4.4 Protein Denaturing + Folding
1/23
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
24 Terms
native conformation
balance between enthalpic + entropic forces, driven by hydrophobic effect
denatured protein
when the tertiary + seconday strucutre is disrupted
ways to denature protein
-heat
-denaturants
-organic solvents
-detergents
-acid/bases
-mechanical forces
-disruption at air + water interface
chaotropes
denaturant, small molecules that work by disrupting h-bonds + interact with the protein surface
ex: urea, guanidium chloride
why was levinthal’s paradox, the theory that proteins fold thru random sequential search for correct structure proven wrong
proetin folding happens way to fast
what is protein folding considered 2-state only
it happens very abruptly bc partially unfolded proteins are very unstable, when one part of the structure is dirupted it destabilizes the remaining structure
cooperative folding
many parts of a protein fold simulatenously in an interconnected way rather than each region independently
two theories of protein folding
1: structural nucleation
2: thermodynamic folding funnel
1:strucutral nucleation
local structures begin to form, based on local prefrences
2:hydrophobic collapse
molten globule state forms, protein rapdily shrinks to dense blob
3: stabilization of secondary structure
final functional form of protein, alpha helicies + beta sheets are precise, internal side chains pack together + water is expelled from portein core
Hierarchical protein folding
1:structural nucleation
2:hydrophobic collapse
3:seconday strucutre stabilization
first step of 2-D folding funnel
burst phase, rapid hydrophobic collapse to molten globule
what conclusions can be drawn from Christain Anfinsens expirements
-protein can be denatured + fold again
-native state is the most thermodynamically stable
urea- chaotrope
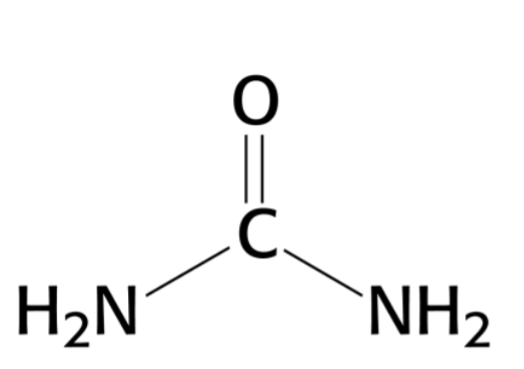
guandium chloride
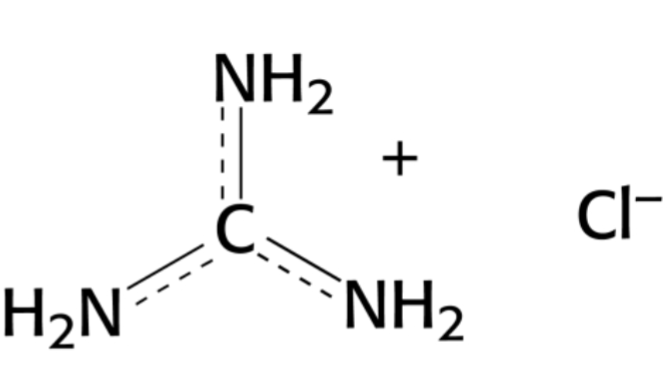
2-D folding funnel steps
1: burst phase
2: rearrangements to maximize enthalpic benefit + find native strucutre (noncovalent interactions)
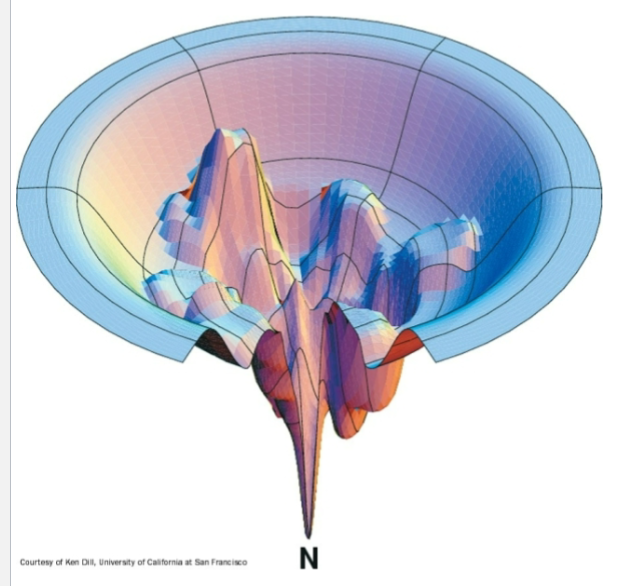
Thermodynamic folding funnel
-depth = ΔG
-width =avalible conformation space for the protein chain
wells= semi-stable folding intermediates
context dependent structure leads us to believe that…
misfolding is possible, as several different conformations are possible in different contexts
amyloids
long highly ordered fibrils, beta sheets gros perpendicular to fibrils
protein misfolding diseases
-alzheimers disease
-huntington’s disease
-cystic fibrosis
-cataracts
-prion disease
molecular chaperones
-inhibit inappropiate interactions between potentially complementary surfaces + disrupt unsuitable liasions
When an organism like E. coli is grown at increased temperature, the expression of chaperone proteins increases.
From a Biochem 5613 perspective, briefly, why
an increase in energy (for example, increased temperature) will lead
to an increase in dynamic motion, and potentially protein unfolding. Chaperone proteins counteract this by
preventing aggregation, and assisting proteins in attaining the native fold. This helps organisms survive
increased temperatures
molten globule
nonpolar side chains are
buried, and not exposed to solvent – but the protein is not taking on a well-formed structure, and there is
significant dynamic motion in the protein interior, rather than the smaller motions (“breathing”) of a typical
protein. Often in the molten globule state, there is still water dispersed through the protein core, and the buried
salt bridges or hydrogen bonding interactions that define specificity in structure are not yet formed