Lecture 12- Dental Structure
1/43
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
44 Terms
Main 5 components for formation of tooth:
enamel
dentin
DEJ
pulp
cementum
3 most important tooth formation (odontogenesis- dentin formation) stages in embryo (6 weeks):
bud, cap, bell
The transitions from one morphological stage to the next (lamina → bud → cap → bell) are visualized in three dimensions, showing how cells proliferate, differentiate, and fold to form the enamel organ and dental papilla
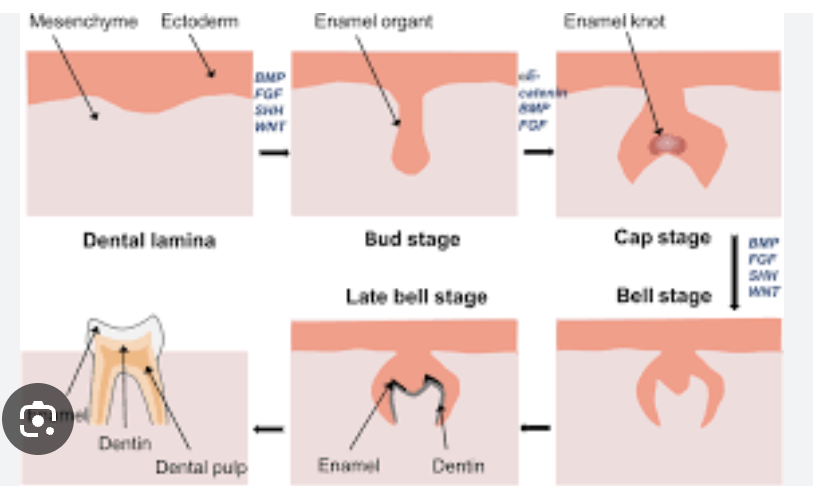
Primary Epithelial Band
The primitive oral epithelium thickens in the jaws as a continuous band
This later splits into dental lamina (inner) and vestibular lamina (outer)
Dental Lamina
Invagination of epithelium into mesenchyme where future teeth will form
Bud Stage
The epithelial cells proliferate into a bud-like structure still connected to the lamina
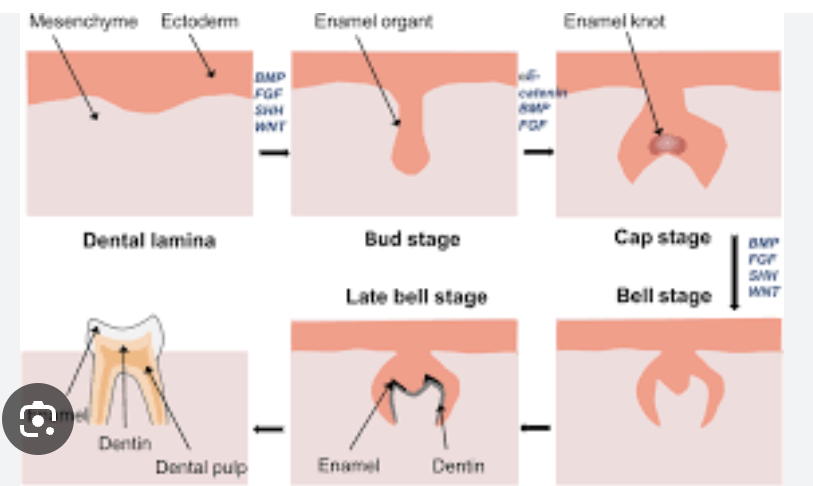
Cap stage
The “bud” becomes more complex and shows a concavity on its inner surface |
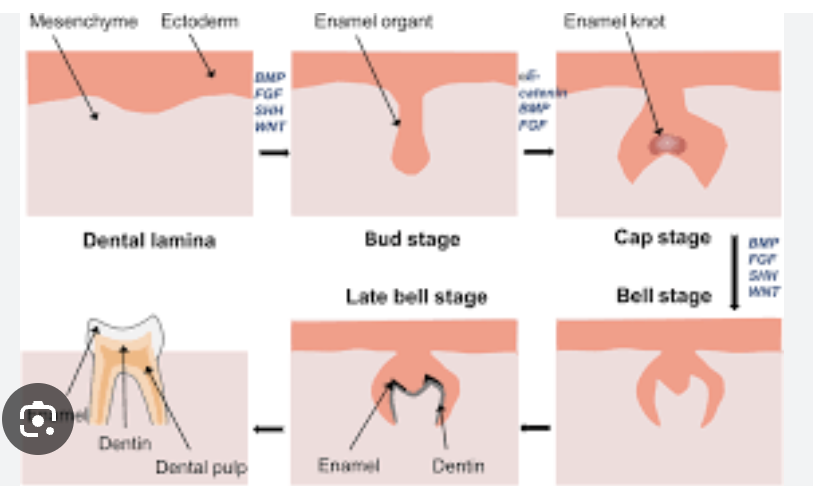
bell stage
The tooth germ takes on a bell shape; morpho differentiation continues
The inner enamel epithelium begins to determine the shape of the crown; enamel knots (non-dividing signaling centers) regulate cusp formation
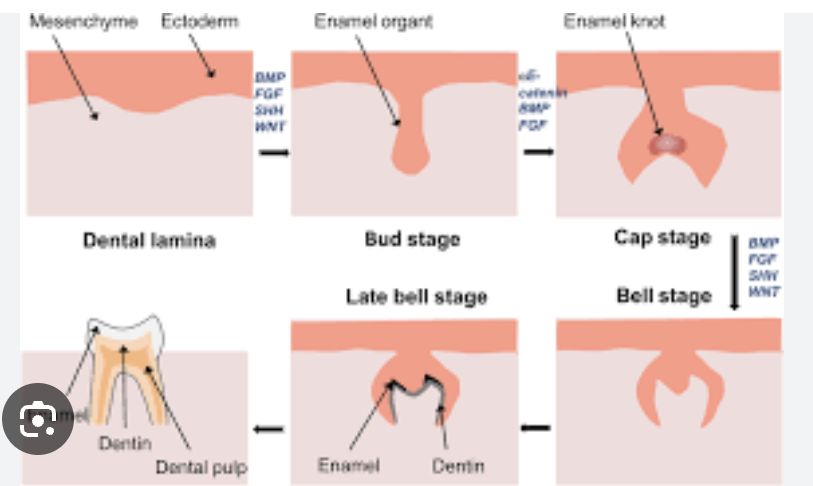
Amelogenesis
the process of forming the enamel layer of teeth.
