Proteins pt.1
1/71
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
72 Terms
Dietary goal of carbohydrates
increase intake of non-digestible CHO
Recommended CHO intake by Health Canada
45-65%
Recommended fat intake by Health Canada
25-35%
Recommended protein intake by Health Canada
10-30%
What is the essential carbohydrate
No specific “essential” CHO
Dietary goal of lipids
decrease total fat intake (especially saturated and industrial trans fat)
increase monounsaturated (MUFA) and ω-3 fats; Mediterranean diet high in MUFA b/c of olive oil
improve ω-6/ω-3 ratio
General purpose of protein
Provides amino acids (AAs) for protein synthesis
Source of energy (if needed); body tries to avoid on a huge extent as it doesn’t want to lose proteins
Substrate for glucose synthesis; main purpose
Average consumption in North America of protein in calories
~16% of daily calories
Types of AAs in humans
21 proteinogenic AAs (includes selenocysteine)
All but selenocysteine are part of the standard genetic code
Non-proteinogenic AAs also exist (e.g., some neurotransmitters like GABA), but these are not used to make protein
9 AAs are considered “essential” or “indispensible” for humans
Proteinogenic AA
refers to an AA that is incorporated into a protein during translation
Where are proteins found in our bodies
Mainly blood (RBC), connective tissue, eye lens
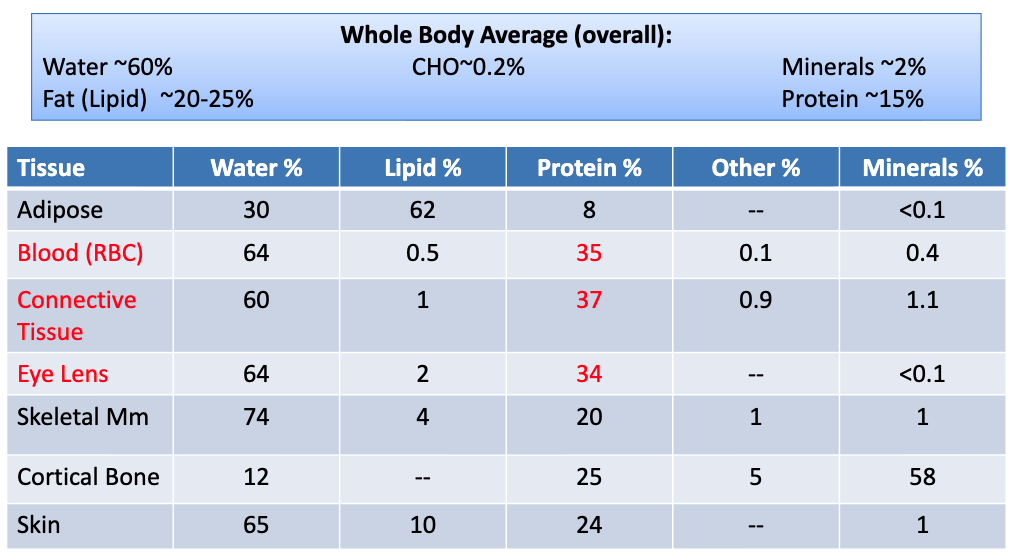
Amino Acid Structure
this can be considered the monomer
Types of amino acids in the body
Standard Amino Acid
Non-Standard Amino Acid
Standard Amino Acids
All are used to make protein
20 AAs are encoded in the genetic code (except for selenocysteine)
Non-Standard Amino Acids
Many exist in the body, but they are rarely used to make
proteins
Usually formed by post-translational modification of other AAs or as intermediates in the metabolic pathways of standard AAs
For example, the GABA neurotransmitter is a metabolite of the amino acid glutamate
*don’t talk about in this course
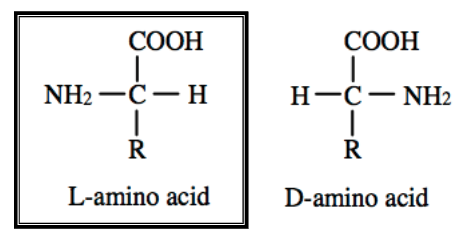
AA Enantiomers
•D vs. L enantiomers
•All standard AAs exist as enantiomers; except for glycine
•L configuration of AAs is naturally occurring
•D configuration of AAs is made through post-translational modifications; mainly used by bacteria
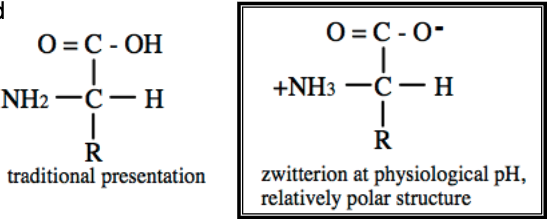
How are AAs Zwitterions
•At physiological pH, AAs are ionized; Protonated amine group and Deprotonated carboxyl group
•No net charge (except R group)
•This increases polarity; i.e., makes AAs more water soluble
How are peptide bonds formed in AAs?
