Biochemistry Exam 3
1/254
Earn XP
Description and Tags
Locking in!!
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
255 Terms
What are lipids?
A highly reduced carbon-rich substance, nonpolar organic solvents.
Lipids can be both
hydrophobic (nonpolar) and amphiphatic (nonpolar & polar regions)
Three common fatty acid lipids are
Triaglycerols (triglycerides)
Glycerophospholipids
Sphingolipids
A common non-fatty acid containing lipid is
Sterols
The active functions of lipids are
Energy storage
Membrane structure
Electron carriers
Emulsifying agents
Horomones
What is a fatty acid?
A caboxcylic acid with a hydrocarbon chain
Which FA has no C-C double bonds?
Saturated
Which FA has at least one C-C double bond?
Unsaturated
Which FA has ONLY one C-C double bond?
Monounsaturated
Which FA has two or more C-C double bonds?
Polyunsaturated
In standard nomenclature, what carbon is named 1? What are the names of carbon 2 and 3?
1 - Goes to the carboxyl carbon
alpha
beta
What does the nomenclature 18:1^Δ9
It is an 18-carbon fatty acid with one double bond between c-9 and c-10
FA nomenclature specifies what?
The chain length (starting @ carboxyl carbon) and # of double bonds (n)
Polyunsaturated fatty acids start their numbering at
the methyl carbon (carbon most distant from carboxyl group) aka the omega (Ω) carbon.
Because of this, double bonds are numbered relative to the Ω carbon
Polyunsaturated fatty acids with a double bond between C3 and C4 and called
omega-3 fatty acids
What essential nutrients do omega-3 fatty acids have?
a-linolenic acid (ALA), docosahecaneoic acid (DHA), and eicosapentaenoic acid (EPA)
What are essential fatty acids?
fatty acids that cannot be synthesized by the body but must be obtained from the diet
Melting points of FA are determined by
length and degree of unsaturation of the hydrocarbon chain
The longer the FA chain (at same degree of unsat.), the _____ the melting point
higher
The ____ the double bonds in a FA (assuming length is same) the higher the melting point
fewer
For saturated FA, their most stable arrangement is what?
Very closely packed - almost crystalline array
The kink found in unsaturated fatty acids is a result of
cis double bonds
Tight packing between these FA have less extensive interactions, taking less energy to disrupt them→ lower melting point
unsaturated fatty acids
Triacyglycerols (trigylcerides) contain what?
Three FA molecules esterified to three hydroxyl groups of glycerol
The primary storage form of body fat is
triacyglycerols
The abbreviation and melting point for Stearic Acid is
18:0 , 70 C
The abbreviation and melting point of Oleic acid is
18:1 , 16 C
The abbreviation and melting point of linoleic acid is
18:2 , -5 C
The abbreviation and melting point for Linolenic acid is
18:3 , -11 C
Triaglycerols are very
nonpolar
What specialized enzymes catalyze the hydrolysis of the ester in triaglycerols in order for the FA to be used as fuel?
lipases
Triacylglycerols wich in unsat. FA and are generally liquid at room temperature are called
oils
Triacylglycerols rich in saturated fatty acids and are generally semisolids or solids at room temperature are called
fats
What are the two major classifications for lipids?
Storage lipids (triacylglycerols) and Structural lipids
What do storage lipids do?
They are used primarily to store energy, they are neutral and nonpolar
What do structural lipids do?
they provide structure for membranes, they contain polar and nonpolar groups
What are the two main types of structural lipids?
Glycerophospholipids and sphingolipids
What are the primary constituents of cell membranes?
Glycerophopholipids
Glycerophospholipids differ from triacylglycerols in that
only two of the hydroxyl groups of glycerol are esterified to FA
The third hydroxyl group in glycerophospholipids contains a _____ group that is connected to a given “head group” via a phosphodiester bond
phosphate
The difference between sphingosine and glycerophospholipids is
the backbone of sphingosine is NOT glycerol
Sterols contain
four fused rings (steroid nucleus) and a hydroxyl group
What is the structural basis for all biological membranes?
Lipid bilayers
What makes lipid molecules self-sealing and flexible?
Their noncovalent interactions
The direction lipids face in bilayers are
polar head groups contact aqueous medium
nonpolar tails point toward interior
What defines the external boundaries of cells and separates cellular compartments?
