Cell Physiology of Disease
1/185
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
186 Terms
The Plasma Membrane
Two molecules thick consisting of amphipathic phospholipids with embedded proteins. It contains important features that can regulate cellular processes including cell-cell signaling, apoptosis, cell division, membrane budding, and cell fusion.
Phospholipid
Have two tails connected by a glycerol and then a phosphate head group. They are very nonpolar at the tail and quite soluble at the head group.
Glycolipid
Have two tails connected by a glycerol and then a sugar attached.
Cholesterol
Has a hydroxyl group connected to a non-polar tail. The hydroxyl group gives it orientation because of its polar nature.
Phospholipid Head, Base, and Tail Groups
Head Groups: amines, serine, inositol
Base Group: glycerol, sphingosine
Tail Groups: C16 for saturated and C18, 20, and 22 for unsaturated
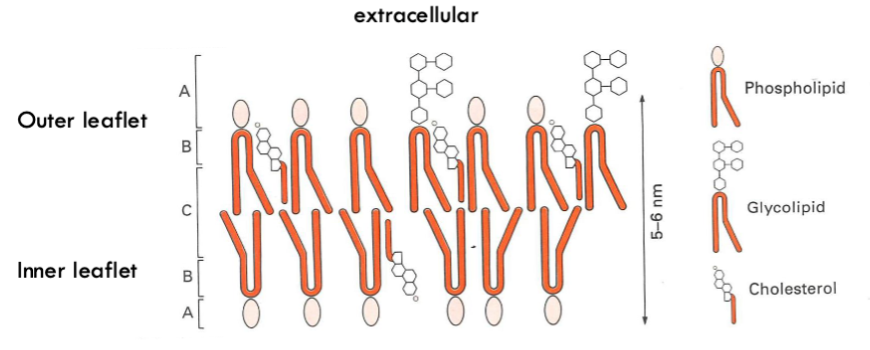
What are the different areas in this diagram?
A. polar region where head groups influence packing
B. Non-polar region where fluidity is influenced by cholesterol and alky chain saturation
C. Fluid non-poar region
Unsaturated vs. Saturated Fats
Fatty acid tails often have double bonds (unsaturated) which put kinks in them to create more fluidity in the membrane.
Cholesterol Content in Membrane
As cholesterol content increases the membrane will become more rigid because the cholesterol molecules stack together
Normally it makes it more fluid though
Different Composition of Inner and Outer Leaflets
There are more negatively charged lipids in inner leaflet
Similar amount of cholesterol
More phosphatidylcholine in the outer layer
Choline is bigger than ethanolamine so creates bigger gaps to create curvature
More phosphatidylethanolamine in the inner layer
Phosphatidylserine is found predominantly in the inner layer (it is negatively charged) this creates a charge imbalance so that it’s more negative on the inside
More polyunsaturated lipids on the inner layer
Different Composition of Leaflets Between Cell Types (Epithelial and Neurones)
There is a lot more cholesterol in neuron leaflets which makes neuronal membranes are less fluid
Different levels of saturation
Lipid Rafts
Areas of the cell membrane that have a different composition with less fluidity and more thickness and are involved in trafficking and signalling processes.
Features of Lipid Rafts
Membrane proteins can’t move in rafts but can move in other sections
Rafts only exist in the outer leaflet
Rafts typically contain sphingolipids and cholesterol and more saturated fats
Signalling Differences in Lipid Rafts
Many proteins associate with rafts to facilitate signalling, whilst others may avoid rafts to maintain slower rates of signalling.
Function of receptors, ion channels, and pumps/transporters
For a cell to be able to control and respond to its environment.
4 Types of Transport Across Cell Membranes
Simple diffusion, carrior-mediated facilitated diffusion, channel-mediated facilitated diffusion, and osmosis.
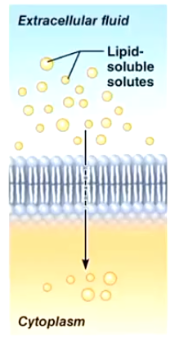
Simple Diffusion
Lipid-soluble molecules diffuse directly through the cell membrane.