Enamel is the hardest tissue in the body, made mostly of mineral (hydroxyapatite) with small amounts of organic matrix and water.
Enamel formation begins after dentin formation has started (dentinogenesis), because ameloblasts depend on signals from the underlying dentin layer and dentin-forming cells (odontoblasts)
Inner enamel epithelium (IEE): from the enamel organ, these cells differentiate into ameloblasts -> specialized epithelial cells that produce enamel matrix proteins and coordinate mineralization
stages of Amelogenesis
1) Secretory Stage (Matrix Formation) - Ameloblasts secrete enamel matrix proteins & create scaffold
2) Maturation / Mineralization Stage - initially secreted enamel matrix becomes increasingly mineralized & hydroxyapatite crystals grow
3) Transitional / Modulation Stage - Ameloblasts change their behavior (morphology) to support final mineral uptake
4) Post-maturation / Protective Stage - After enamel is fully mineralized, ameloblasts ultimately become part of the reduced enamel epithelium and serve a protective role until tooth eruption
composition of enamel
88% inorganic (hydroxyapatite)
10% water
2% organic

inogranic structure of enamel:
Enamel consists of hydroxyapatite crystallites 25 nm thick, 100 m wide and 500-1000 m long. (look like needles)
Crystallites are arranged into 5 um diameter rods that are encapsulated by 1 um thick protein rich sheaths.
Enamel rods are arranged parallel in a direction perpendicular to the dentin-enamel junction
from dentin to the outer enamel surface.
• Within the rod units, the directional arrangement of the apatite crystallites varies.
Crystallites in the central part of the rod are parallel to the rod axis while those near the edge of the rod usually have an angle of near 15°-45° to the longitudinal axis of the rods.
organic structure of enamel: (control the growth enamel)
Enamel organic components consist of proteins and enzymes
Proteins: Amelogenin, Ameloblastin, Enamelin, and Tuftlelin
Enzymes: matrix metalloproteinase (MMP), Proteinase, Phosphatase
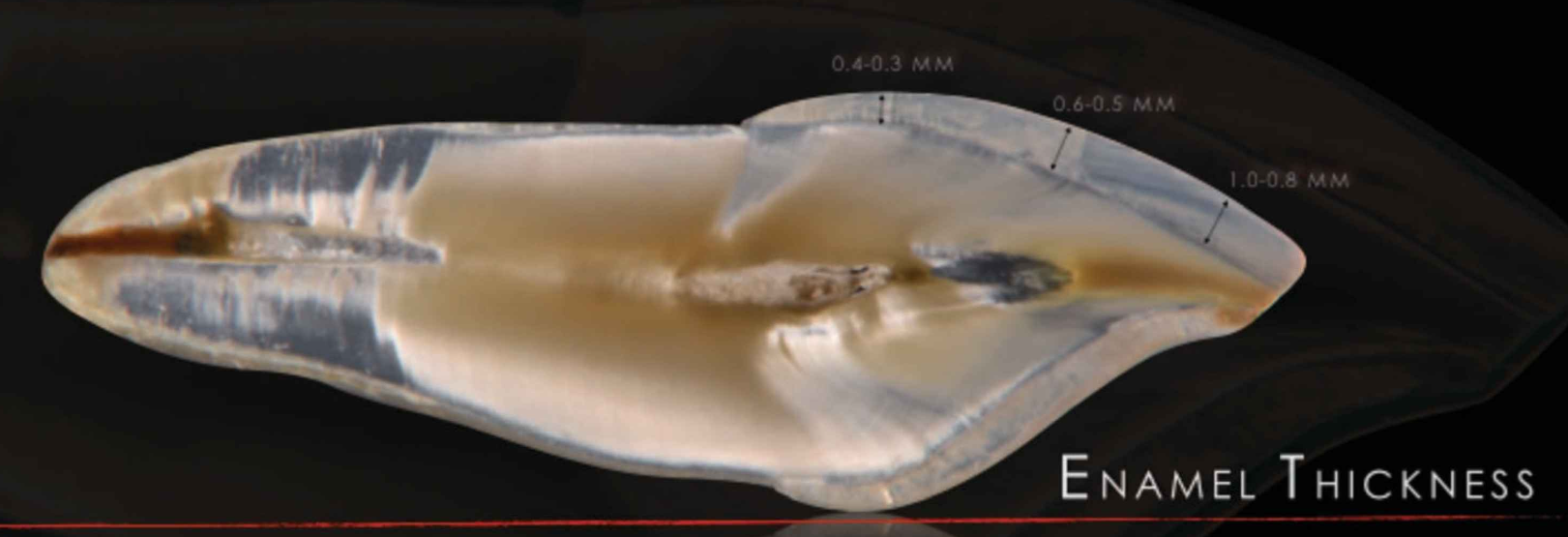
enamel thickness
gingival/ cervical third: 0.4-0.3 mm
middle third: 0.6-0.5 mm
junction of middle third/ incisial: 1- 0.8 mm
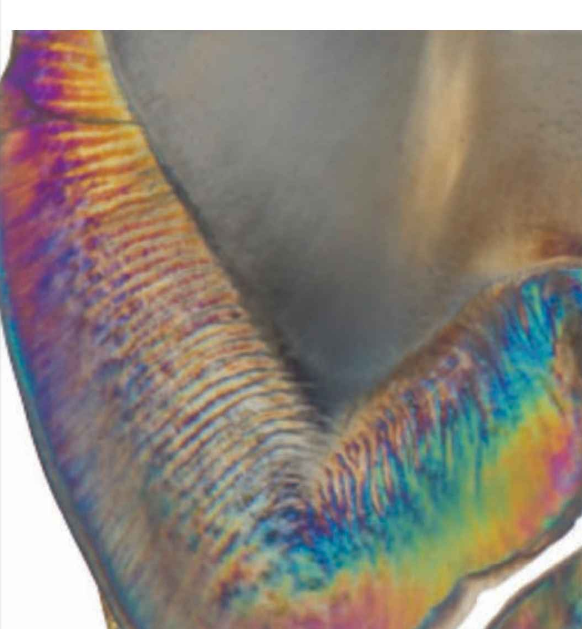
striae of retzius
the striae of Retzius are incremental growth lines or bands
seen in tooth enamel.
