CHEM 261 Unit 7 - Reactions (Questions)
1/18
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
19 Terms
As the size of the halogen increases in an alkyl halide, bond strength ___ and bond length ___.
Decreases, increases
Where is the electrophilic center in an alkyl halide? Why?
Carbon attached to the halogen, halogen is electronegative and pulls the electrons of the carbon– halogen bond in its direction making the carbon electropositive
Which reagant/intermediate concentrations will have an effect on the rate of reaction?
Ones that are involved in the rate determining step
In an SN2 reaction, which molecules are involved in the rate determining step? Why?
The nucleophile and the electrophile. Collide to react and thus the concentrations of the nucleophile and electrophile determine how much will react
What is the rate reaction of an SN2 reaction?
k[Nu-] [electrophile]
Explain the reaction mechanism for SN2 reactions (step mechanism, stereochemistry, reversibility).
Concerted mechanism (one step), configuration at the stereogenic center of the substrate is inverted in the reaction, irreversible
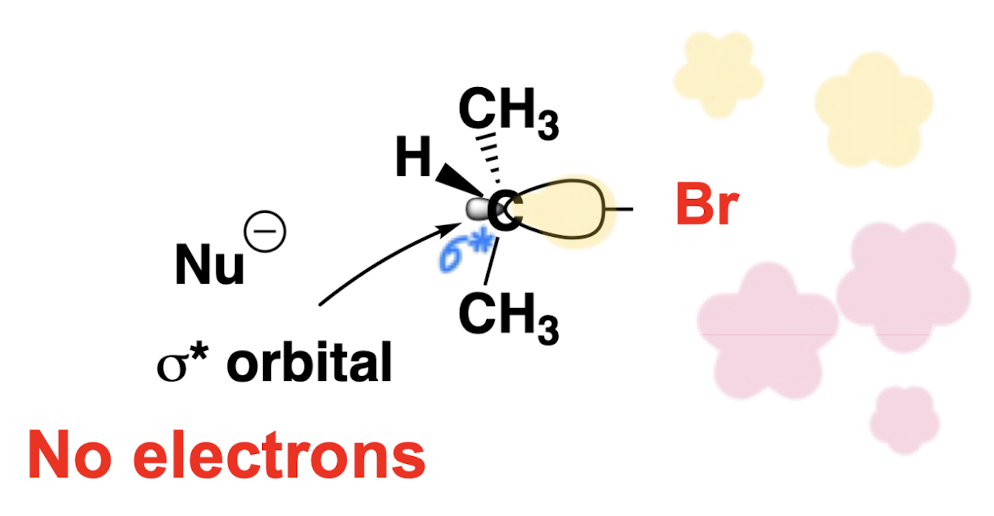
Explain inversion of the substrate atom in an SN2 reaction mechanism.
The nucleophile approaches the carbon bearing the leaving group from the back side (directly opposite the leaving group), attacking the sigma star orbital of the carbon bearing the leaving group. The stereochemistry of the substrate is thus inverted
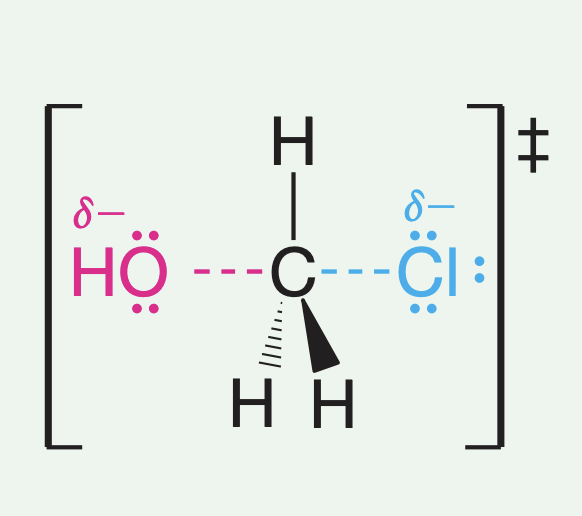
Does the SN2 reaction have transition states or intermediates? Why? What is the transition state or intermediate?
