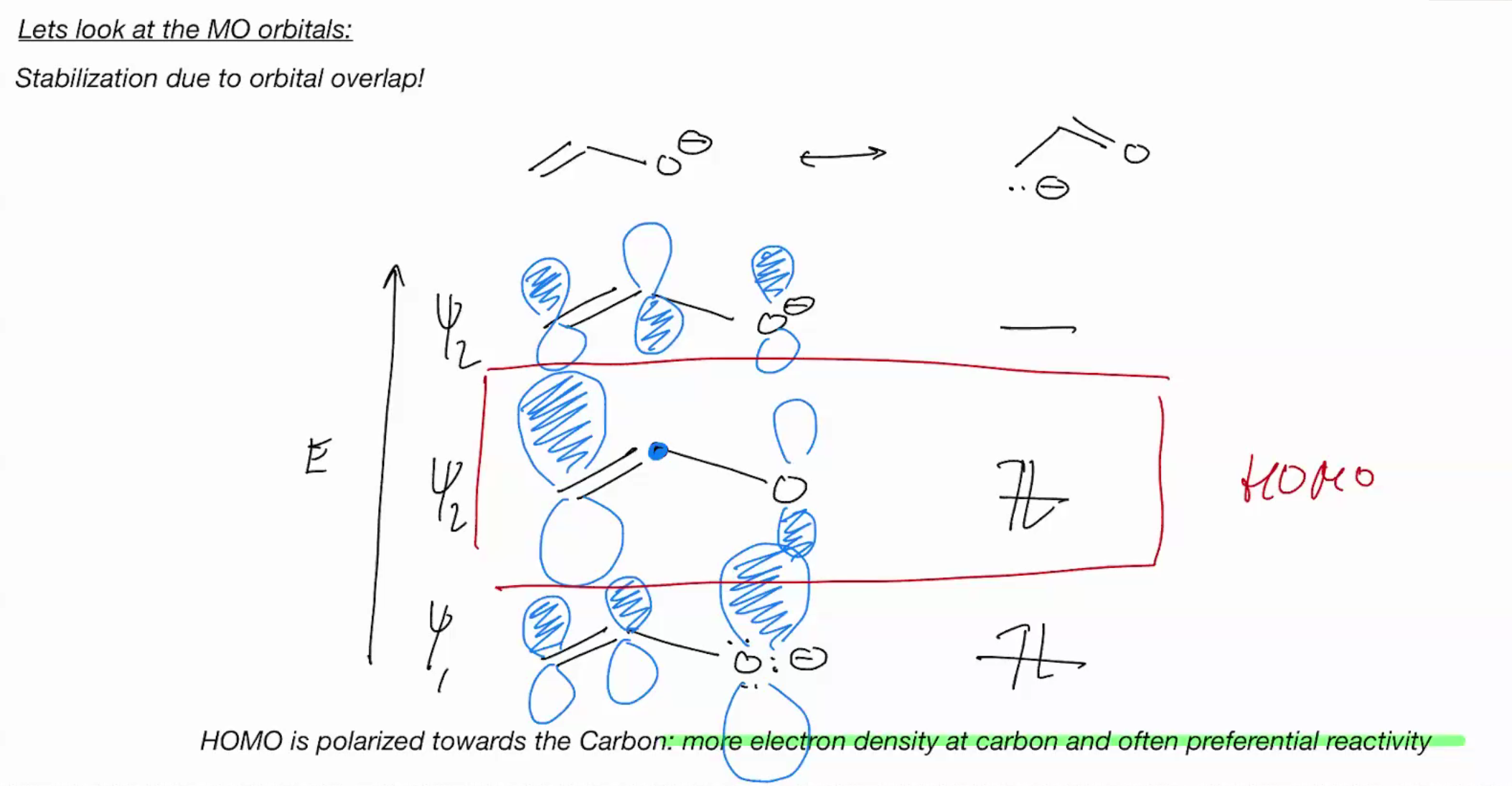
it will react at carbon because it more electron dense (what will though?)
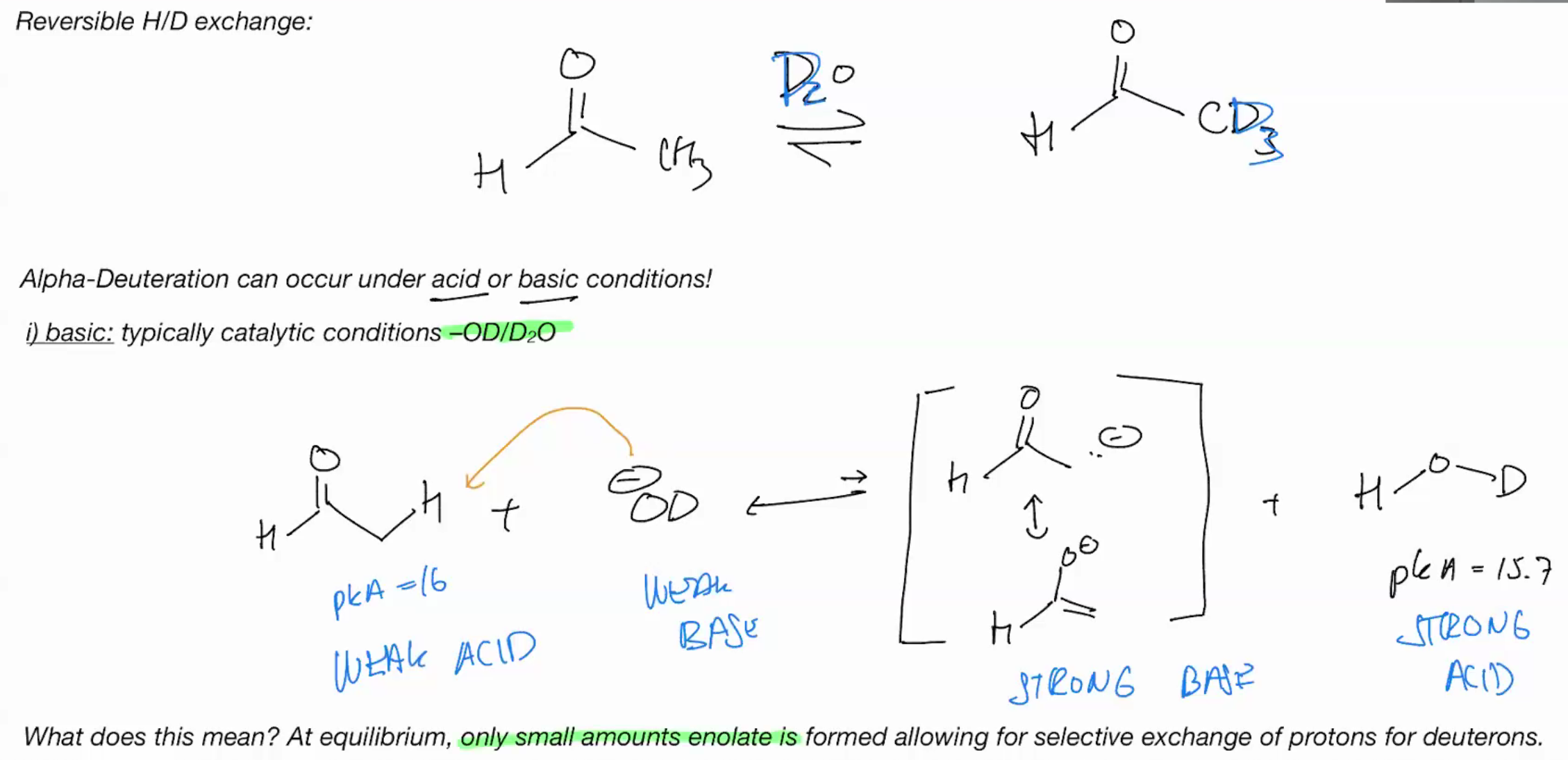
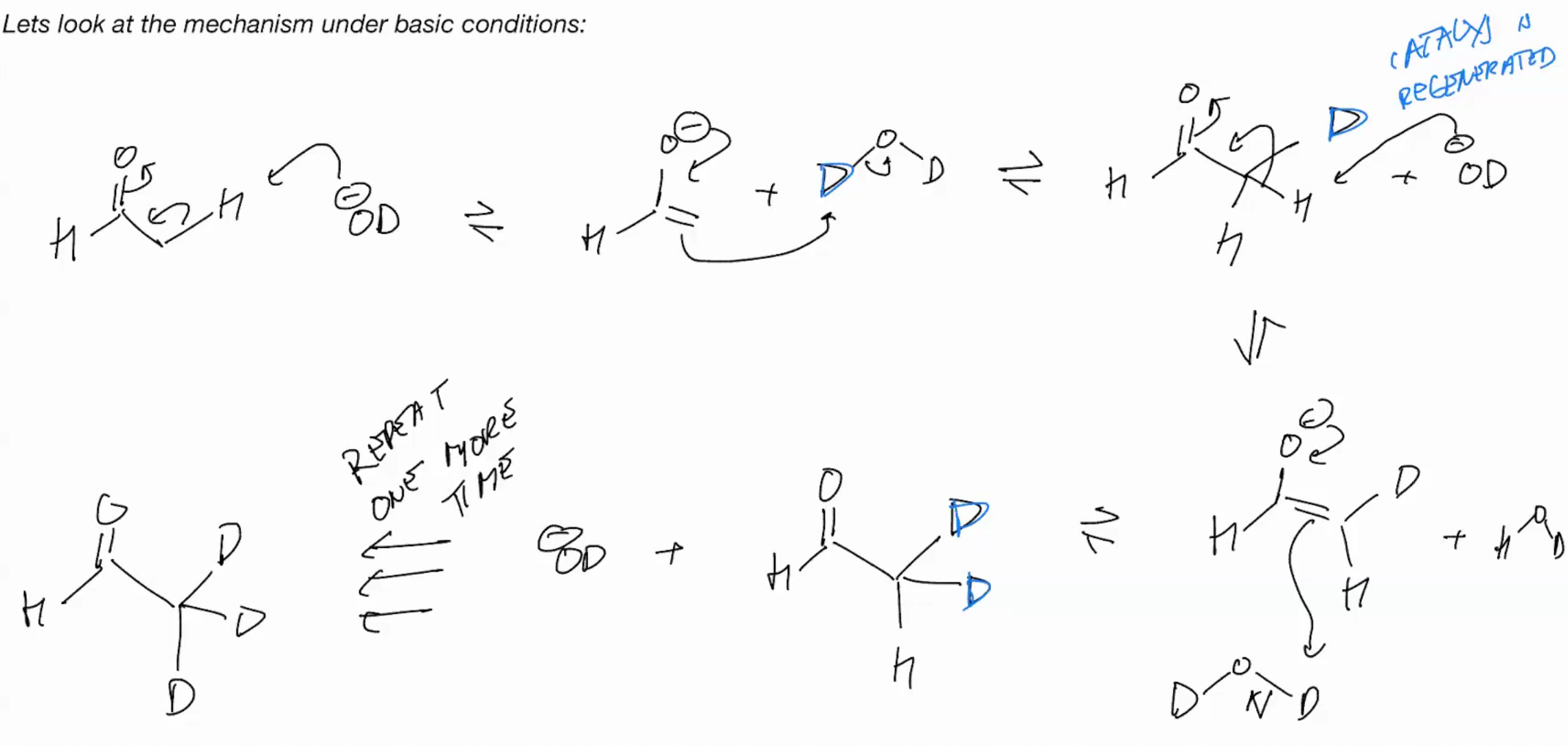
Alpha Dueteration under BASIC