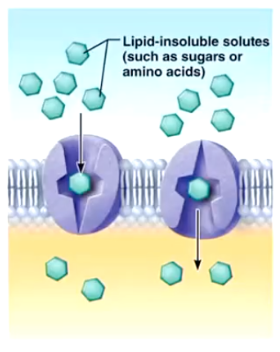
Carrier-Mediated Facilitated Diffusion
Lipid-insoluble solutes such as sugars and amino acids diffuse through the membrane via protein carriers specific for them which change shape upon binding.
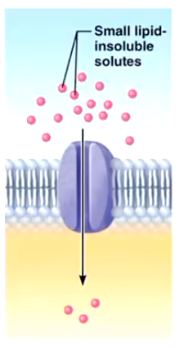
Channel-Mediated Facilitated Diffusion
Small lipid-insoluble solutes travel through a channel protein. Most ions are transported this way and are selected on basis of size and charge.
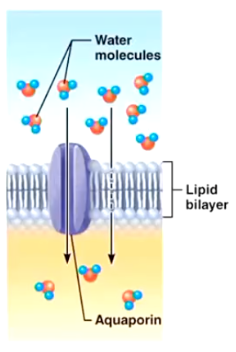
Osmosis
Diffusion of a solvent such as water through aquaporins or through the lipid bilayer.
Two Types of Transporters
Passive and Active Transporters
Passive Transporters (Facilitated Diffusion)
Concentration gradient drives the diffusion of a substrate across the membrane and no energy is required.
Example of a Passive Transporter
Glucose Transporter 2 (GLUT2)
High capacity, low affinity (Km ~20mM)
GLUT2 is upregulated after a meal and allows glucose to passively diffuse down its electrochemical gradient
Defects in the SLC2A2 gene (that encodes GLUT2) are associated with a glycogen storage disease called Fanconi-Bickel syndrome
Active Transporters (Pumps)
Requires energy to move a substrate against a concentration gradient and a membrane protein assists in this process. Two types: primary and secondary
Primary Active Transporters
Energy for the transport process is derived from ATP hydrolysis performed by ATPases which are a arge family of enzymes that catalyse the breakdown ATP – ADP + Pi – release energy. 4 Types include ABC, P-type, F-type, and V-type.
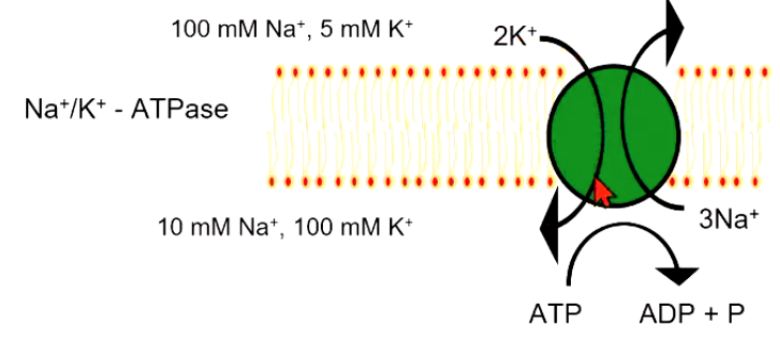
Primary Active Transporter Example
Na+/K+-ATPase where the pump moves sodium and potassium ions in opposite directions, each against its concentration gradient. In a single cycle of the pump, three sodium ions are extruded and two potassium ions are imported into the cell.
Secondary Active Transporters
Couple the movement of solute to the co-transport and/or counter-transport of ions.
