BMS 527 exam 2 quizzes
1/125
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
126 Terms
The minimum amount of energy needed for a reaction to proceed
Activation energy is:
A. The difference in energy between the substrates and products
B. The minimum amount of energy needed for a reaction to proceed
C. The energy supplied by the enzyme
D. Total energy of a reaction
Organic e.g. NAD+
Coenzymes are:
A. Inorganic e.g. metal ions
B. Organic e.g. NAD+
Inorganic e.g. metal ions
Cofactors of enzymes tend to be:
A. Organic e.g. NAD+
B. Inorganic e.g. metal ions
False
ΔG =35 kJ. The reaction is thermodynamically favorable. True or false?
True
ΔG =-35 kJ. The reaction is thermodynamically favorable. True or false?
I and V
Substrate X is hydrophobic. The active site of an enzyme that catalyzes a reaction involving substrate X will most likely contain which of the following amino acids where the hydrophobic part of the substrate binds?
A. E and D
B. K and R
C. S and Y
D. I and V
are inherently chiral
Stereospecificity of enzymes is a result of the fact that enzymes:
A. speed up reactions
B. are inherently chiral
C. are inherently achiral
D. drive reactions towards equilibrium
the orthosteric site
The part of an enzyme where a substrate binds is called the active site or:
A. the substrate
B. the allosteric site
C. the enzyme substrate complex
D. the orthosteric site
the enzyme substrate complex
Enzymes bound to a substrate can be called:
A. the apoenzyme
B. the enzyme substrate complex
C. the active site
D. the active enzyme
Covalent
Which of the following is not a "standard" force through which enzymes interact with substrates?
A. Electrostatic
B. H bonding
C. Covalent
D. Hydrophobic
L-amino acids
Enzymes are made of:
A. L-amino acids
B. D-amino acids
Lower the activation energy of reactions
One function of enzymes is to:
A. Lower the activation energy of reactions
B. shift the equilibrium of reactions
C. Be converted to facilitate reactions
D. Convert D to L amino acids
Amino acids and RNA
An enzyme is made from:
A. Amino acids
B. RNA
C. DNA
D. Lipids
No net change in the amount of reactant or product occurs
A reaction at equilibrium means:
A. Both reactants and products are at equal concentrations
B. No net change in the amount of reactant or product occurs
C. The concentration of products is equal to the concentration of reactants
D. The reaction is complete and no more reaction occurs
involves a net release of energy
An exergonic reaction:
A. Proceeds spontaneously
B. involves a net input of energy
C. involves a net release of energy
D. Is never spontaneous
energy cannot be created nor destroyed, only transformed
The first law of thermodynamics is:
A. Energy is created and destroyed, not transformed B. Entropy increases over time C. The universe tends toward disorder D. Energy cannot be created nor destroyed, only transformed
ΔH -TΔS
Gibbs free energy is equal to:
A. ΔH -TΔS
B. ΔT -HΔS
C. H -T x S
D. ΔS -TΔH
the substrate induces tertiary and quarternary conformational changes in the enzyme to fit the active site
The induced fit hypothesis of enzyme mechanism states that:
A. the enzyme has a preformed complementary binding site for its substrate
B. the substrate induces tertiary and quarternary conformational changes in the enzyme to
fit the active site
C. the substrate undergoes a conformational change to fit the enzyme active site
D. the enzyme covalently binds the substrate
The active site is preformed to be complementary to the substrate
The lock and key hypothesis of enzyme function states that:
A. The active site is preformed to be complementary to the substrate
B. The enzyme adapts to accomodate the substrate
C. The active site is preformed to fit any molecule
D. Involves tertiary and quaternary structural alterations in the enzyme
The universe tends toward disorder
The second law of thermodynamics is
A. Entropy decreases over time
B. Energy cannot be created nor destroyed, only transformed
C. Enzymes are biological catalysts
D. The universe tends toward disorder
a high energy, unstable intermediate form of the substrate
The transition state of an enzyme catalyzed reaction is:
A. a highly stable form of the enzyme substrate complex
B. a high energy, unstable intermediate form of the substrate
C. a high energy, unstable intermediate form of the enzyme
D. a low energy, stable intermediate form of the enzyme
Joules
Units of energy used in biochemistry are:
A. Joules
B. Grams
C. Watts
D. Meters
Enzymes shift the equilibrium of a reaction
Which of the following is not true of enzymes
A. Enzymes shift the equilibrium of a reaction
B. Enzymes increase the rate of a reaction
C. Enzymes are not themselves consumed during a reaction
D. Enzymes speed up the establishment of an equilibrium
The rate of enzyme catalyzed reactions
Enzyme Kinetics is the study of what?
A. The thermodynamics of enzyme cataysed reactions
B. The rate of enzyme catalyzed reactions
C. The amount of product an enzyme can make
Plateau
In Michaelis-Menten kinetics, when the substrate concentration significantly exceeds (>>>) the enzyme concentration, the velocity of a reaction will:
A. Increase
B. Decrease
C. Plateau
D. None of the above
The velocity of the reaction at the maximum enzyme rate
In the Michaelis-Menten model of enzyme kinetics, Vmax is equal to what?
A. The substrate concentration at the maximum reaction rate
B. The substrate concentration at half the maximum reaction rate
C. The enzyme concentration at half the maximum reaction rate
D. The velocity of the reaction at the maximum enzyme rate
Km
When V =1/2 Vmax, the substrate concentration for that reaction is equal to what?
A. Km
B. Kd
C. Vmax
D. 1/2 Vmax
B
Enzyme A has a higher Km than Enzyme B. Which enzyme has a higher affinity for its substrate?
A. A
B. B
C. Both the same
D. Cannot tell from this information
Enzyme C
Enzyme C has a lower Km than Enzyme A. Which enzyme has a higher affinity for its substrate?
A. Enzyme A
B. Both the same
C. Enzyme C
D. Cannot tell from this information
False
An enzyme with a high Km is "easy" to saturate with substrate (compared to an enzyme with low Km). True or False?
Below Km
At a constant enzyme concentration, increases in the substrate concentration have the greatest effect on velocity when the substrate concentration is _________?
A. Below Km
B. Above Km
C. Constant throughout
D. Enzyme specific
allosterism
The process by which the interaction of a chemical or protein at one location influences the function of the same macromolecular complex at a distinct site is known as what?
A. Feedback loop
B. Inhibition
C. Covalent modification
D. Allosterism
homoallosteric effectors
Effectors that bind at the active site of one subunit and generate effects in the active site of another subunit are called:
A. Heteroallosteric effectors
B. Covalent modifiers
C. Homoallosteric effectors
D. Cooperative modifiers
Negative heterotrophic allosteric effector
2,3-bisphosphoglycerate (2,3-BPG) is a ___________ of hemoglobin?
A. Negative heterotrophic allosteric effector
B. Positive heterotrophic allosteric effector
C. Negative homotrophic allosteric effector
D. Positive homotrophic allosteric effector
True
The Y intercept on a Lineweaver-burk plot tells you 1/Vmax. True or False?
True
The x intercept on a Lineweaver-Burk plot tells you -1/Km. True or false?