mechanism , D is labeled heavy and stronger so it prefers to grab H
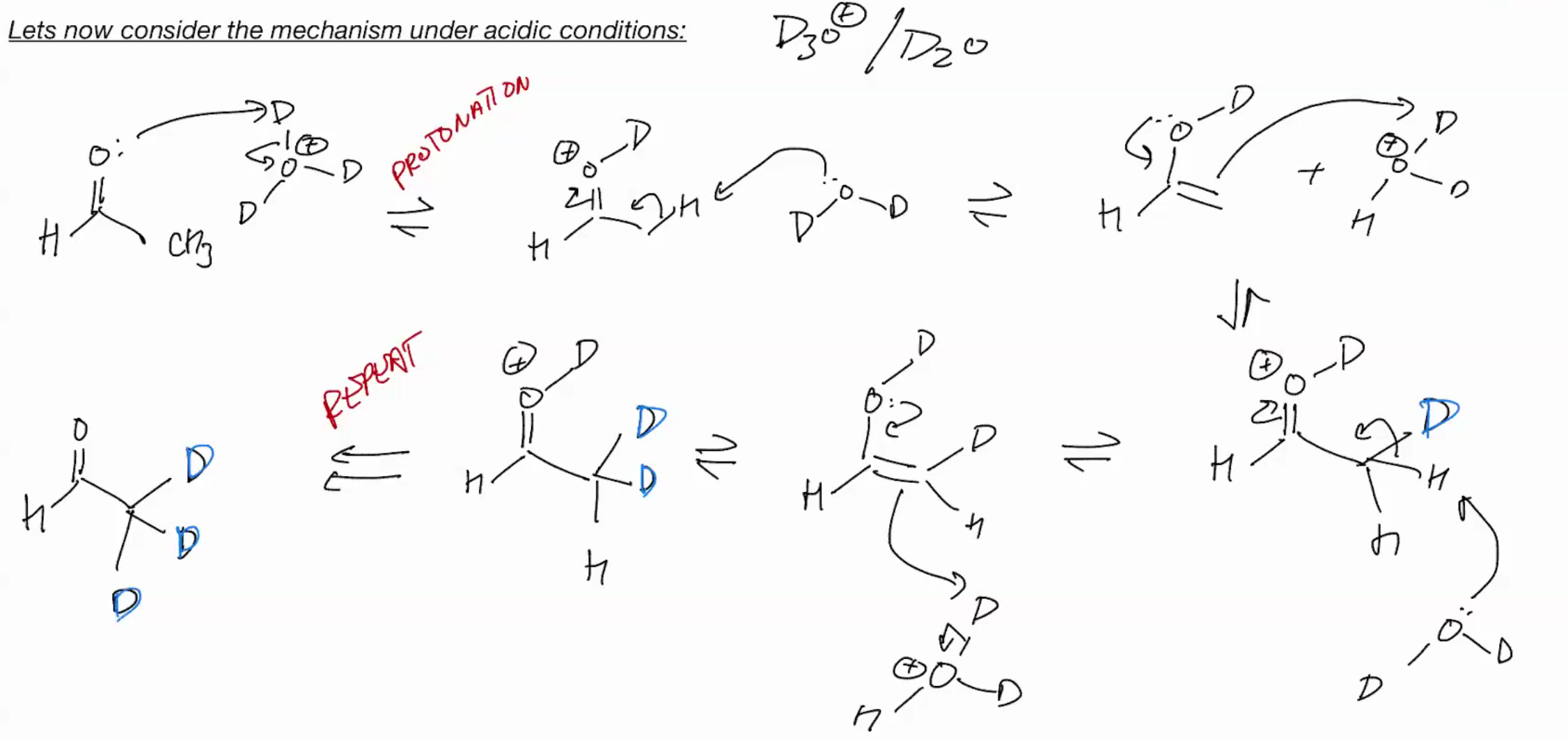
Alpha Dueteration under Acidic
catalytic acid or base
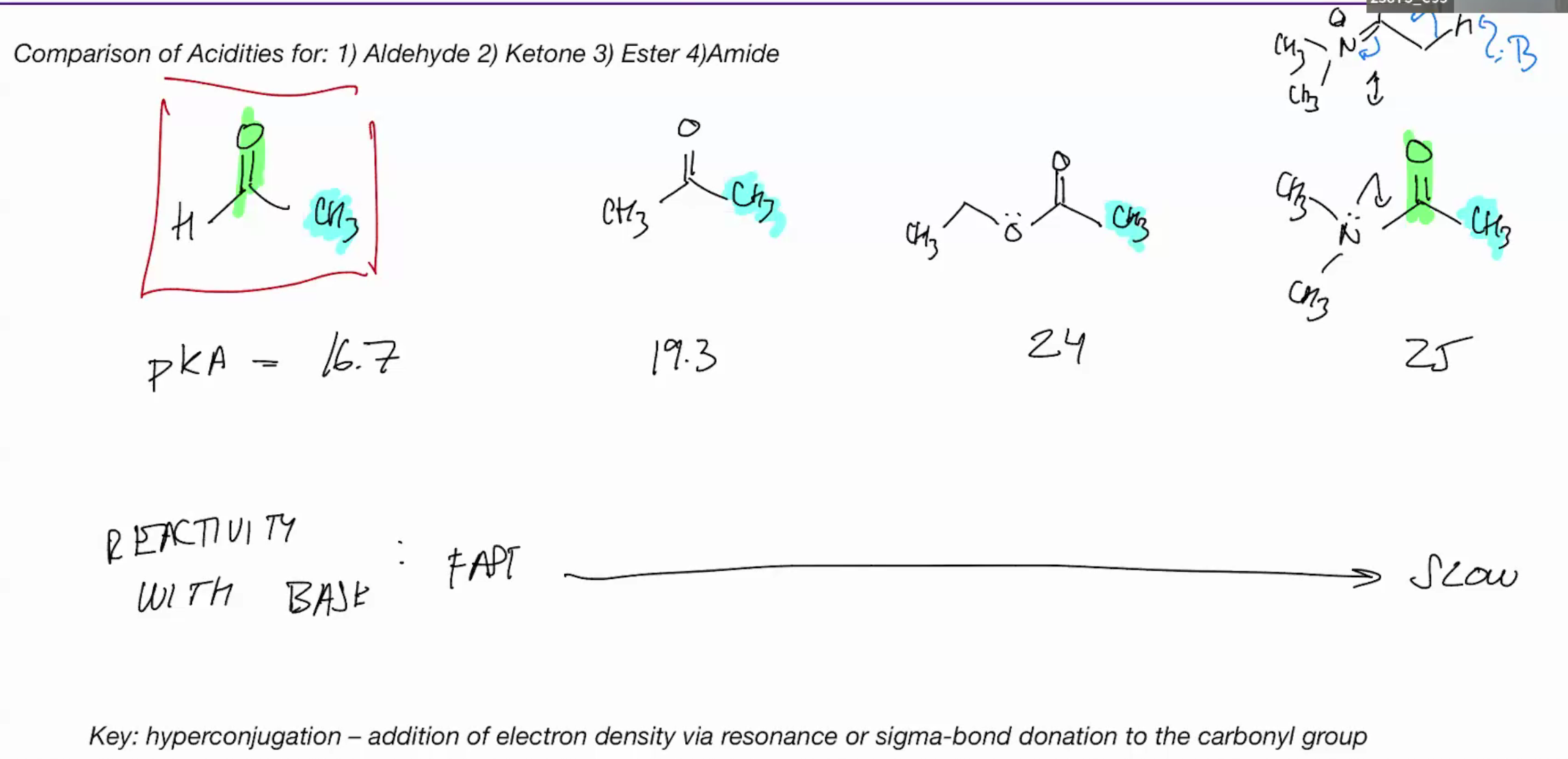