Concerted makes transition states, partial C-O and C-X bonds that arrange in a planar fashion as well as the carbon undergoing a hybridization change from sp3 to sp2. Inversion begins to happen
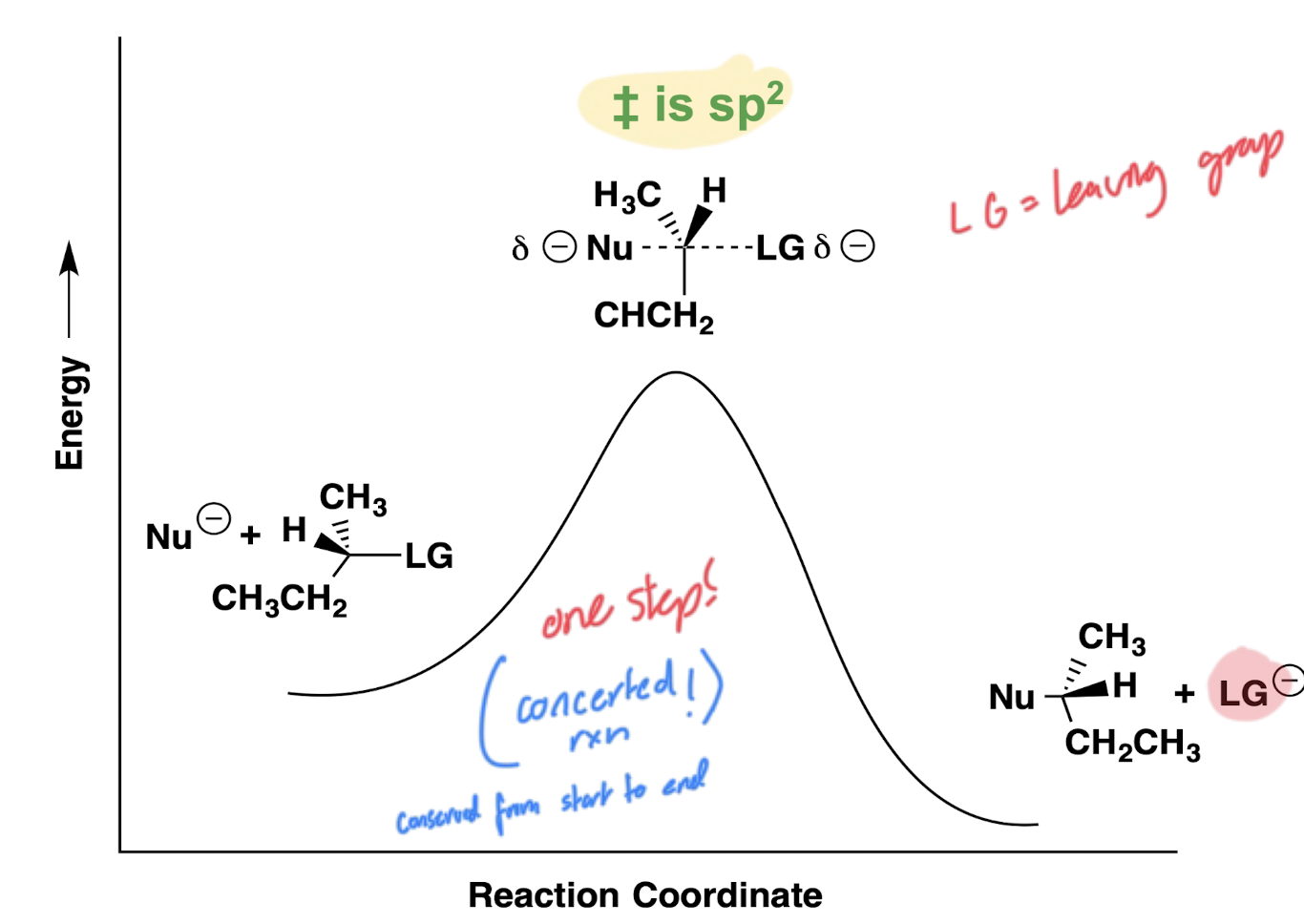
Describe how an energy graph of an SN2 reaction would look.