They represent the incremental pattern of enamel (like a tree), the successive apposition of different layers of enamel during crown formation

ENAMEL CROSS-STRIATIONS
Cross-striations demarcate the amount of enamel deposited by ameloblasts in a single day. The average rate is approximately 4 µm/day in humans.
The average distances between cross-striations in human teeth are about 2.5 µm at the DEJ and 6.5 µm at the enamel surface.

HUNTER-SCHREGER BANDS (HSBS)
A set of 10 or more layers of enamel rods composes the Hunter–Schreger Bands (HSBs).
HSBs are due to the synchronous decussation of enamel rods in the horizontal plane and are probably caused by the reflection of light by the interprismatic material.
They are most concentrated in regions exposed to greater demand, such as:
The occlusal surfaces of posterior teeth (for chewing forces)
The surfaces of maxillary and mandibular canines (for guiding mandibular movement)
enamel rods are ____ on external surface and on internal surface they move into ____ directions/ inclinations
parallel; different

outer enamel
Enamel rods are mostly oriented parallel to each other and perpendicular to the dentin-enamel junction (DEJ).
The outermost layer may contain aprismatic enamel (enamel rods not well defined and very dense)
This is the area of enamel with the highest modulus of elasticity

inner enamel:
Closer to the DEJ, the enamel rods decussate in layers (or bands) like S.
This area of enamel has a lower modulus of elasticity compared to the outer enamel → less rigid to help disipate loads
Contains increased organic content
Contains enamel tufts
Enamel Decussation (how enamel dissipates)
Decussation: Constitutes a structural reinforcement of teeth
Caused by crossing enamel rod bundles within alternating bands that follow a sinusoidal path
Helps prevent crack propagation
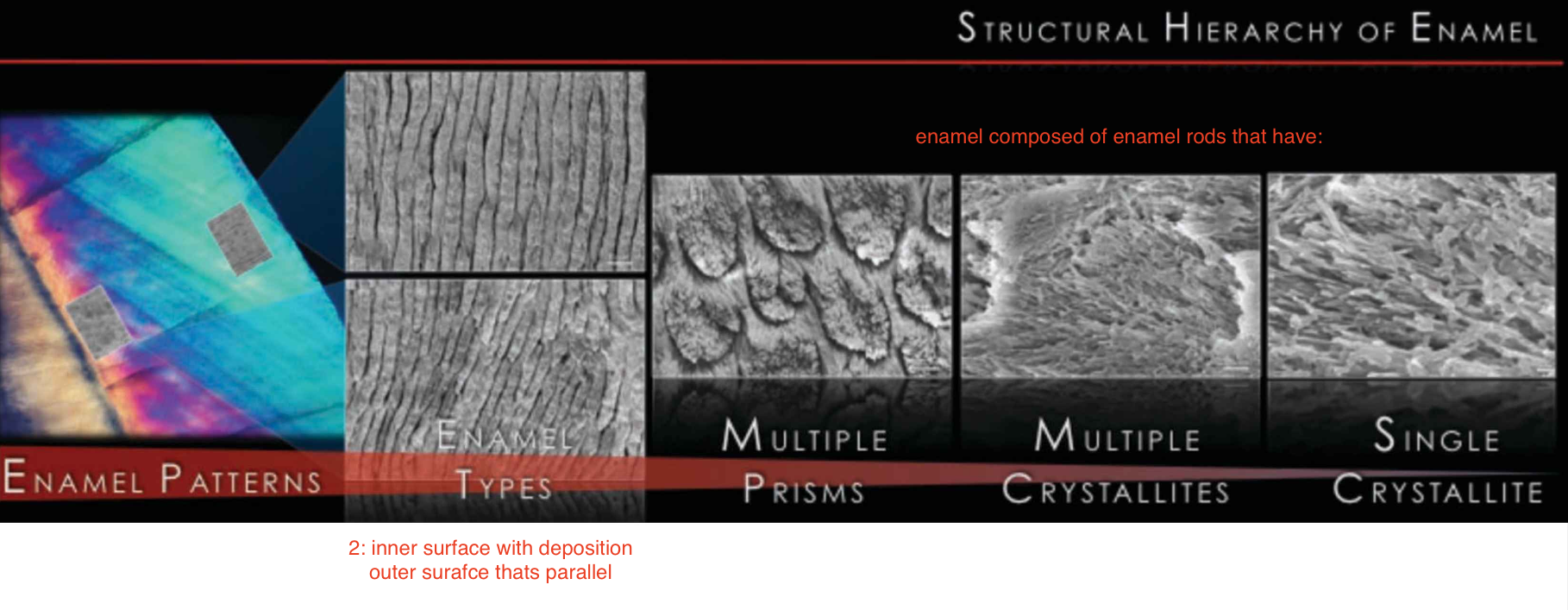
structural hierachy of enamel
enamel patterns → 2 types: inner surface with deposition & outer surafce thats parallel → enamel composed of enamel rods that have: multiple prisms, multiple crystallites, single crystallite
why are enamel rods perpendicular into inner surface?