Acidity comparisions (on the right u can have resonance structures so its harder to make the desired enol bc of those resonance forms/slower
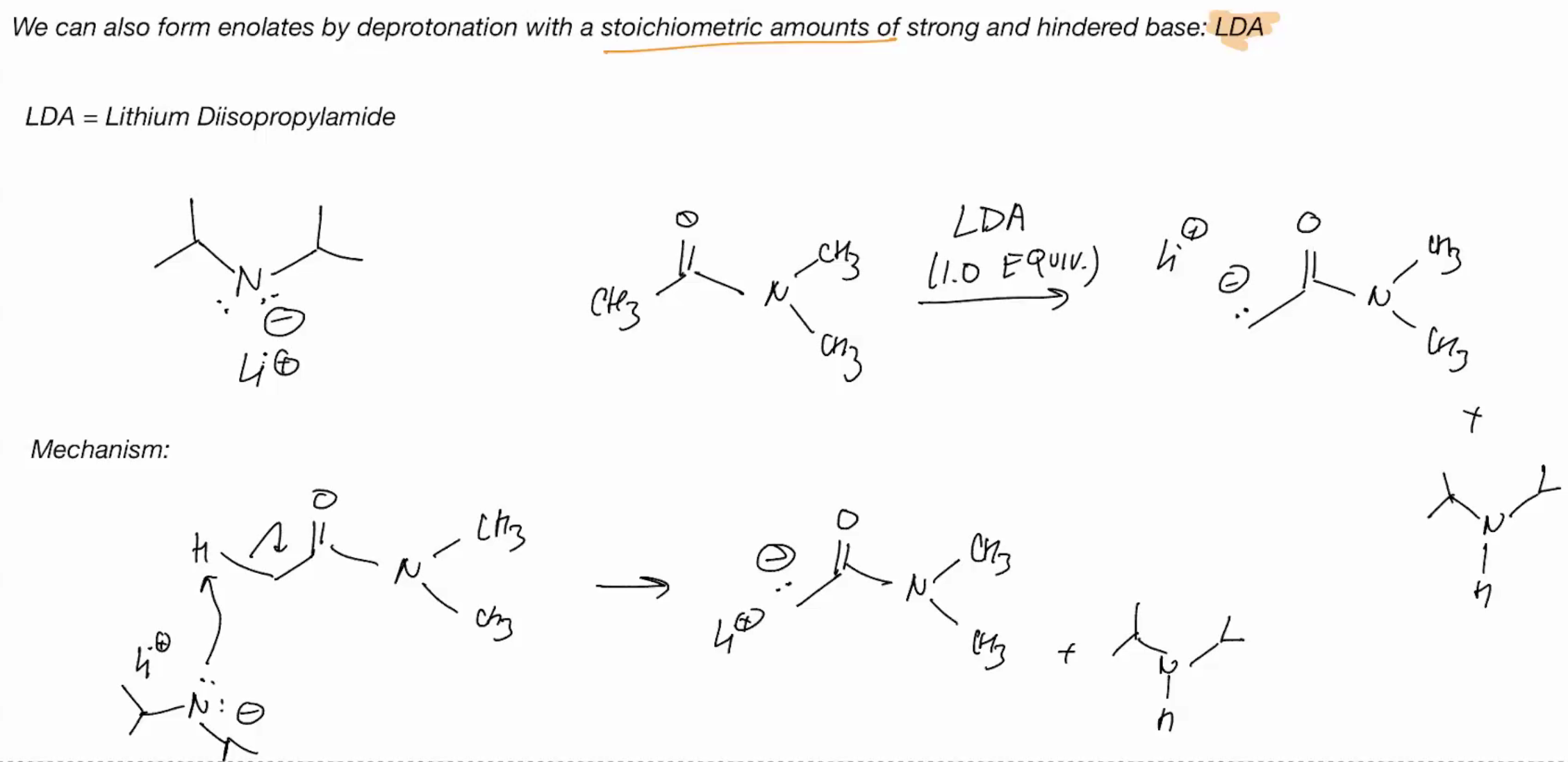
Enolates by derotonation