Two major types of secondary active transporters
Neurotransmitter transporters and Cl- transporters
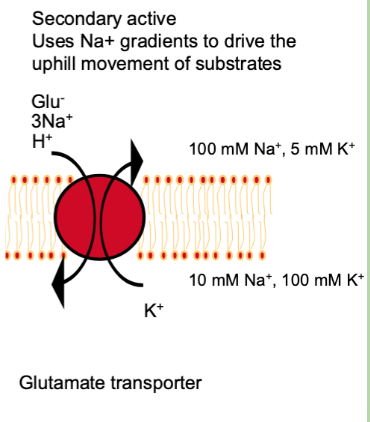
Neurotransmitter Transporters
All Na+ - coupled transporters
Can also be coupled to Cl-, H+, K+
Glutamate, GABA, dopamine, noradrenaline, serotonin, glycine
Important for clearing neurotransmitters from the synaptic cleft
Associated with neurological disease and important drug targets
Ion and water regulation
Cl- Transporters
Other Na+/K+ transport proteins
Regulate ion and water movement across cell membranes
5 Types of Receptors and Channels
GPRCs, Ligand-gated ion channels, voltage-gated ion channels, mechanosensitive channels, and leak channels.
GPRCs
Activate a signal transduction cascade on the inside of the cell via coupling to G-proteins inside cell. Ex. beta-adrenergic receptors
Beta-Adrenergic Signalling
Adrenaline binds to receptor, which induces a change in receptor association with the subunit of the G-protein
GDP is released and replaced with high energy GTP
The alpha subunit (purple) and the beta-gamma subunits dissociate
Alpha and beta-gamma subunits modulate the activity of intracellular enzymes or ion channels
Ligand-Gated Ion Channels
Ligand binding opens a channel to allow ions to move in or out of the cell. LGIC are a large family that allow for the flow of ions (Na+/K+/Ca2+/Cl-) down their electrochemical gradient.
Family of LGICs
Family of Cys-loop receptors
Cationic (serotonin, nicotinic Ach)
Anionic (GABA, glycine)
Ionotropic glutamate receptors (NMDA, AMPA, Kainate)
Voltage Gated Ion Channels
Changes in voltage across the membrane activate the channel. Large family of proteins with a characteristic structure that allow for the movement of Na+, K+, and Ca2+.
Mechanosensitive Channels
Channels open and close in response to mechanical stimuli. All membrane proteins can be affected by pressure exerted from the membrane, Msc are gated by changes in membrane pressure or ‘stretch’.
Leak Channels
Non-gated channels that are always open. Most are leak K+ channels that permit K+ to flow out of the cell to maintain membrane potential (-ve inside).
Cystic Fibrosis
The most common single gene disease that primarily affects the lungs, but also affects the bowel, sweat glands, pancreas and liver. It is usually diagnosed in the first two months of life, often during the newborn screening process that babies undergo at birth.
Incidence of CF in Australia
3,738 people in Australia living with CF
2,124 adults + 1614 kids
57% males
Main Symptoms of CF
Respiratory Dysfunction: infections, obstruction, and destruction
Digestive problems: pancreatic enzymes don’t reach GIT as duct blockage and they also get cysts
High NaCl sweat
Causes of Symptoms of CF
CF patients have reduced Cl- flux/currents conductance in their cells
CF patients also have altered Na+ flux/currents in their cells
Inheritance of CF
Autosomal recessive disease:
No mutations: 100% protein function
One gene allele with F508del mutation: 50% protein function
Both alleles with F508del mutation: <1% function
One gene allele with F508del + one allele with other mutation: generally ~<1-5%
CF Gene
Encodes a protein called CFTR (Cystic Fibrosis Transmembrane Conductance Regulator) which functions as a transmembrane Cl-/HCO3- channel.
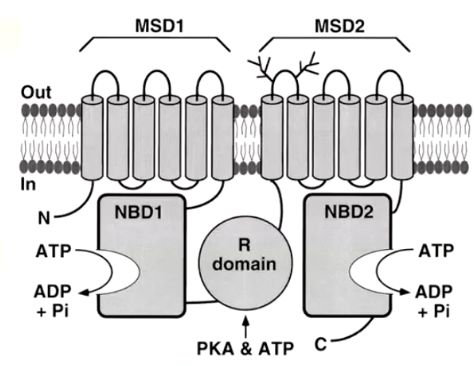
Structural Domains of CFTR
Two transmembrane domains (TMD)
Two nucleotide binding domains (NBD)
One Regulatory domain (R)
How does CFTR open?
