BIOL3611 FA1
1/76
Earn XP
Description and Tags
**Includes questions from Quiz 3: On Ch.1.3 and 1.4
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
77 Terms
If a solution has a proton concentration of 10-11 M, what is the hydroxide ion concentration? Please choose the correct answer from the following choices, and then select the submit answer button.
Group of answer choices
10-11 M
3 M
0.001 M
0.01 M
0.001 M
The measure of randomness is called: Please choose the correct answer from the following choices, and then select the submit answer button.
Group of answer choices:
the hydrophobic effect
the spontaneous effect
enthalpy
entropy
entropy
Complete the following statement: Microorganisms and _____ appeared before the oxygen-containing atmosphere formed.Please choose the correct answer from the following choices, and then select the submit answer button.
Group of answer choices
There is no correct answer because microorganisms did not appear until after the oxygen-containing atmosphere formed.
plants
human beings
cells with nuclei
dinosaurs
cells with nuclei
If an acid has a pKa of 4, then which one of the following BEST indicates the pH at which the acid will be 90% protonated?Please choose the correct answer from the following choices, and then select the submit answer button.
Group of answer choices
5
10
4
3
3
What is the charge of DNA at each phosphate?Please choose the correct answer from the following choices, and then select the submit answer button.
Group of answer choices
−1
−2
1
0
-1
What might be an effect if DNA nucleotide bases in a double helix were held together by covalent bonds rather than hydrogen bonds?Please choose the correct answer from the following choices, and then select the submit answer button.
Group of answer choices
Replication of DNA, which requires separation of the two strands, would require a larger input of energy.
Purified helices would require very low temperatures to melt apart the two DNA strands.
Transcription of DNA, which requires separation of the two strands, would require a lower input of energy.
The helix would be less stable.
Replication of DNA, which requires separation of the two strands, would require a larger input of energy.
Water is a versatile solvent because of its hydrogen-bonding properties. Which of the following will be the LEAST soluble in water?Please choose the correct answer from the following choices, and then select the submit answer button.
Group of answer choices
molecules with hydrogen bond acceptors
hydrophobic compounds
polar molecules
charged compounds
hydrophobic compounds
What property of the DNA structure proposed by Watson and Crick is one of the MOST important to the role of DNA as the genetic material?Please choose the correct answer from the following choices, and then select the submit answer button.
Group of answer choices
With the sugar–phosphate backbone on the outside of the helix, the positive charges help make the DNA molecule more soluble in an aqueous environment.
Because the two strands of a DNA molecule are held together by covalent bonds, they can be easily separated and rebound when necessary for replication.
The structure is compatible with any sequence of bases.
Because of base-pairing, the sequence of bases on one strand is identical to the other strand.
The structure is compatible with any sequence of bases.
If I used the appropriate tool in an application to visualize molecular structures, I expect that I could measure and confirm that the H-bonds that stabilize the protein structure are approximately 2 Angstroms in length.
2 Angstroms is equal to _______ nm.
Group of answer choices
20
0.2
200
0.02
none of the other options are suitable because it is actually a smaller value than 0.2 nm
0.2
Considering the following sequence:
5'- C A G G T G G T -3'
Write out the opposite strand for use in the question below.
Which one of the following is true regarding this short 8-base-pair segment of sequence?
This double-stranded molecule:
Group of answer choices
has 4 adenines.
would have an increased Tm if you could replace two of the G's shown with A's (i.e. new sequence 5'-CAAATGGT-3' and you had a new appropriate opposite strand sequence that fully paired with this new one)
is stabilized by 21 H-bonds.
has 16 purines.
absorbs light at 260nm more efficiently while in double-stranded form than the corresponding single strands would.
is stabilized by 21 H-bonds.
What might be an effect if DNA nucleotide bases in a double helix were held together by covalent bonds rather than hydrogen bonds?Please choose the correct answer from the following choices.
Group of answer choices
Replication of DNA, which requires separation of the two strands, would require a larger input of energy.
Purified helices would require very low temperatures to melt apart the two DNA strands.
Transcription of DNA, which requires separation of the two strands, would require a lower input of energy.
The helix would be less stable.
Replication of DNA, which requires separation of the two strands, would require a larger input of energy.