Condensation Reaction (removal of water)
The carboxyl group of one AA reacts with amino group of another AA, releasing H2O
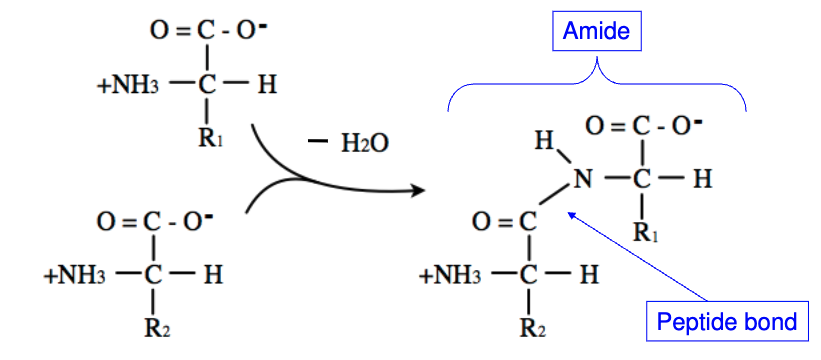
How do you break a peptide bond
Add H2O (hydrolysis reaction)
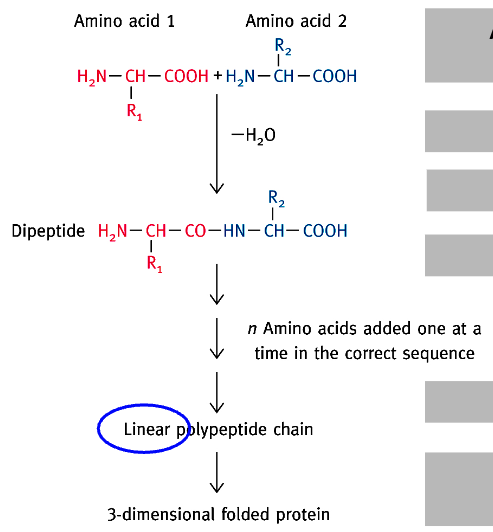
Protein Synthesis: From AA to Protein
2 AAs = Dipeptide
3 AAs = Tripeptide
approx. 50 AAs = oligopeptide
>50 AAs = polypeptide
1 or more polypeptide(s) = a biologically active protein
*Correct folding assisted by chaperone proteins
Misfolded proteins result in:
metabolic diseases, diabetes, obesity
results in accumulation of misfolded proteins
Are peptide and proteins interchangeable
No, peptides are linear and biologically non-functional
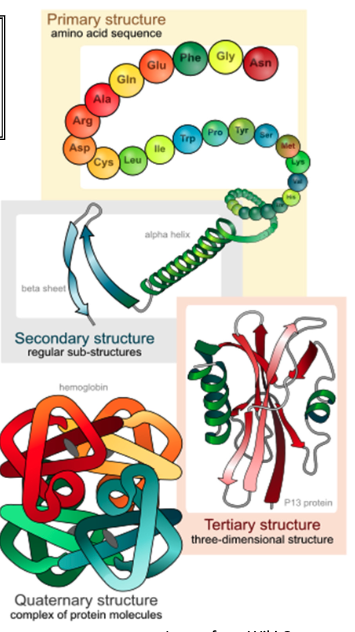
Primary Structure of proteins is determined by
DNA sequence
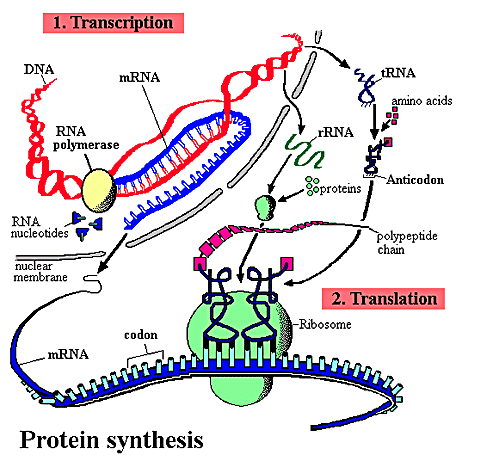
Primary structure of proteins characteristics
Primary structure refers to a polypeptide chain of AAs
held together by peptide bonds; translation is helped by chaperone proteins in the cell
A polypeptide chain has a carboxyl and amino terminus; always start counting from amino end
Secondary structure of proteins is determined by
the hydrogen bonds that create amore stable structure
bonds don’t involve side chains only backbone atoms
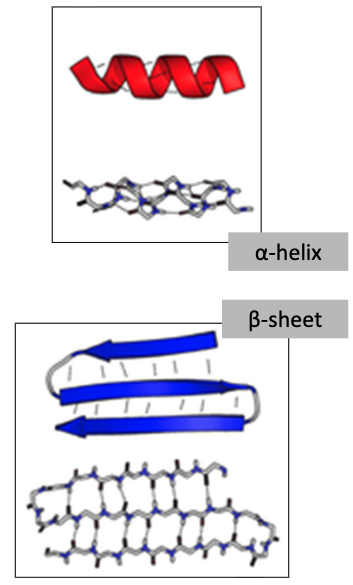
Two types of secondary structure proteins and their characteristics
α-helix