LDA = dissopropylamide (strong and hindered base)
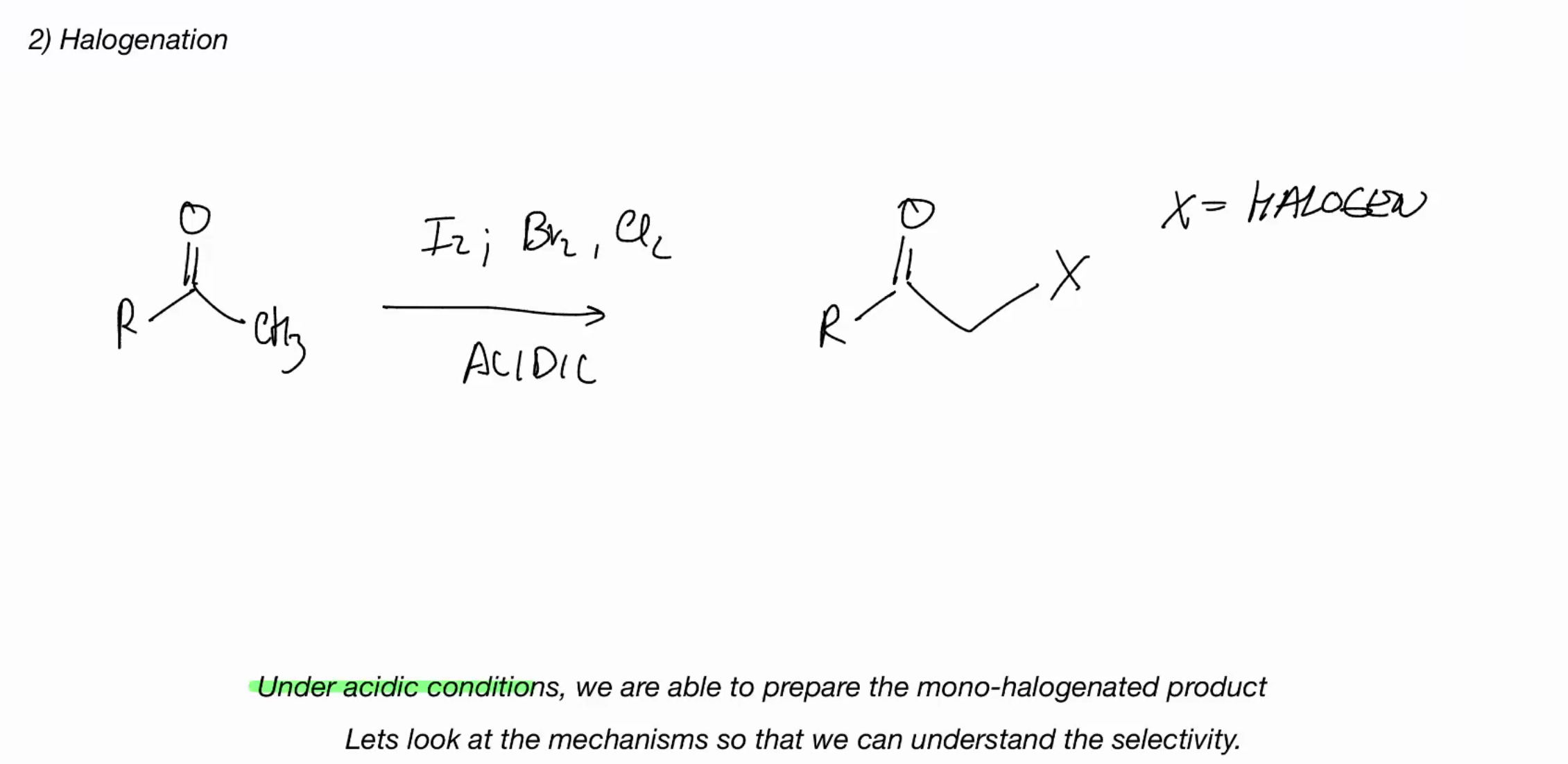
Halogenation (ACID)
Mechanism w acid (same as dueteraton)
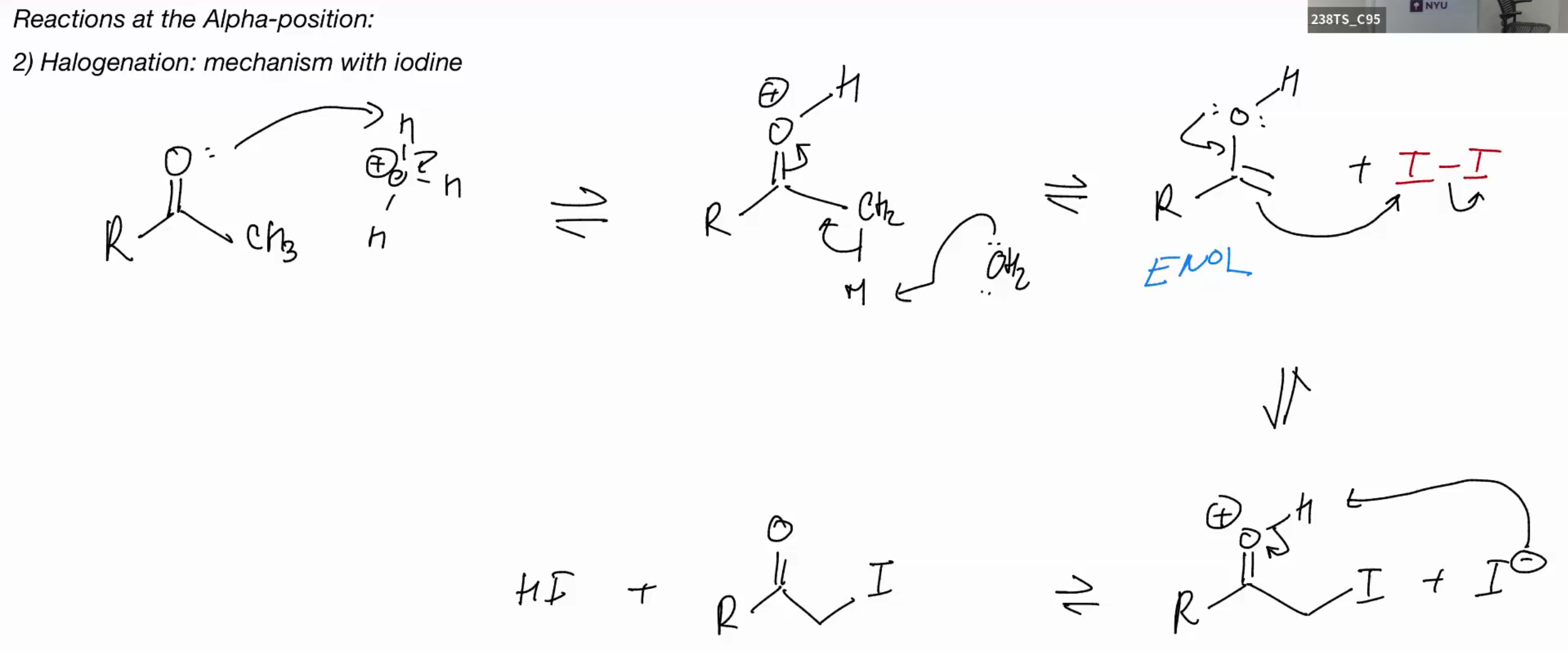
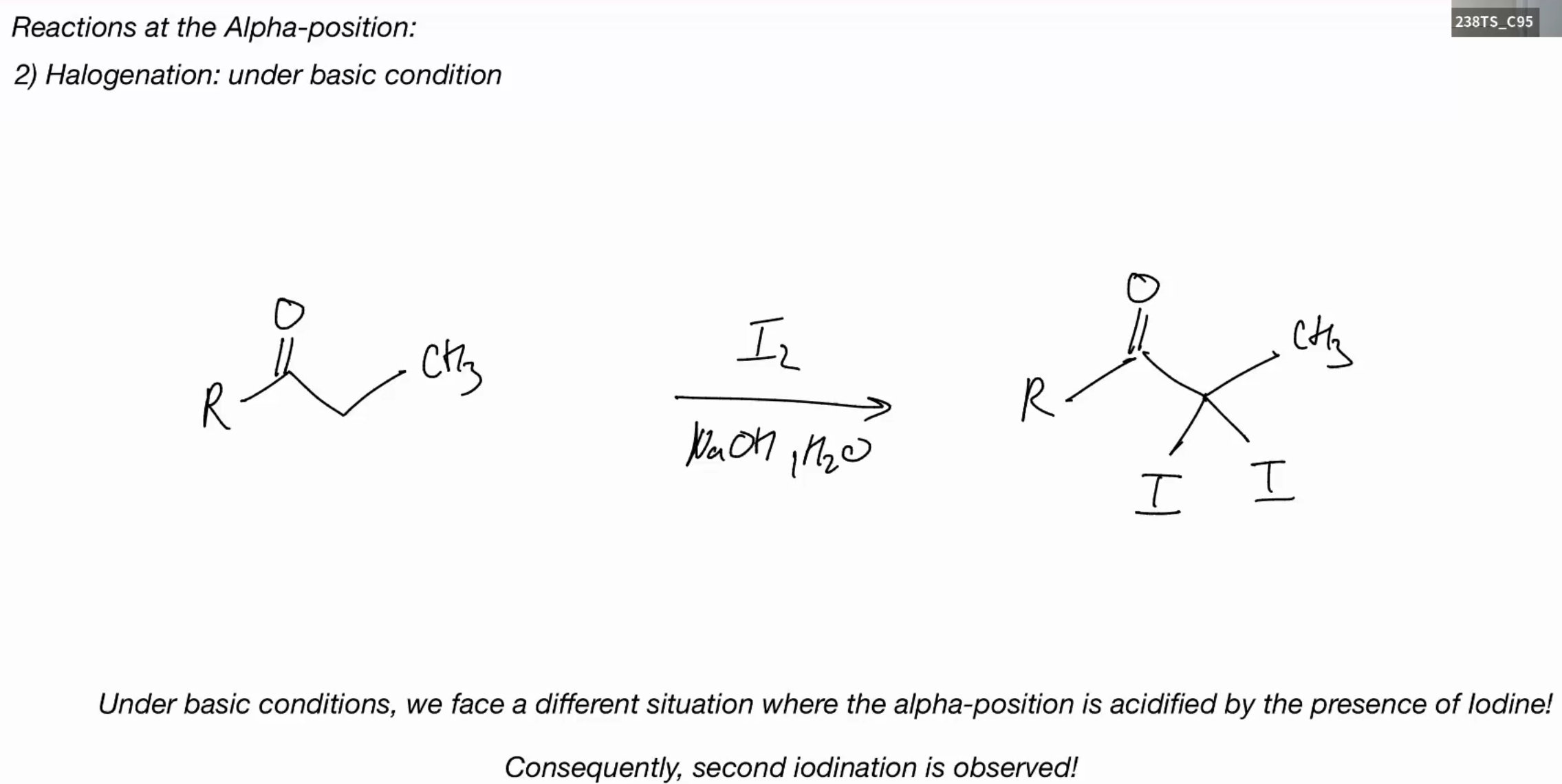