Biological membranes
A biological membrane consists of
proteins embedded in/or associated with a lipid bilayer
Which membrane proteins contain hydrophobic regions embedded in the hydrophobic lipid bilayer as well as penetrate or span the bilayer
Integral membrane proteins (trans membrane proteins)
Which membrane proteins are associated with the membrane through change-change or hydrogen bonding interactions to integral proteins/membrane lipids as well as readily dissociate from membranes
Peripheral membrane proteins
What are functions of membrane proteins?
mediate movement of ions and polar molecules across a membrane
generate proton gradients for ATP production
respond to extracellular signals and communicate them to cell interior
Hydrophobic (fat soluble) molecules can pass through the membrane’s hydrophobic interior by
simple diffusion
Cell membranes are essential impermeable to
ions and large polar molecules
Ions / large polar molecules require this to form passages through hydrophobic barrier
membrane transporter proteins
Three types of integral membrane transporter proteins are
Channels
Passive transporters
active transporters
What are the transmembrane proteins with aqueous pores that extend across the membrane called?
Channels
In channels, the ion movement is
down the concentration gradient
How much energy do channels require?
None
Which is faster? Channels or membrane transporters?
Channels
Passive transporters are
transmembrane proteins
Passive transporters move the solute
down its concentration gradient
How much energy do passive transporters require?
None
Channels are usually
not saturable
Carrier proteins are
saturable
Active transport requires energy to move a solute
up the concentration gradient
Active transport can be powered by
a direct source of energy (e.g. ATP, electron transport, light)
What are the characteristics of simple diffusion?
No protein carrier
Unable to saturate with solute
DOWN the conc. gradient
No energy required
What are the characteristics of Channel facilitated diffusion?
Unable to saturate with solute
DOWN conc. gradient
No energy input required
What are the characteristics of Passive transporter facilitated diffusion?
Able to saturate with solution
DOWN the conc. gradient
No energy input required
What are the characteristics of Active transport?
Able to saturate with solute
UP the conc. gradient
Energy input required
The membrane consists of
Lipids
Proteins
Carbohydrates
Fats are digested by enzymes called
lipases
Lipases degrade what?
fats to free fatty acids and a glycerol
Where does glycerol from degraded fats travel to?
The liver
Fatty acids become bound to what? (a protein)
serum albumin
Fats → Glycerol → ( in liver cell) (Glycolysis) →
pyruvate
Fat → glycerol → (in liver cell) (gluconeogenesis )→
glucose
Fat → Fatty Acids → (Fat synthesis) →
Fats
Fat → fatty acids → (FA oxidation) → acetyl coA → (citric acid cycle) →
CO2 + H2O
Fat → Fatty acids → (membrane lipid synthesis) →
glycerophospholipids , sphingolipids
What are the three steps FA needs to complete to be degraded?
Activation of FA to a fatty acyl-CoA
Uptake into mitochondria
Degradation (beta-oxidation) in the mitochondrial matrix
Prior to oxidation, what must FA do?
must be activated and transported to the mitochondria where the necessary enzymes are located
Before free FA can pass across the barrier, the must first be converted into
fatty acyl-CoA derivativesW
When free fatty acids are converted to acyl-CoA derivatives, what is consumed?
The equivalent of 2 ATP in the conversion of ATP to AMP
True/False: Fatty acyl-CoA esters cannot cross over the inner mitochondrial membrane. (why?)
True. They must be attached to a molecule of carnitine first (to form acyl-carntine)
What are the four pathways in beta-oxidation?
Oxidation
Hydration
Oxidation
Thiolysis
For each pathway in the beta-oxidation pathway, what is released and produced?
Each pathway releases two carbon fragments (acetyl coA) and return the FA that has 2 fewer carbons than the starting one
1 FADH2 and 1 NADH are produced
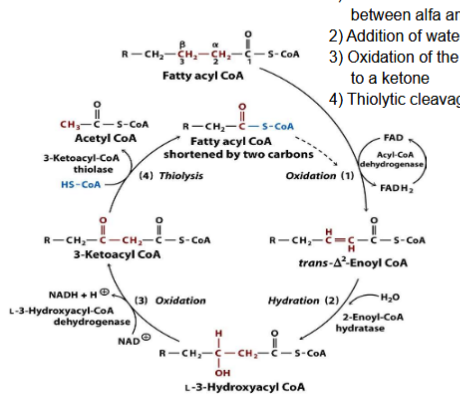
What happens during oxidation?