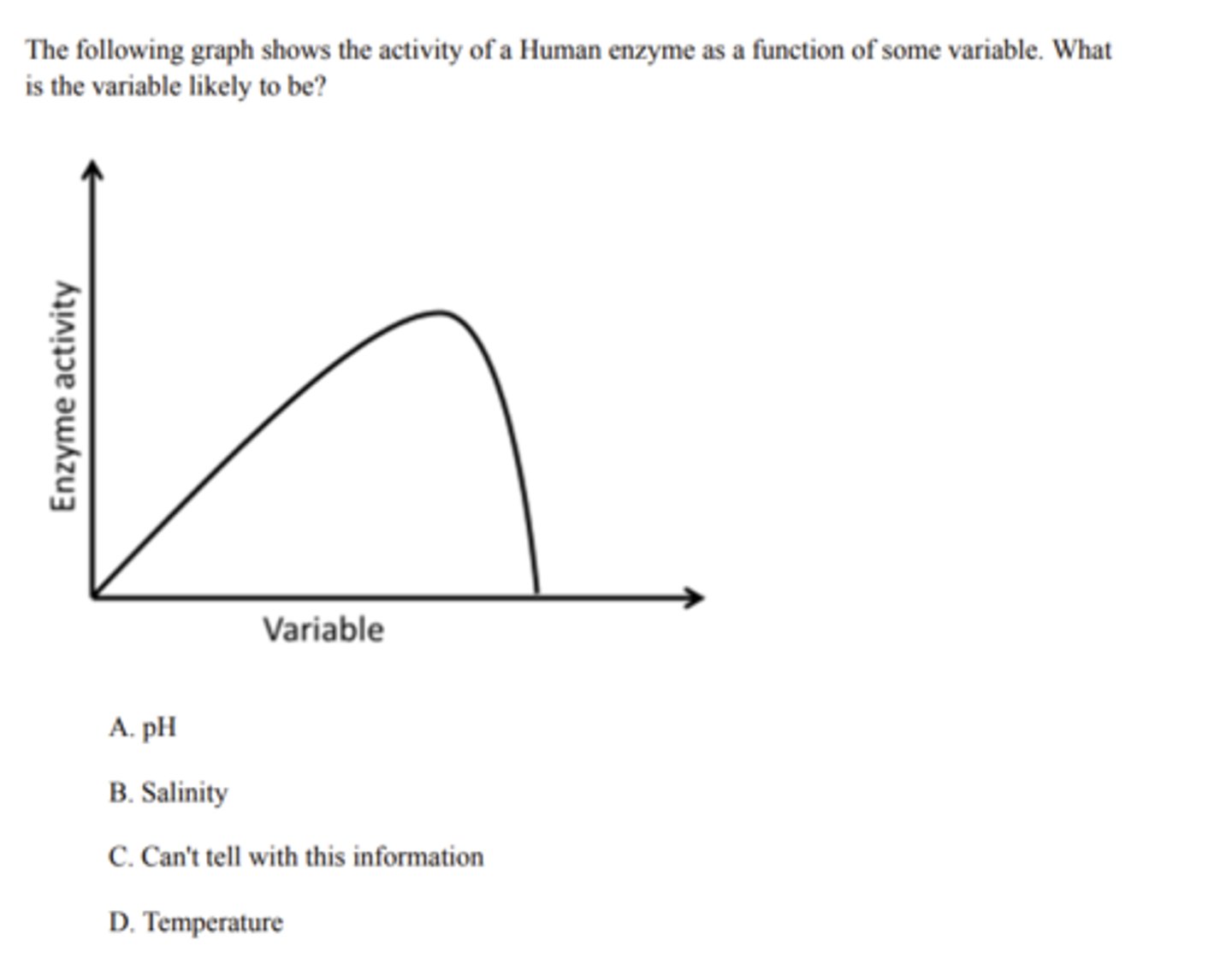
Temperature
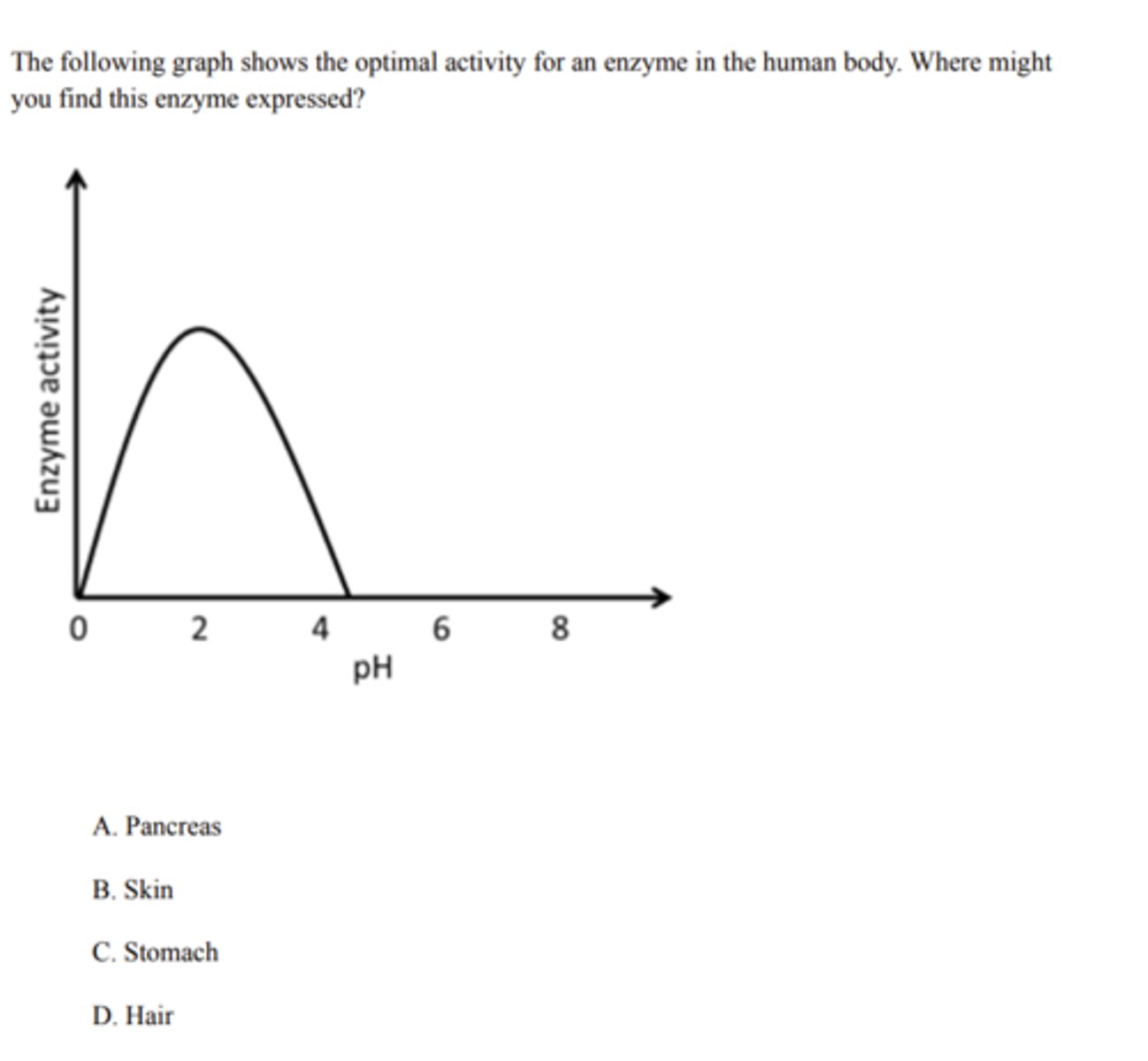
Stomach
The Kcat of the diseased enzyme is lower than the healthy enzyme
Gaucher's disease is a genetic disorder in which a cerebroside accumulates in cells due to a defect in the enzyme lysosomal glucocerebrosidase (LGase).