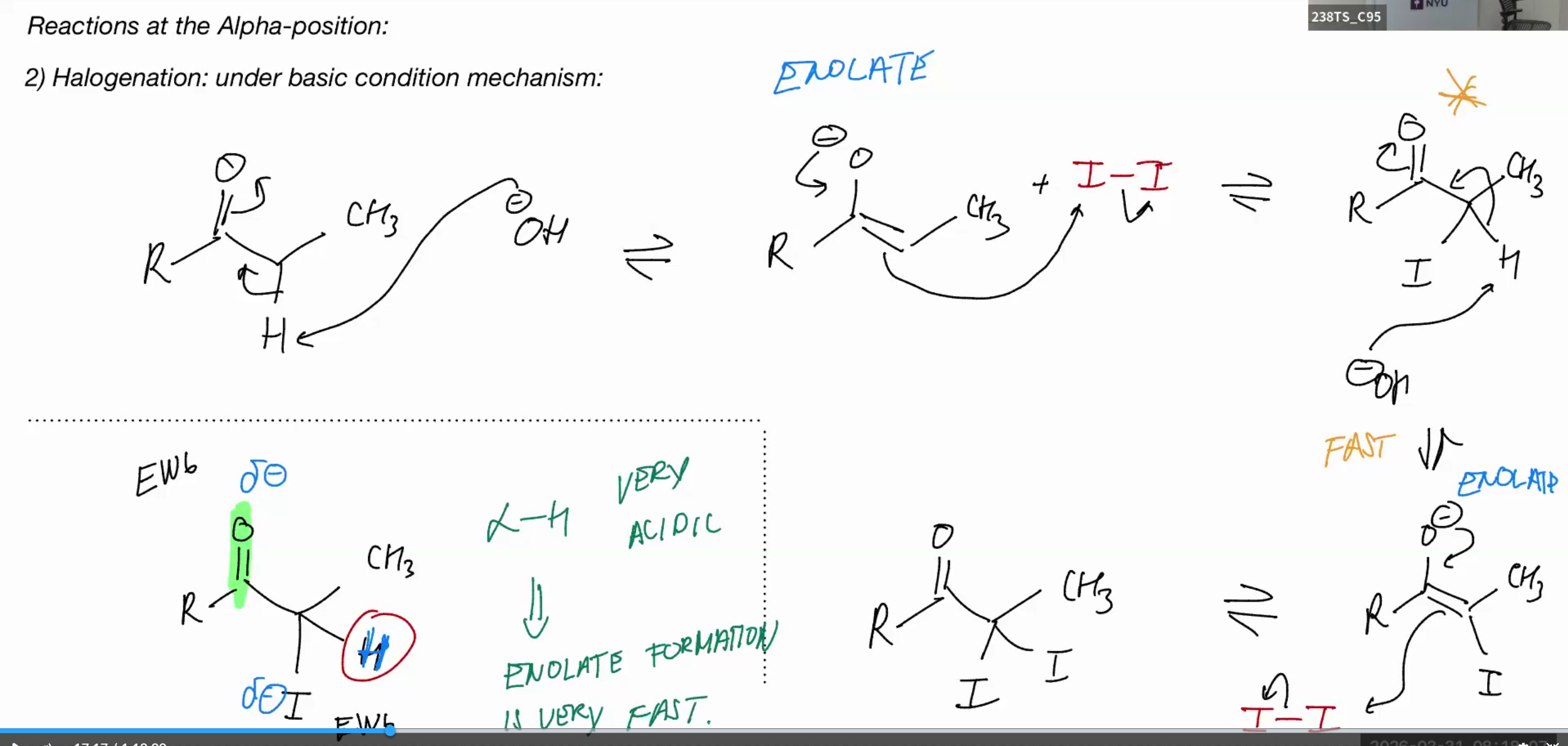
Halogenation (BASIC)
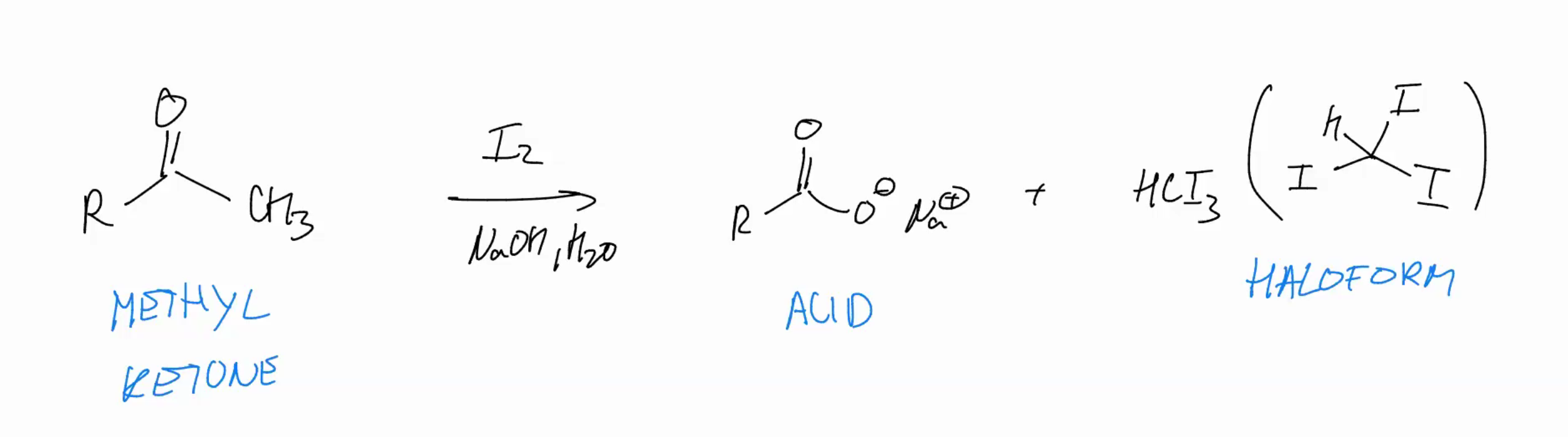
Haloform
start w ketone add base
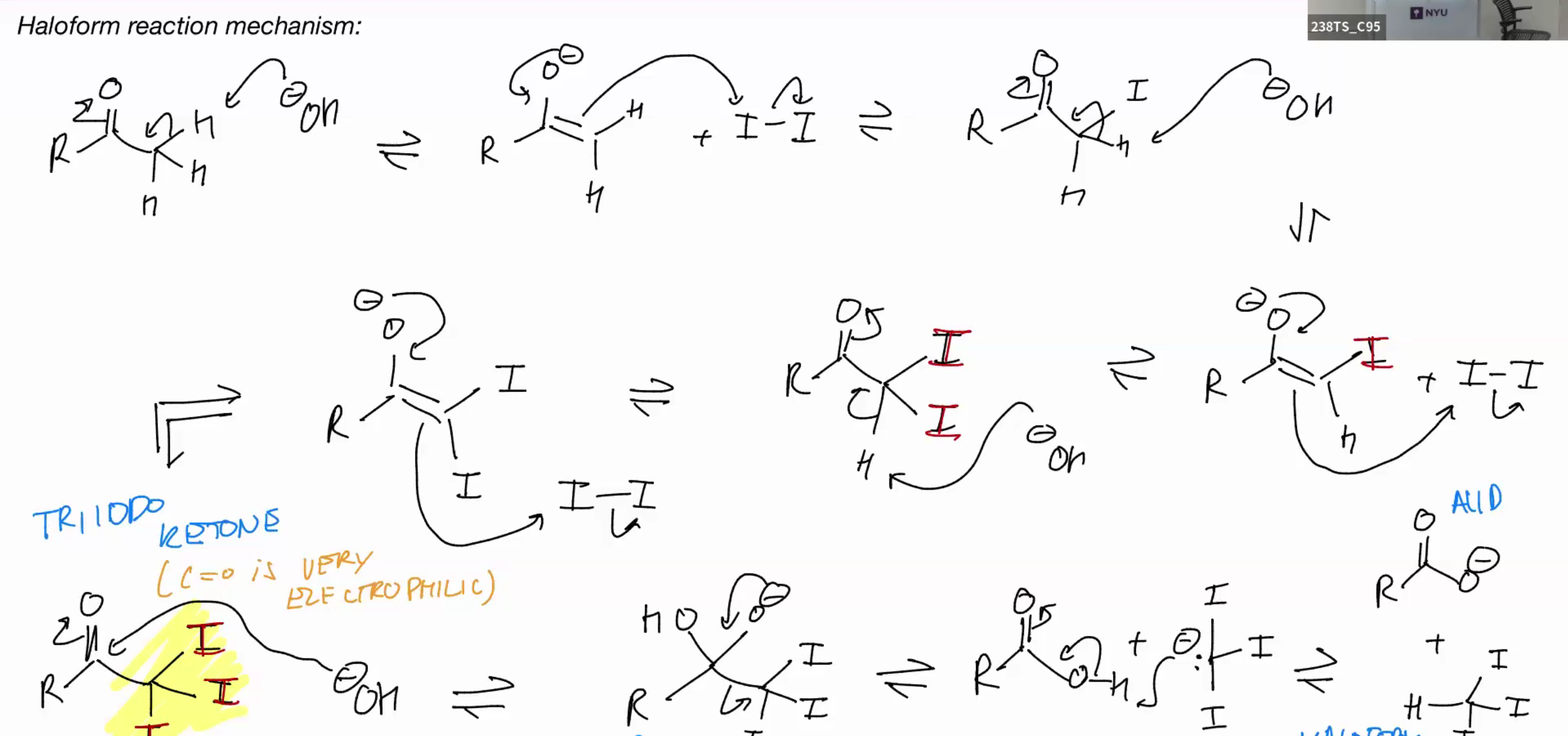
