Physics Quarterly
1/129
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
130 Terms

C. II and III only

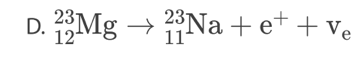
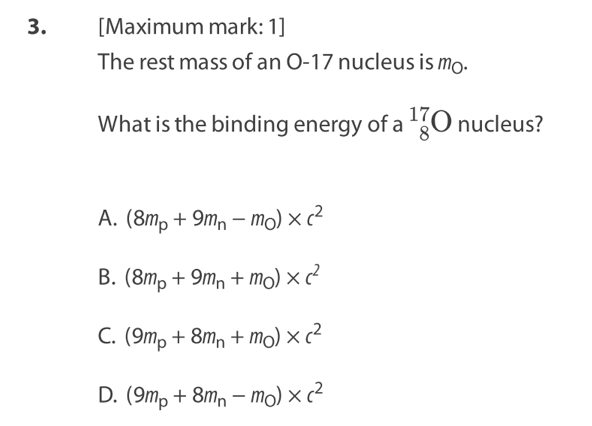
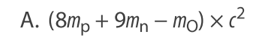

A


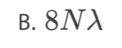

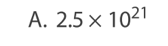



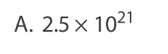
Which statement about atomic nuclei is correct?
The density is...
A. directly proportional to mass number.
B. inversely proportional to nuclear radius.
C. inversely proportional to volume.
D. constant for all nuclei.
D. constant for all nuclei
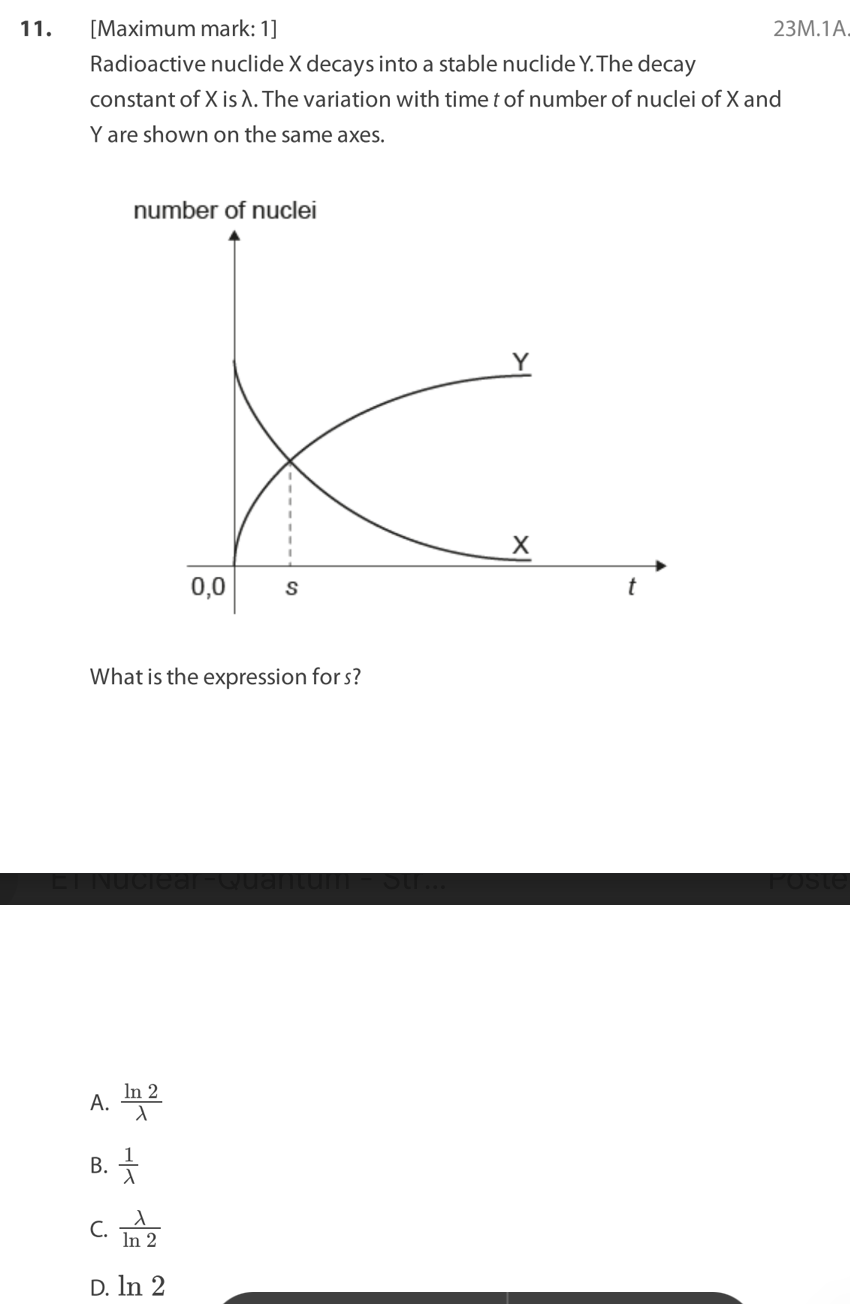
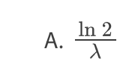

D. the probability that a nucleus decays per unit time in the radioactive
sample.
Three statements about radioactive decay are:
I. The rate of decay is exponential.
II. It is unaffected by temperature and pressure.
III.The decay of individual nuclei cannot be predicted.
Which are correct?
a. I and II only
B. I and III only
C. II and III only
D. I, II and III
D. I, II and III
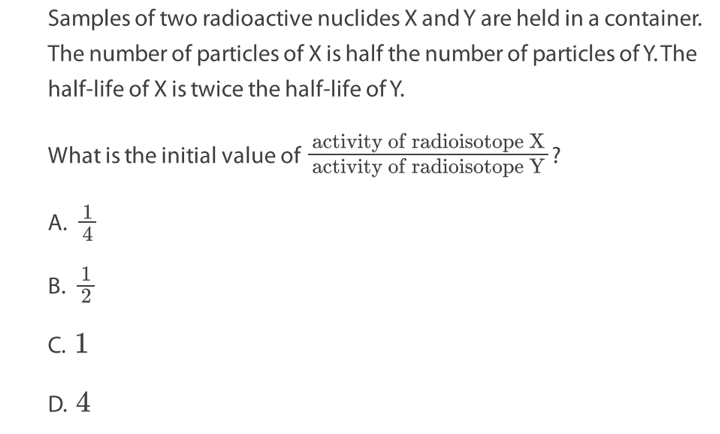


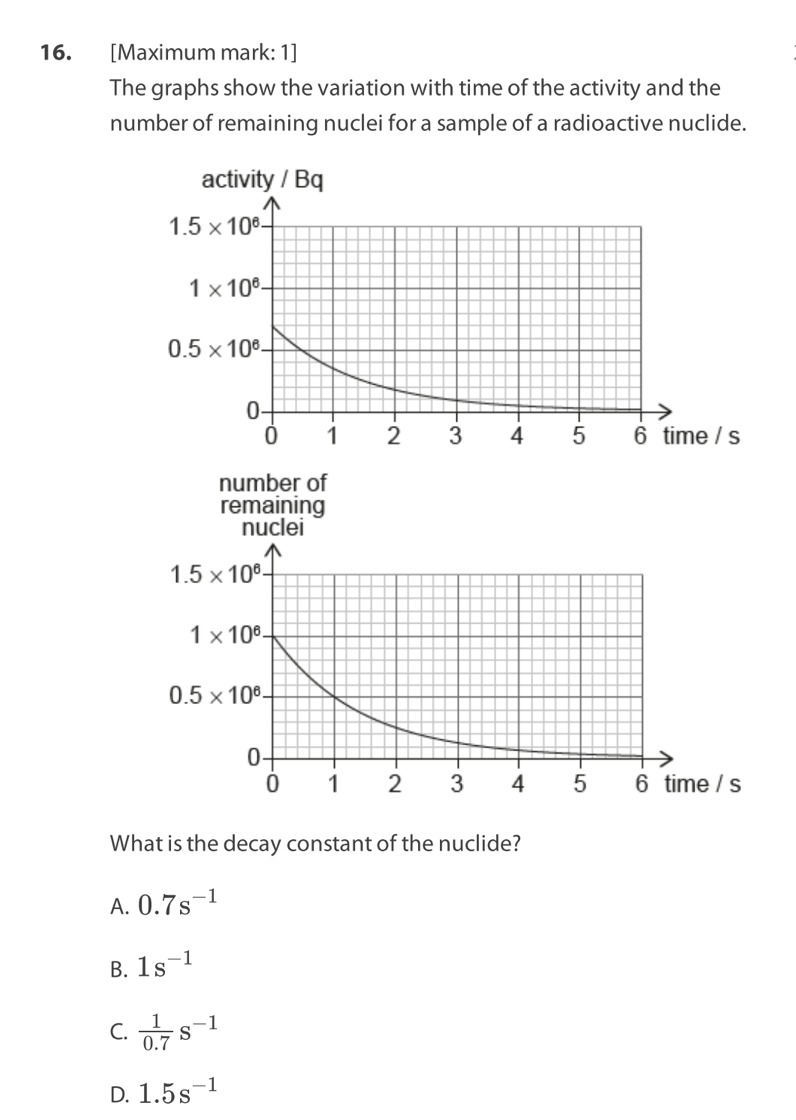

What was a reason to postulate the existence of neutrinos?
A. Nuclear energy levels had a continuousspectrum.
B.The photon emission spectrum only contained specific wavelengths.
C. Some particles were indistinguishable from their antiparticle.
D.The energy of emitted beta particles had a continuousspectrum.
D.The energy of emitted beta particles had a continuousspectrum.