DNA has multiple pKa's, but we looked at only one example site. Which one of the following is true according to our discussions of this site in particular and/or of DNA's pH-sensitivity in general?
Group of answer choices
DNA's 'most susceptible site for deprotonation' (i.e. the one that has the pKa that we discussed) is the 2'C of the sugar.
DNA's A, C, G, and T are referred to as nitrogenous 'bases' because they contain many -OH groups.
DNA's phosphates tend to have positive charge at high pH.
DNA is most likely to be 50% single-stranded and 50% double-stranded at pH 7.
DNA is more likely to be single-stranded than double-stranded at low pH (e.g. pH 4) due to over-protonation of what should normally be H-bond acceptors
DNA is more likely to be single-stranded than double-stranded at low pH (e.g. pH 4) due to over-protonation of what should normally be H-bond acceptors
A single stranded DNA molecule can
Group of answer choices
interact by homology only with its complement
interact by homologous sequences only with itself and fold
have partial homologous interactions with itself or a second DNA/RNA molecule if sequence allows
interact with only a second molecule of DNA if the sequence permits
have partial homologous interactions with itself or a second DNA/RNA molecule if sequence allows
Adenine, Guanine, Cytosine and Thymine
Group of answer choices
are nitrogenous bases
together with sugar and phosphate constitute the building blocks of only DNA
Cytosine and Guanine are purines
Cytosine and thymine are purines
are nitrogenous bases
The length of covalent and non-covalent bonds is important to consider because
Group of answer choices
covalent bonds such as those found in the nitrogenous bases are always constant
non-covalent bonds are critical for molecules to have a given shape
both covalent and non-covalent bonds restrict or allow for structure
Van der Walls are a covalent type of bonds critical for DNA strcuture stability
both covalent and non-covalent bonds restrict or allow for structure
Take a careful look at the relationships shown below. Where do they come from?
Group of answer choices
They are key for the equilibrium of water
They are well known
They come from Kw
The concentration of pure water in most conditions is 55.5M
They come from Kw
The Meselson and Stahl experiment (also known as pulse chase) showed that
Group of answer choices
generation zero showed a heavy N15 band at the bottom of the tube
the first generation showed no heavy N15 DNA
the first generation showed an intermediate size band representing N15-N14(light) mix
the first generation showed a size band representing only N14(light)
The first three are true
The first three are true
Which of the following is NOT true of DNA?
Group of answer choices
DNA is antiparallel.
DNA has a backbone made of alternating sugars and phosphates
DNA is a linear polymer containing deoxyribose sugars
The sugars in single stranded DNA are all oriented in the same direction which means a single strand of DNA does NOT have directionality
Deoxyribose is the sugar in DNA.
The sugars in single stranded DNA are all oriented in the same direction which means a single strand of DNA does NOT have directionality
Which of the following best describes the structural backbone of nucleic acids
Group of answer choices
… – base – sugar – base – sugar – …
… – base – phosphate – base – phosphate – …
… – base – phosphate – sugar – base – phosphate – sugar – …
… – phosphate – sugar – phosphate – sugar – …
… – phosphate – phosphate – sugar – phosphate – phosphate – sugar – …
… – phosphate – sugar – phosphate – sugar – …
How does a nucleotide differ from a deoxynucleotide?
Group of answer choices
Nucleotides are found in DNA, whereas deoxynucleotides are found in RNA
Purines are only found in nucleotides.
Nucleotides contain a deoxyribose sugars.
pyrimidines are only found in DNA
deoxynucleotides are found only in DNA
deoxynucleotides are found only in DNA
Which of the following statements is NOT true for DNA polymerases?
Group of answer choices
Requires a primer.
uses an RNA template
Direction of synthesis is 5' -> 3'.
Uses a DNA template
uses an RNA template
What are the minimal components needed for DNA replication?
Group of answer choices
DNA polymerase, DNA or RNA primers with a 3'OH
DNA polymerase, DNA or RNA primers with a 3'OH, nucleotides
magnesium ions
DNA polymerase, DNA or RNA primers with a 3'OH and deoxynucleotides
None of these
None of these
What is the pH of a solution with proton concentration 0.001M?
Group of answer choices
3
2
11
12
None of the others is correct
3
What is the log (base 10) of 100,000,000?
Group of answer choices
8
-8
10
4
None of the others is correct
8
What is the antilog (base 10) of -4?