• An amino group makes a hydrogen bond with a carboxyl group 4 AAs down the chain, creating a helical shape in the polypeptide.
β-pleated sheets
• An amino group makes a hydrogen bond with a carboxyl group in the folded back polypeptide chain.
• Can be parallel or anti-parallel.
Tertiary structure
corresponds to the arrangement of the secondary structure in 3D space
consists of one polypeptide chain which allows for most protein to be biologically active
Involves interactions between AA side chains (near or far); ex. disulfide bonds can form between cysteine AAs
Hydrophobic AAs tend to be placed towards the centre of a protein to help ensure that the protein is water soluble
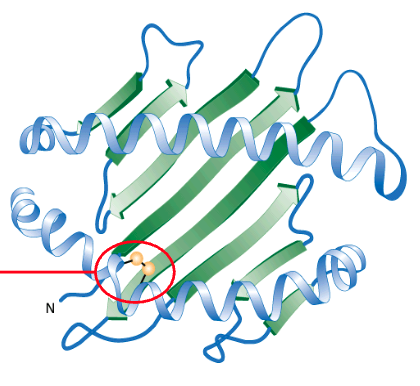
Quaternary Structure of Proteins
corresponds to a combination of 2 or more tertiary structures that are required to make a functional protein
individual structures are referred to as “subunits”
Forms a multi-subunit complex (i.e.,multiple polypeptides); e.g., insulin, immunoglobulins
Not all proteins need a quaternary structure to be biologically active, but some do
Native Protein
corresponds to a protein in its normal 3D conformation where they are biologically active
How can proteins be denatured
heat
salt treatment
detergents
pH (stomach acid)
What happens when a protein is denatured
loses its bioactivity
affects 2°, 3°, and 4° structures (but not 1°); ex. disulphide, alpha-helices, beta-sheets etc.