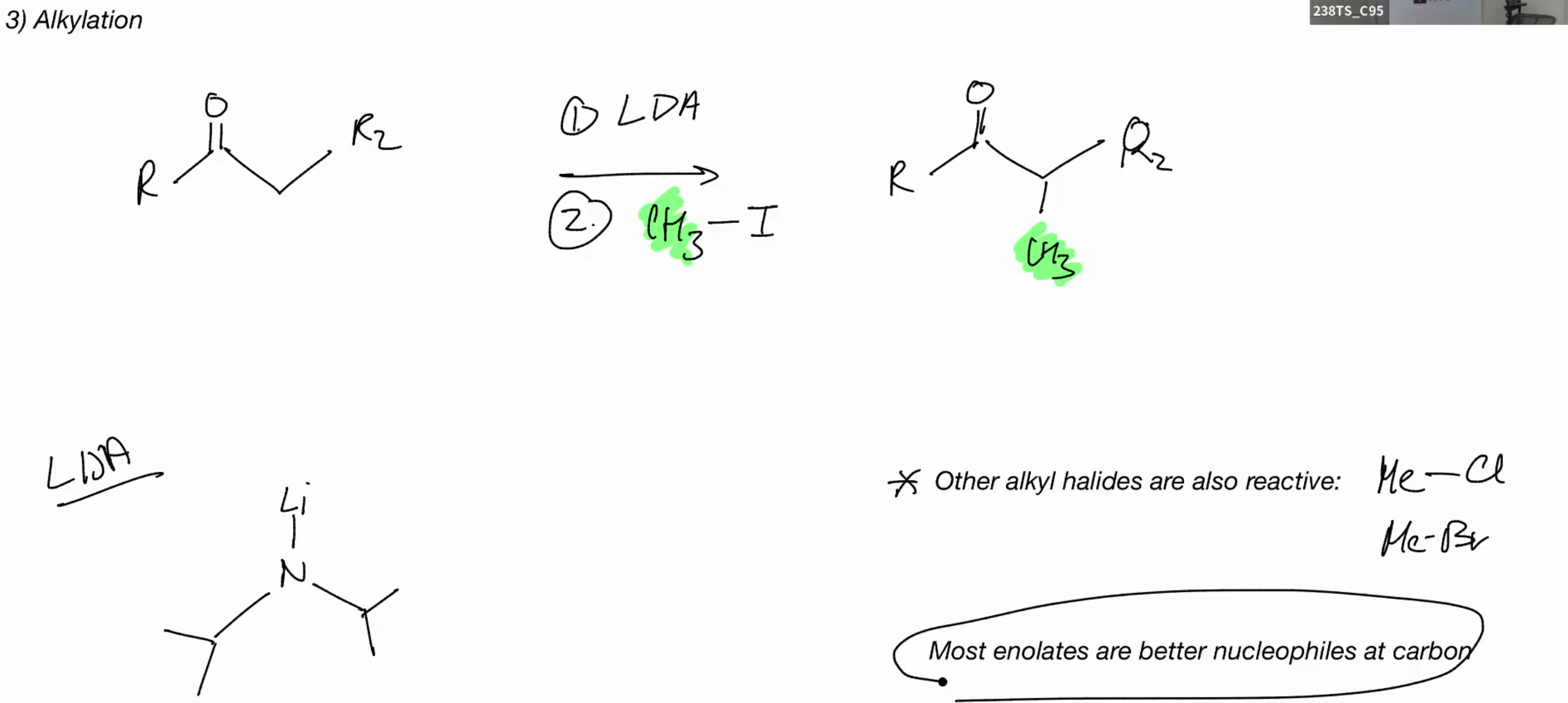
Alkylation
LDA good for monosubstituted
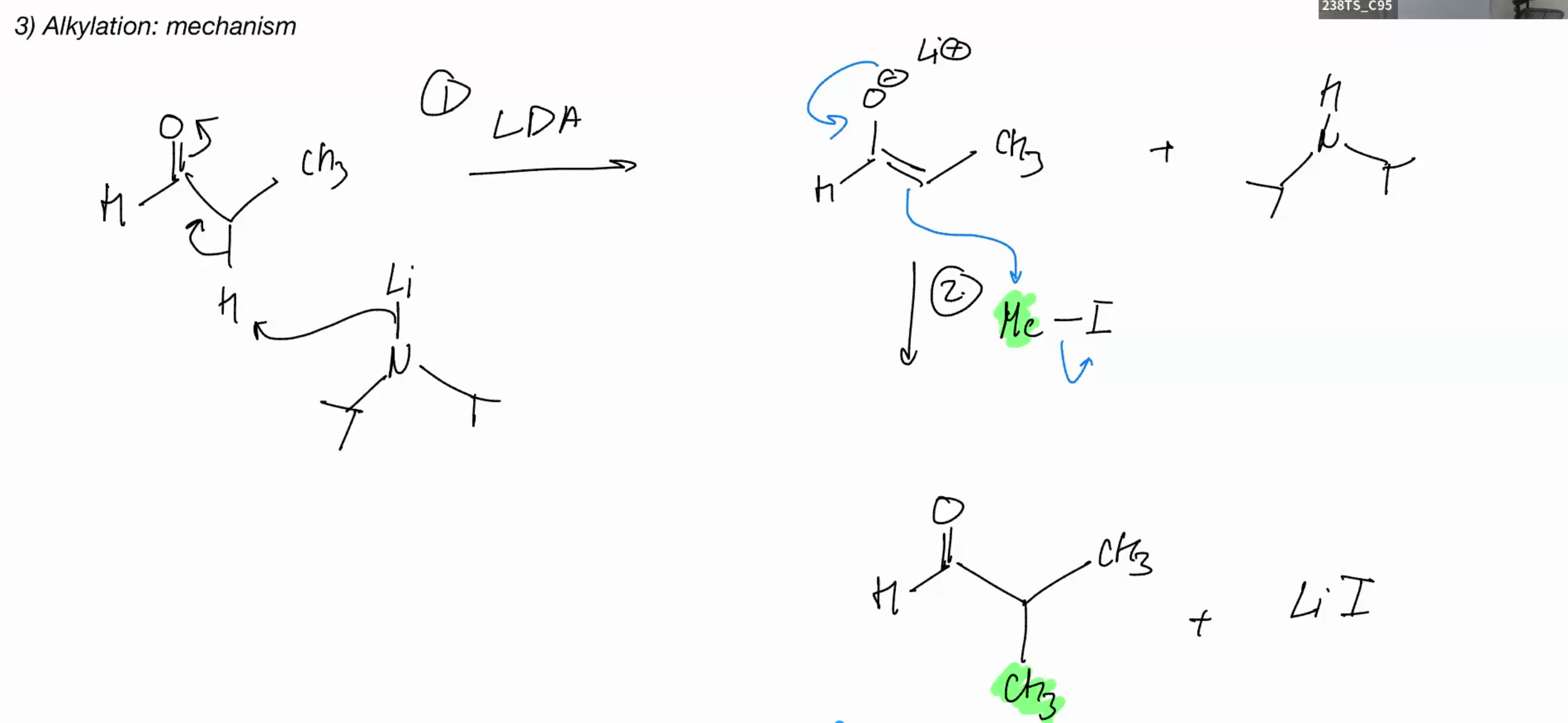
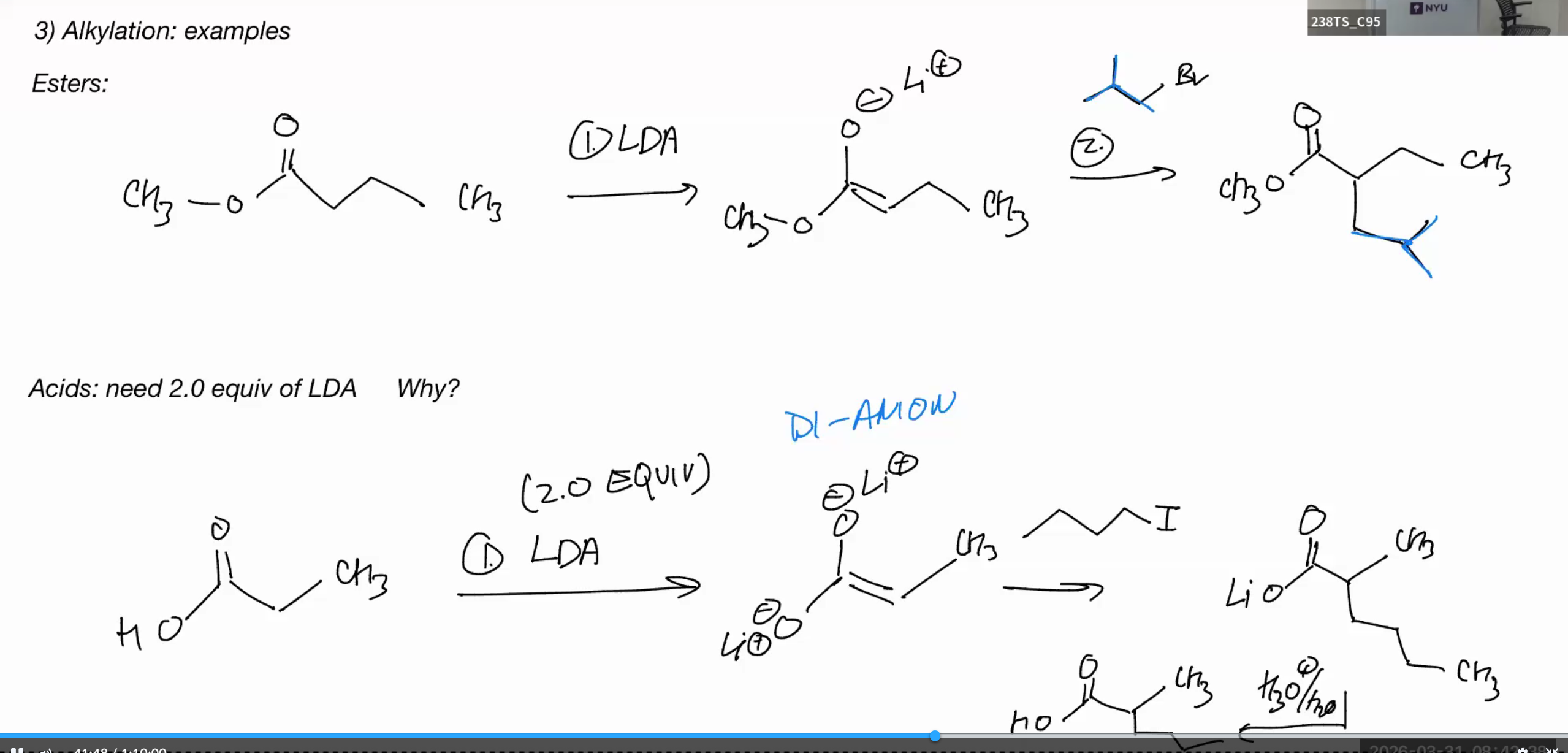
Alkylation examples
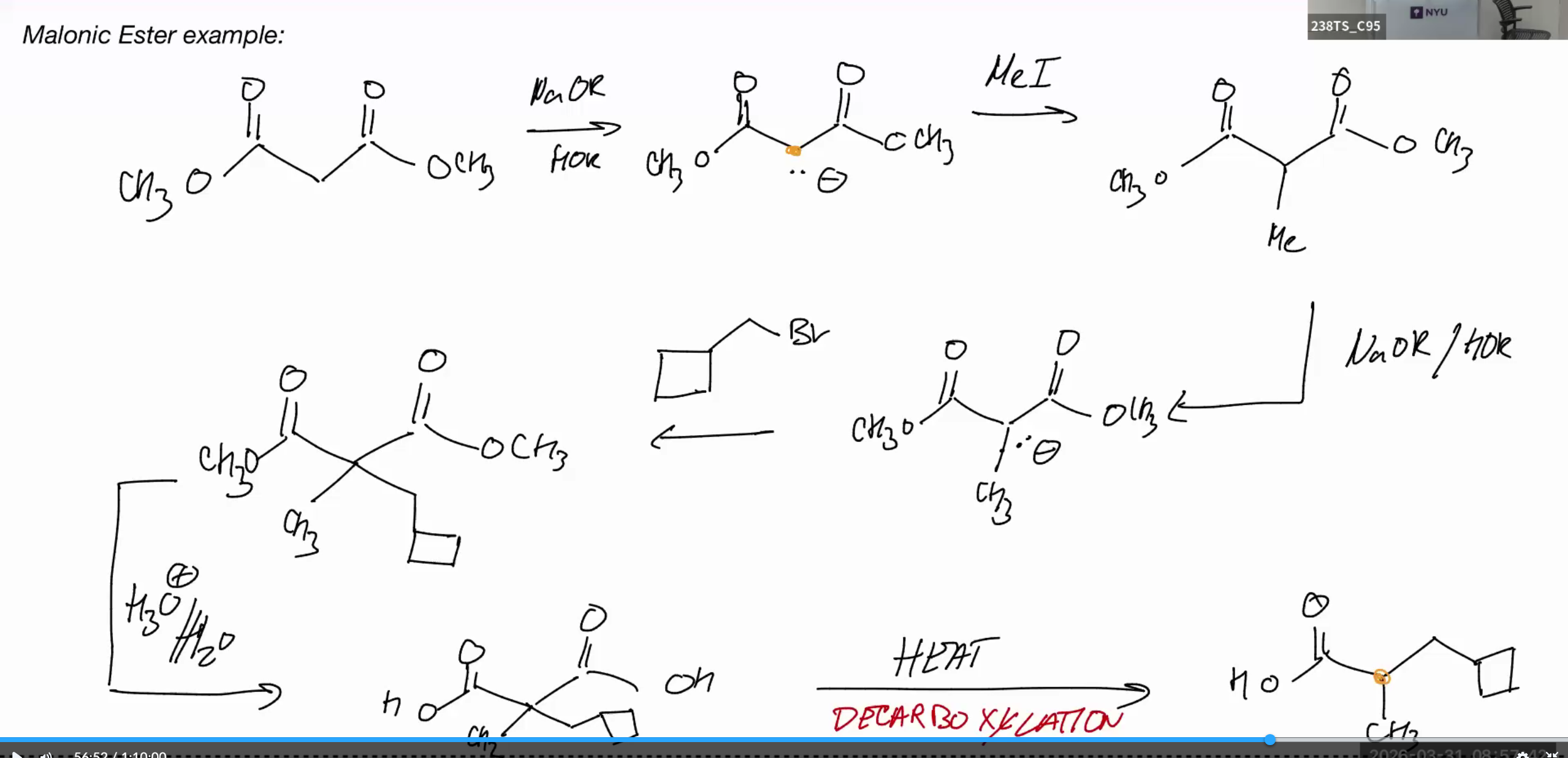