Group of answer choices
.0001
4
10,000
0.4
None of the others is correct
10^x —> .0001
What is the antilog of 1?
Group of answer choices
10
-1
0.1
1
Undefined
10
If a functional group has a pKa of 9, then what is the deprotonated:protonated ratio for this functional group at pH 4?
Group of answer choices
1:100,000
100,000:1
1:10,000
10,000:1
None of the others is correct
1:100,000
If a functional group has a pKa of 3, then what is the (approximate) likelihood that this functional group will be protonated at pH 5?
Group of answer choices
1%
10%
50%
90%
99%
1%
(The functional group might have been the terminal carboxyl group of a protein, and the environment might have been a slightly acidic organelle like the Golgi complex.) Hopefully you also gave your answer a logical test: does it make sense that a functional group would be mostly deprotonated when the pH is two units higher than the pKa? Yes! And although the actual mathematical answer given by the Henderson-Hasselbalch equation is a 1:100 ratio of deprotonated:protonated (which isn't precisely 1%), sometimes we find it more intuitive to think in percentages rather than ratios. So in this example, we could state approximately that at pH 5, carboxyl groups are about 99% deprotonated and 1% protonated and get a pretty good mental image of their likely protonation status
At what pH is a functional group expected to be ~90% deprotonated if its pKa is 4?
Group of answer choices
5
2
3
4
6
5
Smile! You are on the right track with Henderson-Hasselbalch questions! You found the pH where there is a 10:1 ratio of deprotonated:protonated functional group (which is almost mathematically the same as saying that there is 90% deprotonated and 10% protonated). Make sure to do a logical check – does it make sense to say that deprotonation is favored when the pH is higher than the pKa? Yes! This is always a good logical test of your answer to do on exams….
Which amino acid has the molecular formula C3H7NO3?
Group of answer choices
Ser
Ala
Gln
Lys
Met
Ser
Of course we never memorize amino acid formulas! We are trying to make deductions using the following steps: (1) What atoms does every amino acid have, factoring in the central carbon, the alpha-amino group, and the alpha-carboxyl group? (2) Ignoring the hydrogens, what's left over, and what deductions does that allow us to make??
Which amino acid has an expected net charge of +1 at pH 7?
Group of answer choices
Lys
Asn
Glu
Trp
all four of the listed amino acids
Lys
Great! For side chain pKa’s, only consider those listed in Table 2.1 (remember, we are looking only at those truly classified as acidic/basic, and skipped Cys and Tyr), and group them generally as either being acidic (pKa ~3-4), highly basic (pKa ~11-12), or moderately basic (Histidine, with pKa of 6). For one that has positive charge at pH 7, we want one that's nitrogen-containing and has a high pKa. Also take note that your textbook, unless otherwise noted, aims to show molecules at their most likely charge at pH 7.
What is the most likely net charge of the dipeptide Asp-Glu at pH 10?
Group of answer choices
None of the others is correct
+1
0
-1
-2
None of the others is correct
First, remember that deprotonation is favored when the pH is above the pKa. Next think about the functional groups that any dipeptide has: one amino group (pKa ~8-9), and one carboxyl group (pKa ~3-4). Then note that both side chains contain carboxyl groups as well (each has pKa ~3-4). pH 10 is higher than every pKa under consideration, meaning that deprotonation is favored for all. This means that all three carboxyl groups will have -1 charges and the amino group will have no charge, for a net charge of -3. (-1-1-1 = -3)
What is the net charge of the tripeptide Ser-Arg-Gln at pH 2?
Group of answer choices
+2
+1
-1
0
None of the others is correct
+2
Strong finish! It looks like you’ve accounted for all of the functional groups, and noted that the alpha-carboxyl group will be protonated at such a low pH, and that the alpha-amino group and the Arg side chain will be protonated as well. The charges come only from the alpha-amino group (+1) and the Arg (+1).
The covalent link between a DNA base and the rest of the nucleotide always directly involves one of the base:
carbon atoms.
nitrogen atoms.
hydrogen atoms.
phosphorus atoms.
nitrogen atoms —> The covalent carbon–nitrogen bonds define the structures of the bases themselves.
How many hydrogen bond donors and acceptors are on cytosine that are participating in hydrogen bonds between guanine and cytosine?
