chem 7
1/64
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
65 Terms
what is a hydrocarbon
organic compound containing hydrogen and carbon only
what is the homologous series
a group of organic compounds that react in a similar way
what is a alkane n general formula
C-C single bond
CnH2n+2
they r saturated compounds - each carbon atom forms 3 single covalent bonds
part of homologous series
first 4 alkanes
methane, ethane, propane, butane
what properties change as the hydrocarbon chain gets longer
the shorter the chain, the less viscous it is
the shorter the more volatile the hydrocarbon is - eg lower BP
the shorter the carbon chain, the more flammable it is
when does complete combustion occur
when theres plenty of oxygen
formula for complete combustion
hydrocarbon + oxygen →carbon dioxide + water
why are hydrocarbons used at fuels
due to the amount of energy released when they combust completely
what is crude oil and hows it formed
a fossil fuel, formed from the remains of plants and animals, mainly plankton that died millions of years ago and were buried in mud
over millions of year, w high temperature and pressure, the remains turn to crude oil
It is a mixture of lots of different hydrocarbons, most of which r alkanes
how does fractional distillation work
oil is heated until most turned into a gas, the gases enter a fractional column (the liquid bit is drained off)
theres a temperature gradient in the column (hot at bottom and gets cooler as u go up)
LONGER hydrocarbons have high BP and they condense into liquids and drain out of the column early on at the bottom. The SHORTER ones have lower BP and condense and drain out later on, near top of column
u end up w the crude oil mixture separated out into different fractions, each fractions contains a mixture of hydrocarbons that contain a similar number a carbon atoms n similar BP
crude oil uses in modern life
fuel for most modern transport - diesel oil, kerosene, heavy fuel oil and LPG (liquid petroleum gas)
the petrochemical industry uses some of the hydrocarbons from crude oil as a feedstock to make new compounds for things like polymers, solvents, lubricants and detergents
what r organic compound
compounds containing carbon atoms
what is cracking and why good
splitting up long chain hydrocarbons
short chain hydrocarbons r flammable so make good fuels and are in high demand and long chain ones form thick gloopy liquids which r not as useful
cracking also produces another type of hydrocarbon, alkenes - they r used as a starting material when making lots of other compounds and to make polymers
what type of reaction is cracking
thermal decomposition reaction - breaking molecules down by heating them
heating long chain hydrocarbons to vaporise them
what is catalytic cracking
the vapour is passed over a hot powdered aluminium oxide catalyst
the long-chain molecules split apart on the surface of the specks of catalyst
what is steam cracking
mix them w steam and then heat them to a v high temp
what is the petrochemical industry
The petrochemical industry uses fractions from crude oil to make chemicals such as polymers, solvents, detergents and fuels.
2 methods used by the petrochemical industry for cracking hydrocarbons
Catalytic cracking
hydrocarbons are heated
passed over a catalyst
makes smaller alkanes and alkenes
Steam cracking
hydrocarbons are heated with steam
also breaks them into smaller molecules
alkenes and characteristics
have a C=C bond
have a double bond between 2 of the carbon atoms in their chain
it means that alkenes have 2 fewer hydrogens compared w alkanes containing the same number of carbon atoms, making them unsaturated
alkenes general formula
CnH2n
what makes alkenes more reactive alkanes
the double C=C bond can open up to make a single bond, allowing the 2 carbon atoms to bond w other atoms
first 4 alkenes
ethene, propene, butene, pentene
how do alkenes burn and why
with a smoky flame
when burnt in air they tend to undergo incomplete combustion
CO2 and H2O are still produced but u can also get carbon and carbon monoxide (CO) which is poisonous
incomplete combustion formula
alkene + oxygen → carbon + carbon monoxide + co2 + water
results in a smoky yellow flame and less energy being released compared to complete combustion
how to alkenes react
via addition reactions
all have functional group C=C, so they all react in similar ways and u can suggest products of a reaction based on how alkenes react in general
the carbon-carbon double bond will open up to leave a single bond and a new atom is added to each carbon
what is addition of a hydrogen called
hydrogenation
hydrogen can react w the double-bonded carbons to open up the double bond and form the equivalent, saturated, alkane
what can alkenes also react with
halogens - bromine, chlorine and iodine
the molecules formed are saturated w/ the C=C carbons each becoming bonded to a halogen atom
what do alkenes do to bromine water
turn it from orange to colourless
alkanes have no effect, only alkenes
what does steam react with alkenes to form
alcohols
when alkenes react with steam, water is added across the double bond and an alcohol is formed
how is ethanol produced from ethene and steam
Ethene reacts with steam in the reactor to produce ethanol.
Word equation: ethene + steam → ethanol
The reaction mixture then passes into a condenser, where it is cooled.
Ethanol and water condense into liquids because they have higher boiling points.
Unreacted ethene does not condense because it has a lower boiling point, so it stays as a gas.
The unreacted ethene is recycled back into the reactor so it can react again.
This reduces waste and improves yield.
The liquid mixture still contains ethanol and water, so they are separated by fractional distillation.