Malonic Ester example
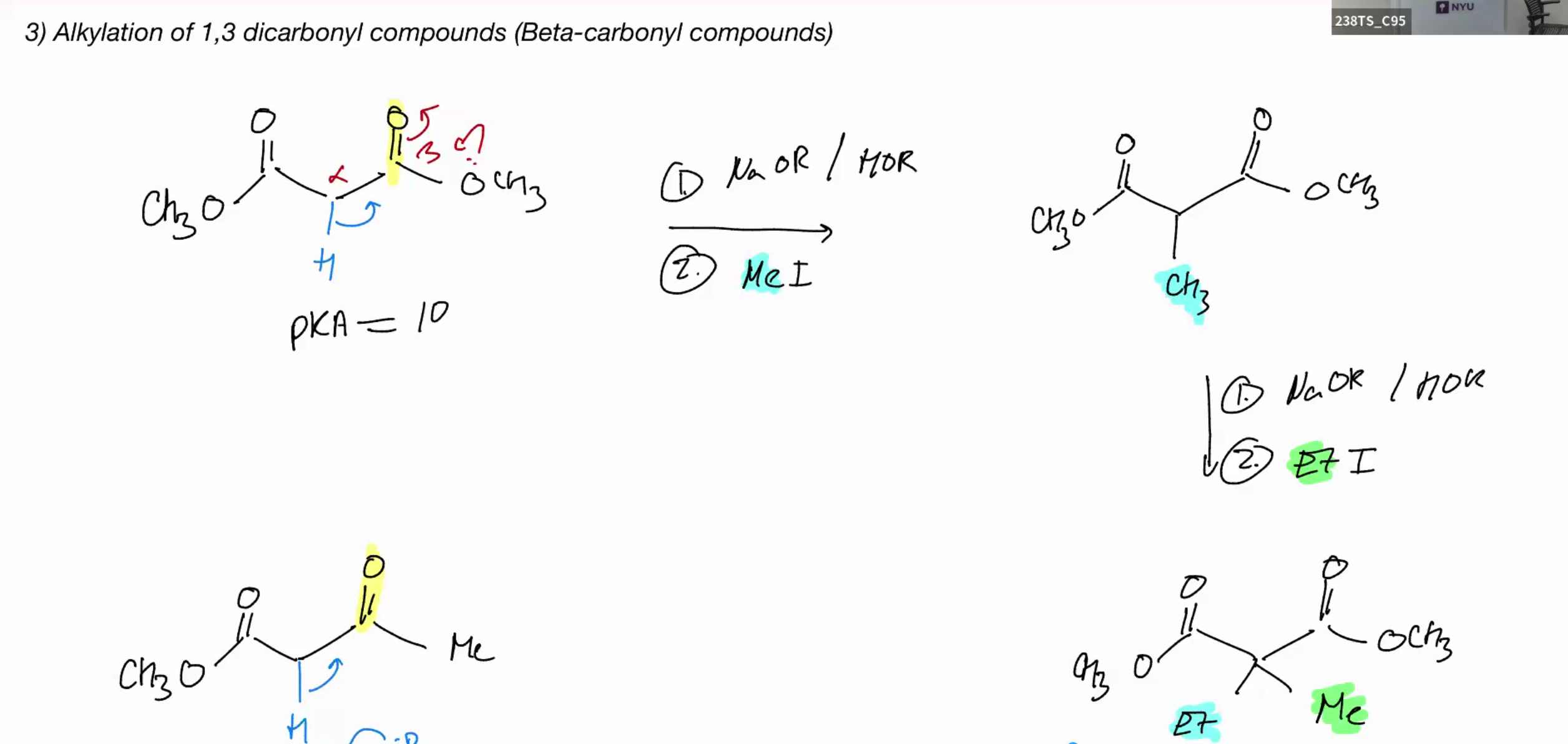
Alkylation of 1,3 dicarbonyl compound (beta carbonyl compounds)
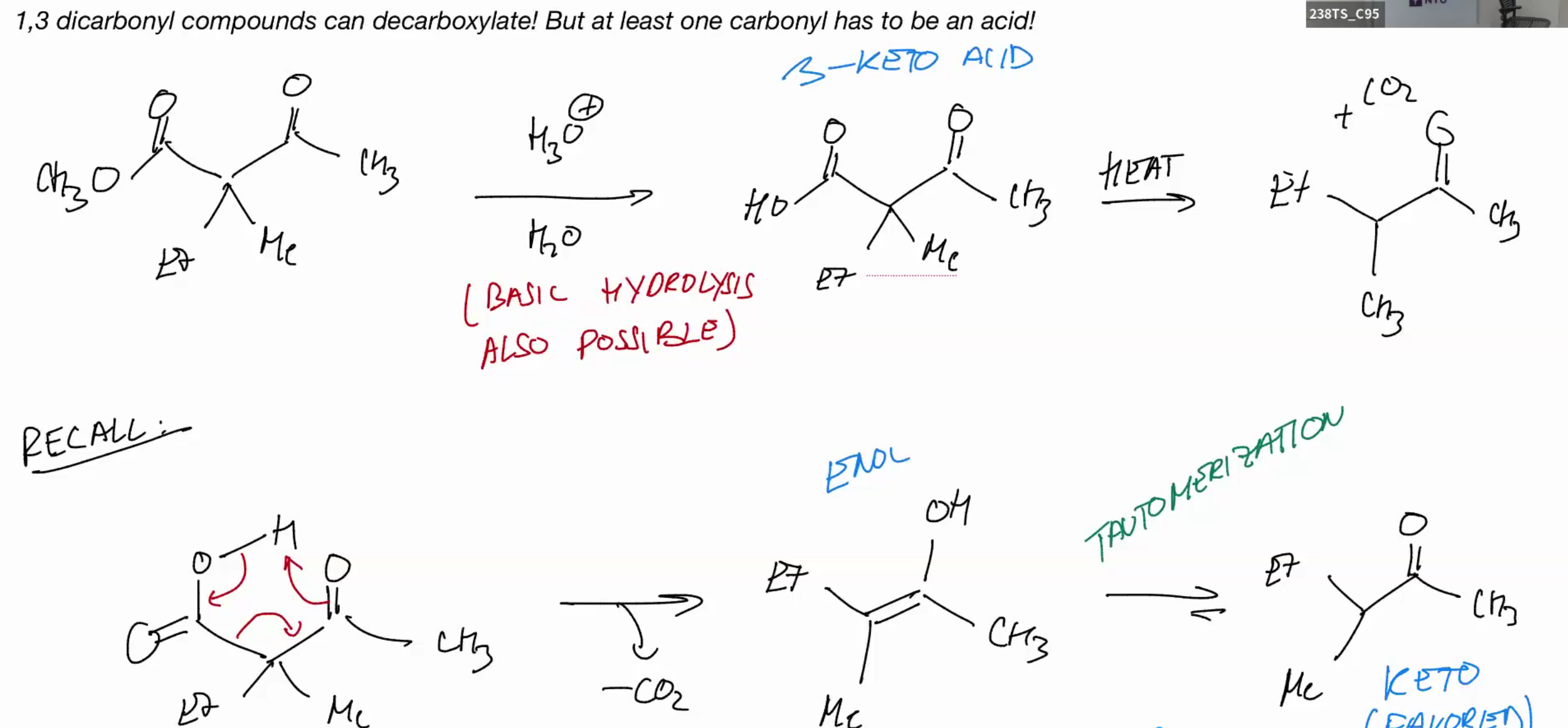
1,3 dicarbonyl compound can be decarboxylated (one carbonyl has to be an acid)