LGase normally resides in the cytopasms of cells where it catalyses the breakdown of its substrate cerebroside to its products sphingosine, fatty acids and monosaccharides. However in Gaucher's disease, a missense mutation in amino acid 370 substitutes N for S (LGaseN370S ) and renders the enzyme less active and unable to efficiently convert substrate to product as quickly as the healthy enzyme. The cerebroside subsequently accumulates via hydrophobic interactions and forms toxic fibrils.
Which of the following statements about the Gaucher's disease enzyme LGaseN370S is true?
A. The diseased enzyme has an exposed hydrophobic amino acid leading to the formation
of toxic fibrils
B. The Kcat of the diseased enzyme is lower than the healthy enzyme
C. A Michaelis-Menten plot cannot be drawn for this enzyme
D. The Kcat of the diseased enzyme is higher than the healthy enzyme
HGPRT

Because competitive inhibitors can be outcompeted by increasing substrate concentrations
Why is Vmax unaffected by competitive inhibitors?
A. Because competitive inhibitors always irreversibly stop enzyme activity
B. Because competitive inhibitors are allosteric
C. Because competitive inhibitors don't affect Km
D. Because competitive inhibitors can be outcompeted by increasing substrate concentrations
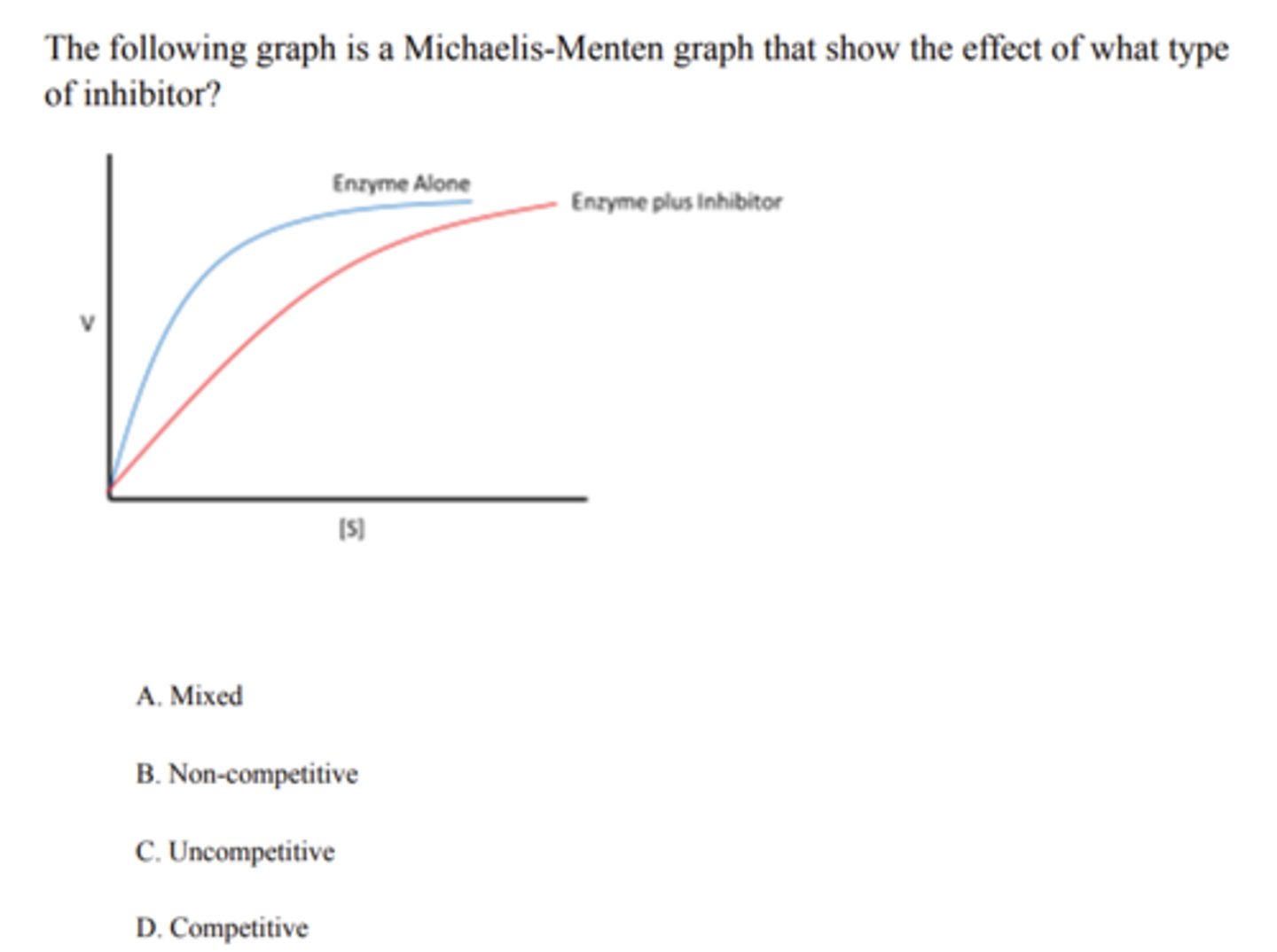
Non-competitive
Cyanide is an example of what type of inhibitor?
A. Competitive
B. Covalent
C. Uncompetitive
D. Non-competitive
Covalent modification of a serine in the enzyme active sit
Diisopropylfluorophosphate (DIFP) is an irreversible inhibitor of acetylcholinesterase. How does DIFP inhibit this enzyme?
A. Covalent modification of a serine in the enzyme active site
B. Covalent modification of a methionine in the enzyme active site
C. Non-covalent competitive inhibtion of the enzyme
D. Allosteric inhibition of the enzyme
Plateau
When the substrate concentration >>>enzyme, the velocity of a reaction will:
A. Increase
B. Decrease
C. Plateau
D. None of the above
Feed forward effect
A pathway of reactions proceeds A to B, B to C and C to D. Each step is catalyzed by an enzyme. The product B increases the conversion of C to D by specifically activating the C-D converting enzyme. What mechanism is this an example of?
A. Feedback loop
B. Feed forward effect
C. Allosteric regulation
D. Covalent modificatio
feedback loop
A pathway of reactions proceeds A to B, B to C and C to D. Each step is catalyzed by an enzyme. The product D decreases the production of B from A by inhibiting the enzyme responsible for that reaction. What mechanism is this an example of?