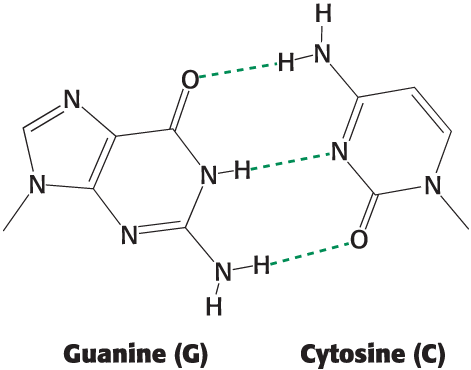
one donor and two acceptors
two donors and one acceptor
two acceptors and two donors
one donor and one acceptor
one donor and two acceptors
What property of the DNA structure proposed by Watson and Crick is one of the MOST important to the role of DNA as the genetic material?
With the sugar–phosphate backbone on the outside of the helix, the positive charges help make the DNA molecule more soluble in an aqueous environment.
Because of base-pairing, the sequence of bases on one strand is identical to the other strand.
Because the two strands of a DNA molecule are held together by covalent bonds, they can be easily separated and rebound when necessary for replication.
The structure is compatible with any sequence of bases
The structure is compatible with any sequence of bases
Estimate the length of a carbon–carbon triple bond.
1.54 × 10−10 m
Estimate the length of a carbon–carbon triple bond.
1.54 × 10−10 m
1.2 × 10−10 m
1.35 × 10−8 m
1.34 Angstroms
1.8 Angstroms
1.35 × 10−8 m
1.34 Angstroms
1.8 Angstroms
1.2 × 10−10 m —> A typical single carbon–carbon covalent bond has a bond length of 1.54 Å, where 1 Å = 10−10 m. A double bond is 1.34 Å; a triple bond will be even shorter and stronger.
Why does a DNA strand have directionality?
The order of bases along a DNA strand consists of a repetition of the sequence GATC.
Each sugar is oriented in the same way in the sugar–phosphate backbone.
The phosphate groups attach only to one side of the base.
The bases all protrude from the sugar–phosphate backbone
Each sugar is oriented in the same way in the sugar–phosphate backbone.
pKa
The susceptibility of a proton to removal by reaction with a base
When pH=pKa, the deprotonated and protonated concentrations are the same as the deprotonation process is halfway
pH > pKa
Deprotonation occurs when the pH of the solution is greater than the pKa of the molecule (acids with lower pKa values lose their protons more easily) —> the environment is basic relative to the compound so the compound would lose their proton
pH < pKa
Protonation occurs when the pH of the solution is lower than the pKa of the molecule, as the environment is acidic relative to the compound so the environment donates a proton to the compound
Biochemical similarities in key processes from bacteria to humans suggest:
that bacteria are very different to mammals
that there is a common ancestor to life on earth
the common ancestor is likely to have appeared after microorganisms were detected on earth
biochemical similarities do not mean anything
that there is a common ancestor to life on earth
Avery's experiment demonstrated that DNA was the carrier of the genetic information because:
Isolated DNA from one bacterium will transmit a trait to a bacterium that did not have it
Nucleases, DNA hydrolyzing enzymes, kill the transforming ability
Proteases, protein hydrolyzing enzymes, do NOT affect transforming ability
All answers are correct
All answers are correct
Adenine, Guanine, Cytosine and Thymine
are bases found on DNA and RNA
together with deoxysugars and phosphate constitute the building blocks of nucleic acids
Cytosine and Guanine are purines
Cytosine and thymine are purines
all of them are incorrect
all of them are incorrect
The nitrogenous bases
are covalently attached to sugar-phosphate in DNA
the sugar-phosphate are on the inside of the double helix
are the DNA backbone
base pair to each other on the inside of the DNA helix by covalent bonds
are covalently attached to sugar-phosphate in DNA
Water in biochemistry is important because
contributes to the specificity of base pairing
it can make hydrogen bonds to produce highly ordered structures
it is the solvent of life
all answers are correct
all answers are correct
A single stranded DNA molecule can
interact by homology only with its complement
interact with homologous sequences only with itself and fold
have partial homologous interactions with itself or a second DNA/RNA molecule if sequence allows
interact with only a second molecule of DNA if the sequence permits
have partial homologous interactions with itself or a second DNA/RNA molecule if sequence allows
Take a look at the DNA image below.
