Molecular Geometry
1/25
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
26 Terms
Molecule
2 or more elements that are chemically combined
Compound
2 or more different elements chemically combined
London Dispersion Forces
Found in all molecules
very weak
Only IMF in nonpolar molecules
attraction due to temporary dipoles
Dipole-Dipole Forces
Found in all polar molecules
Has partial positive and negative charges that attract each other.
Hydrogen Bonding
Very strong dipole-dipole forces (Hydrogen has FON)
strongest IMF possible in polar covalent molecules
Ion-Dipole/Molecule-Dipole
Attractions between an ion and a water molecule that is stronger than an ionic bond
responsible for ionic compounds dissolving in water
Ion
Charged particle formed when an atom gains e- or loses e-
Isotope
Elements w/ the same number of protons but different number of neutrons
Ground State
Most stable or lowest energy arrangement of electrons
Electrons fill the lowest energy level first
Fill each new orbital w/1 electron before beginning to pair
Linear
2 bonding pairs
180 degrees
Trigonal Planar
3 bonding pairs
120 degrees
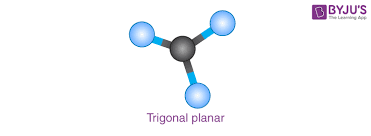
Bent
2 bonding pairs
1 nonbonding pair
120 degrees
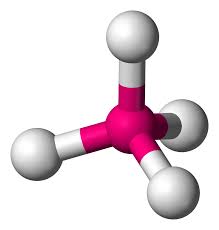
Tetrahedral
4 bonding pairs
109.5 degrees
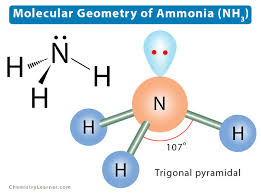
Trigonal Pyramidal
3 bonding pairs
1 nonbonding pair
109.5 degrees
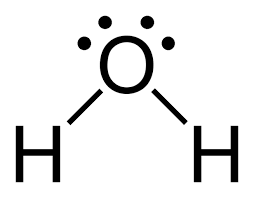
Bent
2 bonding pairs
2 nonbonding pairs
109.5 degrees or 104.5
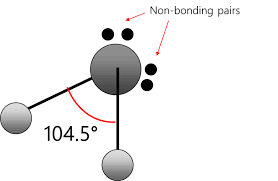
Molecular Geometry
Describes what molecule actually looks like
Electronic Geometry
Takes into account all charged groups (Bonds+Lone pairs)
Polar
uneven, asymmetric distribution of charge/atoms
Nonpolar
Even, symmetric distribution of charge/atoms
Relationship between polarity + solubility
“Like dissolves Like”
substances dissolve in other substances w/similar IMFs
Universe favors low energy and high entropy
Transferring electrons (IONIC)
Metal (Lose electrons easily) + Nonmetal (attract electrons, hold them tightly)
Sharing Electrons (COVALENT/MOLECULAR)
Nonmetals (similar electronegativity and Ionization energy)
Difference in electronegativity determines polarity
Mobile Electrons (METALLIC)
Metals only (Sea of electrons)
Bonds Breaking
Equation - H2 + energy = 2H
Energy - Endothermic
Stability - decreasing
Like - Divorce
Bonds Forming
Equation - 2H = H2 + energy
Energy - Exothermic
Stability - Increases
Like - Marriage