Phosphorylation by cAMP/PKA (R) allows ATP hydrolysis (NBDs) to open the channel (TMDs).
Disease-Causing CF Mutations
Over 2500 mutations known. 700 documented as disease-causing
F508Del most common which impacts how the two NBDs interact, so the protein doesn’t fold properly and hence not trafficked to the membrane. Hence, expressed but trapped in Golgi.
Other mutations affect expression and/or function
6 Classes of Disease-Causing CF Mutations
Class I: Early termination of CFTR
Class II: Defective trafficking, doesn’t reach plasma membrane
Class III: reaches plasma membrane but no function
Class IV: reaches plasma membrane but defective function/Cl transport
Class V: normal CFTR but less of it
Class VI: less CFTR as protein is unstable
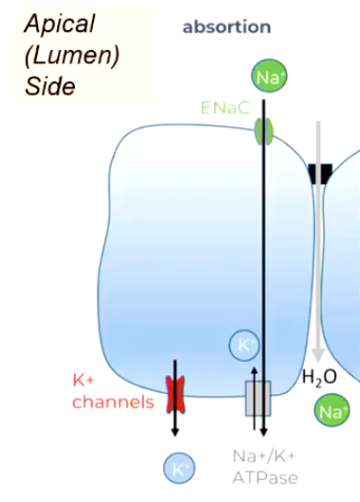
Absorption of Fluid Across Respiratory Epithelia
Start with Na+ pump (basolateral)
Na gradient enables Na+ influx (apical)
Water follows salt by osmosis
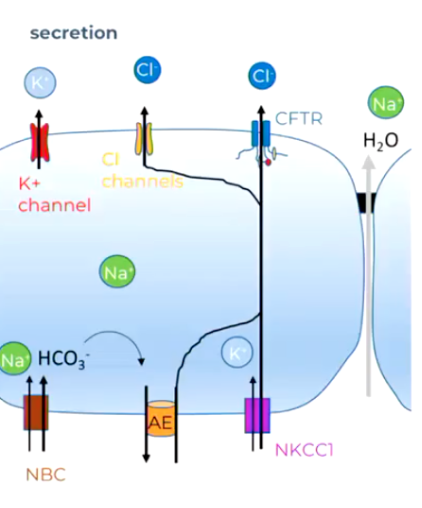
Secretion of Fluid Across Respiratory Epithelia
Start with Na+ pump (basolateral)
Na gradient enables Cl- influx (NKCC, basolateral)
Enables Cl efflux (CFTR, basolateral)
Water follows (osmosis)
Normal Fluid Balance in Respiratory System
Cl - / HCO3- secretion through CFTR drives osmotic flow of water
Absorption of Na + through ENac balances this osmotic gradient and amount of water. ENaC usually inhibited by CFTR
CF Fluid Balance in Respiratory System
There is less fluid in the airways and thick mucus means less clearance, and more bacterial infection, inflammation & degradation
Less Cl - / HCO3- secretion with less osmotic flow of water
Excess absorption of Na+ and water further dehydrates airways.