if enamel rods were all parallel into inner surface then whole tooth would break so thats why inner surface goes into dif directions to block the crack from propogating

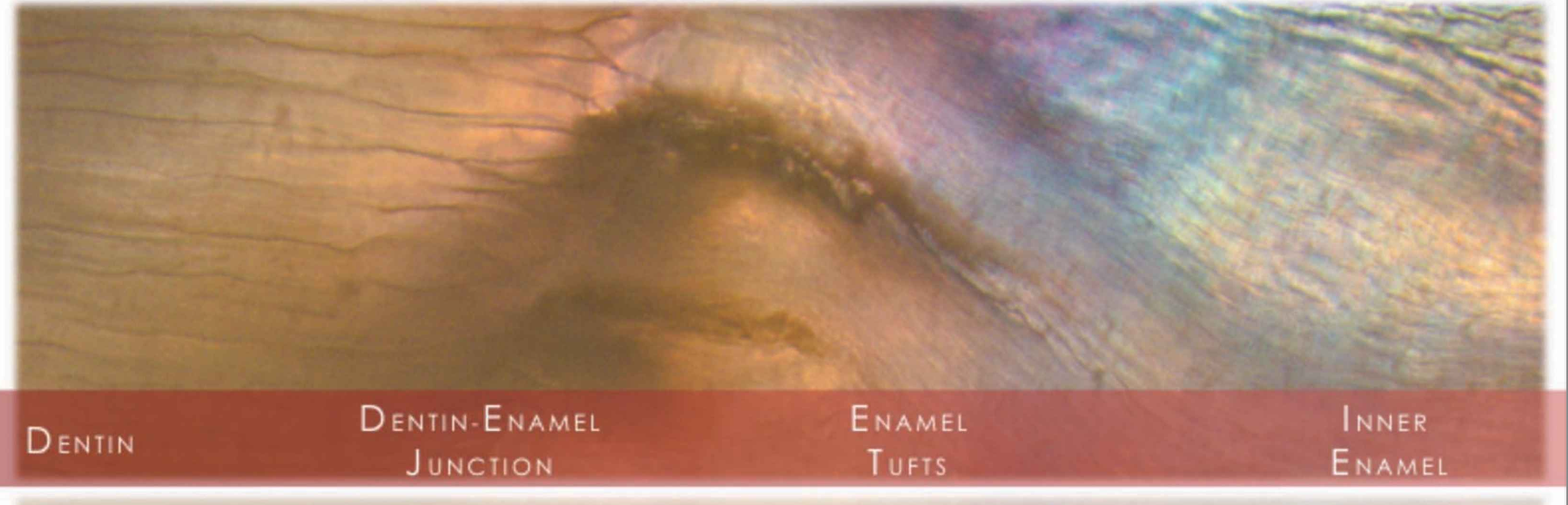
enamel tufts
Brush-like structures extending outward from the dentin-enamel junction (DEJ)
Hypomineralized regions containing increased residual enamel matrix proteins, likely due to changes in the direction of adjacent enamel rods originating from different areas of the scalloped dentin-enamel junction
May assist in the resilience of enamel

enamel lamellae
Fissure-like linear enamel defects containing proteins, proteoglycans, and lipids
Extend along the longitudinal axis of the tooth, perpendicular to the dentin-enamel junction (DEJ)
proteins that are not fully absorbed

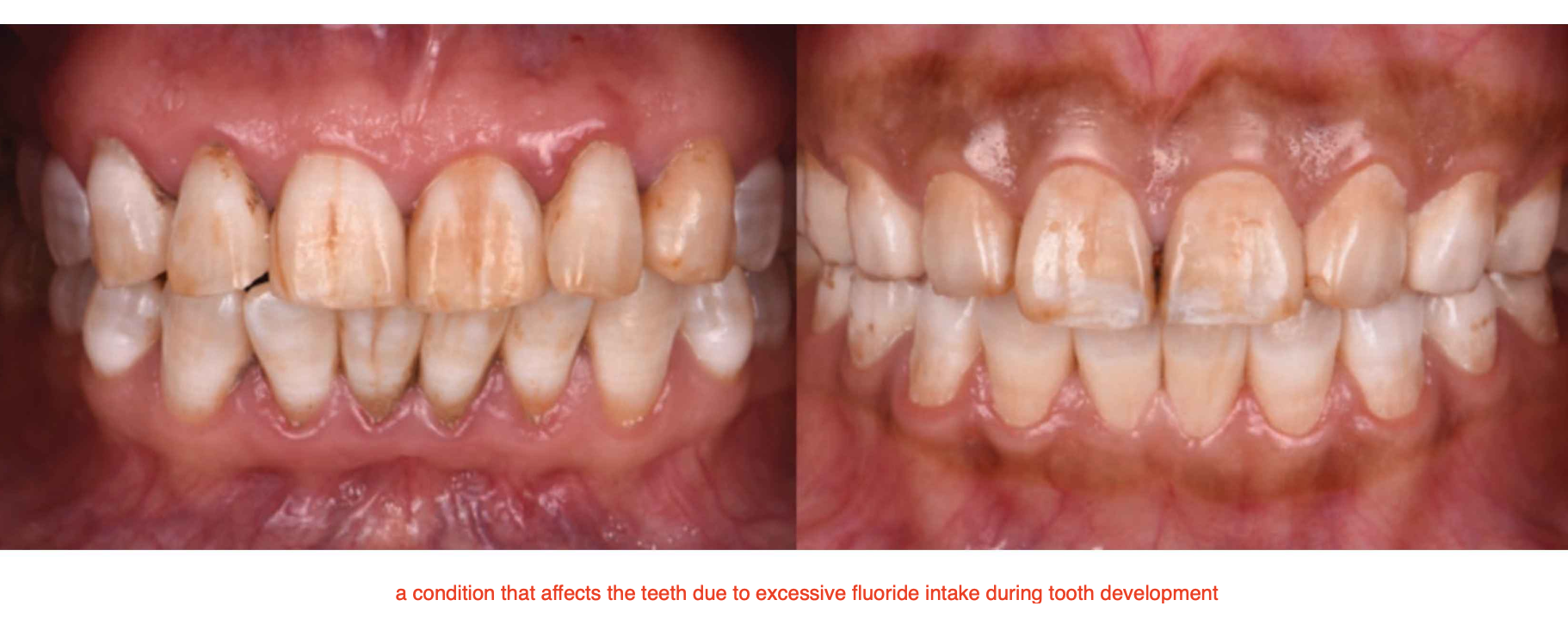
dental fluorosis
Areas of increased enamel porosity along the striae of Retzius (so excessive fluoride)
Hypomineralized lesions that can extend throughout the enamel
Can lead to pits, bands, attrition, abrasion, and loss of extensive areas post-eruptively
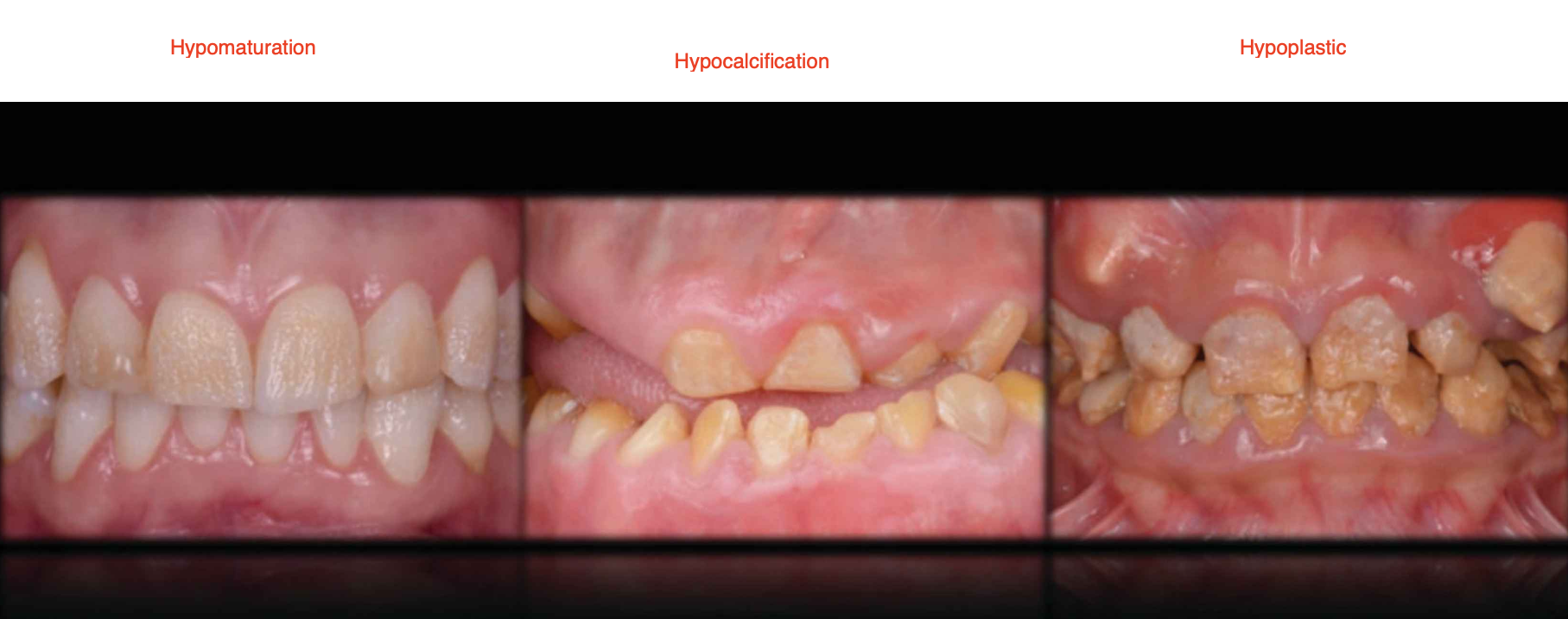
Amelogenesis Imperfecta
Cause: Mutations in AMELX, ENAM, MMP20, and FAM83H genes
Types:
Hypomaturation: Defect in the final growth and development of the tooth enamel (chips)
Hypocalcification: Defect in the initial stage of enamel formation, followed by defective tooth growth (soft)
Hypoplastic: Defects in the amount of enamel thin