A. Feedback loop
B. Feed forward effect
C. Covalent modification
D. Phosphorylation
irreversible
Acetylation of an amino acid function group in the active site of an enzyme is one potential mechanism of what type of inhibitor
A. Irreversible
B. Reversible
the Km is unaffected by the inhibiter
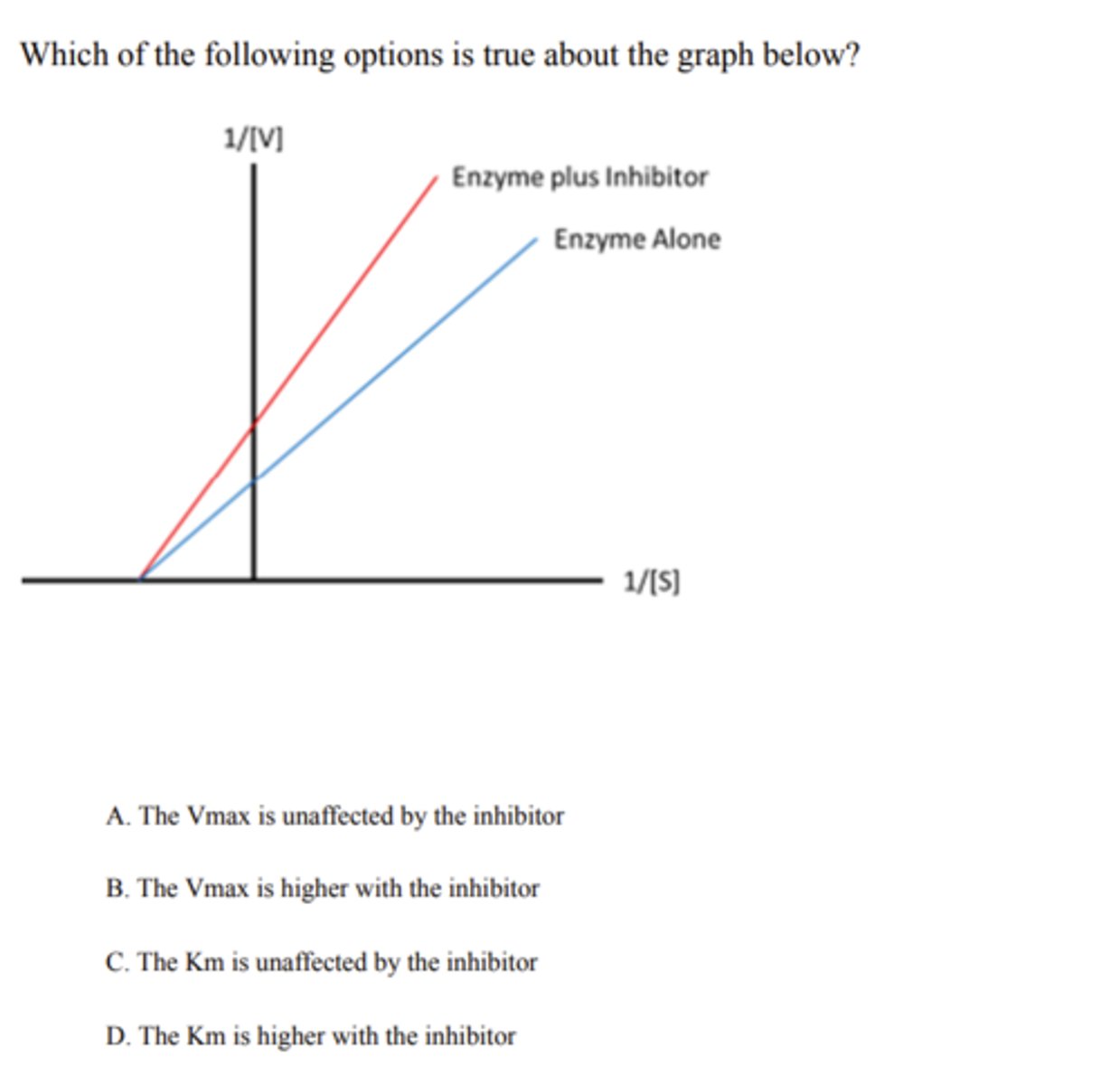
competitive
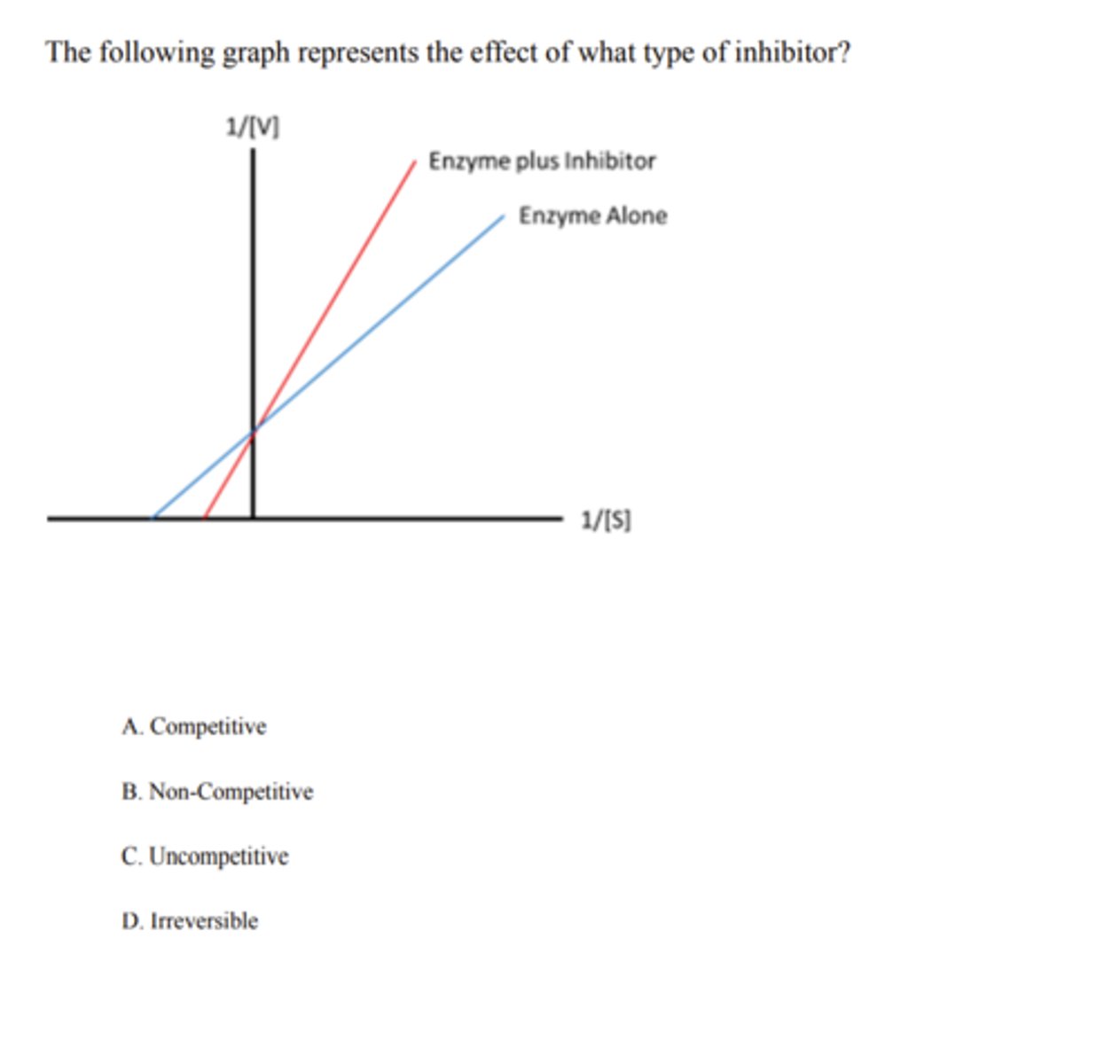
uncompetitive
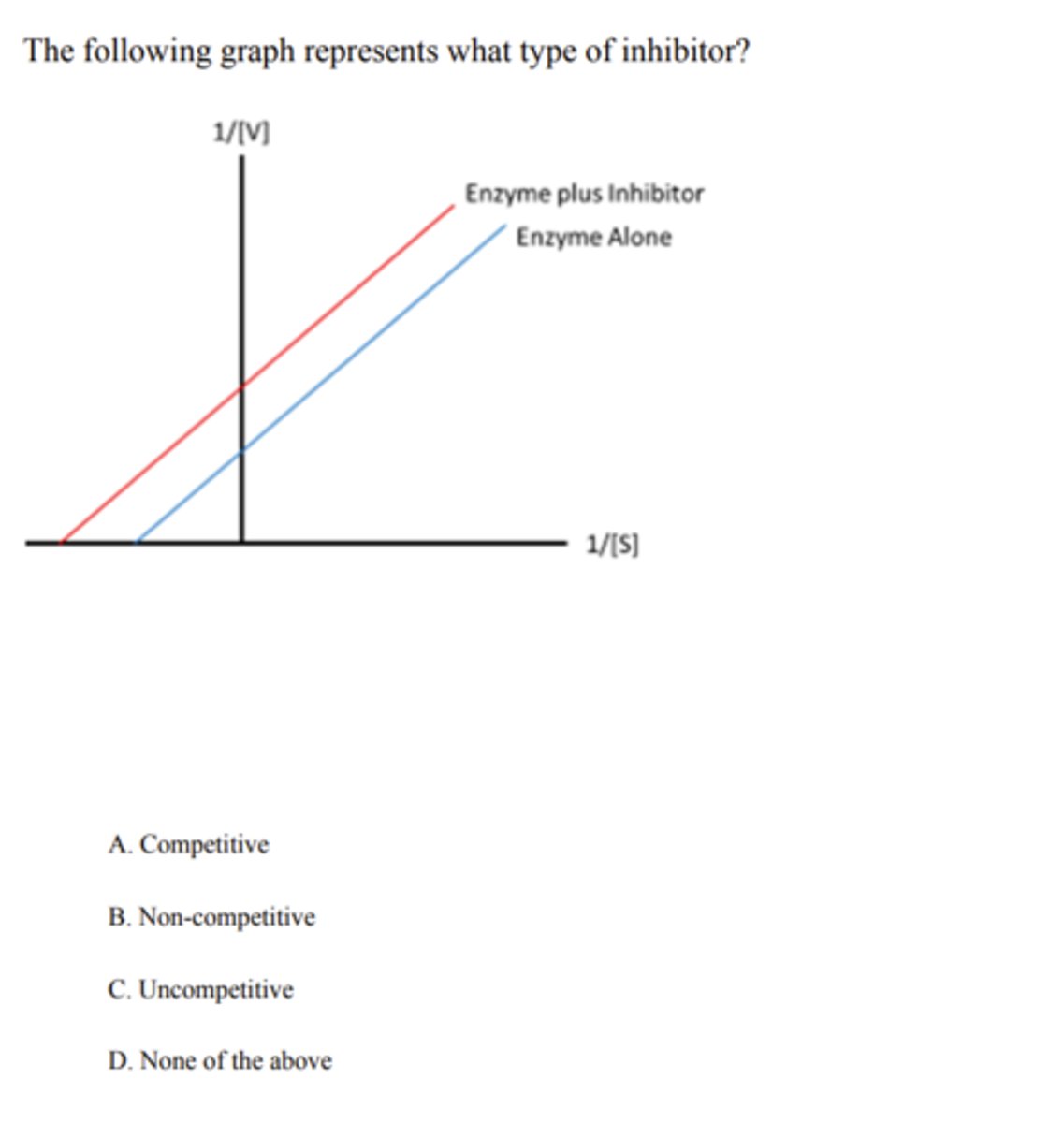
competitive
Methotrexate is a _______ inhibitor of dihydrofolate reductase
A. Mixed
B. Competitive
C. Non-competitive
D. Irreversible
competitive
Y
Which of the following Amino acids can be phosphorylated?
A. W
B. Y
C. M
D. F
The enzyme cannot be phosphorylated
You have discovered a new enzyme. When this enzyme is phosphorylated on tyrosine residue 245, the enzyme becomes active and functions to promote neuronal cell survival. You observe that in a neurological disease, the enzyme is mutated, such that the important Y residue at position 245 becomes an F. Why might this mutation lead to neuronal cell death?