Normal Sweat Production
Secretory Gland Component: secreted as iso-osmotic NaCl (~100 mM, plus some urea and lactate)
Absorbing duct Component: epithelial re-absorb Na+ and Cl- so that ([NaCl] is reduced to 10-30 mM)
Both controlled by sympathetic nerve control – activates the transporters
Secretory Gland Component
Na pump establishes Na+ gradients (basolateral membrane)
NKCC co-transporter (symport) brings Na+ in with Cl- (and K+) (basolateral)
Cl- diffuses across apical membrane, K+ diffuses back into ISF
Na+ and water follow through gap junctions
Absorbing Duct Component
CFTR Cl - channel opens and Cl - flows across apical membrane
Na+ channel (called ENaC) opens and Na + also flows across apical membrane
Cl - diffuses back across basolateral membrane, Na+ pumped across
This part of epithelia has no water channels and tight gap junctions (so water not re-absorbed)
CF Disfunction of Sweat Glands
CF causes sweat glands to produce sweat with excessively high salt (sodium chloride) levels because faulty CFTR proteins cannot reabsorb chloride from sweat as it moves up the duct. Leads to dehydration…
Two Main Aspects of Ion Channel Function
Gating = opening and closing
Permeation = ion passage
Gating of CFTR
CFTR opens when PKA phosphorylates the R domain (via cAMP signaling) and ATP binds and is hydrolyzed at the NBDs, enabling channel gating.
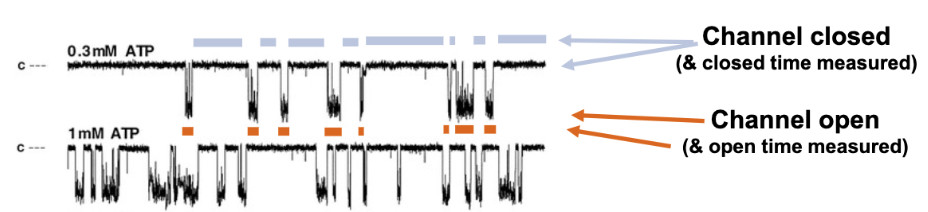
Channel Open Probability (Po)
= (open time)/(open + closed time)
Open and Closed Ion Channel States
Open (ion-conducting) and closed (non-conducting)
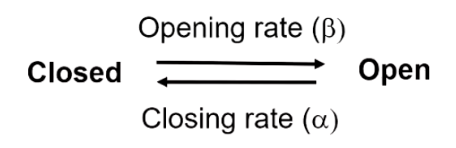
Opening and Closing Rate of an Ion Channel
β (s⁻¹) = rate the channel opens (C → O)
α (s⁻¹) = rate the channel closes (O → C)
Mean Open and Closed Time
The rates determine how long the channel stays in each state:
Mean open time = 1 / α
Mean closed time = 1 / β
F508del Mutation
This mutation means that CFTR does not express in the plasma membrane
Significantly reducing its ability to transport chloride ions even when it reaches the plasma membrane. This mutation results in a severely reduced open probability (Po), which is approximately one-third or less than that of wild-type CFTR.
Aims of Drug Application for Cf
Prevent the misfolding of mutant CFTR proteins = Correctors
Improve the gating of mutant CFTR proteins that do get to the plasma membrane = Potentiators
Increase the number of stable CFTRs at the plasma membrane = Amplifiers and Stabilizers
1989 Cf Dug Development
CFTR gene is cloned. Disease mutations identified and can be characterized using patch clamp recordings and other models.
2000 Cf Drug Development
Aurora Bioscience (later acquired by Vertex) partners with CF Foundation to develop CF drugs that can counteract.
Current Cf Drug Developments
Extended approvals of Trikafta & 1/day Alyferek for younger CF patients and in other countries. Research for improved drugs and ways to target the remaining 10% of patients not helped by Trikafta.
Key Cf Drugs
Kalydeco (ivacaftor, 2012) - approved for 4% of CF patients (G551D mutation).
Orkambi (lumacaftor/ivacaftor, 2015) - approved for 35% of homozygous F508del mutation patients.
Symdeko (tezacaftor/ivacaftor, 2018) - introduced with fewer side effects.
Trikafta (elexacaftor/tezacaftor/ivacaftor, 2019) - approved for 90% of CF patients aged over 12
High Throughput Screening for CFTR Modulators
In HTS assays, cells engineered to express CFTR (often mutant forms such as F508del CFTR mutation) are exposed to thousands of compounds in multi-well plates. Automated robotic systems, fluorescence-based reporters, and imaging technologies measure changes in CFTR activity, typically by detecting chloride or iodide ion movement across the cell membrane. Potentiators give a bigger fluorescent response.