One peak due to the concerted mechanism
The larger the activation energy, the ___ the reaction rate.
Slower
Explain how to improve the rate of a reaction by altering the energy of the reactants.
Using better (more reactive) nucleophiles and electrophiles gives them higher energy, which will lower the energy needed to reach the transition state and cause a faster reaction
Explain how to improve the rate of a reaction by altering the energy of the transition state.
Choosing a good (reactive) solvent lowers the energy of the transition state, meaning less energy is required and the reaction goes faster
Explain how the substrate/electrophile affects the rate of an SN2 reaction.
The more substituents on the alkyl halide, the slower the rate of reaction as the nucleophile has more difficulty locating the correct carbon atom (with the halogen), which raises the energy of the transition state (Ea)
Compare the reaction rates of allylic and benzylic halides to those of methyl halides.
React faster than methyl halides because resonance delocalization into the double bonds of allylic and benzylic halides delocalize the positive charge that develops in the transition state
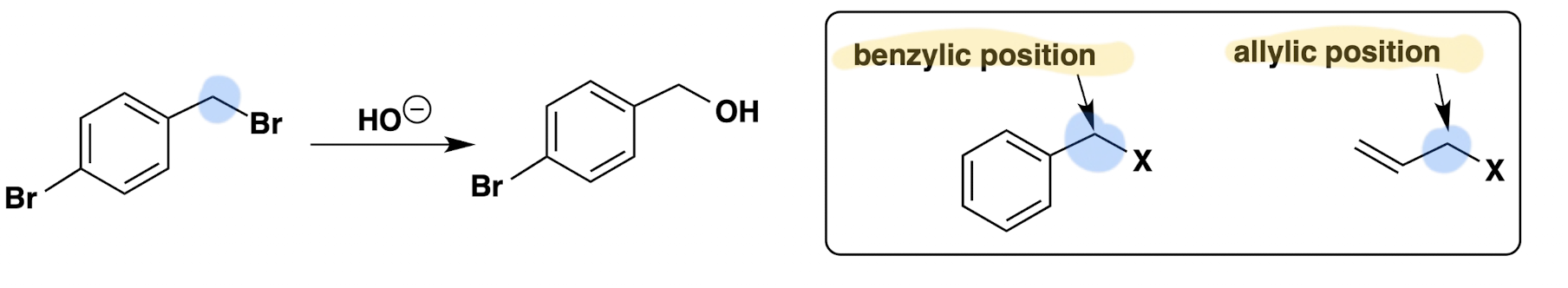
Can SN2 reactions happen at sp2 or sp carbons?
No, it needs to be able to attack from the backside
What are the general trends of nucleophilicity? How does the nucleophile affect the rate of an SN2 reaction
Stronger Lewis base is a better nucleophile, - charge is better than neutral
Increases down a group
Increases as electronegativity decreases
Reversibility of the reaction (forms an ion)
Good nucleophiles have higher energy, which reach the transition state faster and thus increase the rate or reaction
What are the general trends/rules of leaving group effectiveness? How does the leaving group affect the rate of an SN2 reaction?
Good leaving groups need to be able to stabilize the negative charge from the bond broken during the reaction
Weak bases are the best leaving groups (from strong conjugate acids)
The larger an atom, the higher its polarizability, the better it is able to stabilize a negative charge
Acid activated hydroxyl groups generate a very good leaving group C-OH2+
Good leaving groups lower the energy of the transition state, increasing the rate by dispersing the negative charge of the transition state
Substitutions must be carried out in ___ solvents. Why?
Polar, nucleophiles are typically charged species that cannot be dissolved in nonpolar solvents
How does the solvent affect the rate of an SN2 reaction?
Polar protic solvents stabilize the charge on the nucleophile via the formation of solvent cages through hydrogen bonding, lowering its energy and giving less energy to reach the transition state, decreasing the rate. Polar aprotic solvents do not significantly stabilize the nucleophile, meaning reactions go faster in these solvents