Generation of a double bond between alpha and beta bonds
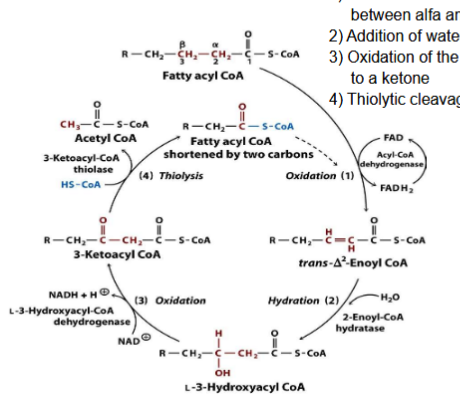
What happens during Hydration?
Addition of water to the double bond
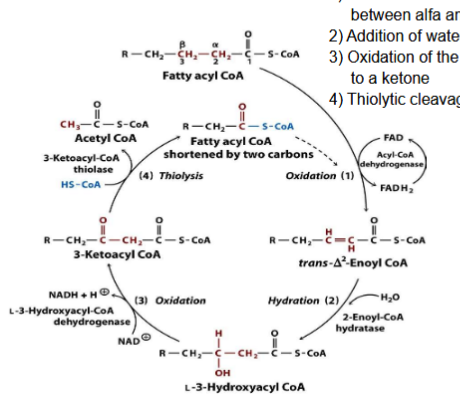
What happens during Oxidation (2)?
Oxidation of the beta-hydroxylacyl-CoA to a ketone
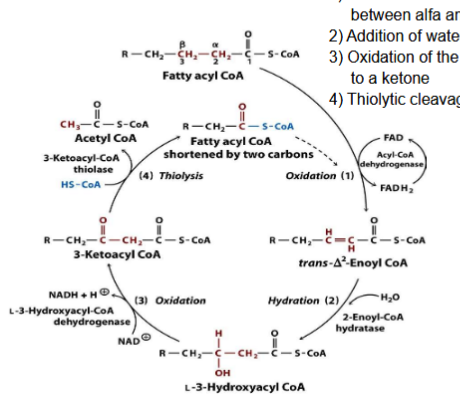
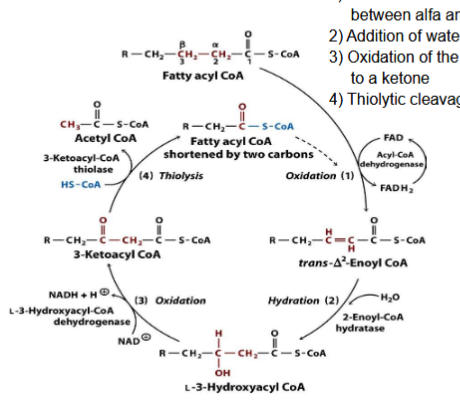
What happens during Thiolysis
Thiolytic cleavage by CoA
Each turn of the beta oxidation pathway forms what?
an Acetyl-CoA
The number of acetyl-CoA units formed is equal to
the number of carbons in the fatty acid, divided by 2
The last acetyl-CoA is formed without what?
formation of NADH and FADH2. This is because the last step forms two acetyl-CoA if the fatty acid has an even number of carbons
The number of ___ and ___ units formed is less than the number of acetyl-coA formed.
NADH , FADH2
Palmitic acid:
CH3-(CH2)14-COOH —— 16C
When palmitic acid is beta-oxidized, what is produced?
8 acetyl-CoA, +AMP + 2Pi + 7 FADH2 + 7 NADH
Oxidation of 1 glucose (6 C) to CO2 and water yields how many ATP’s?
32
Oxidation of palmitate ( 16 C) yields how many ATP’s?
106 ATP → 106 ATP /16 carbons → 6.6 ATP per carbon
True/False: Oxidation of fatty acids yields more energy than Carbohydrates?
True
Free fatty acids are transported in the blood bound to
serum albumin protein