Chapter 7 - Quantum Theory of Atoms
1/16
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
17 Terms
Crest
Place of MAX disturbance
Trough
Place of MIN disturbance
Node
Place of 0 disturbance
Amplitude
MAX output of wave measured from center
Wavelength
distance from crest to crest or trough to trough (measured in m, symbol λ)
Frequency
How many waves pass by per second (s-1 or Hz, symbol v)
Speed of wave equation
FREQUENCY x WAVELENGTH
s-1 x m = m/s
Electromagnetic Spectrum
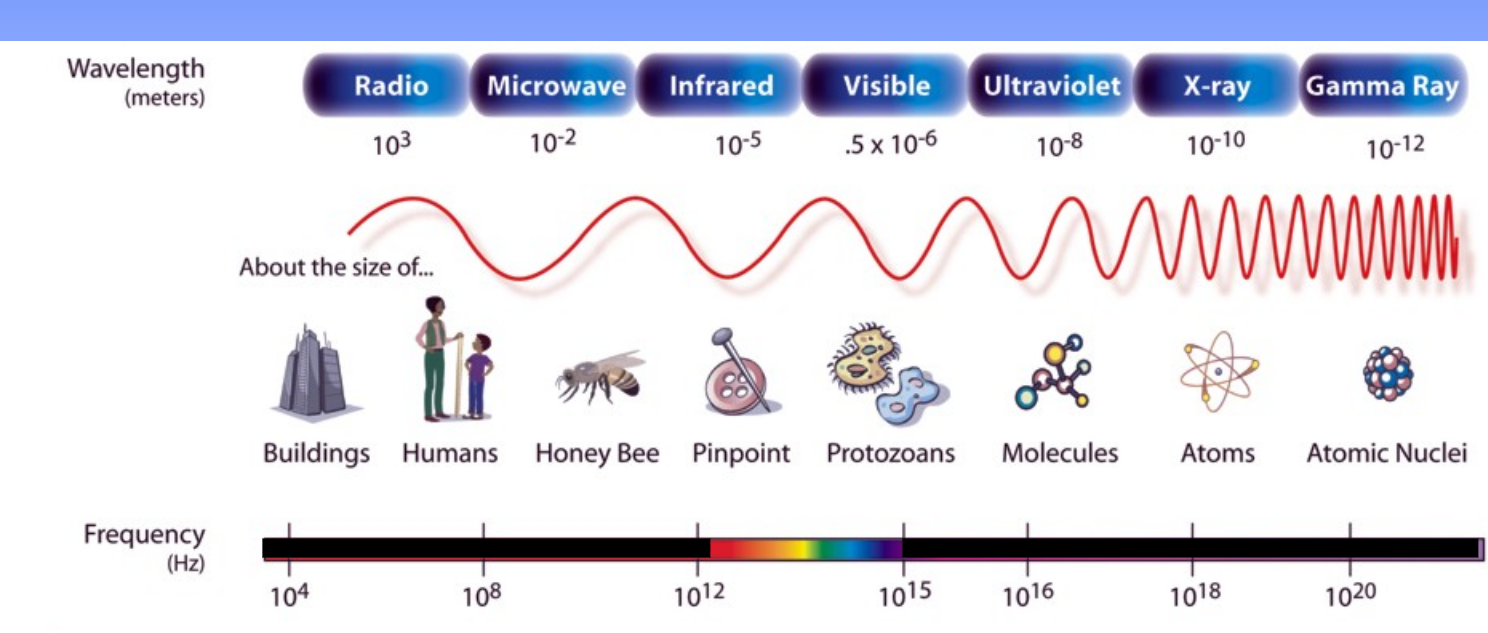
Light waves…
1) travel at speed of light
2) made of electromagnetic fields that “wave” at e/o
3) C = vλ (speed)
Particles of light are called
photons
Energy of photon equation
E = hv
E → energy of photon
h → 6.626 × 10-34 Js
E = hc/λ
v = c/λ
Explain the Bohr Model of an Atom
Electron gains energy → jumps to an excited state for higher energy level
Electron loses energy → no “excited state” because it releases photon of light (fall back down)
En = -2.18 × 10-18 J / n2
rn(radius of electron’s orbit at level n) = (52.9pm)n2
Wavelength equation for photon
λ = 91.1 nm / (1/n2 f - 1/n2 i )
nf = energy level final
ni = energy level initial
What’s ground state
Arrangement of electrons in atom where all are in lowest possible energy level
What’s excited state
Any arrangement of electrons in an atom where 1+ electrons gained energy and reach higher energy levels
Quantum mechanical model of atom
Electrons act like waves λ
“Electron density” is ψ² (probability of where electrons are)
Electrons exist in orbitals (regions of high probability)
Orbitals described in 4 quantum #s (n, l, ml , and ms )