Ethanol is collected first because it has a lower boiling point than water.
what are plastic made of
long chain molecules, polymers
formed when lots of small monomers join together
usually carbon based w/ alkene monomers
what is polymerisation
when lots of small molecules, monomers, join together to form a long molecule, a polymer
what r addition polymers made from
unsaturated monomers
the monomers that make up addition polymers have a double covalent bond
lots of unsaturated monomer molecules, alkenes, can open up their double bonds and join together to form polymers chains - this is addition polymerisation
what does addition polymerisation require
high pressure and a catalyst
what are alcohols and their general formula
-OH group
general formula:
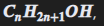
first 4 alcohols
methanol, ethanol, propanol, butanol
alcohol charcteristics
flammable and undergo complete combustion is air to produce CO2 and water
all soluble in water and their solutions have a neutral pH
also can react w/ sodium, one product of this is hydrogen
what happens when alcohols r heated
they are flammable
complete combustion
produce CO2 and WATER
what do different alcohols form
different carboxylic acids
what are alcohols used as and examples
solvents and fuels
methanol and ethanol used as solvents in industry this is bc they can dissolve most things water can dissolve as well as substances that water cannot dissolve
first 4 alcohols used as fuels bc they burn cleanly and not smelly
what is ethanol made by and for what
fermentation for alcoholic drinks such as wine or beer
uses an enzyme in yeast to convert sugars into ethanol, co2 also produced
happens fastest at 37 degrees in a slightly acidic solution and under anaerobic condtions
fermentation word equation
sugar →ethanol + carbon dioxide
→is yeast
carboxylic acids functional group
-COOH
first 4 carboxylic acids
methanoic, ethanoic, propanoic, butanoic
how do carboxylic acids react
like any other acid, with carbonates to produce salt, water and carbon dioxide
what happens when carboxylic acids dissolve in water
they ionise and release H+ ions resulting in an acidic solution
but they don’t ionise completely, so form weak acidic solutions
have a high pH (less acidic) than aqueous solutions of strong acids with the same concentration
esters functional group and how formed
-COO-
formed from an alcohol and a carboxylic acid
an acid catalyst is usually also used
how are esters made from carboxylic acids
alcohol + carboxylic acid →ester + water
→is acid catalyst
example of 1 ester
ethyl ethanoate
how r polymers made by condensation polymerisation
involves monomers which contain different functional groups
the monomers react together and bonds form between them, making polymer chains
for each new bond that forms, a small molecule (water) is lost
THIS IS WHY ITS CALLED CONDENSATION POLYMERISATION
how r addition and condensation polymerisation different
addition:
only 1 monomer type containing a C=C bond
only 1 product formed
functional groups involved in polymerisation: carbon-carbon double bond in monomer
condensation
2 monomer types each containing 2 of the same functional groups OR one monomer type with 2 different functional groups
2 types of product - the polymer and a small molecule (water)
functional groups involved in polymerisation: 2 reactive groups on each monomer
3 naturally occurring polymers
proteins (amino acids)
DNA molecules (nucleotide polymers)
simple sugars (eg starch or cellulose)
what 2 groups do amino acids have
amino group (NH2) and an acidic carboxyl group (COOH)
example of amino acid
glycine
how do amino acids form proteins
they form polymers known as POLYPEPTIDES via condensation polymerisation
the amino group of an amino acid can react with the acid group of another, and so on, to form a polymer chain. For every new bond formed, a water molecule is lost
1 or more long chains of polypeptides are known as PROTEINS
the order of the amino acids in their polymer chains is what gives proteins their properties and shapes
what r dna molecules made from and how
nucleotide polymers
dna found in every living thing, contains genetic info, it is large and double helix shaped
DNA is made of 2 polymer chains of monomers called nucleotides. each contains a small molecule, a base, 4 different types of bases (A, C, G, T)
the bases on the different polymer chains pair up with eachother and form cross links keeping the 2 strands of nucleotides together, and making it double helix shaped
order of bases = code of an organisms genes
how do simple sugars form polymers
sugars are small molecules that contain carbon, oxygen and hydrogen
sugars react together through polymerisation reactions to form larger polymers, like starch which living things use to store energy, and cellulose found in plant cells
Explain how oxides of nitrogen are produced when burning fuels.
high temperatures ignore pressure
(cause) nitrogen (from air) and oxygen (from air) to react
In the first step, crude oil is heated and hydrocarbon vapours are formed.
Explain how kerosene is produced from these hydrocarbon vapours.
there is a temperature gradient in the (fractionating) column
(so) kerosene condenses
at the level (in the column) corresponding to kerosene’s boiling point (range)
Explain how fractions are obtained from crude oil by fractional distillation.
crude oil is heated to vaporise (the hydrocarbons)
there is a temperature gradient in the (fractionating) column
(so) the gases condense at different levels
because of their) different boiling points
Explain why sulfur impurities are removed before petrol is burned in car engines.
(when burned sulfur impurities) produce sulfur dioxide
(which) causes acid rain
Name the process which produces smaller hydrocarbon molecules from heavy fuel oil.
Give the conditions used in this process.
(name of process) cracking
(conditions) high temperature
steam / catalyst
Give the result of the test when carbon dioxide is bubbled through limewater.
calcium hydroxide
method to show different dyes in an ink
draw pencil start line
• place spot of ink on start line
• name suitable solvent
• place solvent in beaker
• place paper in solvent so solvent is below start line
• use a lid
• allow solvent / dyes to travel up paper (until near top)
• dry + count spots
Give the general name for what dna is made of
nucleotides