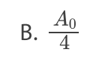

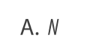

C.7100 Bq
Photons of discrete energy are emitted during gamma decay.Thisis
evidence for
A. atomic energy levels.
B. nuclear energy levels.
C. pair annihilation.
D. quantum tunneling.
B. nuclear energy levels.
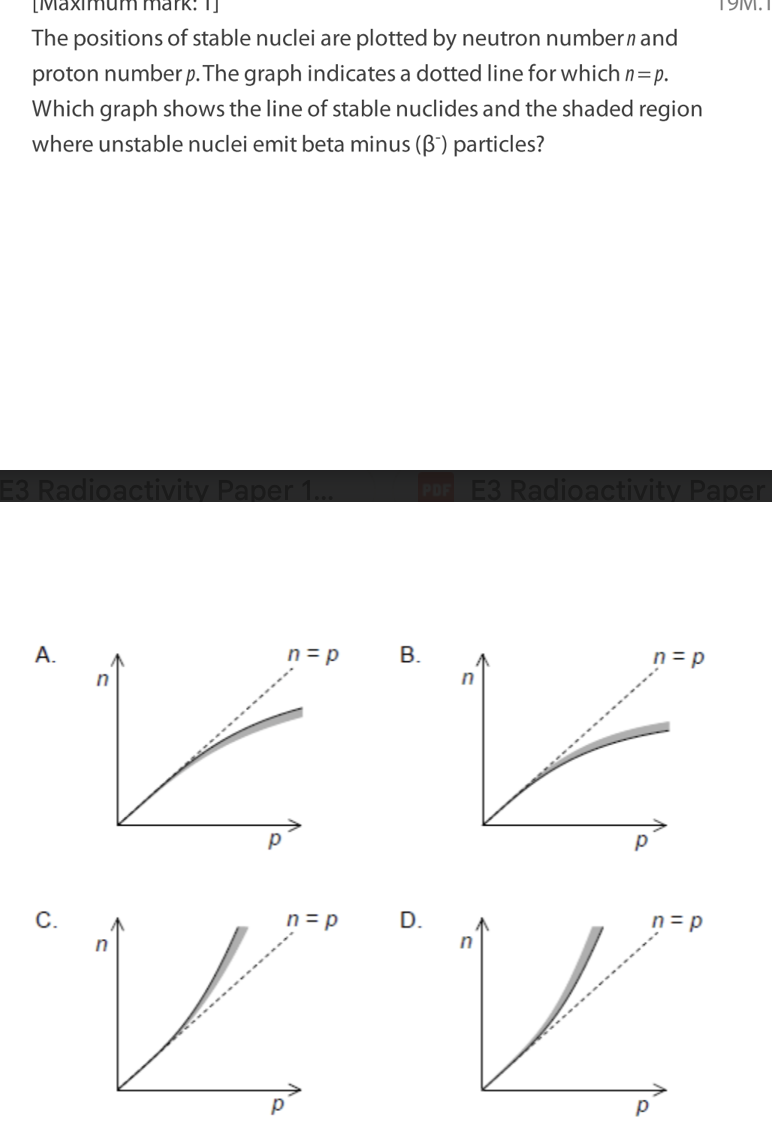
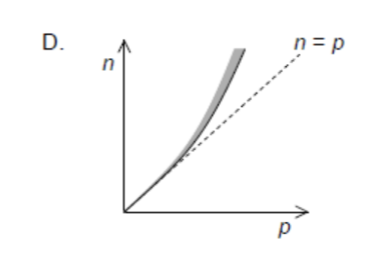

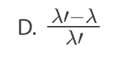

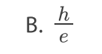
Which statement is correct about Compton scattering of a photon by an
electron?
A. The energy of the photon decreases.
B. The wavelength of the photon decreases.
C. The momentum of the photon is unchanged.
D. The combined momentum of the particlesincreases.
A. The energy of the photon decreases.



C. 2
An electron is accelerated from rest through a potential difference of 3.8
kV.
The de Broglie wavelength of the electron after acceleration is
A. 0.021 mm
B. 0.021 μm
C. 0.021 nm
D. 0.021 pm
C. 0.021 nm
The Compton effect can be explained using
A. conservation of momentum
B. kinematic equations applied in two dimensions
C. the concept of a photon
D. the wave theory of light
A. conservation of momentum
A beam of X-rays of wavelength 100.00 pm isscattered from a block of
carbon. Radiation is observed at right anglesto the incident beam.
What isthe Compton shift for the observed radiation?
A. 0.1024 pm
B. 2.4322 pm
C. 2.4322 nm
D. 0.1024 nm
B. 2.4322 pm
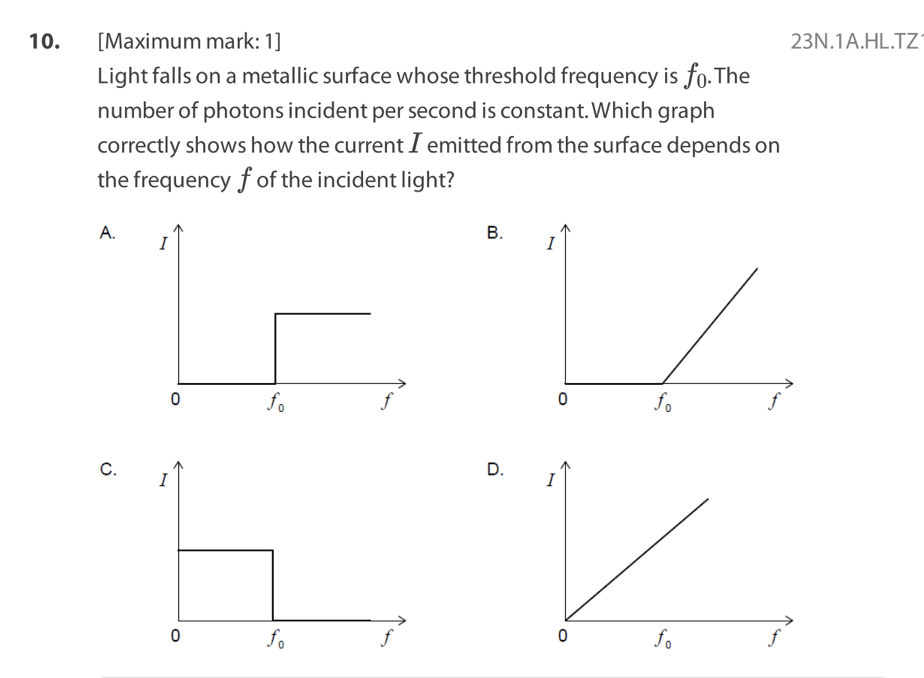
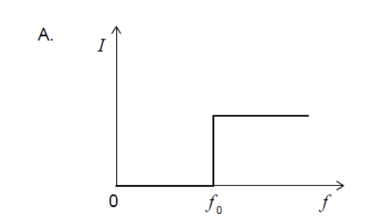
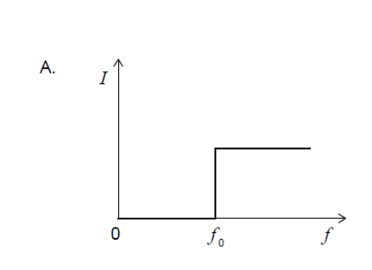
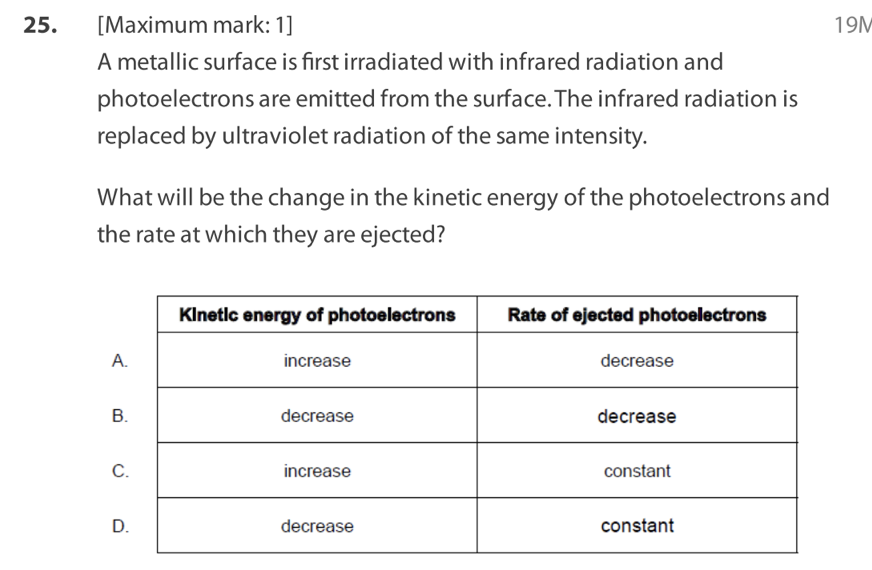
Light of intensity I and frequency , which is above the threshold
frequency, is directed at a polished metal surface.The following three
claims are made about electrons ejected from the surface.
I. The ejection is almost instantaneous.
II. The number ejected is proportional to I.
III. Their kinetic energy is equal to Planck’s constant multiplied by .
Which statements are correct?
A. I and II only
B. I and III only
C. II and III only
D. I, II and III
A. I and II only


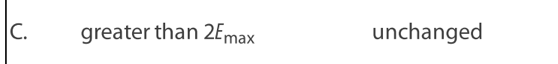

C. 3K
What is evidence for wave–particle duality?
A. Line spectra of elements
B. Electron-diffraction experiments
C. Rutherford alpha-scattering experiments
D. Gamma-ray spectra
B. Electron-diffraction experiments
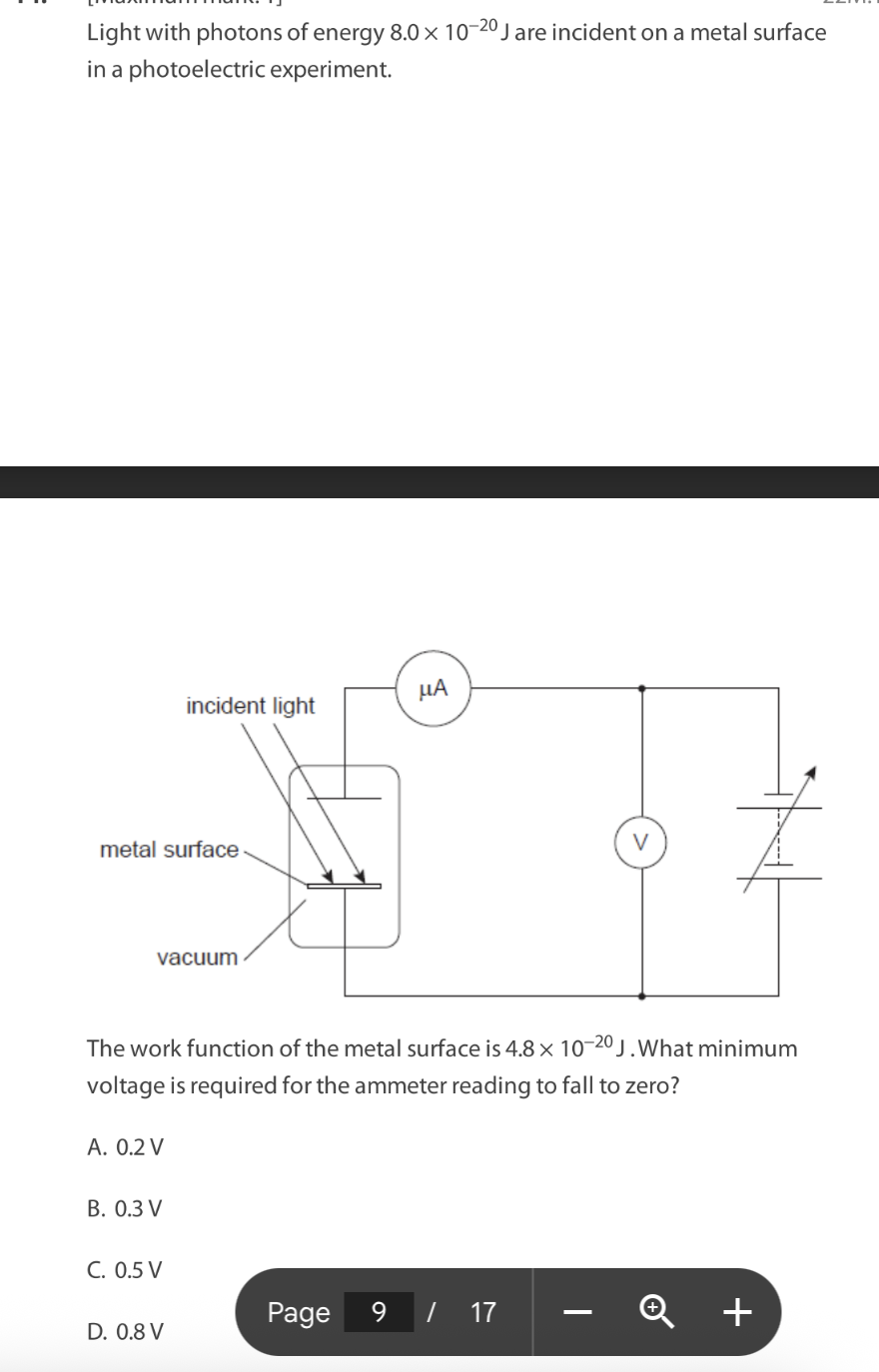