can be treated with flouride (must be ingested)
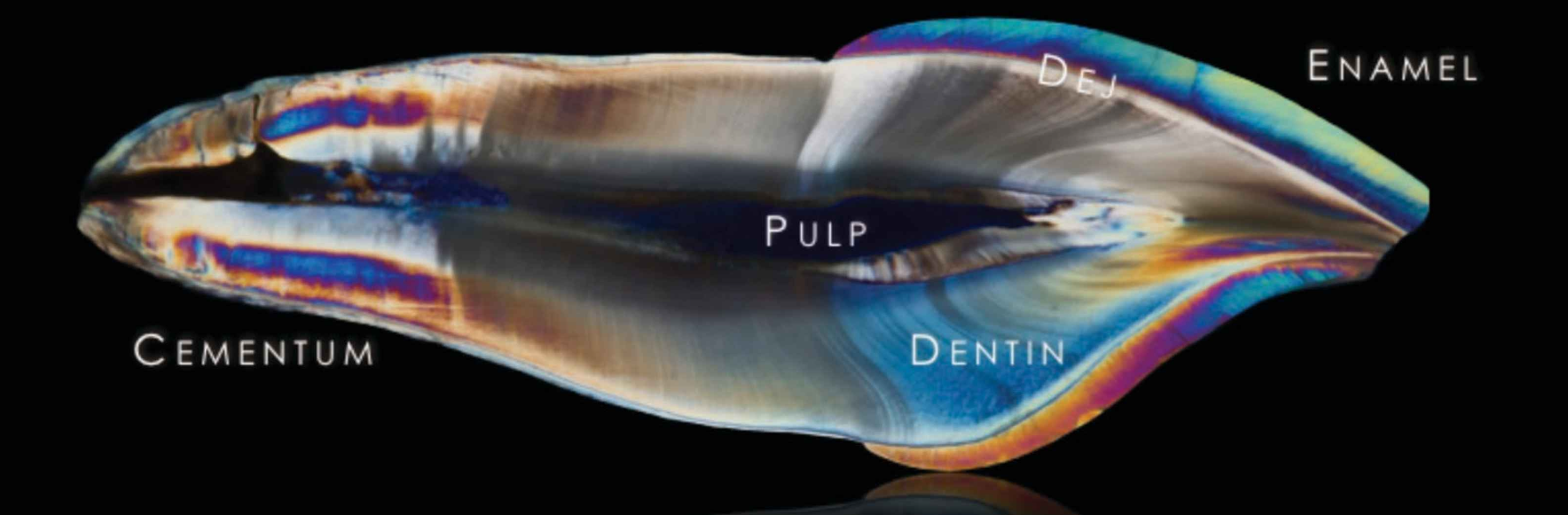
dentin-enamel junction DEJ
The dentin-enamel junction (DEJ) unites two dissimilar calcified tissues: enamel and dentin
Three-dimensional scalloped appearance with its convexities directed toward the dentin and concavities directed toward enamel.
Scallop size: 30 pm on incisors and 43 pm on molars
Prevent enamel cracks from propagating across the interface, thus preventing catastrophic tooth fractures → if crack reaches dentin the whole tooth will crack


Dentin composition:
50% inorganic (hydroxyapatite)
30% organic
20% water (more than enamel)
Dentin- organic microstructure
Type I Collagen (90%) (small amounts of Type III and Type V collagen are present 1-3%)
• Non-collagenous components (10%): phosphorylated and non-phosphorylated proteins,
proteoglycans, lipids, growth factors
• Proteins: Amelogenin, Osteonectin, Osteocalcin
• Enzymes: Matrix metalloproteinases (MMP-1, -2, -3, and -9), tissue inhibitors of metalloproteinases
(TIMPS), Acid & alkaline phosphates
Dentin- inorganic microstructure
Crystallites: 2-5 nm in thickness and 60nm in length and randomly fill interfibrillar spaces (between collagen fibers)
• Intertubular crystallites have a needle-like appearance.
They are located either at the surface the collagen fibrils, and parallel with the collagen fibril axis.
3 structures in dentin:
1) dentinal tubules: holes from DEJ to pulps (blue)
2) peritubular dentin: around tubule, very calcified area (green)
3) intertubular dentin: dentin between each tube (red area)
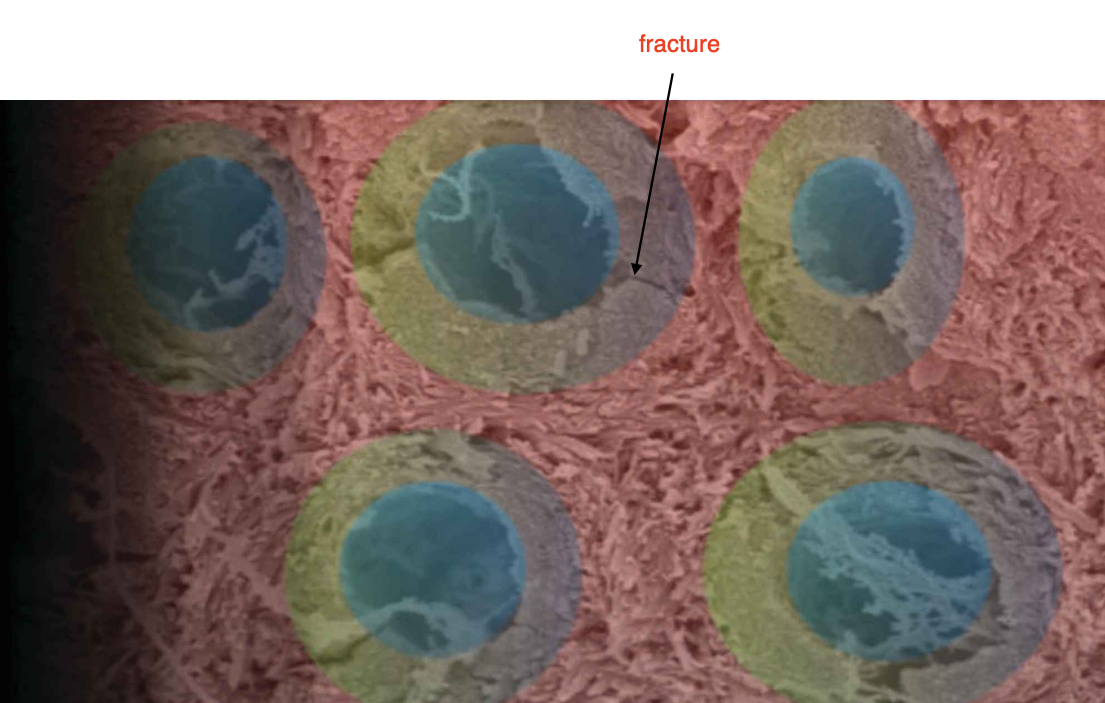
intertubular dentin
Organic content (12%): Collagen-rich dentin associated with proteins along and between the collagen fibrils
Inorganic content (88%): Hydroxyapatite crystallites, 2–5 µm wide and 60 nm long
Hardness:
Near DEJ: 0.49–0.52 GPa
Near pulp: 0.12–0.18 GPa
peritubular dentin
Structure: Amorphous network without collagen fibrils, lining the lumina of dentinal tubules
Organic content: 4%
Inorganic content: 96%, composed of 25–30 nm hydroxyapatite crystallites
Hardness: 2.23–2.50 GPa
superficial vs deep dentin
superficial dentin: close to DEJ; 20,000 tubules/ mm^2
deep dentin close to pulp; 40,000 tubules/ mm²
3 types of dentin: primary, secondary and tertiary
primary dentin: Dentin formed from the start of dentinogenesis near the DEJ up to eruption, before the tooth becomes functional
secondary dentin: Dentin formed after the completion of tooth formation and continues to form throughout the remaining life of the tooth
tertiary dentin: Dentin fromed in the presence of aggression: reactionary or reparative
Reactionary Dentin
Formed as a response of odontoblasts and Höehl’s cells to:
Carious decay
Cavity preparation
Abrasion, erosion, or attrition
Cytotoxic molecules from a restorative biomaterial