A. The enzyme cannot be phosphorylated
B. The enzyme is involved in a feedback loop
C. The enzyme is involved in the cell cycle
D. The enzyme cannot be dephosphorylated
Inhibitor A (graph A)
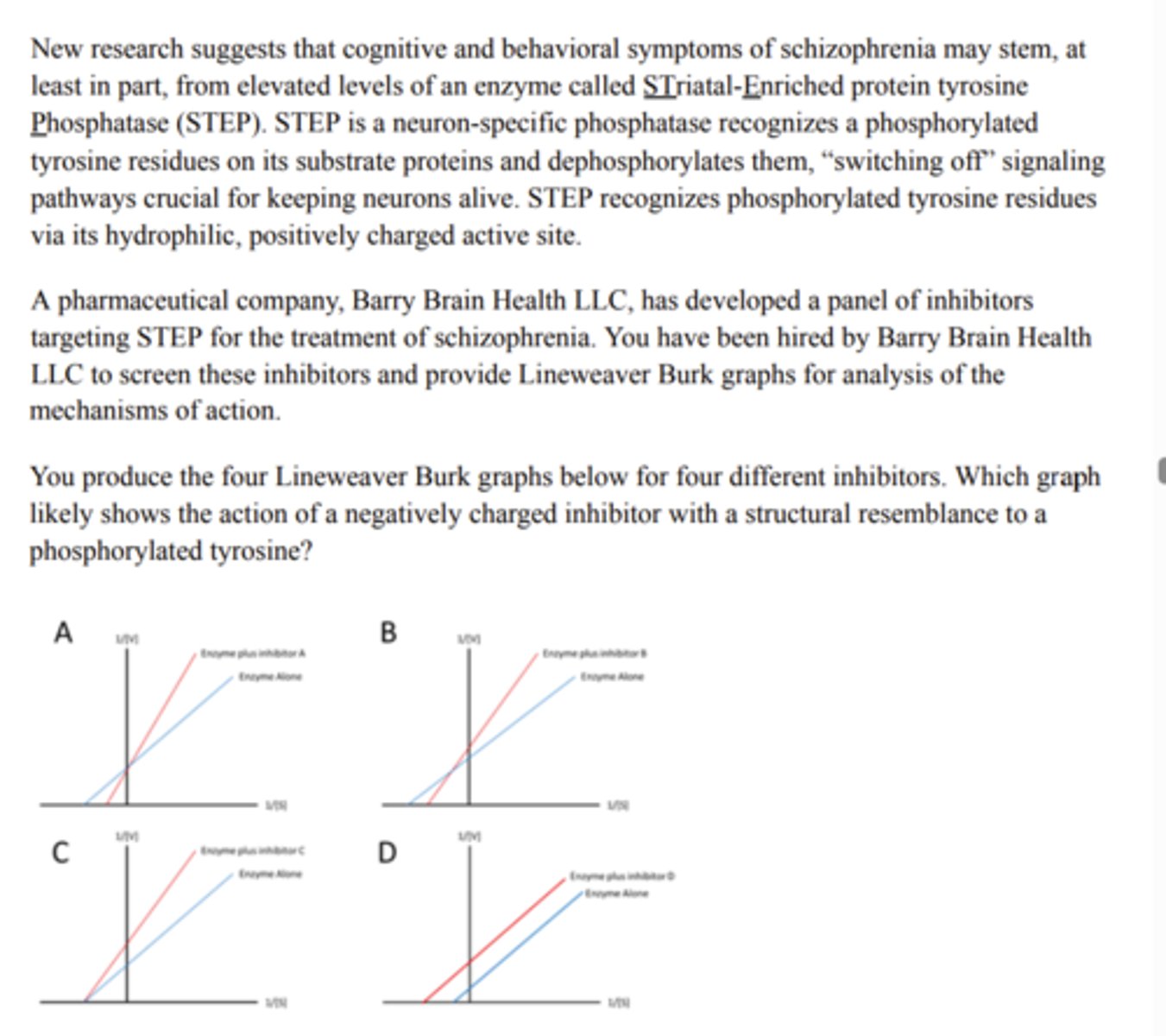
Ping Pong
The enzyme aspartate aminotransferase (AST) performs a reaction that converts aspartate to oxaloacetate AND also converts alpha-ketoglutarate to glutamate. As you are studying the mechanism of this enzyme, you notice that an inhibitor (I) blocks the production of glutamate but not oxaloacetate in healthy human cells (all substrates present). When you test this inhibitor in vitro, you subsequently observe that the inhibitor does not function without the presence of aspartate. What Enzyme reaction mechanism does the above evidence support?