What does ivacaftor (VX-770) do for Cf patients?
Increases the hormone stimulated lumen volumes ie, mucous becomes less thick and sticky
Increases cilia beat frequency
Rapid, significant & sustained improved lung function
Sweat Cl- decreases (below CF cutoff)
BMI (nutrition) increases
Better scores, less hospital visits
What does trikafta a corrector + potentiator do for Cf patients?
It is a highly effective, prescription triple-combination therapy used to treat cystic fibrosis (CF) in patients aged 2 and older with at least one F508del mutation. It significantly improves lung function, reduces coughing, and boosts quality of life, acting as a disease-modifying treatment.
Neonatal Epilepsy
Caused by genetics, is generalized, and can have severe impact.
Late Onset Epilepsy
Cerebrovascular, trauma, neoplasms, neurodegenerative
Mostly focal onset
Higher mortality
Epileptic Seizure
Transient (short) occurrence of signs and/or symptoms due to abnormal excessive or synchronous neuronal activity in the brain.

Epilepsy
A disorder of the brain characterized by an enduring predisposition to generate epileptic seizures, and by the neurobiologic, cognitive, psychological, and social consequences of this condition.
Epilepsy Syndrome
A specific, identifiable type of epilepsy characterized by a cluster of features, including specific seizure types, EEG patterns, and age of onset.
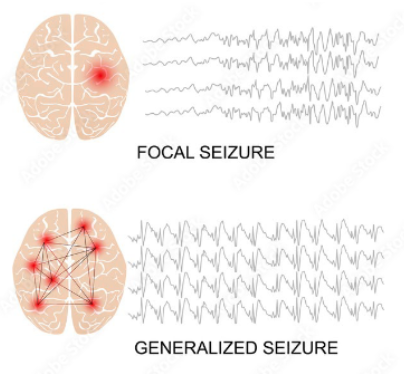
Focal Onset Seizure
Starts in a specific brain region and may be associated with a specific thought, sensation, memory or even action
Can spread to adjacent (“1”), cross-hemispheric (“2”) or subcortical regions (“3”)
Symptoms associated with region
Focal seizure can “generalize”
Generalized Onset Seizure
Results from abnormal electrical activity emerging rapidly across both sides of the brain simultaneously, typically causing consciousness loss
Seizure involves thalamocortical and/or broad cortical changes very early in seizure
Four types include: absence, myotonic, atonic, tonic-clonic
Always unaware
EEG for Seizures
Non-invasive diagnostic tool
Uses electrodes placed outside of the skull (on skin)
Begin as focal or generalized
Ictal vs Interictal
How do seizures start?
Arise from excessive action potentials in neurons (too much excitation) where different Action Potential (AP) firing patterns in response to the same set level of stimulation are seen across the brain.
What do excessive APs in seizures result from?
increased intrinsic excitability, or
increased levels of input at synapses.
Neuronal Connections
Neurons are connected in local and distant circuits through a dense network of synapses. These synapses are either excitatory or inhibitory.
Excitatory Signals
Activates a nerve cell
Increases chance nerve cell fires an action potential
Moves the nerve cell Vm towards voltage threshold
Depolarizing
Inhibitory Signals
Switches off a nerve cell
Decreases chance nerve cell fires an action potential
Moves the nerve cell Vm away from voltage threshold
Hyperpolarizing
Excitatory Synapses
When open allow cations to permeate (Na+ and/or Ca2+) and the postsynaptic response includes cation influx which results in depolarization and an excitatory post synaptic potential.
Excitatory Synapse Neurotransmitter
Glutamate which is detected by ionotropic receptors which include either AMPA (mostly Na+ influx) or NMBA (mostly Ca2+ influx).
Inhibitory Synapses
When open allow anions to permeate (Cl- ) and the postsynaptic response includes anion influx which causes hyperpolarization and an inhibitory post synaptic potential.