*when theres a caries near by the dentin will calcify in response to protect pulp

Reparative Dentin
Formed in response to pulp exposure
Often forms a dentin bridge
can stimulate pulp to create dentin bridge with products
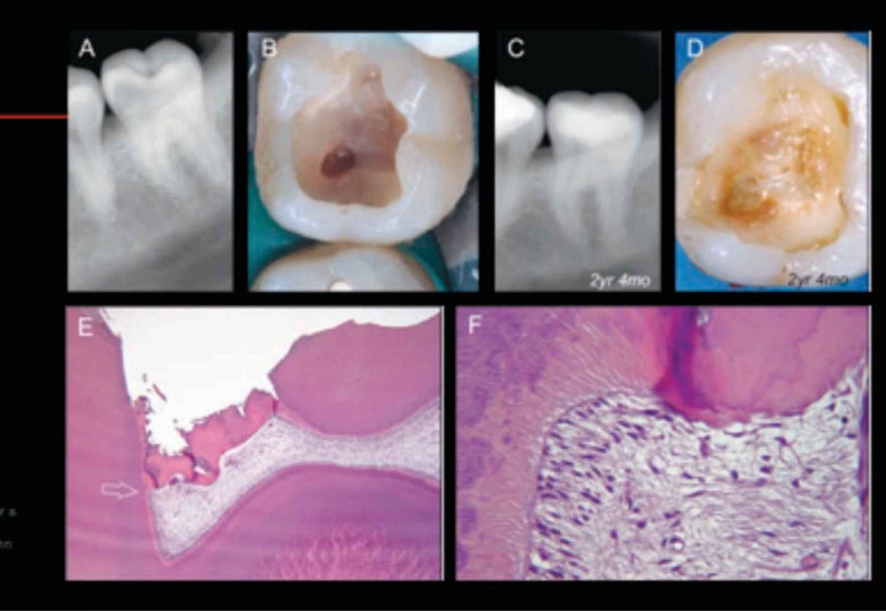
Sclerotic Dentin
Tubular obliteration with rhombohedral crystallites
Dentin becomes hypermineralized (too much minerals)
Common in Non-Carious Cervical Lesions (NCCLs)
Clinically appears as shiny dentin
dental tubules calcified
Tomes' fibers (Odontoblast processes)
Odontoblast (inside of pulp that extends in tubules) processes are the long, slender extensions of odontoblast cells that penetrate into the dentin, extending from the pulp-dentin border.
these processes are involved in dentin formation, mineralizing the dentin matrix and regulating its structure.
They also play a role in mechanosensation (sensing mechanical forces) and participate in the repair of dentin after injury to mature teeth.
Brännström's hydrodynamic theory
is the leading explanation for dentin hypersensitivity, proposing that stimuli like hot, cold, or sweets cause fluid to move within exposed dentinal tubules.
This fluid movement stimulates mechano-receptors and nerve endings in the pulp, which then triggers a short, sharp pain sensation.
The theory is supported by research demonstrating that tubule occlusion can reduce dentin permeability, thereby preventing the fluid flow that causes sensitivity.