A. Ping-Pong
B. Ordered Sequential
C. Random Sequential
D. You can only infer it is a bisubstrate reaction
The catalytically inactive enzyme alone
The definition of an Apoenzyme is:
A. The catalytically active enzyme-cofactor complex
B. The catalytically inactive enzyme alone
C. The catalytically inactive enzyme-substrate complex minus a cofactor
D. The catalytically inactive enzyme-cofactor complex
B12
Coabalamin
B1
Thiamine pyrophosphate
B6
Pyridoxone-5-phosphate
B7
Biotin
B5
Coenzyme A (from Pantothenic acid)
B2
Flavin adenine dinucleotide (from Riboflavin)
B3
Nicotinamide adenine dinucleotide (from nicotinamide)
B9
Tetrahydrofolate (from folate)
Barryzyme operates via an induced fit model when binding to substrate
A newly discovered Enzyme, Barryzyme, is involved in an isomerization reaction that converts glucose into galactose. Crystallographers solve two structures of the Barryzyme enzyme and describe them as follows:
(i) an apoenzyme with a shallow groove, and
(ii) using a non-isomerizable substrate, a substrate-bound form in which loops in the enzyme structure have rotated ~90° compared to the apoenzyme to create a tight pocket around the substrate.
The team also notes the enzyme is highly temperature/pH sensitive during assays.
Which of the following conclusions is MOST justified?
A. Barryzyme uses a lock and key mechanism when binding to substrate
B. Barryzyme operates via an induced fit model when binding to substrate
C. The enzymes sensitivity to changes in temperature prove the reaction is endergonic
D. Binding of substrate is facilitated through allosteric effects resulting in rotation of
loops in the enzyme
Substrate A
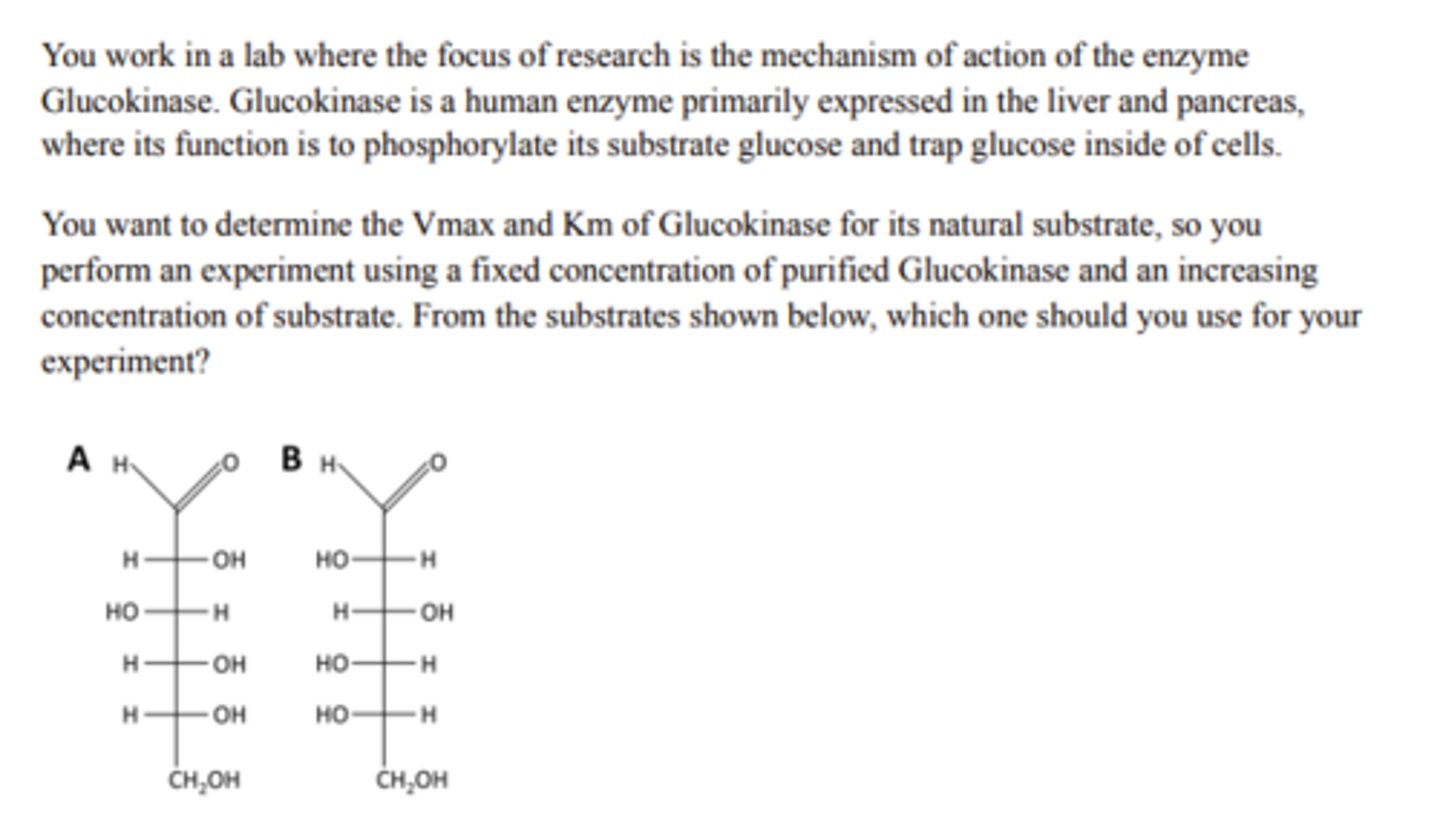
The enzyme would not perform its reaction
An enzyme contains a Glutamate (E) amino acid in it's active site. The optimal conditions for this enzyme are 37C and pH 7.4, because the reaction is entirely dependent upon the active site E residue being negatively charged to perform it's reaction. Which of the following would best describe the effect caused by placing the Enzyme into an environment with a pH of 3.5?
A. The enzyme would perform its reaction better
B. The enzyme would not perform its reaction
C. The enzyme would catalyze a different reaction
D. None of the above
The catalytically active enzyme-cofactor complex
The definition of a holoenzyme is:
A. The catalytically active enzyme-cofactor complex
B. The catalytically inactive enzyme alone
C. The catalytically active enzyme-cofactor-substrate complex
D. The catalytically inactive enzyme-substrate complex minus a cofactor
X-ray crystallography structures of enzymes showing active sites present without the substrate
What evidence exists to support the Lock and Key hypothesis of enzyme-substrate binding?
A. X-ray crystallography structures of enzymes showing active sites that change when a substrate is added
B. X-ray crystallography structures of enzymes showing active sites present without the substrate
C. X-ray crystallography structures of enzymes showing flexible regions of the enzyme
D. X-ray crystallography structure of the enzyme-substrate complex in a transition state
Positive homotrophic allosteric effector
Oxygen is an example of what type of effector of hemoglobin?