A. I and II only
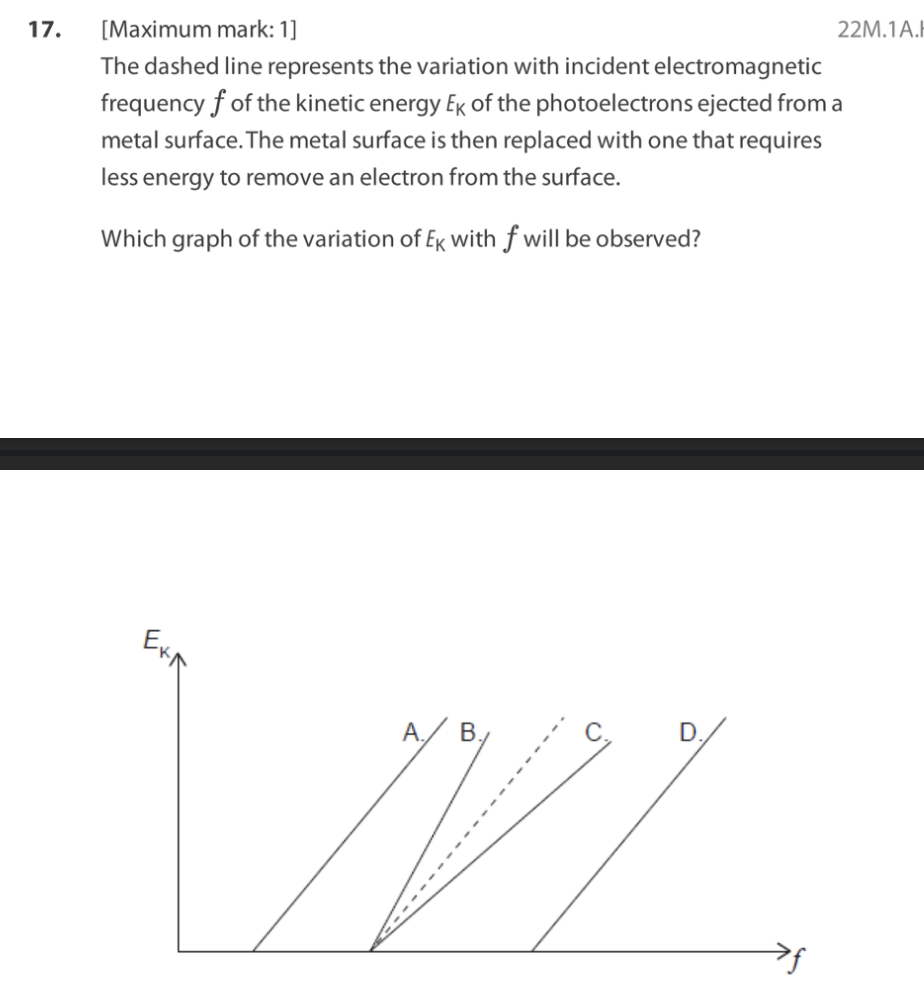
A
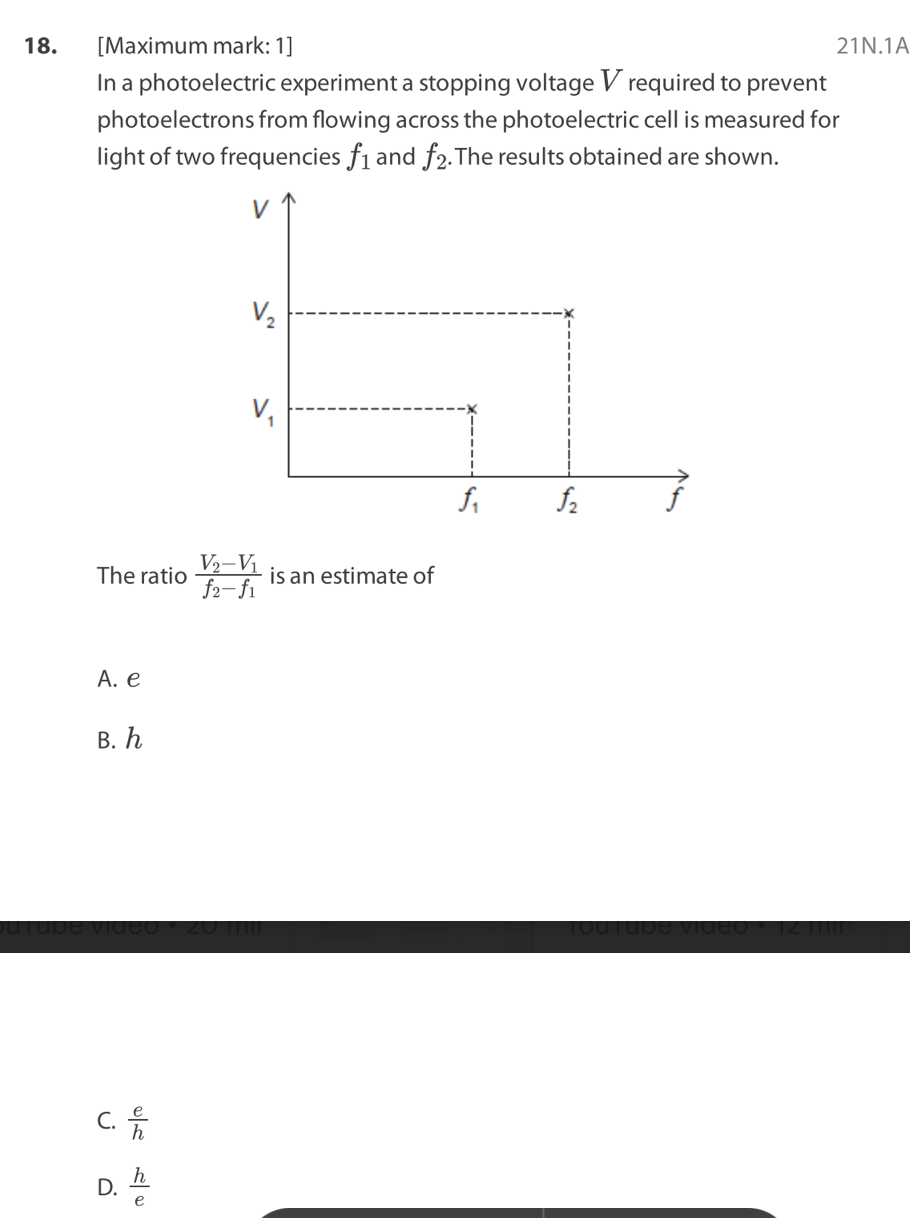
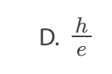

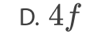

C. II and III only

A. Angular momentum of electrons is quantized


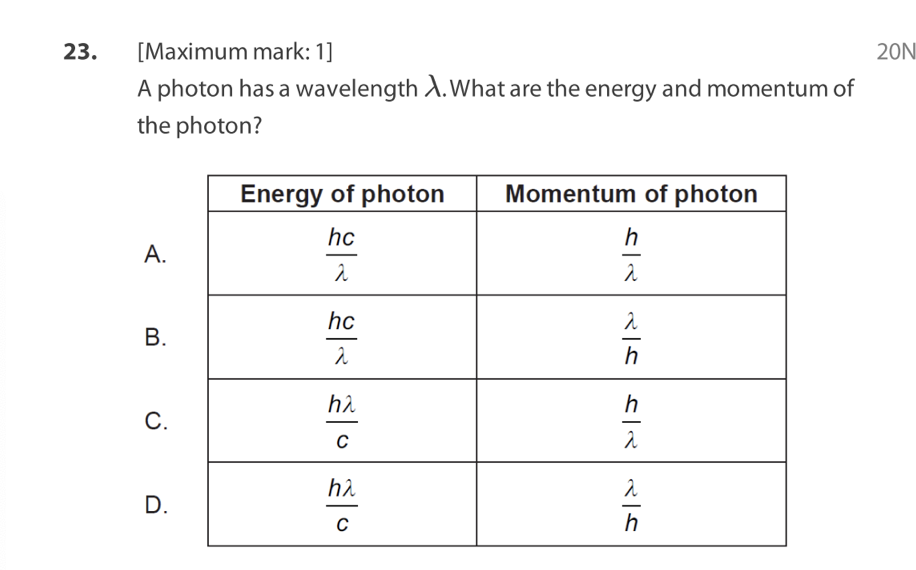
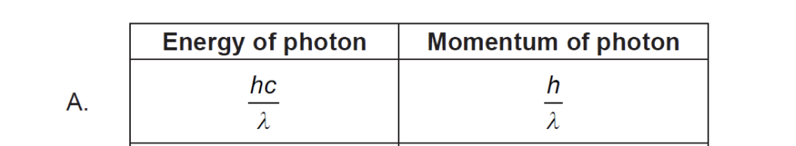


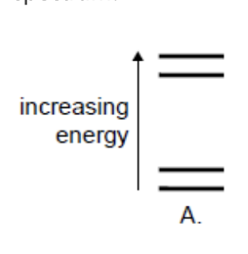

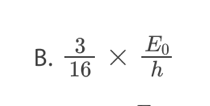

B. The size of the nucleus
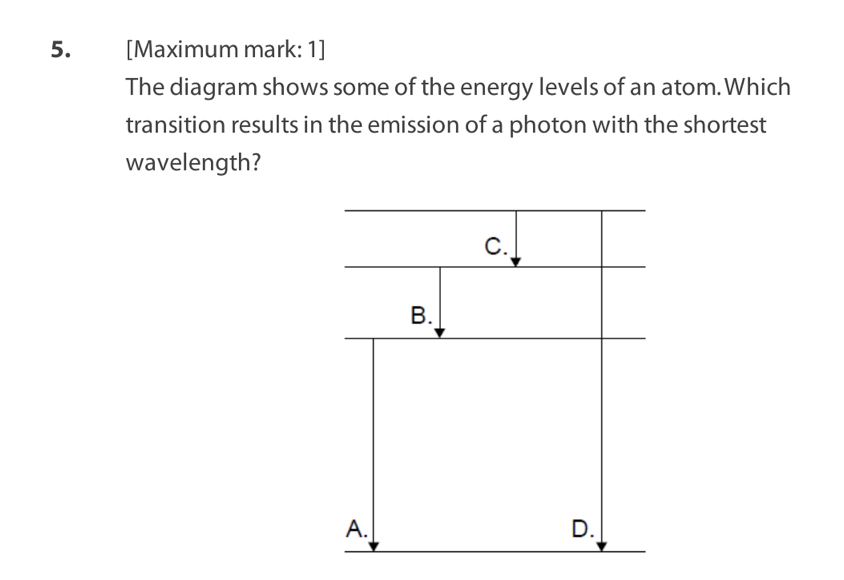
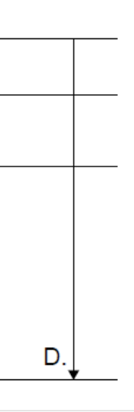

The results obtained from Geiger-Marsden-Rutherford scattering
experiments provide evidence for the existence of the nucleusin
atoms. Deviationsfrom Geiger-Marsden-Rutherford scattering...
I. provide evidence for the existence of the strong nuclear force.
II. allow for an estimation of the radius of the nucleus.
III. occur when alpha particles are incident at low energies.
Which statements are correct?
A. I and II only
B. I and III only
C. II and III only
D. I, II and III
A. I and II only

