Transition Metal
1/29
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
30 Terms
Define a d block element
An element that has a d orbital as its highest energy occupied orbital
Define a transition metal
An element that has a d orbital as its highest energy occupied orbital that has at least one ion with an incomplete d-sub shell
Why does chromium have the electronic configuration of [Ar] 3d^5 4s^1 instead of [Ar] 3d^4 4s² and coppor have [Ar] 3d^10 4s^1 instead of [Ar] 3d^9 4s²
Molecules are more stable in subshells are full or half full
Define a ligand
Species capable of donating a lone pair of electrons and forming a coordinate dative bond to a central metal ion
Define coordinate (dative) bond
Both electrons of shared pair come from the same atom
Define complex ion
Central metal ion attached to one or more ligand
Define coordination number
The number of coordinate bonds formed by a central metal ion
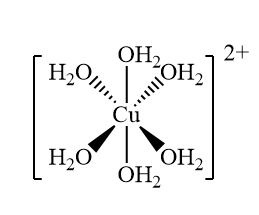
Draw the complex ion [Cu(H2O)6] 2+
Give the bond angle, coordination number and shape
90 degrees, octahedral, 6 coordination
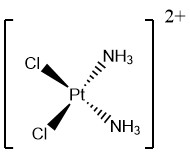
Draw the complex ion Pt(NH3)2Cl2
(Square planar)
Give the bond angle and coordination number
Pt has a charge of 2+
90 degrees, coordination of 4
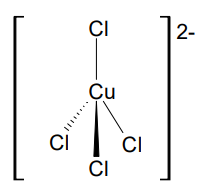
Draw the complex ion [CoCl4]2-
(Tetrahedral)
Give the bond angle and coordination number
109.5 Tetrahedral, 4 coordination
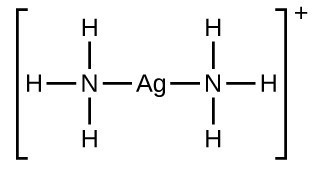
Draw the complex ion [Ag(NH3)2]+
Give the bond angle, coordination number and shape
Linear, 180, coordination 2
What is a ligand substitution reaction
One ligand is replaced by another (often with a colour change)
Define didentate ligands and give an example
Donate 2 lone pairs of electrons to a metal ion
Form 2 coordinate binds with a central metal ion each
Example - C2O4²-
Define a hexadentate ligand
Donates 6 lone pairs of electrons to a metal ion
Forms 6 coordinate bonds with central metal ion
What are the properties of transition metals (oxidation states, in solutions)
Have variable oxidation states because the electrons that sit in 4s and 3d have very close energy levels so electrons are gained and lost using a similar amount of energy
Form coloured ions in solution
Form complex ions
What is the colour of Fe2+ and Fe3+ ions in solution
Fe2+ - Light green
Fe3+ - Orange
What is the colour of Cu+ and Cu2 in solutions
Cu+ - Colourless
Cu2+ - Blue
What happens to Cu+ ions in solution and why
Cu+ has a full 3d subshell so forms colourless compounds.
It is unstable and disproportionates in solutions
Cu2O(s) + H2SO4(aq) → Cu(s) + CuSO4(aq) + H2O
Cu2O(s) (+1)
Cu(s) (0) Brown solid
CuSO4(aq) (+2) Blue sol.
Give examples of small ligands (can fit 6 around a central metal ion)
H2O
NH3
CN-
How many Cl- ligands can fit around one central metal ion
4
Why are transition metals coloured and why do different complexes have different colours
3d orbital energy levels are split by ligands
Absorption of energy in visible light causes electrons to move to higher energy levels
Frequency absorbed is linked to the energy gap between the d energy levels by E=hv
Size of energy gap is affected by the ligands, the central metal ion and its oxidation state
Colour seen is that of wavelengths transmitted not absorbed - complementary colour
Different complexes have different colours due to different number of d electrons and different splitting of the d orbitals. These lead to different wavelengths being absorbed and different complementary colours being seen
How can transition metals act as heterogenous catalysts
Available 3d and 4s electrons are used to form weak bonds with reactant molecules, so reactants adsorb on to the solid metal surface
Bonds within reactants weaken allowing a faster reaction as activation energy is lowered
Weak dative bonds break and products desorb off catalyst surface
How can transition metals act as homogenous catalysts
Variable oxidation states of transition metal allow an alternative route with a lower activation energy
Ions can act as oxidising and reducing agents in the reaction, being regenerated
Show the equations and expected colour changes from the reaction of Fe2+ and Fe3+ with alkali (NaOH or NH3)
Fe2+(aq) + 2OH-(aq) → Fe(OH)2(s)
Pale green sol → Green ppt
Fe3+(aq) + 3OH-(aq) → Fe(OH)3(s)
Orang sol → Brown ppt
How can the amount of Fe3+ be quantified
First reduce Fe3+ to Fe2+ with Zn / H2SO4.
Filter to remove excess Zn
Titrate resulting Fe2+ using acidified potassium mangantate (VII)
Write the equation and expected colour change for the reaction of Cu2+ with NaOH
Cu2+ (aq) + 2OH (aq) → Cu(OH)2 (s) (Precipitation)
Blue sol → Blue ppt
Cu2+ forms a blue complex in water. Write the equation and expected colour change for the reaction of the Cu2+ complex with excess ammonia
[Cu(H2O)6]2+(aq) → Cu(OH)2(s) → [Cu(NH3)4(H2O)]2+ (Precipitation + ligand sub.)
Blue sol → Blue ppt → Deep blue sol.
Overall - [Cu(H2O)6]2+(aq) + 4NH3(aq) ⇌ [Cu(NH3)4(H2O)2]2+(aq) + 4H2O(l)
Blue sol → Deep blue sol.
How can you determine the percentage of Copper in brass
Oxidise a known mass of brass using a strong oxidising agent e.g. conc HNO3
All metals present will be oxidised to form metal nitrates
Neutralise with excess Na2CO3
Add excess potassium iodine solution
2Cu2+(aq) + 4I-(aq) → CuI(s) + I2(aq)
Titrate the I2 produced sodium thiosulphate solution of known concentration
Add starch indicator near end point (
Explain how a calibration curve can be constructed and used to find an unknown concentration
Dilute solution X by known amounts to produce a series of solutions of known concentration
Choose filter of the complementary colour to the solution
Zero the colorimeter using the same solvent in the cuvette
Measure absorbance of different solutions
Plot a graph of absorbance (y) against concentration (x)
Add a line of best fit
Measure absorbance of unknown concentration
Use the calibration curve and absorbance to find the concentration
Write the equation and expected colour change for the reaction of the Cu(OH)2 with excess ammonia
Cu(OH)2(s) + 2H2O(l) + 4NH3(aq) → [Cu(NH3)4(H2O)2]2+(aq) + 2OH-(aq)