keto favored
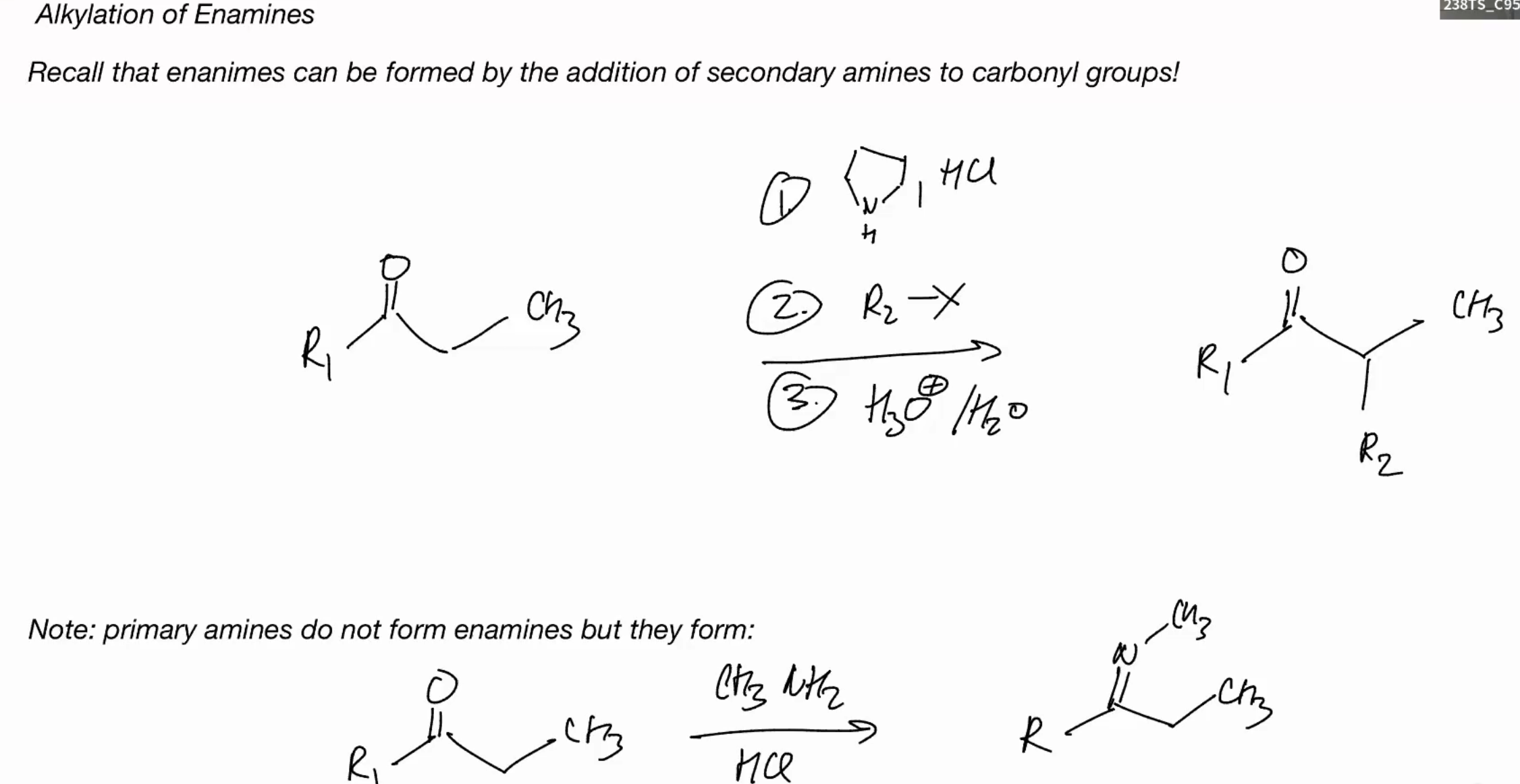
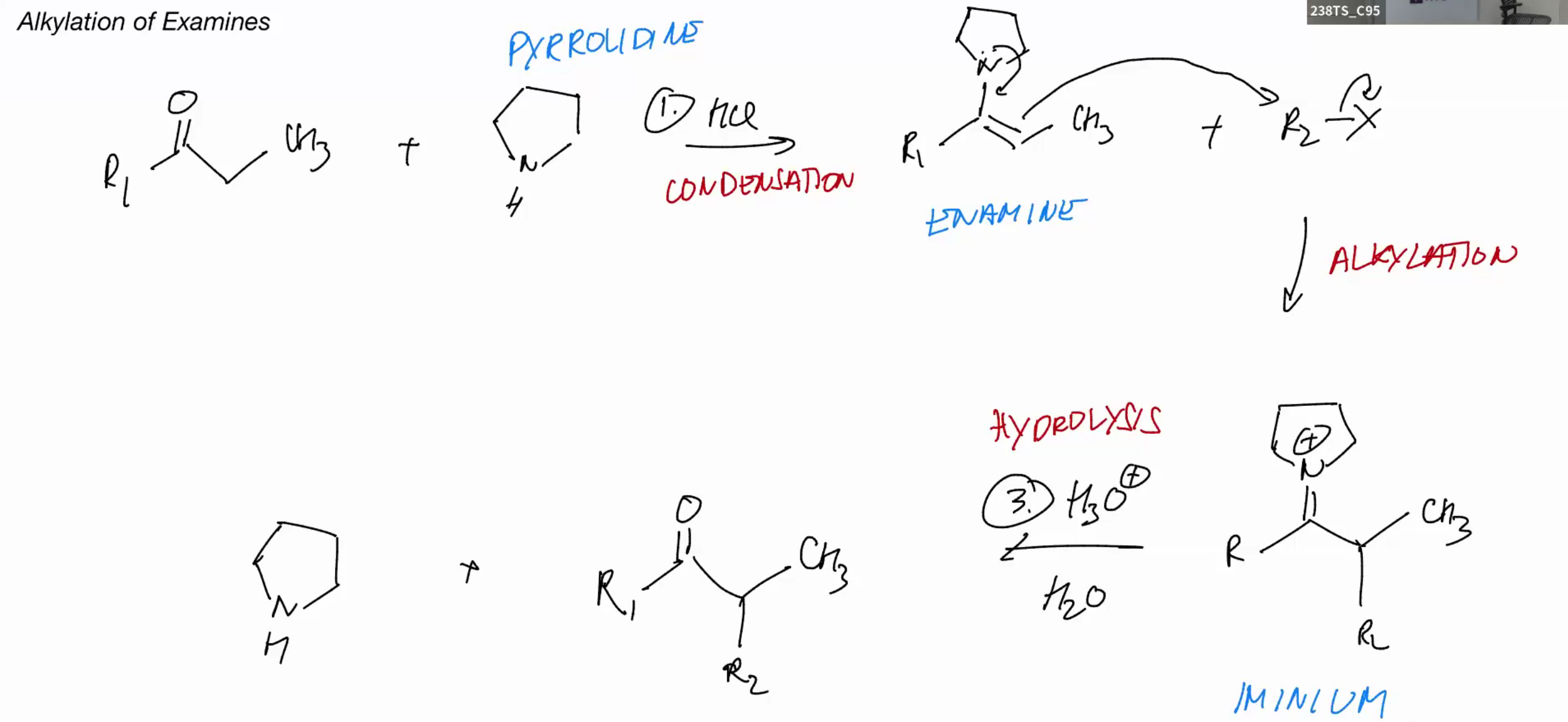
Alkylation of enamines
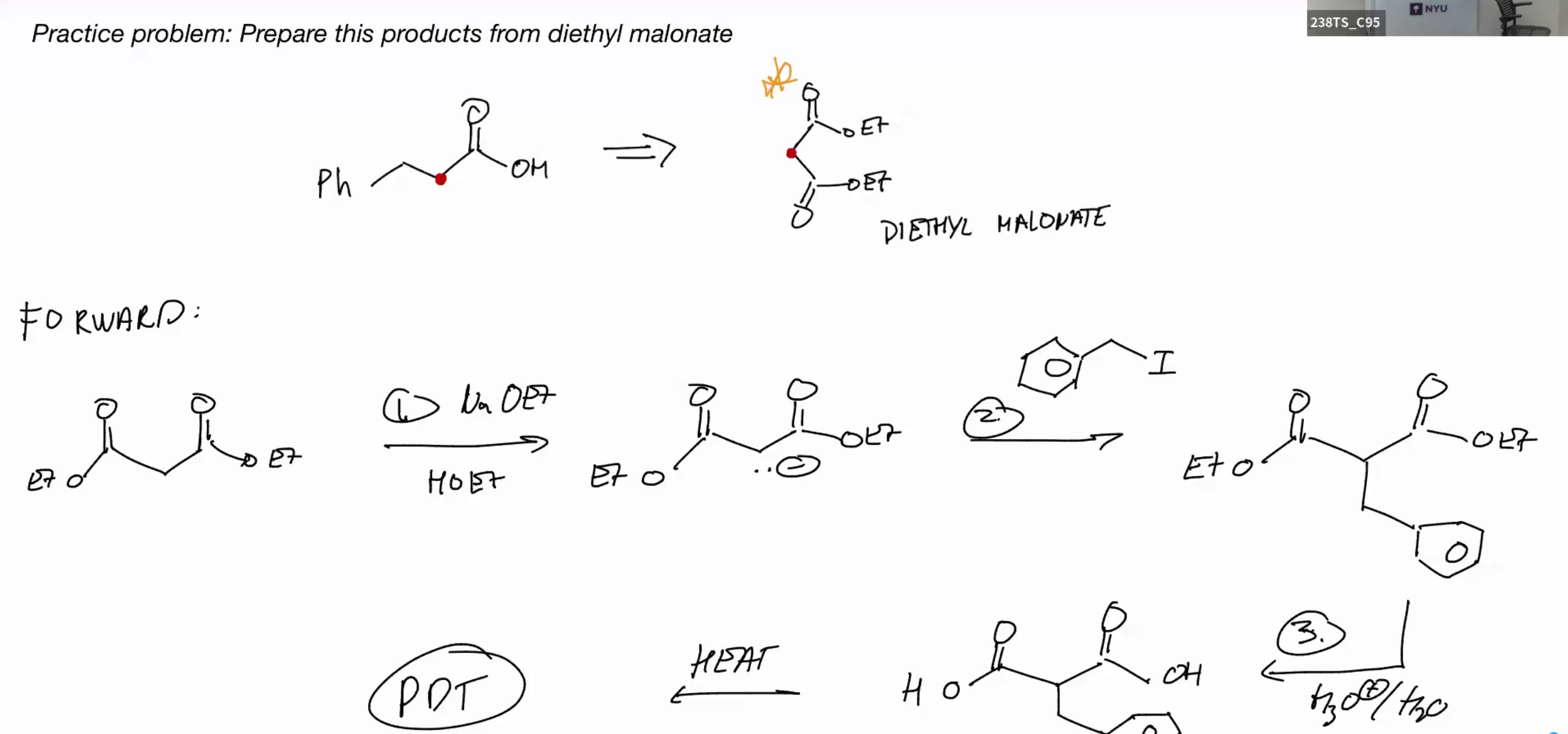
EXAMPLE PROBLEM FOR DICARONYL