C. 2
The results obtained from Geiger-Marsden-Rutherford scattering
experiments provide evidence for the existence of the nucleusin
atoms. Deviationsfrom Geiger-Marsden-Rutherford scattering...
I. provide evidence for the existence of the strong nuclear force.
II. allow for an estimation of the radius of the nucleus.
III. occur when alpha particles are incident at low energies.
Which statements are correct?
A. I and II only
B. I and III only
C. II and III only
D. I, II and III
A. I and II only

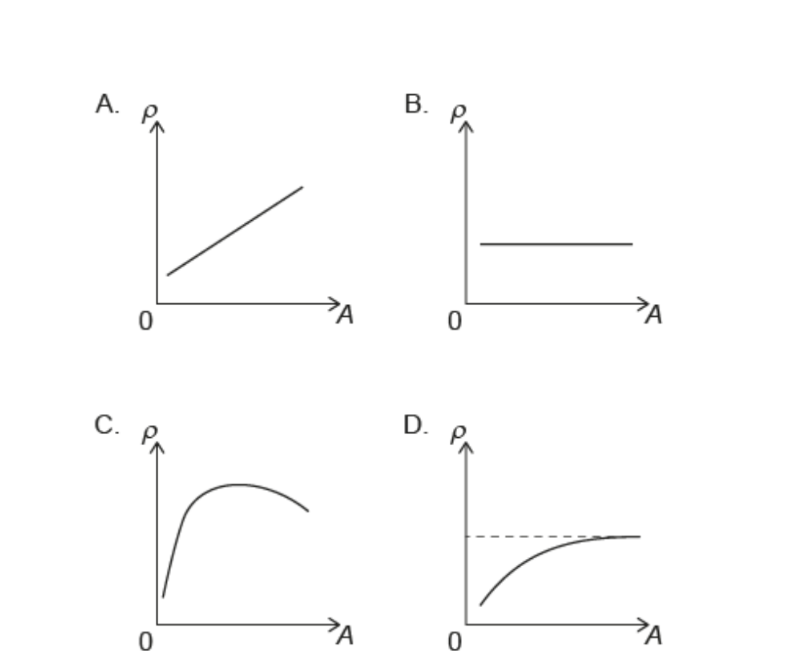
What isthe variation of nuclear density ρ with nucleon number A?

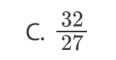


Which emission shows a continuous energy spectrum?
A. Photons during energy transitions between atomic energy states
B. Gamma photonsfrom the nuclei of radioactive isotopes
C. Beta particlesfrom the nuclei of radioactive isotopes
D. Alpha particlesfrom the nuclei of radioactive isotopes
C. Beta particles from the nuclei of radioactive isotopes



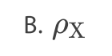

C. 125
Three possible features of an atomic model are
I. orbital radius
II. quantized energy
III. quantized angular momentum.
Which of these are features of the Bohr model for hydrogen?
A. I and II only
B. I and III only
C. II and III only
D. I, II, and III
D. I, II, and III
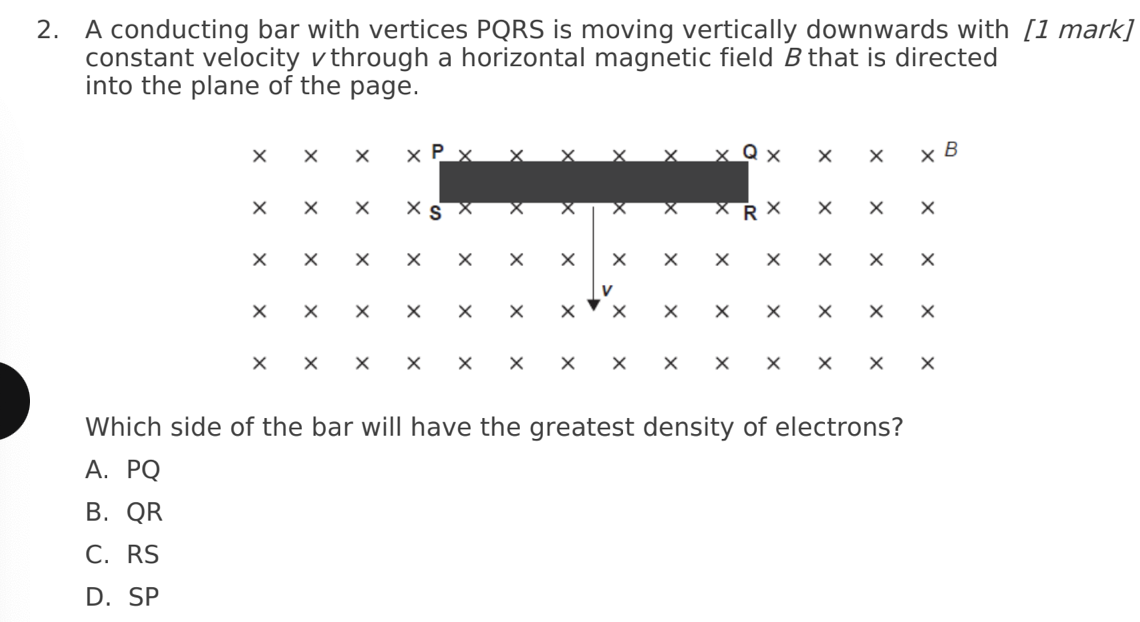
D. SP
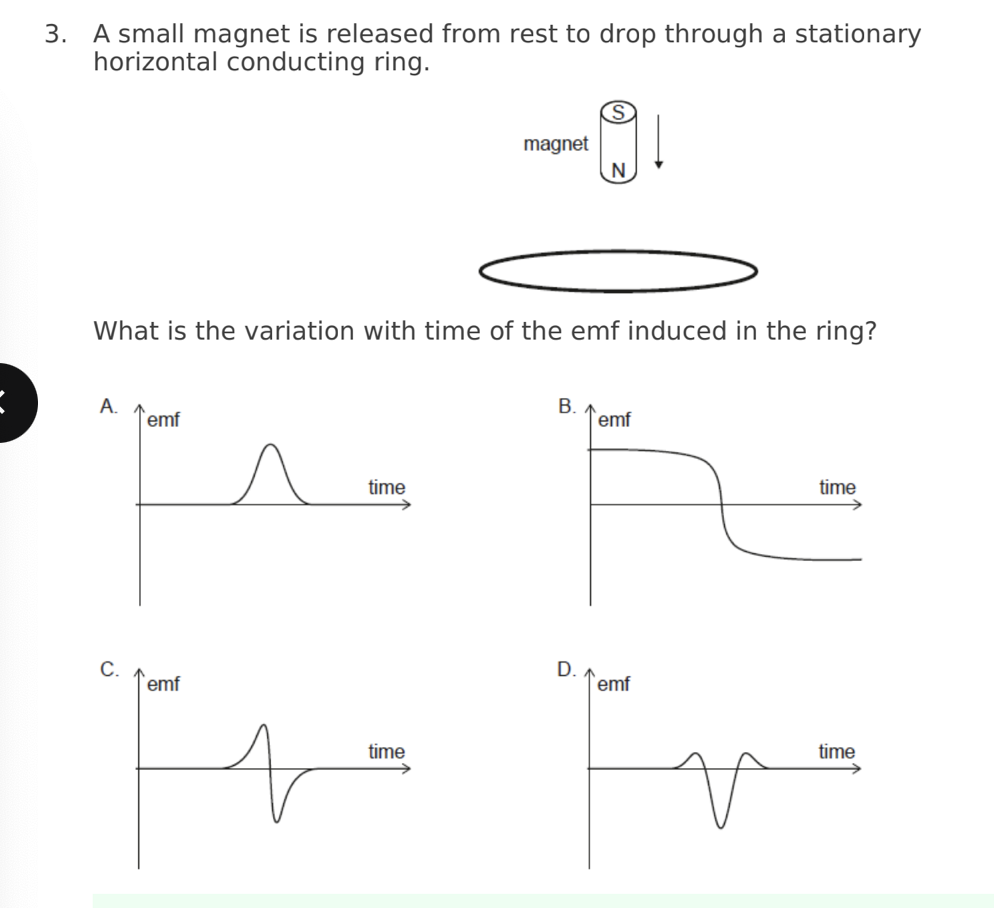
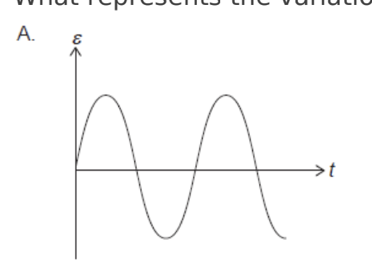
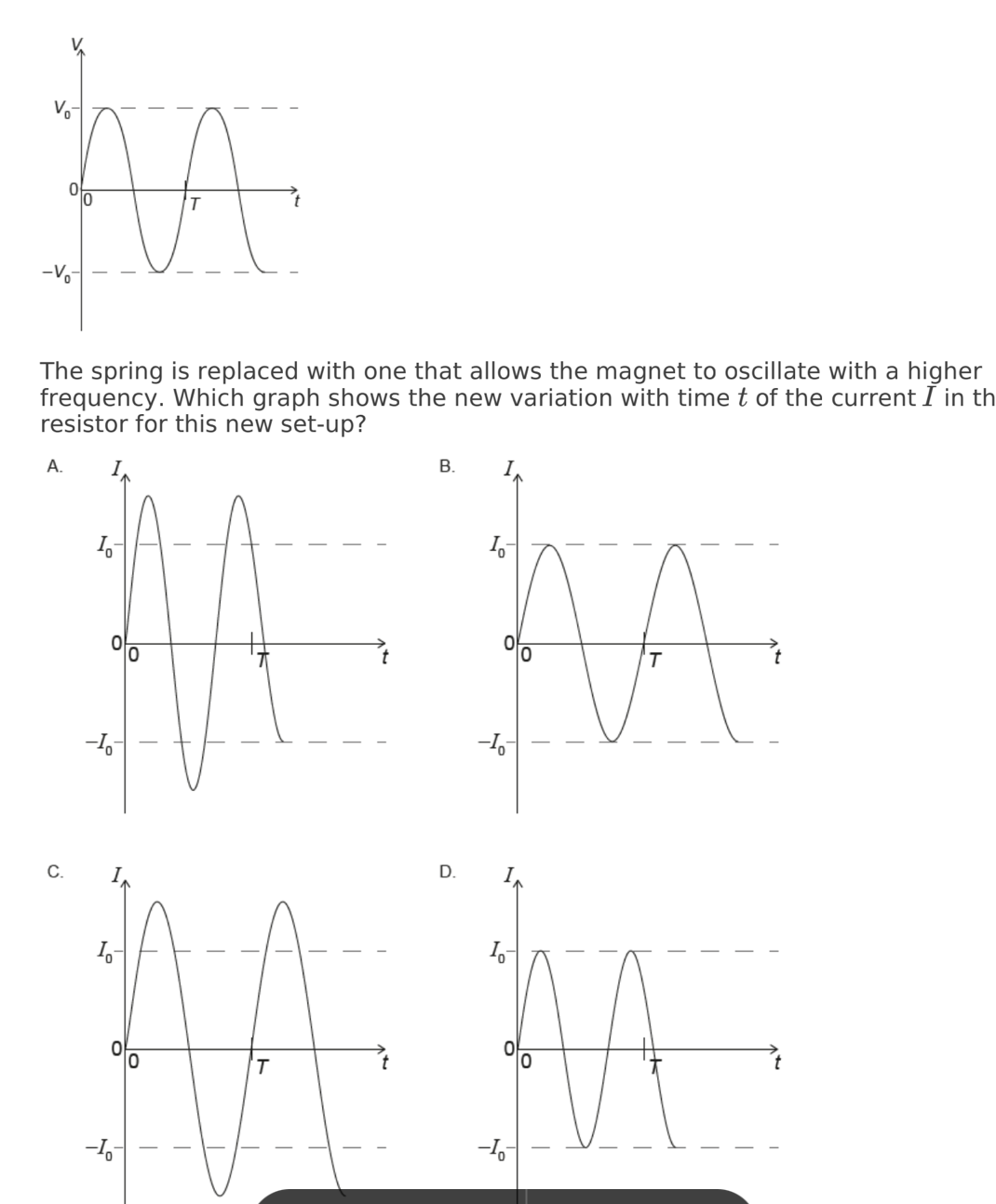
A conducting ring encloses an area of 2.0 cm and is perpendicular to a
magnetic field of strength 5.0 mT. The direction of the magnetic field is
reversed in a time 4.0 s. What is the average emf induced in the ring?
A. 0
B. 0.25 μV
C. 0.40 μV
D. 0.50 μV
D. 0.50 μV
The conservation of which quantity explains Lenz’s law?
A. Charge
B. Energy
C. Magnetic field
D. Mass
B. Energy
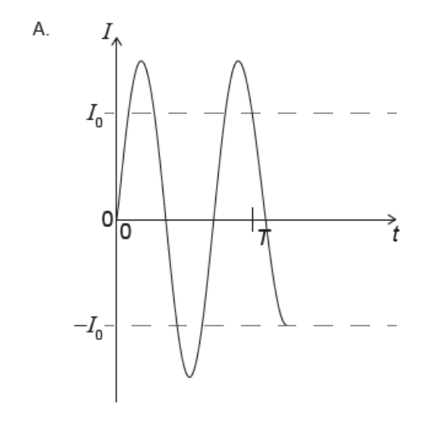
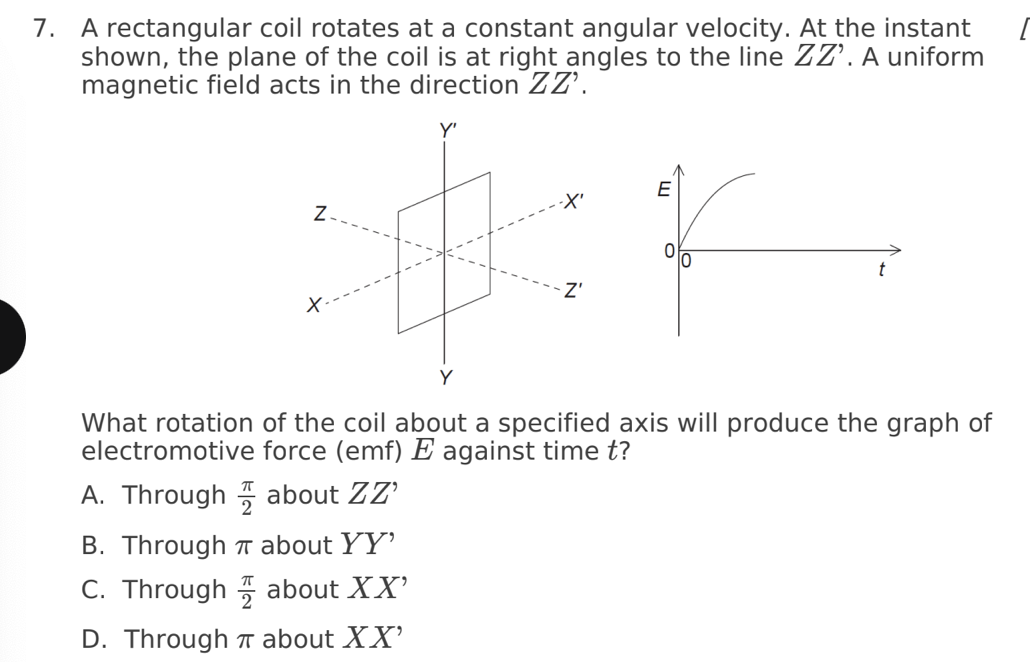
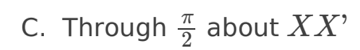
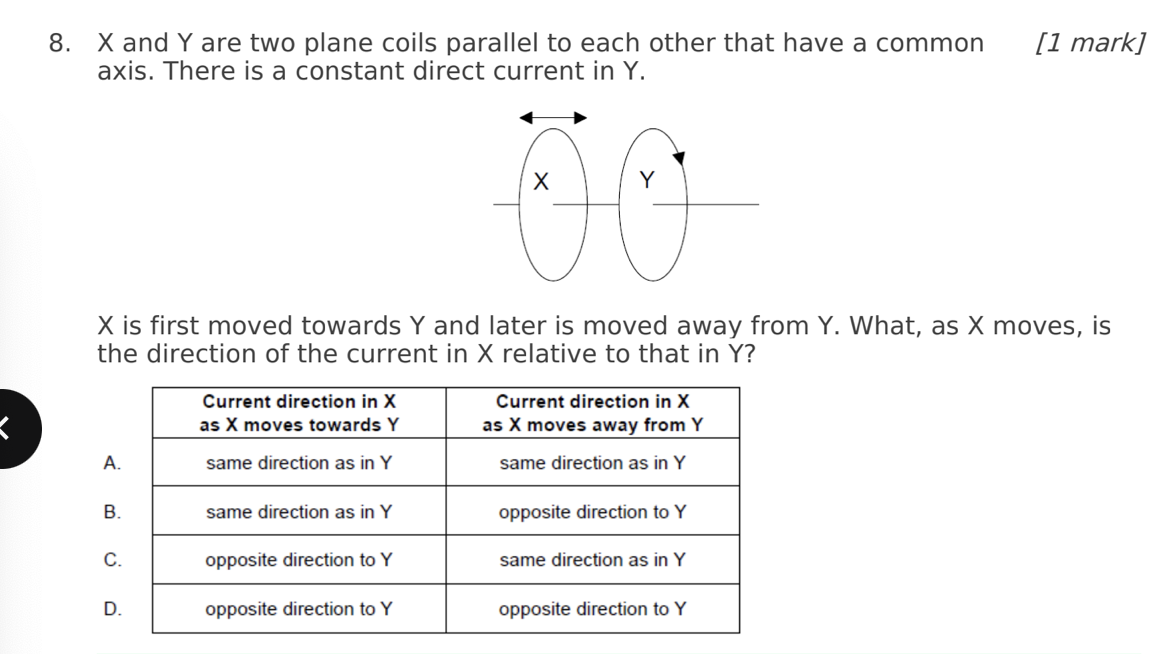

