Chapter III: Intro to organic molecules and functional groups
1/44
Earn XP
Description and Tags
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
45 Terms
Functional Group
an atom/group of atoms with characteristic & physical properties
most organic compounds contain a carbon backbone consisting of C-C and C-H bonds to which _______ ______ are attached
Heteroatoms
atoms other than carbon or hydrogen
π bonds
most commonly occur in C=C and C=O bonds
carbon backbone/skeleton
made of C-C and C-H σ bonds
R
abbreviation of the carbon skeleton

Alkane
a molecule made up of only C-C and C-H bonds. Contains NO polar bonds, lone pairs, or π bonds, & therefore contains NO reactive sites (functional groups)
Example: Ethane (CH3CH3)

Alkene
have a C=C bond as a functional group
Example: Ethylene (CH2=CH2)
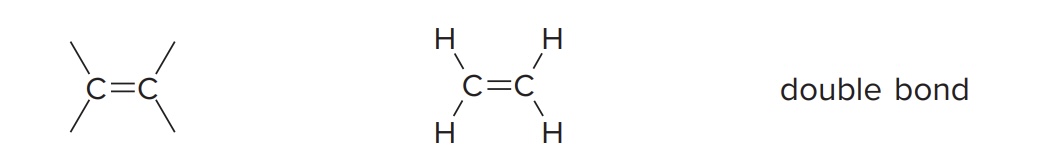
Alkyne
Have a C≡C bond as a functional group
Example: Acetylene (HC≡CH)
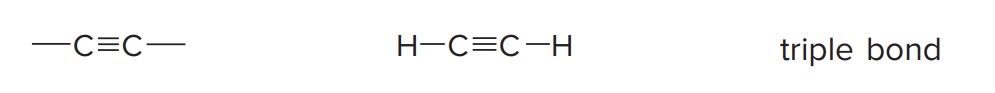
Alcohol
a molecule with a carbon skeleton and an OH group (hydroxy group) that makes the molecule have lone pairs and polar bonds that make it reactive
Example: Ethanol
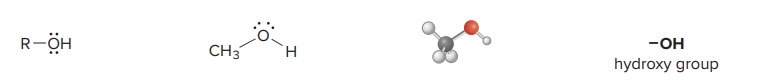
Hydrocarbons
compounds made up only of the elements carbon & hydrogen
Aliphatic Hydrocarbons
a compound/portion of a compound made up of C-C σ and π bonds but not aromatic bonds
