Topic 6b - Reversible Reactions
1/16
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
17 Terms
Physical reactions vs chemical reactions
All physical reactions are reversible
changes of state
Dissolving/crystal
Most chemical reactions are not
A small selection of chemical reactions are reversible
What are all reactions
Exothermic in one direction and endothermic in the other
Reversible reactions
They can be represented like this:
A+B⇌C+D
The double arrow means reversible
Initially rate of forward reaction is high, lots of A+B to collide
Over time the forward rate of reaction decreases as less A+B. But backwards rate increases
Eventually we reach equilibrium, this is when the forward and backward rates are equal, the reaction continues but we observe no further change. This only happens in a closed system
What happens to concentration and rate of reaction for: A+B⇌C+D
At the start of the reaction the concentration and rate of the forward reaction (A+B) are high where as the concentration and rate of the reverse reaction (C+D) is zero
As the reaction continues the concentration and rate of the forward reaction (A+B) are reducing where as the concentration and rate of the reverse reaction (C+D) are increasing
When equilibrium is reached the concentration and rate of the forward reaction (A+B) and reverse (C+D) reactions are both constant
Why do we alter the position of equilibrium and how do we do this
We can alter the position of equilibrium to favour one of the reactions
We can do this by altering one of three conditions:
temperature
Catalyst
Pressure
What is a closed system
If a reversible reaction takes place in a closed system then a state of equilibrium will always be reached. It’s a system with a closed lid, to prevent reactants escaping
What was the name of the chemist that looked at this
Le Chatelier’s
How does temperature affect the position of equilibrium
If we increase the temperature we favour the endothermic direction as there is more energy for the reaction to take in
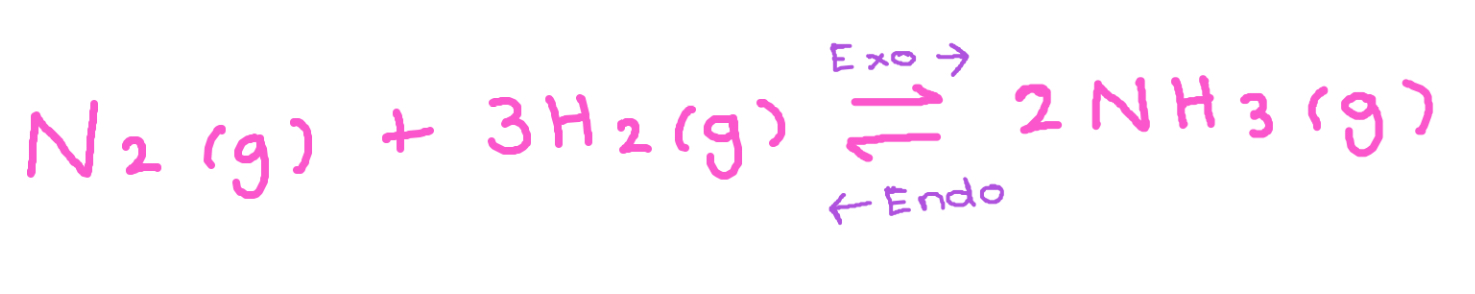
How temperature affects the position of equilibrium for this reaction:
Increase in temp favours endothermic diversion
In this reaction the backwards reaction is endothermic
Equilibrium shifts to favour this reaction to produce more N2 + H2 (Exo →)
How does pressure affect the position of equilibrium
If we increase the pressure that the reaction is happening at then the reaction will favour the reaction that produces less molecules
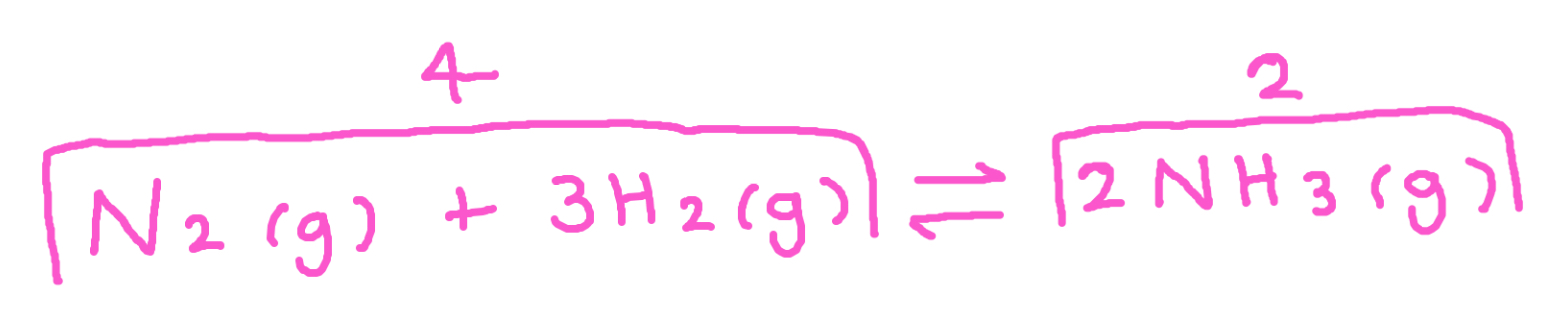
How does pressure affect the position of equilibrium for this reaction
For this reaction, this is the forwards reaction as it produces less molecules (4→2)
Equilibrium shifts to favour this reaction, we produce more NH3
What do catalysts do to any chemical reaction
Speed it up by offering an alternative energy pathway with lowered activation energy requirement
This is exactly the same for reversible reactions, except that the catalyst speeds up both reactions equally
How does catalysts affect the position of equilibrium
It doesn’t! Adding a catalyst has no impact on the position of equilibrium and does not favour either the forward or reverse reactions. However, we do reach equilibrium faster
Exam Eg:
A+B⇌C+D
Explain why the position of the equilibrium lies to the right
More of C than A+B
Exam Eg:
CH4 + H2O ⇌ CO + 3H2
Energy change = +206 (endo)
1) If the temp of this equilibrium was increased, what would happen to the equilibrium yield of hydrogen? - explain why
2) If the pressure of this equilibrium was increased, what would happen to the equilibrium yield of hydrogen? - explain why
1) More H2, why? - increase in temp favours endothermic, this is the → reaction, makes more H2
2) Less H2, why? - increase in pressure favours reaction that produces less molecules in this reaction ←
Exam Eg:
CO + H2O ⇌ CO2 + H2
Energy change = -42 (exo)
1) If the temp of this equilibrium was increased, what would happen to the equilibrium yield of hydrogen? - explain why
2) If the pressure of this equilibrium was increased, what would happen to the equilibrium yield of hydrogen? - explain why
1) Less H2, why? - temperature favours endothermic diversion In ← less H2
2) Stay the same, why? - same number of molecules on both sides
Exam Eg
CO2 + 3H2 ⇌ CH3OH + H2O
If steam was removed from the equilibrium mixture, what happens to the equilibrium yield of methanol? - explain why
Make more methanol, why? - replace lost H2O