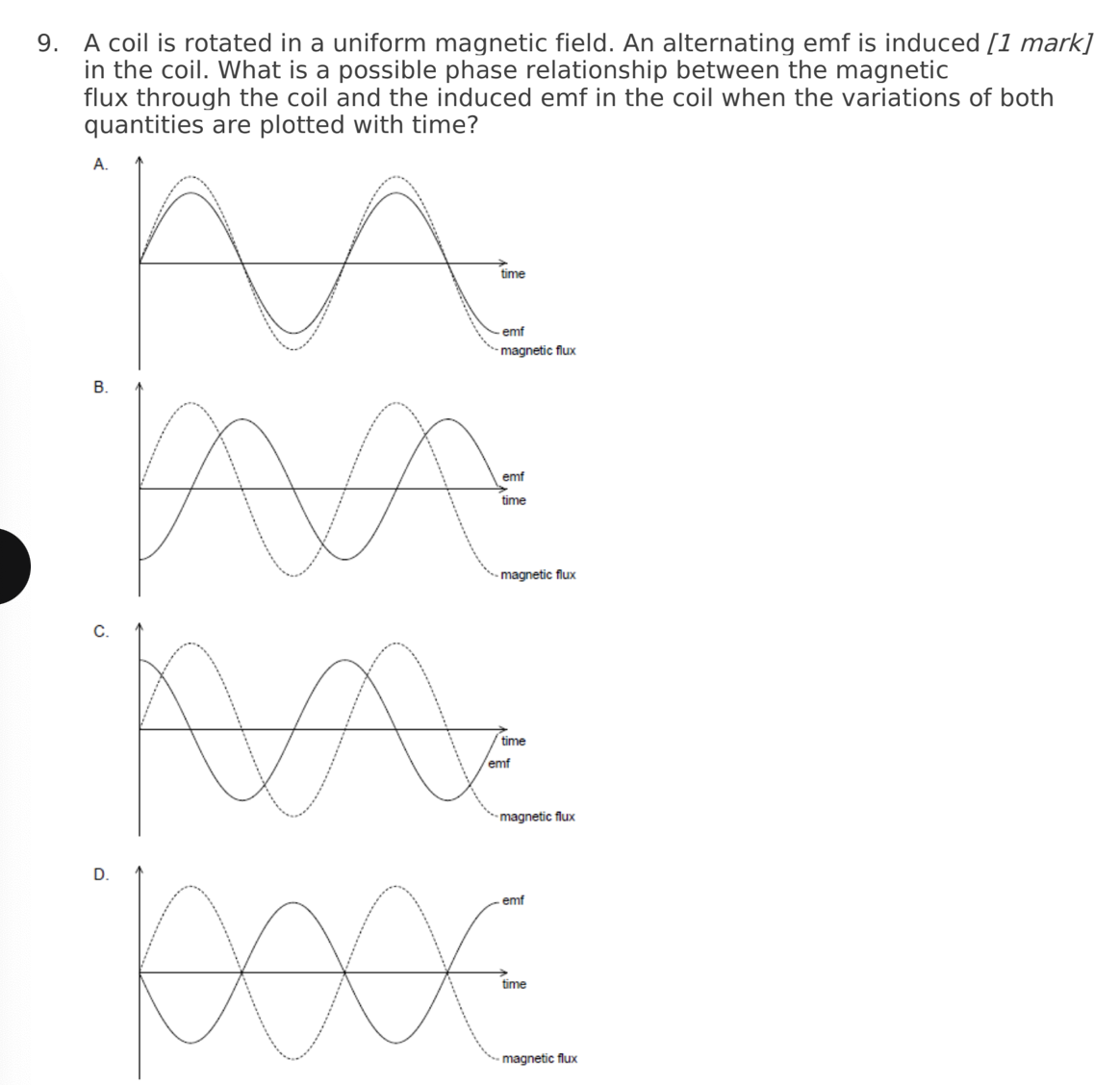
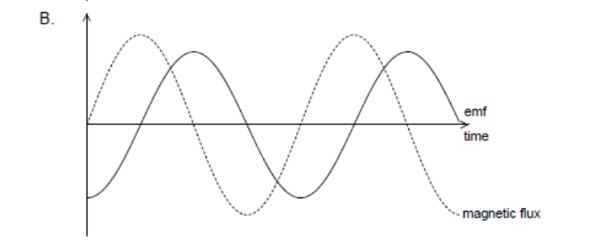
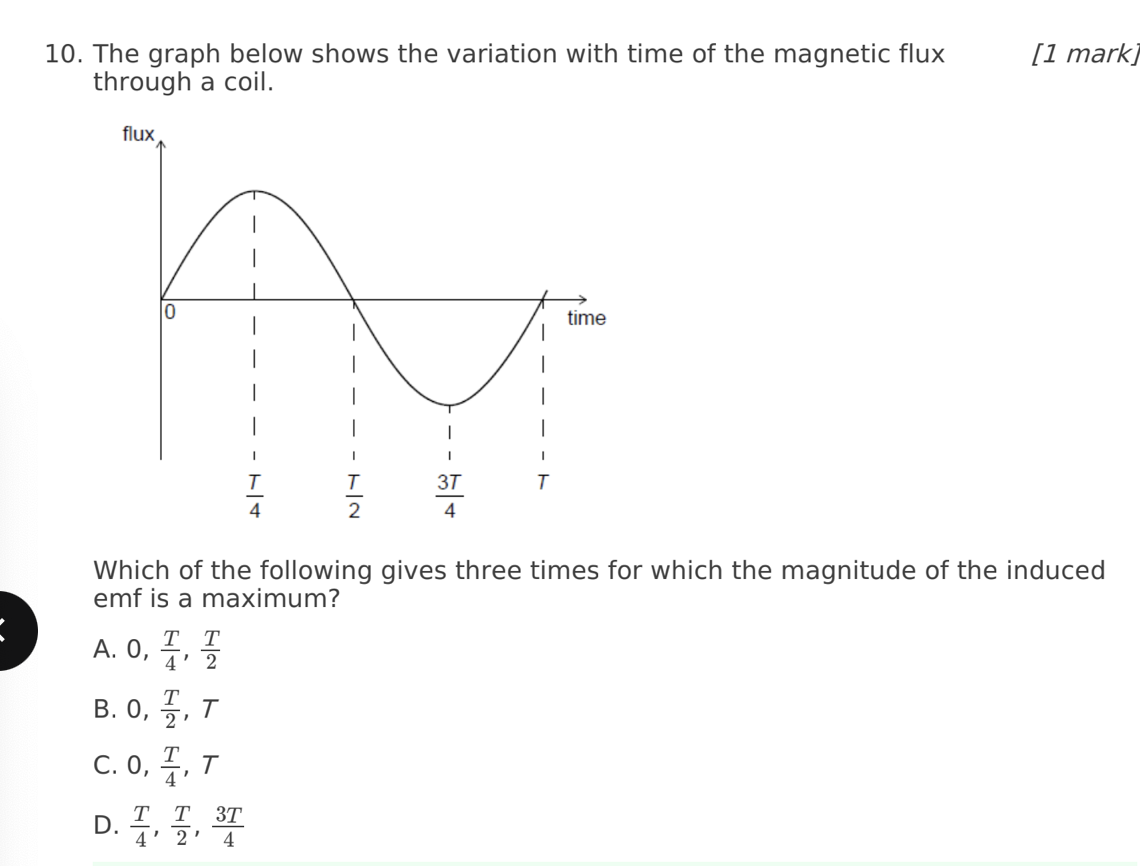
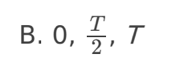
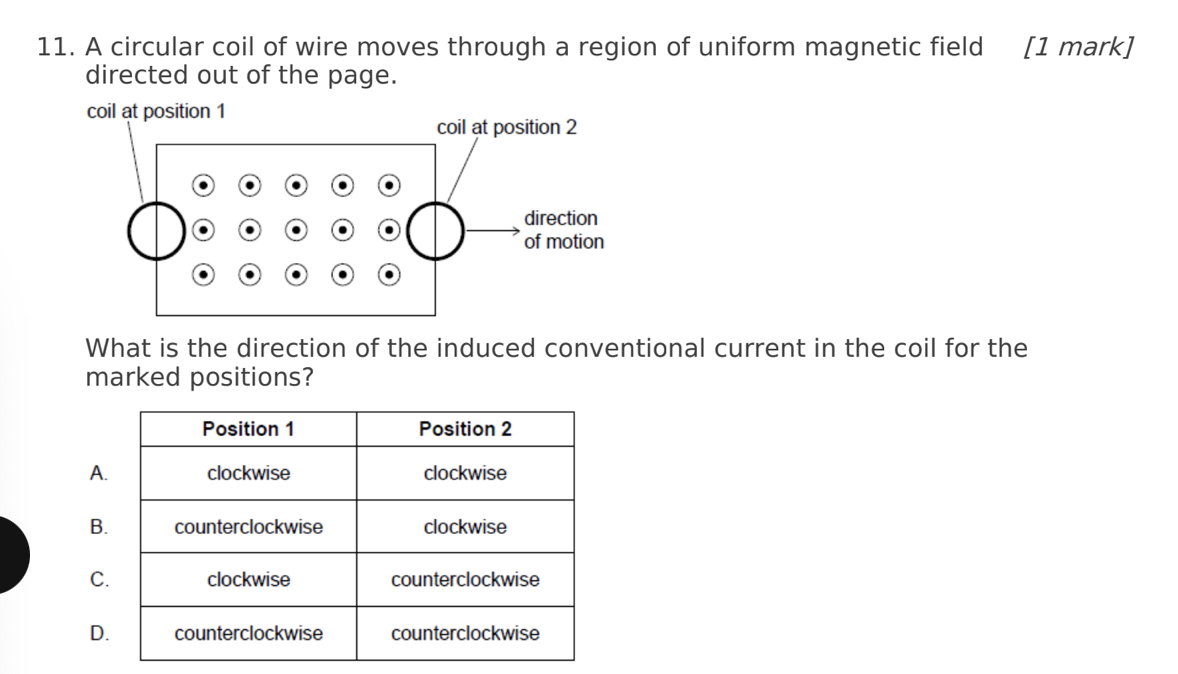