Different ways to classify AA
Essential versus not essential
Basic, acidic, or neutral
Polar versus non-polar
Essential AA (Indispensable)
Not made by the body or can’t be made quickly enough to meet demands
Types of essential AAs
lys
thr
iso
leu
met
phe
trp
val
his
Conditionally essential AAs
Not normally required in the diet in a healthy individual but become essential under specific contexts
ex. a genetic problem (phenylketonuria) or development of disease Cirrhosis
Phenylketonuria
an inborn error of metabolism whereby a person is unable to breakdown Phe into Tyr
A build-up of Phe in the body causes intellectual disability
The solution is to limit Phe in the diet and supplement with Tyr (conditional essential)
Liver disease (cirrhosis)
impairs Phe and Met catabolism
Tyr and Cys are synthesized from Phe and Met, respectively
Tyr and Cys become indispensable in this context
Tyr and Cys become conditionally essential
Non-Essential AAs (or Completely Dispensable)
Can be synthesized in the body and are not essential to obtain from the diet
Basic AAs include:
lysine
arginine
histidine
Properties of Basic AAs
Polar
+ve charged on NH3 group on side chain enables DNA binding
important in histone protein, which interact with DNA
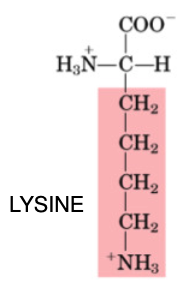
Lysine
•Essential
•Limiting in grain products; small abundance
•Involved in the production of carnitine, which is important for fatty acid metabolism
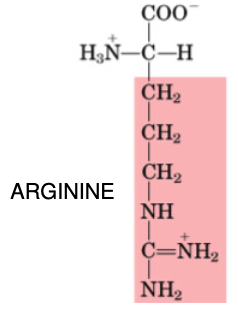
Arginine
•Conditionally Essential in preterm infants unable to synthesize arginine, until gut and intestinal tract can produce
•Non-essential in healthy adults
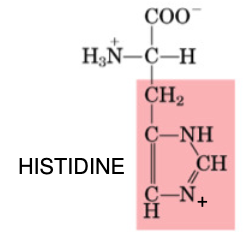
Histidine
•Essential
•Ring structure
•Used to produce histamine (inflammation)
Acidic and Neutral AAs include:
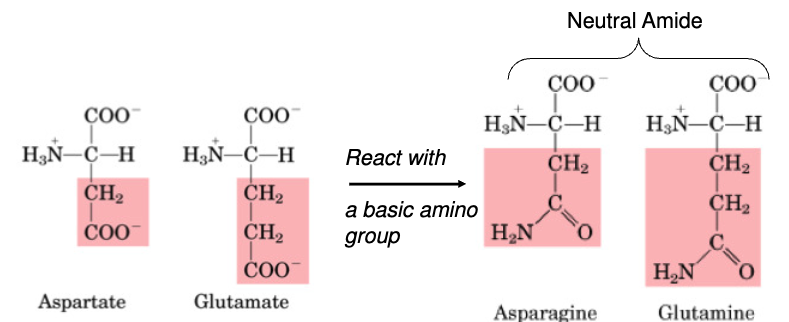
Aspartate
Glutamate
Asparagine
Glutamine
Glycine
Alanine
Properties of acidic AAs
-ve charge on side chain carboxyl group
polar
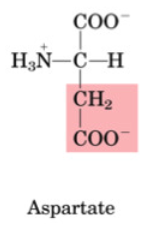
Aspartate
•Non-essential
•Important for amino acid catabolism as it’s involved in transamination
•Transaminated to oxaloacetate (Krebs)
•A “source” of nitrogen in the urea cycle
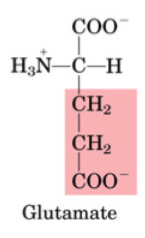
Glutamate
•Non-essential
•Important for amino acid catabolism
•Transaminated to α-ketoglutarate (Krebs)
•Used to produce GABA (neurotransmitter)
Asparagine
non-essential
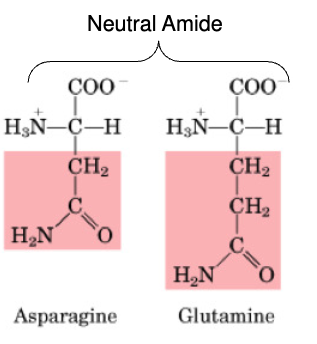
Glutamine
•Non-essential
•Important in AA catabolism because it is an inter-organ carrier of nitrogen (to the liver & kidney)
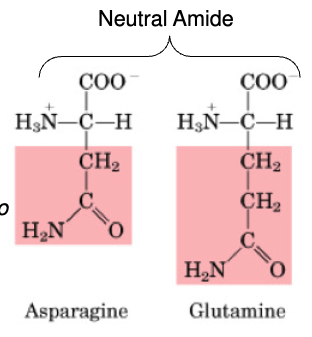
Properties of Neutral AAs
no charged on side chain
non-polar
Aliphatic: C&H atoms joined in straight or branched chains
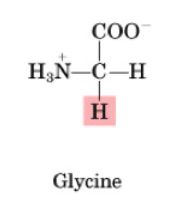
Glycine
•Non-essential
•No enantiomers
•Used primarily to produce porphorin (a component of heme, which is found in hemoglobin); allows RBC to carry O2 around body
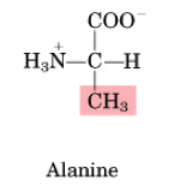
Alanine
•Non-essential
•Important in AA catabolism because it is an inter-organ carrier of nitrogen (to liver & kidney)
•Important role in the glucose-alanine cycle
Branched Chain AAs include:
Leucine
Isoleucine
Valine
Characteristics of branched chain AAs