In the figure the 'W end' label represents 5'.
If each end label (W, X, Y, Z), was replaced with a 5' or a 3' label, then Z would represent a _____ label. Inspection of the nitrogenous base closest to the W label also shows that it is a _____.
3'. Purine
3'. Pyrimidine
5'. Purine
5'. Pyrimidine
5'. Purine
Chargaff's experiments aided in the discovery of the DNA structure because:
Only guanines and adenines have a 1:1 ratio in all organisms
Only adenines and thymines have a 1:1 ratio in all organisms
Guanines and thymines are always in the same proportion
all answers are incorrect
all answers are incorrect
The fact that there is biochemical unity is demonstrated by
Protein diversity and uniqueness among living cells
Protein diversity but evolutionary conservation among living organisms
DNA and protein diversity
Only protein conservation among living organisms
Protein diversity but evolutionary conservation among living organisms
Which of the following domains includes all multicellular organisms?
bacteria
archaea
eukaryotes
prokaryotes
eukaryotes
What is the defining characteristic of eukaryotes?
Lack of a nucleus
Presence of a nucleus
Unicellular nature
Biochemical similarity to Bacteria
Presence of a nucleus
Carl Woese’s discovery in 1977 led to the recognition of which domain?
Bacteria
Eukarya
Archaea
Prokaryotes
Archaea
Prokaryotes belong to which domains?
Eukarya and Bacteria
Bacteria and Archaea
Archaea and Eukarya
Eukarya and Metabolites
Bacteria and Archaea
Why are certain species' biochemical pathways unique?
They have no common ancestor.
They evolved in specific biological niches.
They lack metabolic adaptation.
They belong to the same domain.
They evolved in specific biological niches.
Biochemical pathways help scientists understand:
Evolutionary history
Physical appearances
Unrelated cellular functions
Genetic mutations
Evolutionary history
What kind of organisms are included in the domain Eukarya?
Only microscopic unicellular organisms
Both multicellular and unicellular organisms
Only plants and animals
Exclusively multicellular organisms
Both multicellular and unicellular organisms
Which statement is true about Archaea?
They include all animals and plants.
They are more biochemically like Eukarya than to Bacteria.
They have a well-defined nucleus.
They were the first domain of life
They are more biochemically like Eukarya than to Bacteria.
Blood contains a total concentration of phosphate of approximately 1 mM and typically has a pH of 7.4. Given that the pKa of is 7.21, what is the ratio of [HPO-24] and [H2PO-4] in blood?
Tip: is there an equation that relating pH to pKa?
0.55
1.55
1
impossible to calculate
1.55
Think about why it is important for blood to maintain a stable pH. Choose the incorrect answer form below:
blood contains charged molecules
Blood contains charged molecules that will have altered function at differetn pHs
nothing in blood has the probablity to change its state or charge since it is sterile
Blood is sterile though this is not key for answering this question
nothing in blood has the probablity to change its state or charge since it is sterile
Multiple answers. What does a zwitterionic form of an amino acid mean? Multiple answers
The ionization state is altered by changes in pH
aminoacids in solution (in a test tube) exist at neutral pH as dipolar ions
In the dipolar form the amino group is protonated and the carboxyl form is deprotonated
the dipolar form is present at alkaline pHs
A, B, and C
Multiple answers. What are hydrophobic aminoacids?
these are a group of Aminoacids with nonpolar R groups
these are a group of aminoacids with polar R group
these are a group of aminoacids with a single hydrogen
a group of aminoacids that are averse to water
A and D
Mutliple answers. Glycine, Alanine, Proline, Valine, Leucine, Isoleucine, Methionine, Tryptophan, and Phenyalanine are
aminoacids that have non-polar R groups
aminoacids that have hydrophilic R groups
aminoacids with a a hydrophobic R group
building blocks of proteins
A, C, and D
Multiple anaswers. Which of the following is true about the polar aminoacids Serine, Threonine, and Tyrosine?
The hydroxyl group makes them hydrophilic
The hydroxyl group makes them more reactive than hydrophobic aminoacids
They are not building blocks of proteins
They are uncharged
A, B, and D
Multiple answers. Which of these statements are true about Lysine and Arginine?