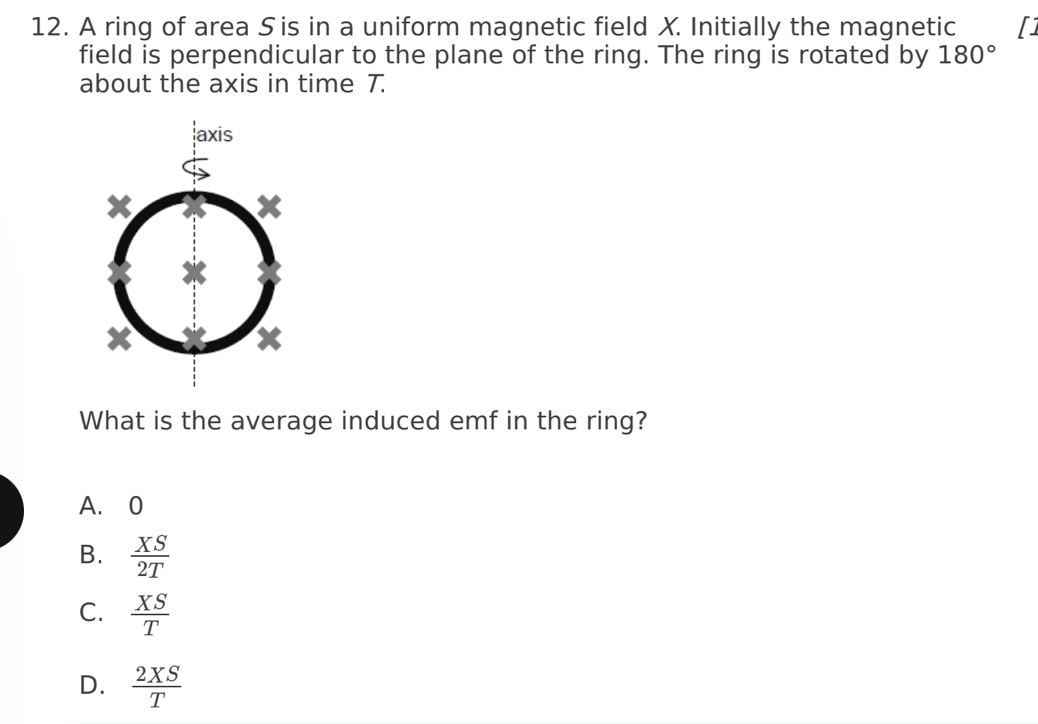
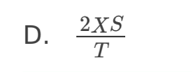
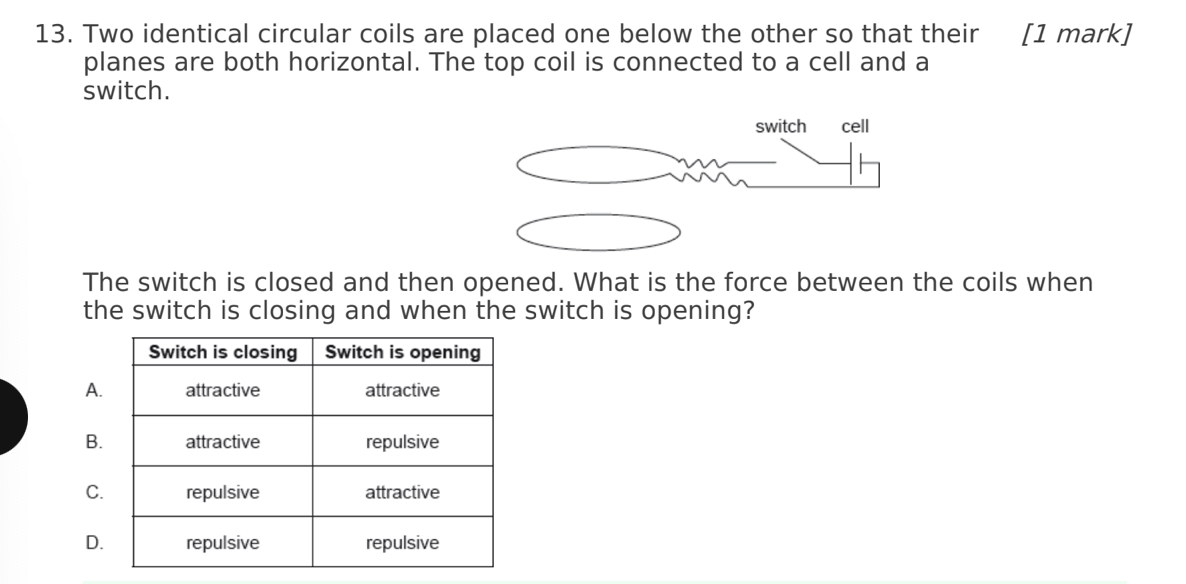