Dentinogenesis Imperfecta
Cause: Mutations in the DSPP gene, which alter the proteins produced, leading to abnormally soft dentin
Clinical features: Teeth are discolored, weak, and more prone to decay and fracture
Types:
Type I:
Occurs in people with osteogenesis imperfecta (brittle bones)
Mutations in COL1A1 or COL1A2
Type II:
Occurs without another inherited disorder
Some families have progressive hearing loss in older age
Most common type of dentinogenesis imperfecta
Type III:
Occurs without another inherited disorder
First identified in families in southern Maryland
Also reported in individuals of Ashkenazi Jewish descent
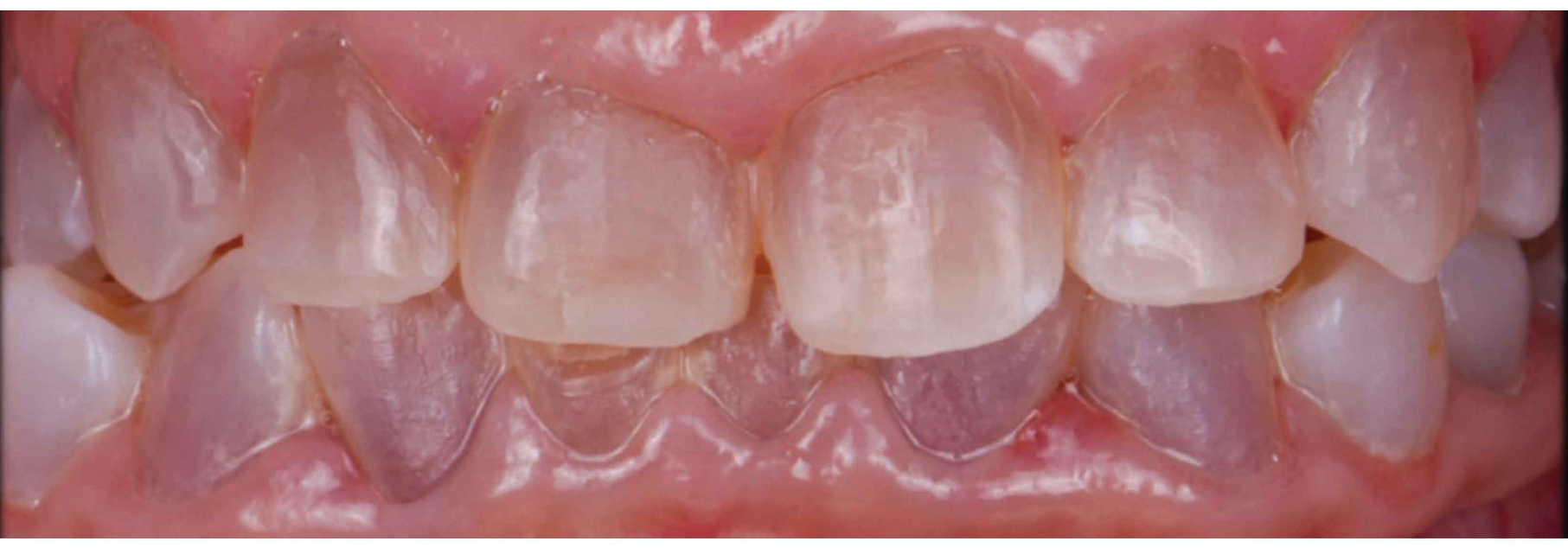
microstructure of dentin in Dentinogenesis Imperfecta
Atubular dentin: The dentin lacks the typical organized tubules and instead presents with occasional, irregular dentin tubules.
Altered composition: It exhibits a higher mineral-to-matrix ratio compared to unaffected dentin, primarily due to a reduced amount of matrix (specifically, amide I of collagen) resulting from decreased type I collagen formation.
Reduced strength: This altered composition leads to lower fracture resistance in the affected dentin.
Genetic association: DI is associated with Osteogenesis Imperfecta and a mutation on the COL1A1 gene, which is involved in collagen production.
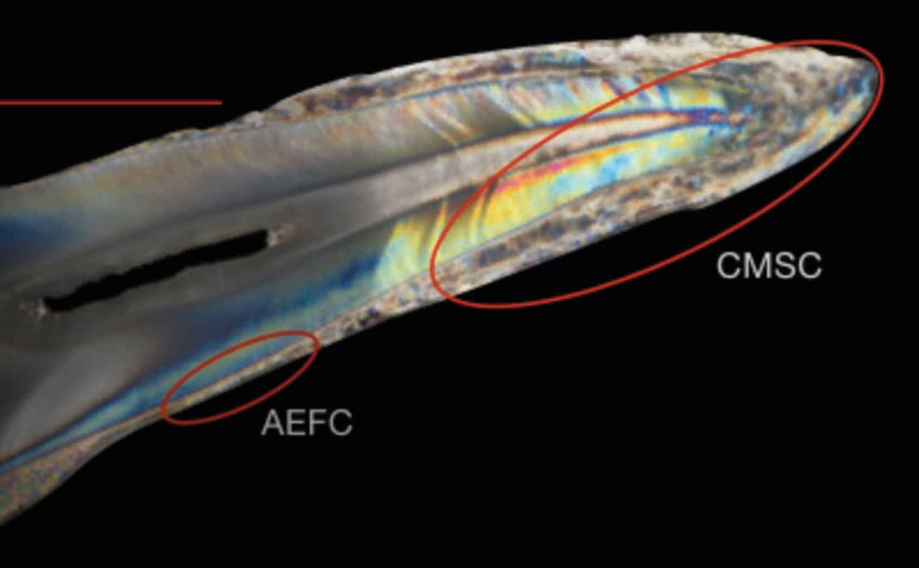
cementum
Mineralized tissue covering the entire root surface
Types:
Acellular Cementum
Thin, covering the cervical root
Also called Acellular Extrinsic Fiber Cementum (AEFC)
20-50 um
Cellular Cementum
Thick, covering the apical root
Also called Cellular Mixed Stratified Cementum (CMSC)
150-200 um
CEMENTUM ENAMEL JUNCTION (CEJ)
the boundary between the enamel (hard outer layer of the tooth) and the cementum
60% - 65% ENAMEL AND CEMENTUM OVERLAPS
30% edge to edge
5-10% enamel and cementum dont overlap
