1.7 Water, 1.8 Inorganic Ions
1/11
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
12 Terms
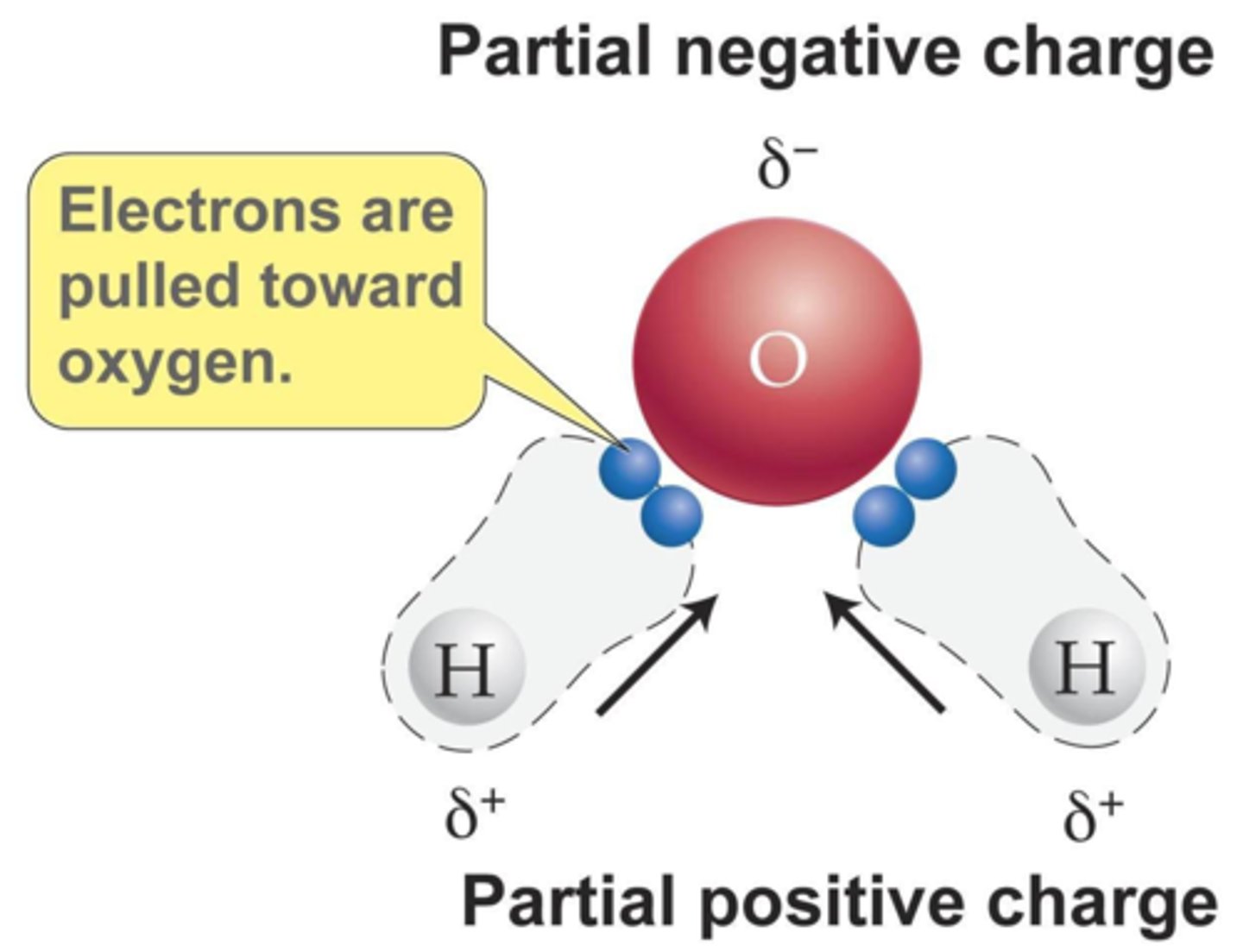
Why is water a polar molecule?
Water is polar because
● oxygen is more electronegative than hydrogen
● attracting electron density more strongly
●creating a slight negative charge δ- on oxygen and a slight positive charge δ+ on hydrogen.
What are 4 biologically important properties of water?
Water is a
● metabolite/solvent for chemical reactions in body
● has a high specific heat capacity
● a high latent heat of vaporization
● exhibits cohesion between molecules.
Why is water significant to living organisms?
Water acts as
● a solvent for polar molecules during metabolic reactions
● helps maintain core temperature
● facilitates cohesion-tension in the transpiration stream.
Describe water as a metabolite
● condensation reaction (removal of a water molecule to form a bond)
● hydrolysis reaction (water is added to break a chemical bond)
describe water as a solvent.
● Water is polar - many ions will dissolve
● allowing for chemical reactions to occur in cells
● transport of metabolites
Describe water 's high specific heat capacity
● many H bonds in water
● alot of energy to break bonds
● temperature of water is stable
● stable habitats, temperature regulation in an organism
Describe water's latent heat of vaporisation.
● large amount of energy required to change its state
● cooling effect for organisms
● little water evaporating loses great amount of heat
What are inorganic ions and where are they found in the body?
Inorganic ions are
● ions that do not contain carbon atoms
● are found in the cytoplasm and extracellular fluid
● often in varying concentrations.
What is the role of hydrogen ions in the body?
● High concentrations of H+ ions lead to low (acidic) pH
● H+ ions interact with hydrogen bonds and ionic bonds in tertiary structure proteins, causing denaturation.
What is the role of iron ions in the body?
● Iron ions (Fe2+) bond to the porphyrin ring to form the haem group in haemoglobin,
●Haem group has binding site to transport 1
molecule of O2 around body in bloodstream.
●4 haem groups per haemoglobin molecule.
What is the role of sodium ions in the body?
● Sodium ions are involved in co-transport for glucose and amino acids absorption in the gut
● involved in the propagation of action potentials in neurons.
What is the role of phosphate ions in the body?
Phosphate ions are components of
●DNA
●ATP
●NADP
●cAMP.