Aromatic Hydrocarbons
named because many of the earliest compounds in this category had strong, characteristic odors
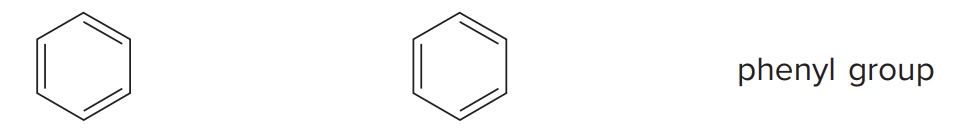
Benzene
Simplest aromatic hydrocarbon. Its 3 π bonds make it a functional group. Called a “phenyl” group when part of a bigger molecule
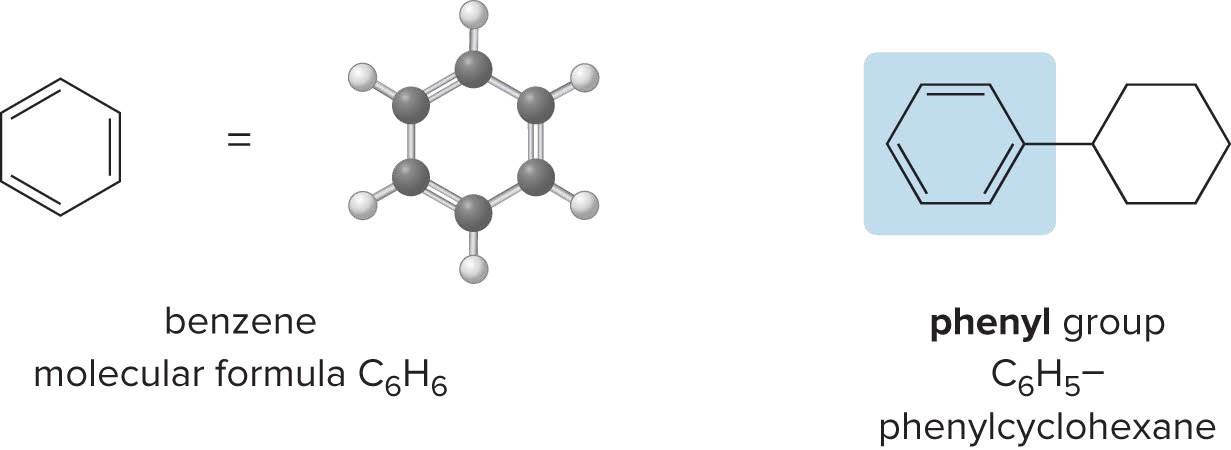
Classification of Carbon Atoms
C atoms are classified by the # of other carbon atoms attached to them
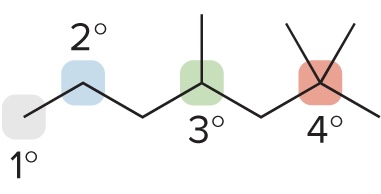
Primary Carbon (1° Carbon)
bonded to one other C atom
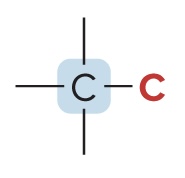
Secondary Carbon (2° Carbon)
bonded to two other C atoms
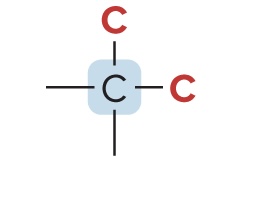
Tertiary Carbon (3° Carbon)
bonded to three other C atoms
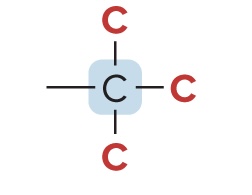
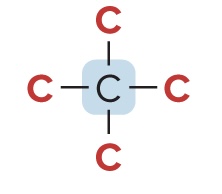
Quaternary Carbon (4° Carbon)
bonded to four other C atoms
Classification of Hydrogen Atoms
H atoms are classified by the type of carbon to which they are bonded
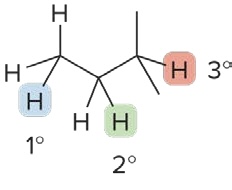
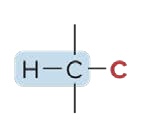
Primary Hydrogen (1° H)
the C atom is bonded to 1 other C atom
Secondary Hydrogen (2° H)
the C atom is bonded to 2 other C atoms
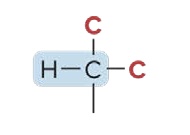
Tertiary Hydrogen (3° H)
the C atom is bonded to 3 other C atoms
Alkyl Halide