no charge on side chain
non-polar
all are branched
All are essential
Not catabolized in the liver, so high levels found in circulation
Promote protein synthesis; bypasses liver so its goes to muscle for signalling
BCAA levels are high in protein supplements
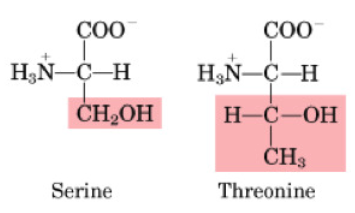
Hydroxylated AAs include:
Serine
Threonine
Characteristics of Hydroxylated AAs
OH-group on side chain is important for protein phosphorylation
polar AA
also includes Tyrosine, but is grouped with aromatic AAs
Ser is non-essential and Thr is essential
OH group on side chain is important for post-translational phosphorylation of proteins
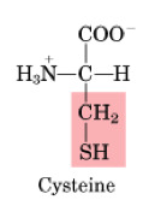
Sulfur-containing AAs include:
Cysteine
Methionine
Properties of sulfur-containing AA
non-polar
contain a sulfur-group
Cysteine
•Non-essential
•Made from methionine
•“Spares” methionine when cysteine consumed in the diet
•Used to form disulfide bonds
•Used in glutathione synthesis (oxidant defence system)
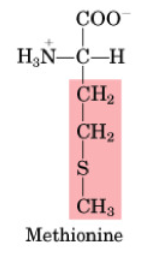
Methionine
•Essential
•1st step in the synthesis of all proteins
•Methionine is limiting in legumes
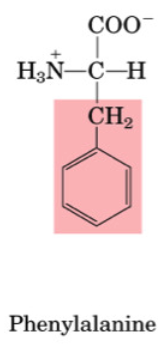
Aromatic AAs include:
Phenylalanine
Tyrosine (only polar aromatic AA)
Tryptophan
Proline
Phenylalanine
•Essential
•Used to make Tyrosine
Tyrosine
•Non-essential
•“Spares” Phe
•post-translational modification of proteins
•Used to synthesize neurotransmitters
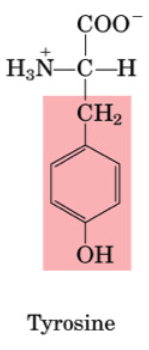
Tryptophan
•Essential
•Used as precursor to make serotonin (mood)
•Used for niacin (Vit B3) synthesis
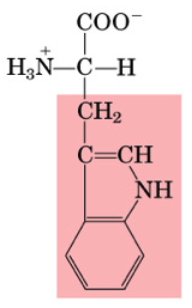
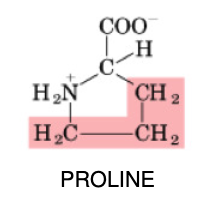
Proline
•Non-essential
•Important for collagen production (extracellular matrix)
•Aliphatic side chain
Where does post-translational modification take place
in polypeptide chains, not free AA
most proteins require some type of modification before they are biologically functional
Types of post-translational modifications
phoshorylation
hydroxylation
gamma-carboxylation
iodination
ADP-ribosylation
Phosphorylation by kinase enzymes include:
– Serine-OH
– Threonine-OH
– Tyrosine-OH
*The OH group is where phosphorylation takes place
Phosphorus dependent
Hydroxylation (creation of new hydroxyl group):
Lysine → hydroxylysine (very important in elastin subunits,
needs copper (copper dependent); associated with aortic rupture)
Proline → hydroxyproline (very important in collagen subunits, needs Vit C (vitamin C dependent); associated with scurvy)
Gamma-carboxylation
Required for calcium homeostasis and blood clotting
Certain proteins are modified to become Ca2+ binding proteins
Another carboxyl group is added to glutamate
Vitamin K dependent
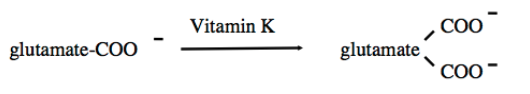
Iodination
– Critical in the formation of thyroid hormones
– Crucial for regulation of the metabolic rate
– About 2 billion humans are iodine deficient
– Iodine dependent
ADP-ribosylation
– Adding ADP-ribose to an acceptor protein
– Critical for DNA repair and regulation of protein function
– Dependent on Vit B3 (niacin); niacin dependent
– Niacin used to form NAD+. When NAD+ is broken down in the cell, ADP-ribose and nicotinamide are the products.