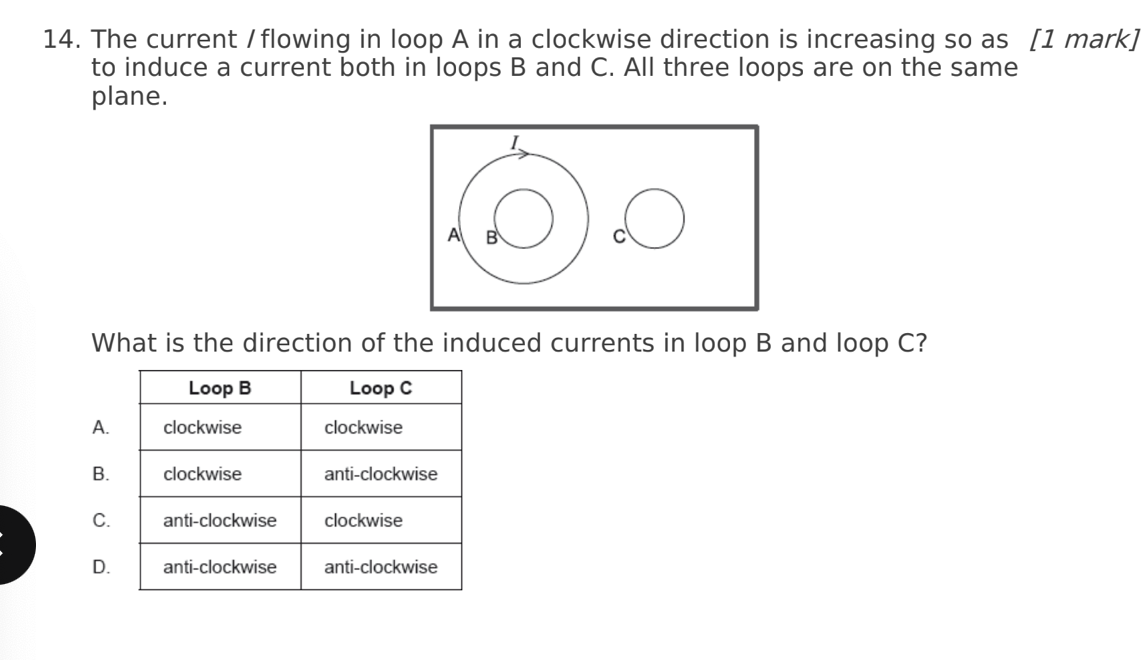

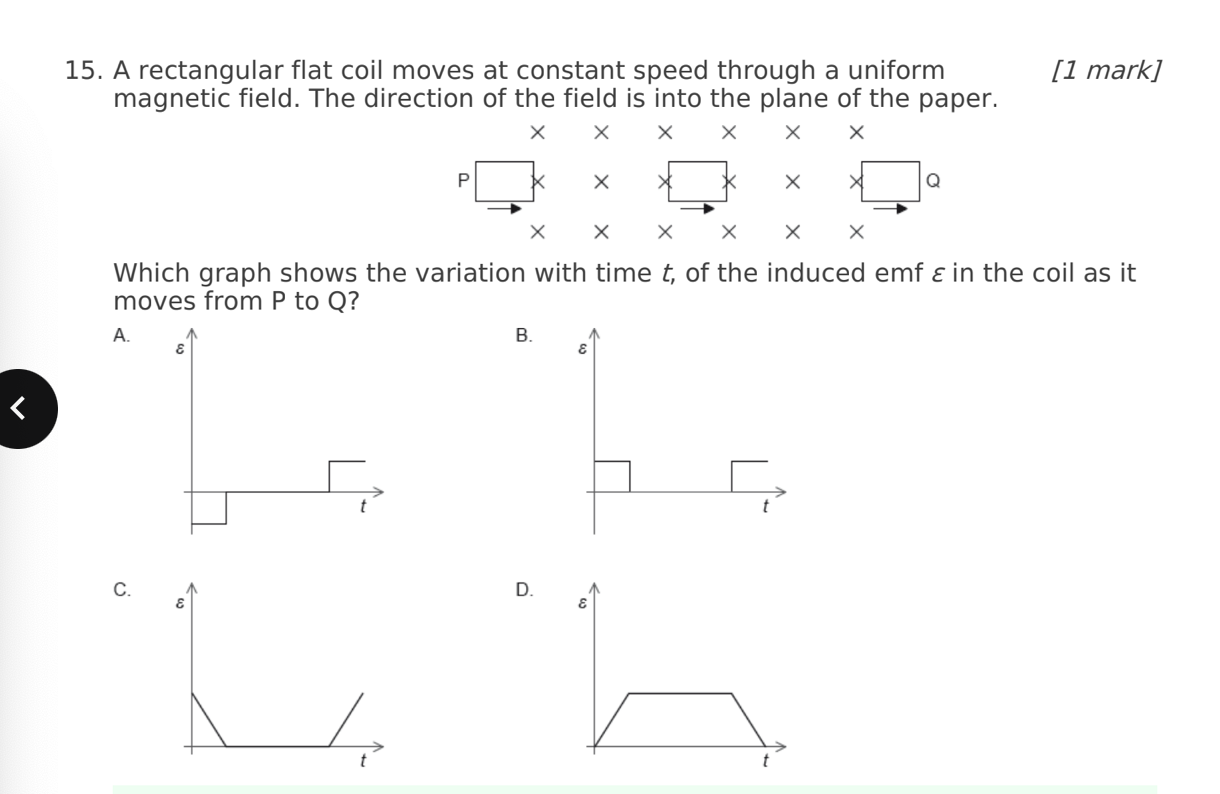
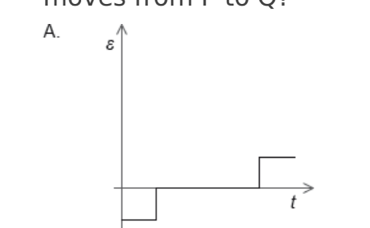

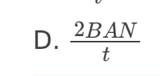
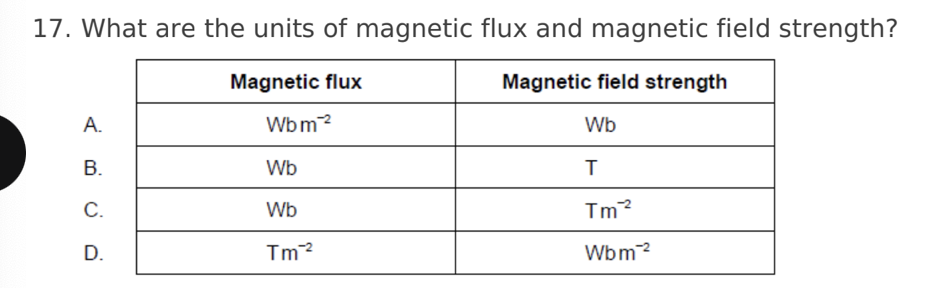

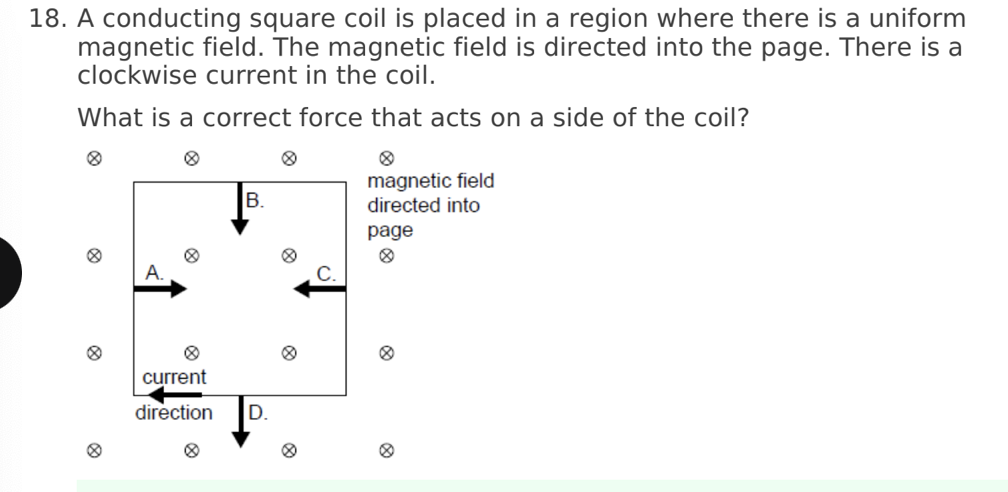
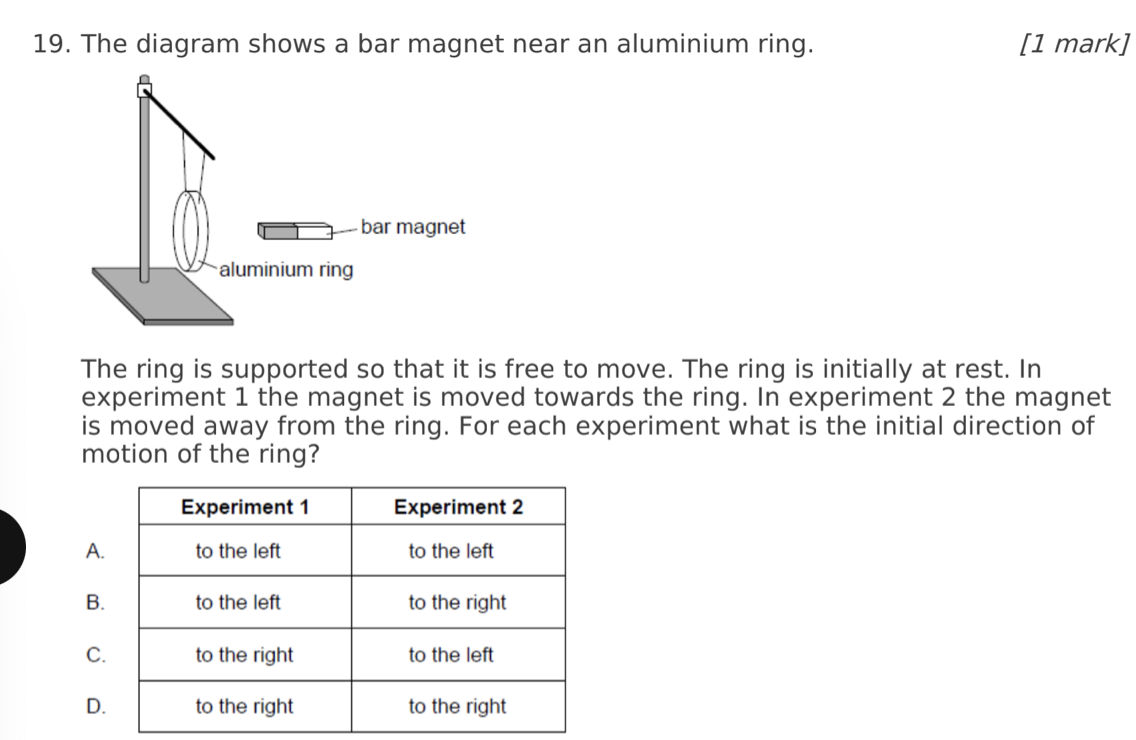