They have R groups made of repetitive CH2
The Arginine R group has more NH2s than Lysine
They are highly hydrophobic
They are highly hydrophylic
A, B, and D
These aminoacids have short R groups compared to methionine. Choose the best answer
Glycine
Alanine
Lysine
Valine
Glycine
In which one of the following conditions would the dipeptide Pro-Lys be most likely to have a net charge of +2?
pH 13
pH 10
pH 6
pH 1
pH 4
pH 1
I was reading recently about a new amino acid that has a pKa of 5
This amino acid will have an overall ______ net charge at pH 7 and would be attracted to a_______________ charged pole
negative; negative
negative; positive
positive; negative
positive; negative
negative; positive
As you have no doubt encountered, internet images/sources can contain errors! Here is one that I just came across:
Of course we are not memorizing chemical formulas! However we can inspect this one, and compare it to our overall amino acid knowledge in order to determine the impact of the error.
Which one of the following best describes a problem with this chemical formula for glutamine, based on what we were supposed to remember about this amino acid (and amino acids in general)?
Knowing what we know about amino acids in general and glutamine in particular, the error in this chemical formula ________.
either impacts the side chain or the alpha-carboxyl group (but there is no way to narrow it down further).
either impacts the side chain or the alpha-amino group (but there is no way to narrow it down further).
must definitely impact the alpha-amino group (none of the other answer options are suitable).
must definitely impact the alpha-carboxyl group (none of the other answer options are suitable).
must definitely impact the side chain (none of the other answer options are suitable).
either impacts the side chain or the alpha-carboxyl group (but there is no way to narrow it down further).
Which one of the following molecules (not side chains) has a most likely net charge of +1 at the indicated pH, according to our coverage?
Glu at pH 12
Trp at pH 2
Tyr at pH 10
Val at pH 6
His at pH 5
Trp at pH 2
Anfinsen experiment demonstrated that protein......... is encoded in a protein's ...............................structure and was dependent on the ................................of Ribonuclease A
structure/secondary/folding
folding/secondary/performance
folding/primary/activity
structure/tertiary/activity
folding/primary/activity
The role of betamercaptoethanol and urea in Anfinsen's experiment is respectively
denaturation of all types of bonds/no role
denaturation of non-covalent bonds/denaturation of covalent bonds
dentaturation of covalent bonds/denaturation of non-covalent bonds
denaturation of hydrogen bonds/denaturation of disulfide bonds
dentaturation of covalent bonds/denaturation of non-covalent bonds
This image is from your book, and we discussed it in class. This image represents
two proteins encoded by two genes
two activities encoded by the same gene
different activities based on the same structure
two activities with the same secondary structures but different tertiary structure
two activities encoded by the same gene
The graph above depicts the interaction of a transcription factor EppR and its cognate binding site on the DNA. The dissociation constant is shown. Choose the incorrect answer
The transcription factor extracellular concentration is independent of the KD
The transcription factor intracellular concentration is represented by the KD
The transcription factor intracellular concentration is at least the value of the KD
The transcription factor intracellular concentration is lower than the KD
The transcription factor intracellular concentration is lower than the KD
A protein X was shown to interact with itself, and with additional different proteins, all of them with a different tertiary structure while the KD measured with the different interacting proteins were in similar ranges. Choose below the best explanation for the data.
Protein X is encoded by a gene able to form different structures and thus to have multiple interacting partners
Protein X is likely an IDP or has ID regions
There is an error in the measurements and
None of the answer is true
Protein X is likely an IDP or has ID regions
Intrisically disordered proteins or proteins with intrinsically disordered regions
All proteins in the proteome contain these regions
adopt a given structure upon interaction with a partner
adopt always an alpha helix structure upon interaction with a partner
adopt always a beta sheet structure upon interaction with a partner
adopt a given structure upon interaction with a partner
The table represents probability of a given aminoacid to be part of an alpha helix, beta sheet or a turn. Choose the incorrect answer
Leu and Ala have similar probabilities to be in alpha helices
Glu is equally likely to be in an alpha helix or in a turn
Alanine is equally likely to be in an alpha helix or a beta sheet
met can be in alpha helices or beta sheets
Alanine is equally likely to be in an alpha helix or a beta sheet