Ether
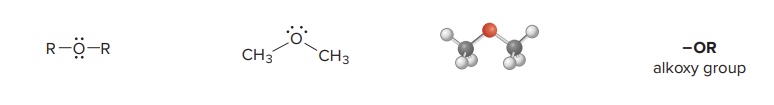
Amine

Thiol
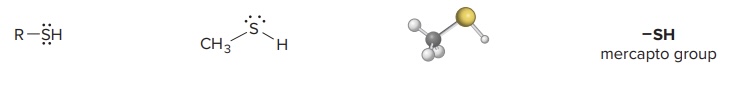
Sulfide

Classification of Alkyl Halides & Alcohols
Classified by the carbon to which they are bonded
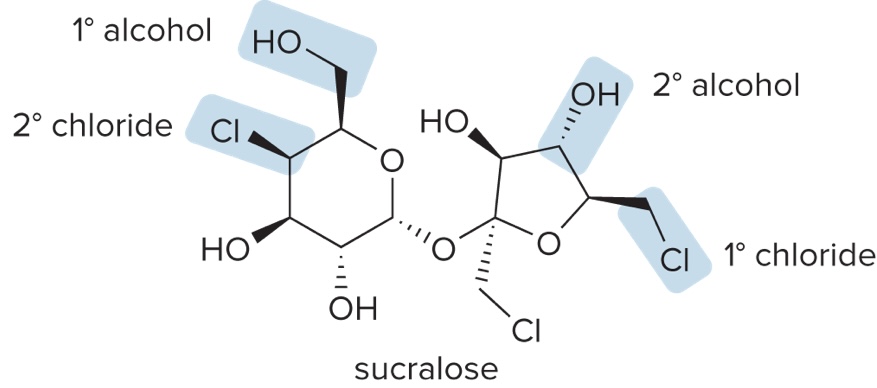
1° alkyl halide/1° alcohol
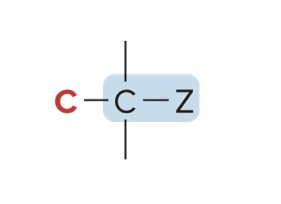
2° alkyl halide/2° alcohol
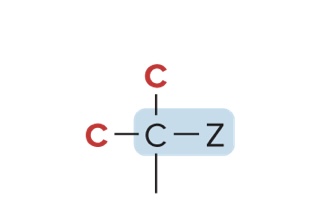
3° alkyl halide/3° alcohol
Classification of Amines
Classified based on the # of carbons bonded to the nitrogen atom
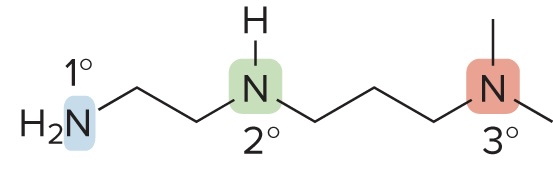
1° amine
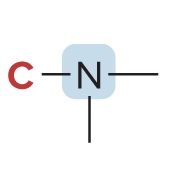
2° amine
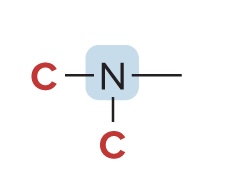
3° amine
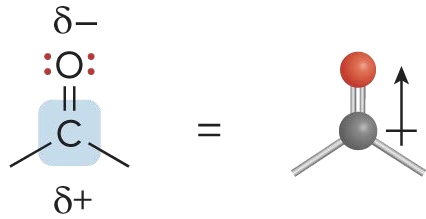
Carbonyl Group
when there is a C=O polar bond, making the group an electrophile. The lone pairs on O allow it act as a nucleophile and base
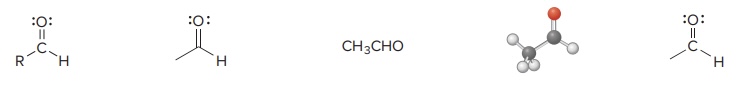
Aldehyde
Ketone
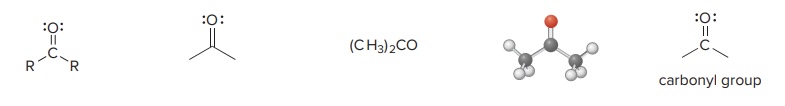
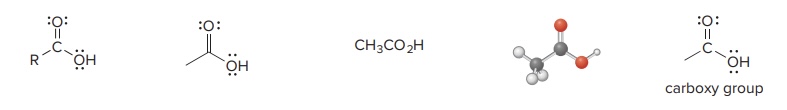
Carboxylic Acid
Ester
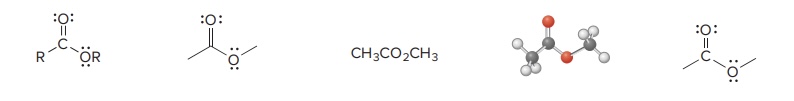
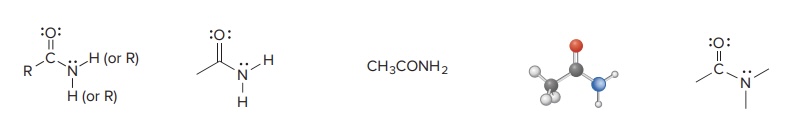
Amide
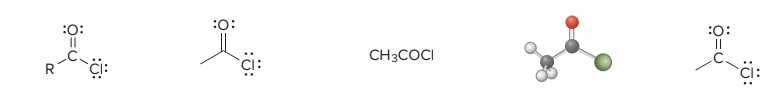
Acid Chloride
Intermolecular Forces
Interactions (attractions) that exist between molecules
Ion-Ion Interactions
Because ionic compounds contain oppositely charged particles, they are held together by extremely strong electrostatic interactions
Van der Waals Forces
very weak interactions caused by momentary changes in electron density in a molecule
The only IMFs present in non-polar Compounds