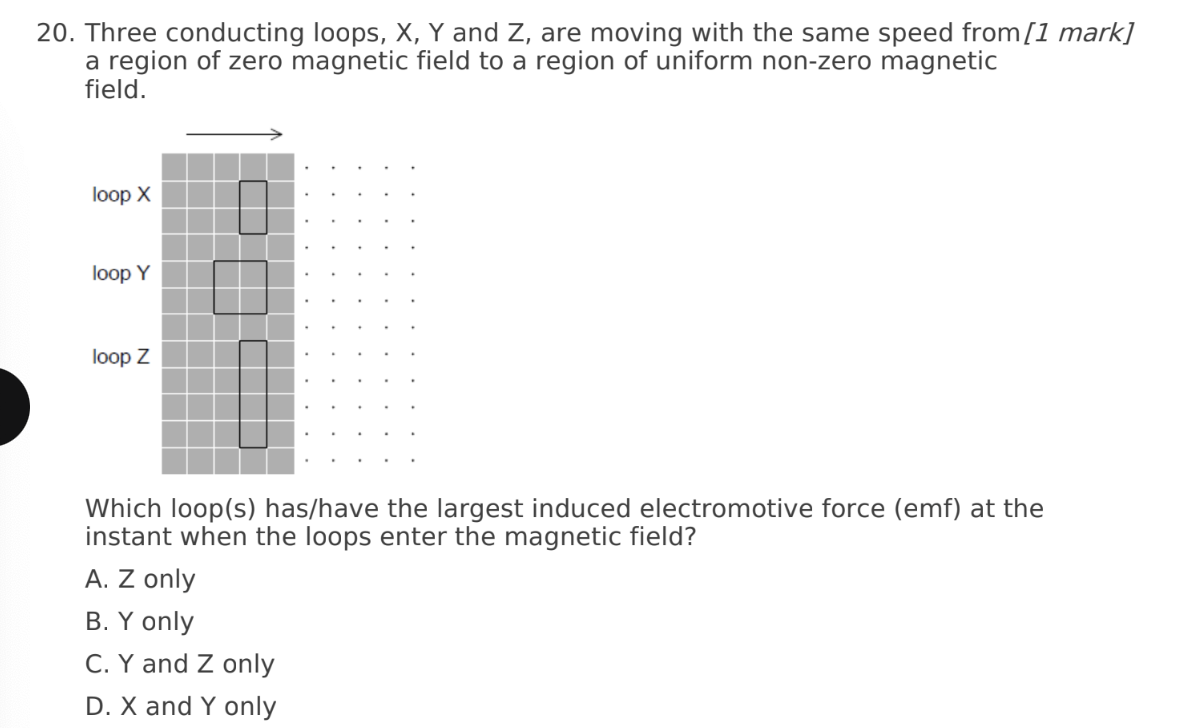
A. Z only

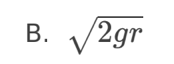

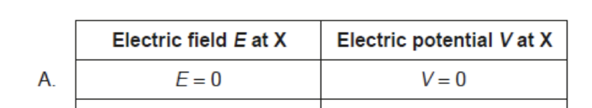
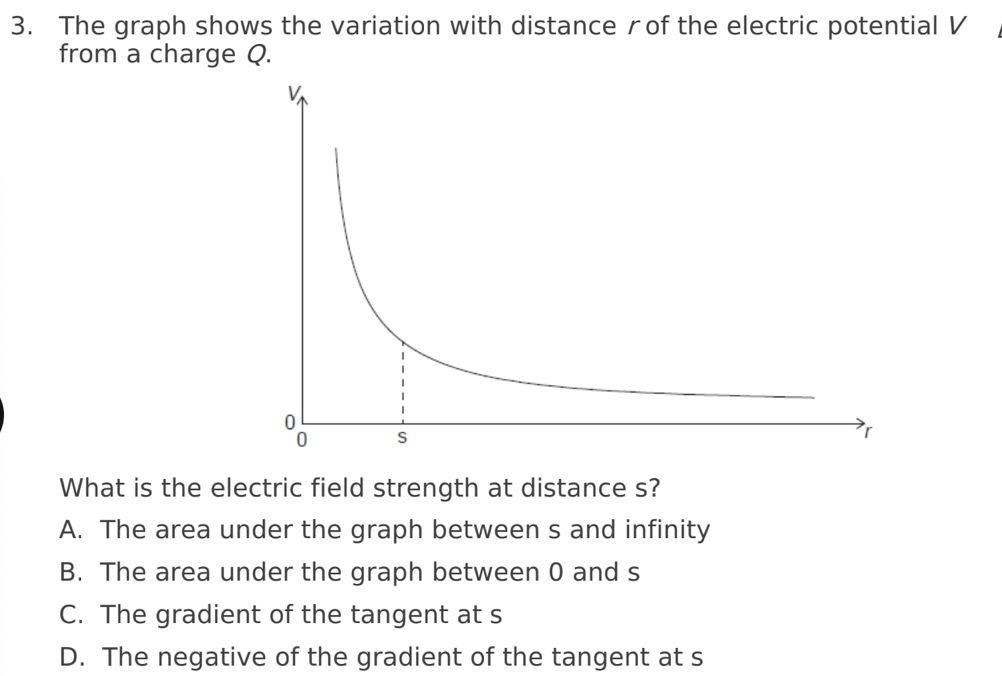
D. The negative of the gradient of the tangent at s

D. 20 μJ

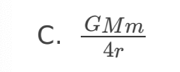

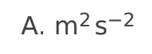
A planet has radius R. The escape speed from the surface of the planet is
v. At what distance from the surface of the planet is the orbital speed
0.5v?
A. 0.5R
B. R
C. 2R
D. 4R
B. R


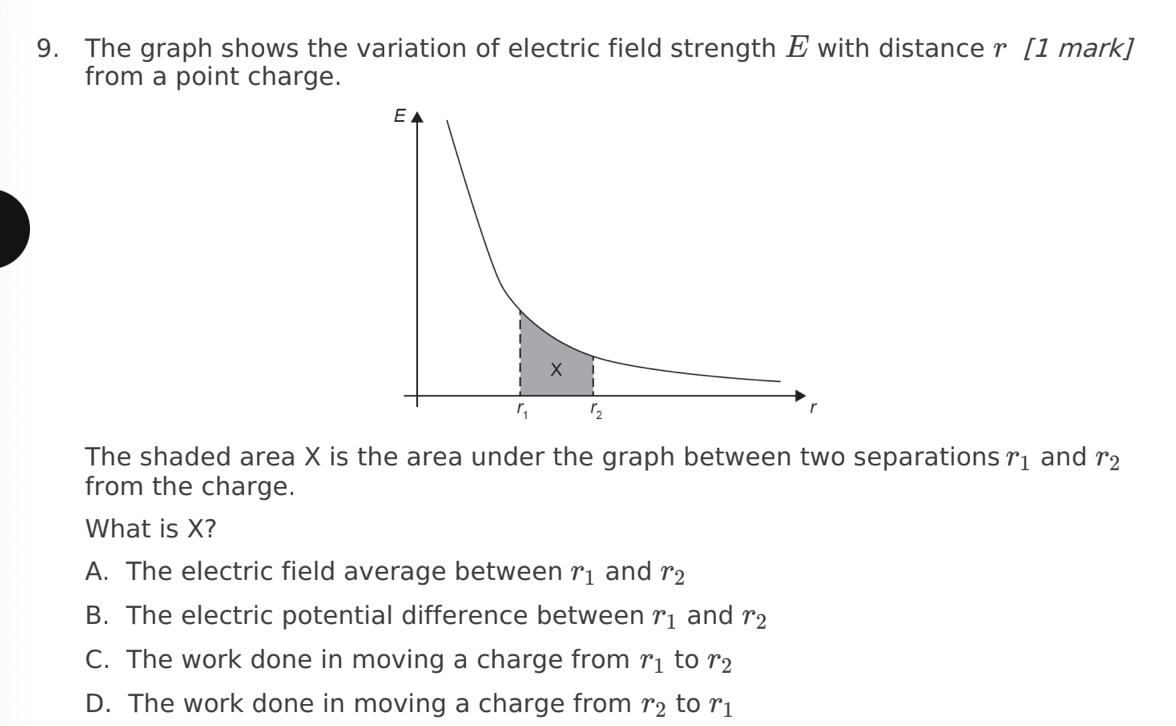


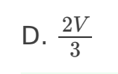

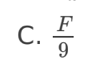
12. An electron enters a uniform electric field of strength E with a velocity v.
The direction of v is not parallel to E. What is the path of the electron after
entering the field?
A. Circular
B. Parabolic
C. Parallel to E
D. Parallel to v
B. Parabolic

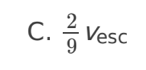





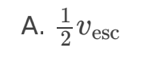
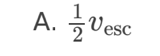
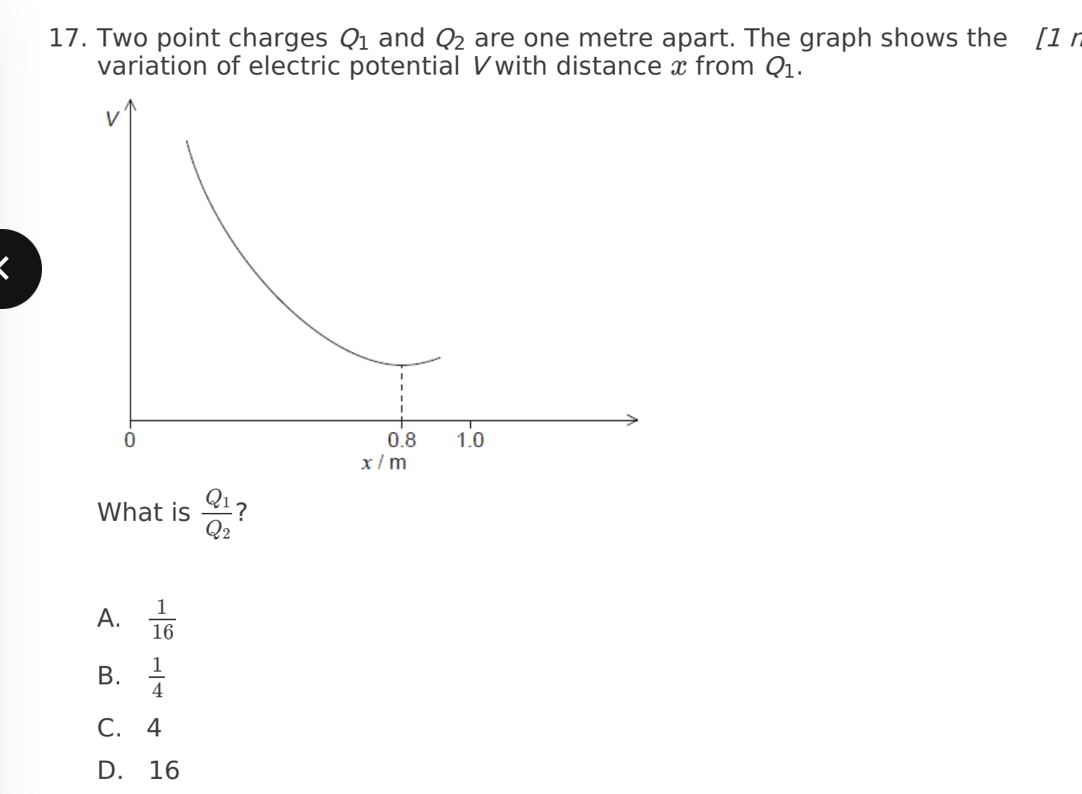
D. 16