A. Positive heterotrophic allosteric effector
B. Negative heterotrophic allosteric effector
C. Positive homotrophic allosteric effector
D. Negative homotrophic allosteric effector
Enzymes make a reaction more thermodynamically favorable
Which of the following IS NOT TRUE of enzymes:
A. Enzymes reduce the amount of activation energy required for a reaction to proceed
B. Enzymes change the rate of a reaction
C. Enzymes make a reaction more thermodynamically favorable
D. Enzymes do not alter the reaction equilibrium or thermodynamic favorability
cofactor
Divalent cations e.g. Mg2+, are an example of which of the following:
A. Coenzyme
B. Cofactor
C. Enzyme
D. Substrate
Inhibitor A
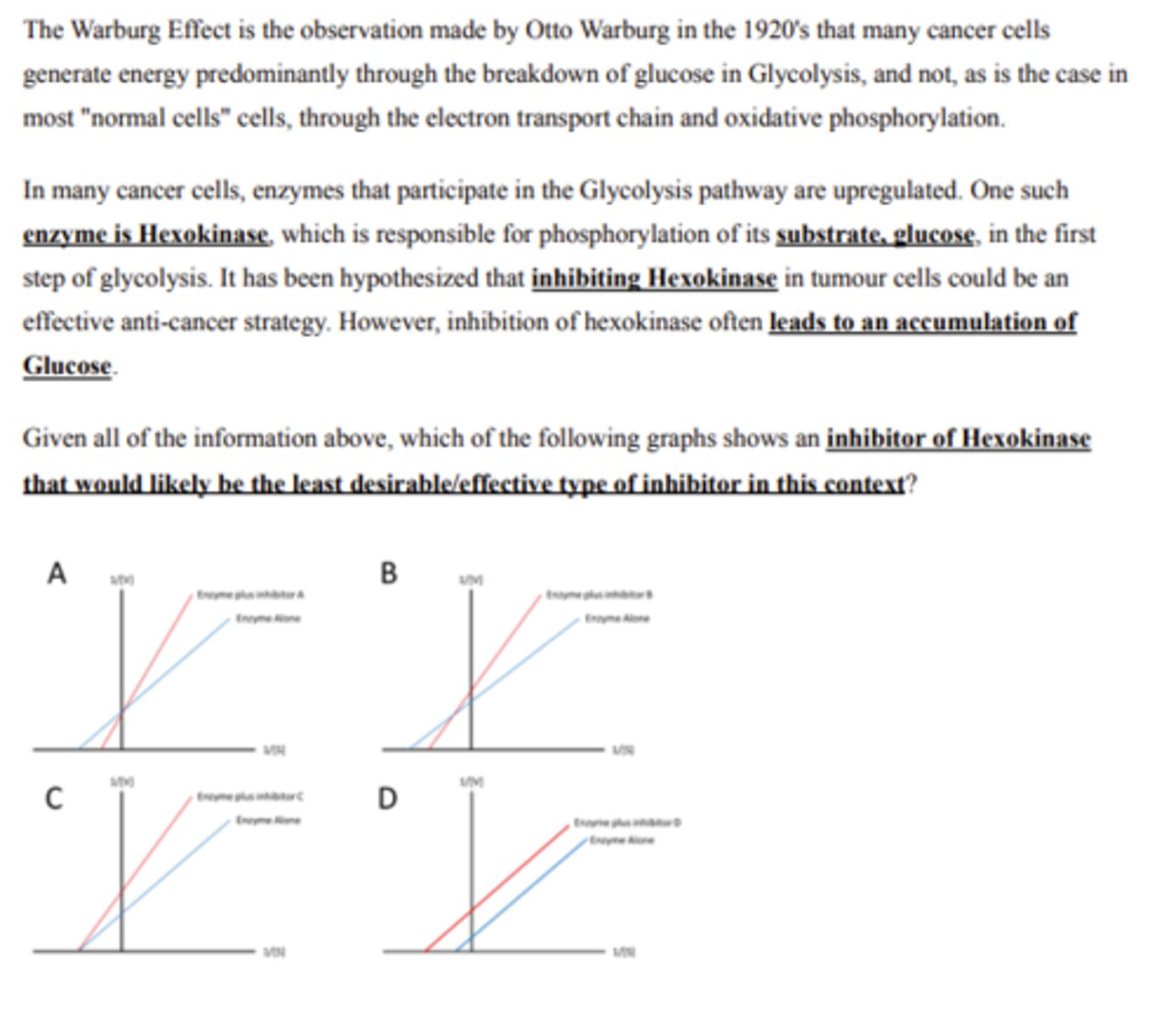
mixed
An enzyme inhibitor that can bind to both the enzyme alone or the enzyme-substrate complex is what type of inhibitor?
A. Competitive
B. Uncompetitive
C. Non-competitive
D. Mixed
uncompetitive
What type of reversible inhibitor binds only to the enzyme-substrate complex?
A. Competitive
B. Uncompetitive
C. Non-competitive
D. Mixed
irreversible
Acetylation of an amino acid function group in the active site of an enzyme is one potential mechanism of what type of inhibitor?
A. Irreversible
B. Reversible
competitive
An inhibitor that increases the apparent Km (decreases the apparent affinity) of an enzyme for its substrate but does not affect the Vmax is what type of inhibitor?
A. Uncompetitive
B. Non-competitive
C. Competitive
D. Mixed
negative allosteric heterotopic effector
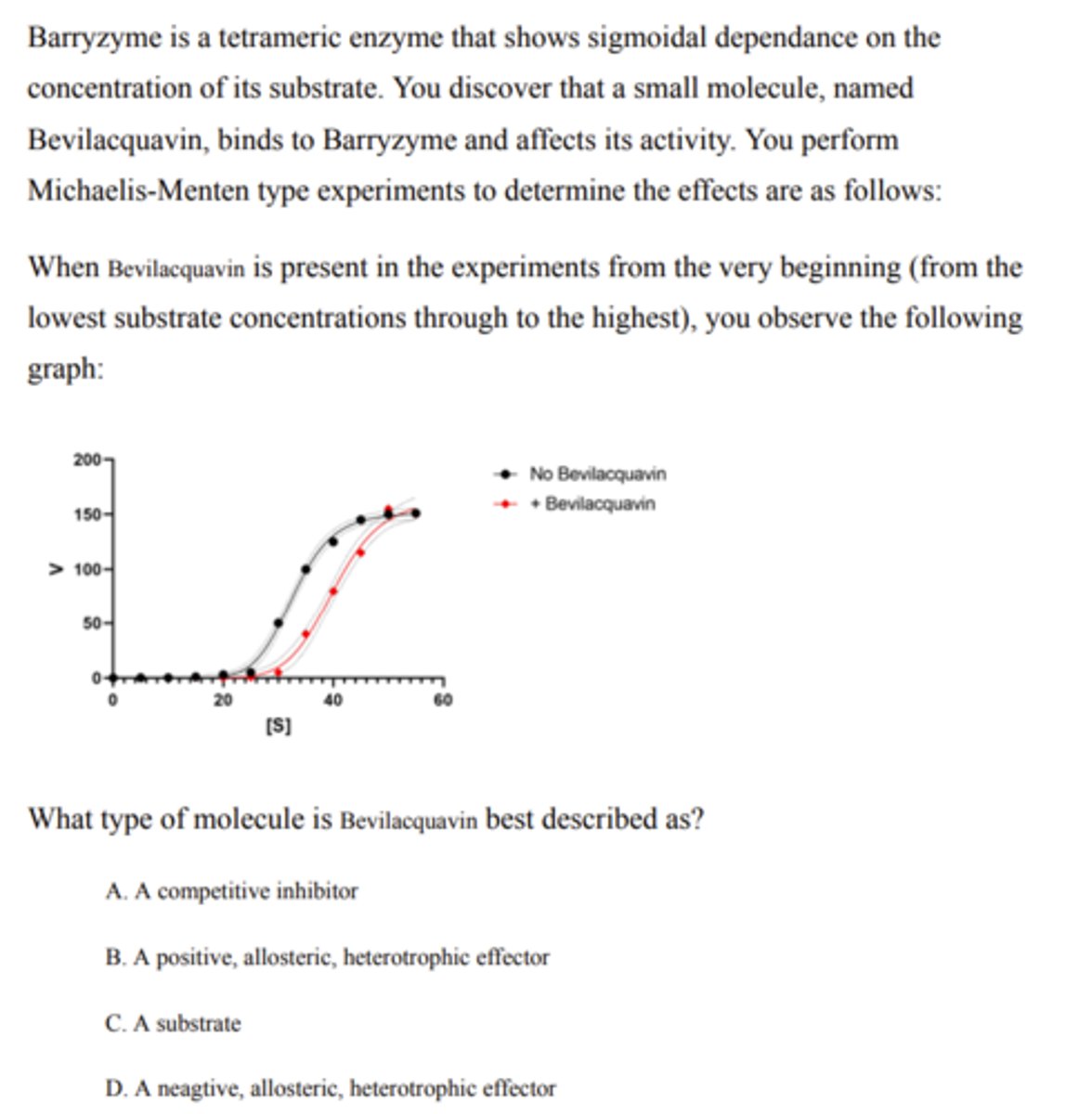
Product produced over time at the maximum rate of enzyme activity
Vmax is a measurement of what?