Inhibitory Synapse Receptor
GABA is the neurotransmitter in the brain, and glycine is also common in the spinal cord. They are detected by ionotropic receptors. Permeant ion for both receptors are chloride ions (Cl - ).
Causes of Epilepsy
Genetics is the most common cause of epilepsy (70-80%), followed by structural (eg cancer, neurodevelopmental, strokes) and then metabolic.
Genes that Cause Epilepsy
Ion channel genes, synaptic support protein genes, mTOR pathway regulator genes, and chromatin remodelling and transcription regulator genes. Include more than 140 genes identified.
3 Typical Genetic Patterns of Epilepsy
De Novo Mutations (not inherited) are the most common
Polygenic/Complex (complex inheritance of risk variants) are less common
Autosomal Recessive (tranditional inheritance patterns) very infrequent
Dravet Sydrome (an epileptic encephalopathy) Symptoms
Prolonged and frequent seizures, behavioral and developmental delays, movement and balance issues, orthopedic conditions, delayed language and speech issues, growth and nutrition issues, sleeping difficulties, chronic infections, and sensory integration disorders.
Causes of Dravet Syndrome
Many genetic causes, but typically (80%) a de Novo loss of function mutation in the voltage-gated Na+ channel gene, SCNA1. SCN1a will be (NaV1.1) is expressed only in inhibitory interneurons (GABA neurons). Therefore, less APs in GABA neurons = less inhibition therefore more excitation.
Drug to Treat SN1a Loss of Function
Rescue of the cellular defect using Hm1a, a specific activator of SCN1a channels. A specific drug derived from Australian funnel web venom.
Factors Affecting Nerve Excitability
Type of synaptic inputs (EPSPs & IPSPs)
Number and activity of synapses
Intrinsic Excitability (how easily a set input causes an AP or increase in APs)
The type of on channels and shape of neurons contributes to intrinsic excitability
What cellular factors cause seizures?
Too little inhibition
Too much excitation
Altered synapse number or efficacy
Increased intrinsic excitability
Changes in APs
GABAA Receptor
Mediates brain inhibition
Target of many anti-epileptic drugs
Seems to fail in severe seizures
100s of gene mutations impacting GABA
What does KCC2 do?
KCC2 (K+ Cl- co-transporter subtype 2) is a transporter that keeps chloride low inside neurons so GABA stays inhibitory, but during seizures KCC2 membrane expression decreases, chloride builds up, and GABA inhibition fails making seizures worse.
KCC2 as a viable therapeutic strategy for epilepsy
In vitro Cl- transport was increased, there were no effects on normal behaviour, and no effects on normal GABA inhibition in isolated nerves.
Ascending Pain Pathways
Detection of stimulus by sensory nerve fibre endings
Processing of signals in the dorsal horn of the spinal cord
Transmission of response to CNS
CNS response
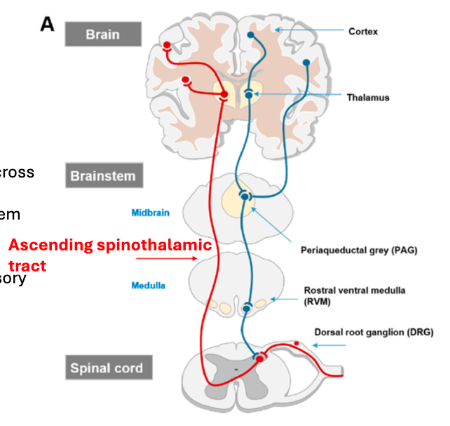
Ascending Pain Pathway Processing in the Brain
Primary afferent nociceptive neurones synapse in the superficial dorsal horn
Axons of nociceptive neurones (second order) cross the midline and ascend the spinoreticular and spinothalamic tracts and project to the brain stem and thalamus
Third order neurones project to the somatosensory region of the cerebral cortex, thalamus and limbic system