A. Substrate concentration at which a reaction occurs
B. Substrate concentration at which the enzyme performs optimally
C. Product produced over time at the maximum rate of enzyme activity
D. Substrate used over time at the maximum rate of enzyme activity
The Kcat of the diseased enzyme is lower than the healthy enzyme
Gaucher's disease is a genetic disorder in which a cerebroside accumulates in cells due to a defect in the enzyme lysosomal glucocerebrosidase (LGase). LGase normally resides in the cytoplasms of cells where it catalyses the breakdown of its substrate cerebroside to its products sphingosine, fatty acids and monosaccharides. However in Gaucher's disease, a missense mutation in amino acid 370 substitutes N for S (LGase N370S ) and renders the enzyme less active and unable to efficiently convert substrate to product as quickly as the healthy enzyme. The cerebroside subsequently accumulates via hydrophobic interactions and forms toxic fibrils.
Which of the following statements about the Gaucher's disease enzyme LGase N370S is true?
A. The diseased enzyme has an exposed hydrophobic amino acid leading to the formation of toxic
fibrils
B. The Kcat of the diseased enzyme is lower than the healthy enzyme
C. A Michaelis-Menten plot cannot be drawn for this enzyme
D. The Kcat of the diseased enzyme is higher than the healthy enzyme
Baby B because the lower catalytic efficiency means the enzyme will clear phenylalanine slower

uncompetitive
An enzyme inhibitor that binds exclusively to the enzyme-substrate complex is what type of inhibitor?
A. Non-competitive
B. Mixed
C. Competitive
D. Uncompetitive
Because competitive inhibitors can be outcompeted by increasing substrate concentrations
Why is Vmax unaffected by competitive inhibitors?
A. Because competitive inhibitors always irreversibly stop enzyme activity
B. Because competitive inhibitors are allosteric
C. Because competitive inhibitors don't affect Km
D. Because competitive inhibitors can be outcompeted by increasing substrate concentrations
covalent
An irreversible inhibitor relies heavily on what type of bonding to its target protein?
A. Hydrogen
B. Van der waals
C. Covalent
D. Hydrophobic
The alpha phosphate is connected to the ribose sugar through a phosphoanhydride bond
Which of the following statements IS NOT TRUE about ATP:
A. Adenine base is connected to the ribose sugar via a glycosidic bond
B. The alpha phosphate is connected to the 5' carbon of the ribose sugar
C. The gamma phosphate is connected to beta phosphate through a phosphoanhydride bond
D. The alpha phosphate is connected to the ribose sugar through a phosphoanhydride bond
All of the above
ATP is a high energy molecule because:
A. Entropy of ADP + Pi is greater than ATP
B. Phosphoryl groups compete for the lone pair of electrons of its bridging oxygen atom, this competition is absent in the hydrolysis products
C. Electrostatic repulsions between the charged groups of a phosphoanhydride destabilize ATP compared to ADP
D. All of the above
A release of energy
Catabolic processes are generally accompanied by:
A. An input of energy
B. A release of energy
C. No change in energy
D. None of the above
An input of energy
Anabolic processes require:
A. An input of energy
B. A release of energy
C. No change in energy
D. None of the above
a reaction is at equilibrium
If ΔG = 0, then:
A. a reaction is favorable
B. a reaction is unfavorable
C. a reaction is at equilibrium
D. None of the above
a reaction is favorable
If ΔG is -ve, then:
A. a reaction is favorable
B. a reaction is unfavorable
C. a reaction is at equailibrium
D. None of the above
A reaction is not favorable
If ΔG is +ve, then:
A. A reaction is favorable
B. A reaction is not favorable
C. A reaction is at equilibrium
D. None of the above
ΔG° assumes "standard" conditions (concentration, temperature, and pressure)
ΔG° differs from ΔG because:
A. ΔG° assumes "standard" conditions (concentration, temperature, pressure and pH)
B. ΔG° assumes "standard" conditions (concentration, temperature, and pressure)
C. ΔG assumes "standard" conditions (concentration, temperature, pressure and pH)
D. None of the above
considers pH
ΔG°' differs from ΔG° because ΔG°':
A. considers time
B. considers volume
C. considers pH
D. None of the above
AMP
Which of the following is not a "classic" electron carrier:
A. NAD
B. NADP
C. FMN
D. AMP
Disulfide
When oxidized, glutathione forms what type of bond with another molecule of glutathione?
A. Anhydride
B. Phosphodiester
C. Glycosidic
D. Disulfide
NADP+
The oxidized form of NADPH is:
A. NAD+
B. NADP+
C. NADH
D. NADPH
All of the above
Electron carriers are frequently involved in intracellular redox reactions. Why would the presence of high concentrations of reducing agents (agents that reduce other molecules) inside of a cell potentially be problematic?
A. Inhibit NAD+ formation
B. Inhibit NADP+ formation
C. Inhibit GSSG formation
D. All of the above
Nicotinamide adenine dinucleotide
Pellagra is a set of diseases characterized by broad, systemic symptoms including inflamed skin, diarrhea, dementia, and mouth sores. Treatment for some Pellagras can be as simple as supplementing the patient's diet with increased vitamin B3.
Given what you have learned in BMS527, a lack of which of the following coenzymes may be the cause of pellagra?
A. Flavin mononucleotide
B. Glutathione
C. Nicotinamide adenine dinucleotide
D. Glutathione
NADH
NADPH
FADH2
From the list below, select all the reduced versions of the cellular electron carriers/coenzymes
(Select all that apply)
A. NAD+
B. NADH
C. FAD+
D. NADP+
E. NADPH
F. FADH2