Orgo Ch. 1 Practice Problems: Structure and Bonding; Acids and Bases
1/28
Earn XP
Description and Tags
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
29 Terms
What is the ground-state electron configuration of each of the following elements:
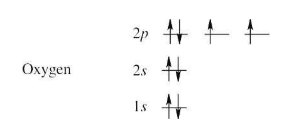
(a) Oxygen
(b) Nitrogen
(c) Sulfur
(a) | To find the ground-state electron configuration of an element, first locate its atomic number. For oxygen, the atomic number is 8; oxygen thus has 8 protons and 8 electrons. Next, assign the electrons to the proper energy levels, starting with the lowest level. Fill each level completely before assigning electrons to a higher energy level. (image attached) Notice that the 2p electrons are in different orbitals. According to Hund’s rule, we must place one electron into each orbital of the same energy level until all orbitals are half filled. Remember that only two electrons can occupy the same orbital, and that they must be of opposite spin. A different way to represent the ground-state electron configuration is to simply write down the occupied orbitals and to indicate the number of electrons in each orbital. For example, the electron configuration for oxygen is 1s2 2s2 2p4. | |
(b) | Nitrogen, with an atomic number of 7, has 7 electrons. Assign these to energy levels. The more concise way to represent ground-state electron configuration for nitrogen: 1s2 2s2 2p3 | |
(c) | Sulfur has 16 electrons. | |
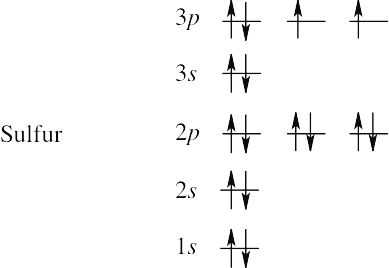 | ||
1s2 2s2 2p6 3s6 3p4 |
How many electrons does each of the following biological trace elements have in its outermost electron shell?
(a) Magnesium
(b) Cobalt
(c) Selenium
The elements of the periodic table are organized into groups that are based on the number of outer-shell electrons each element has. For example, an element in group 1A has one outer-shell electron, and an element in group 5A has five outer-shell electrons. To find the number of outer-shell electrons for a given element, use the periodic table to locate its group. | ||
(a) | Magnesium (group 2A) has two electrons in its outermost shell. | |
(b) | Cobalt is a transition metal, which as two electrons in the 4s subshell, plus seven electrons in its 3d subshell. | |
(c) | Selenium (group 6A) has six electrons in its outermost shell. | |
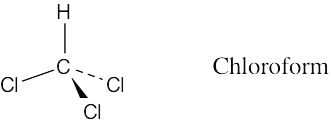
Draw a molecule of chloroform, CHCl3, using solid, wedged, and dashed lines to show its tetrahedral geometry.
A solid line represents a bond lying in the plane of the page, a wedged bond represents a bond pointing out of the plane of the page toward the viewer, and a dashed bond represents a bond pointing behind the plane of the page.
Convert the following representation of ethane, C2H6, into a conventional drawing that uses solid, wedged, and dashed lines to indicate tetrahedral geometry around each carbon (black = C, gray = H).
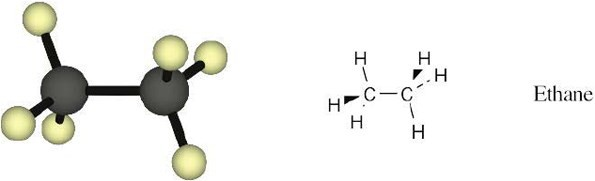
What are likely formulas for the following substances?
(a) CCl?
(b) AlH?
(c) CH?Cl2
(d) SiF?
(e) CH3NH?
Identify the group of the central element to predict the number of covalent bonds the element can form | ||||||||||||||||||||||
(a) | Carbon (Group 4A) has four electrons in its valence shell and forms four bonds to achieve the noble-gas configuration of neon. A likely formula is CCl4. | |||||||||||||||||||||
| ||||||||||||||||||||||
Write line-bond structures for the following substances, showing all nonbonding electrons:
(a) CHCl3, chloroform
(b) H2S, hydrogen sulfide
(c) CH3NH2, methylamine
(d) CH3Li, methyllithium
Start by drawing the electron-dot structure of the molecule. | ||||||||||||||||||||||
(1) | Determine the number of valence, or outer-shell electrons for each atom in the molecule. For chloroform, we know that carbon has four valence electrons, hydrogen has one valence electron, and each chlorine has seven valence electrons. | |||||||||||||||||||||
(2) | Next, use two electrons for each single bond. | |||||||||||||||||||||
(3) | Finally, use the remaining electrons to achieve a noble gas configuration for all atoms. For a line-bond structure, replace the electron dots between two atoms with a line | |||||||||||||||||||||
| ||||||||||||||||||||||
Why can’t an organic molecule have the formula C2H7?
Each of the two carbons has 4 valence electrons. Two electrons are used to form the carbon–carbon bond, and the 6 electrons that remain can form bonds with a maximum of 6 hydrogens. Thus, the formula C2H7 is not possible.
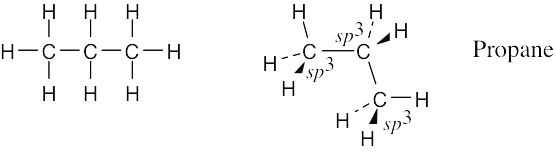
Draw a line-bond structure for propane, CH3CH2CH3. Predict the value of each bond angle, and indicate the overall shape of the molecule.
Connect the carbons and add hydrogens so that all carbons are bonded to four different atoms.
The geometry around all carbon atoms is tetrahedral, and all bond angles are approximately 109°.
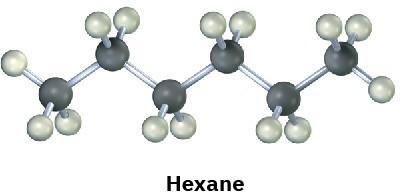
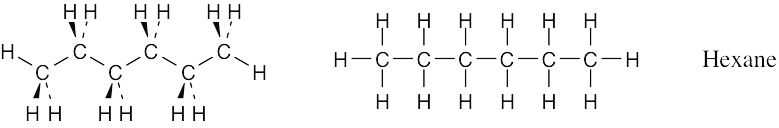
Convert the following molecular model of hexane, a component of gasoline, into a line-bond structure (black = C, gray = H).
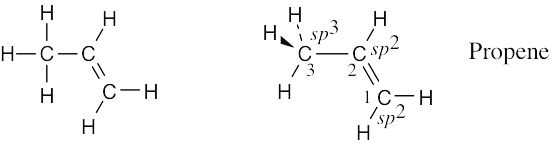
Draw a line-bond structure for propene, CH3CH=CH2. Indicate the hybridization of the orbitals on each carbon, and predict the value of each bond angle.
The C3–H bonds are σ bonds formed by overlap of an sp3 orbital of carbon 3 with an s orbital of hydrogen
The C2–H and C1–H bonds are σ bonds formed by overlap of an sp2 orbital of carbon with an s orbital of hydrogen.
The C2–C3 bond is a σ bond formed by overlap of an sp3 orbital of carbon 3 with an sp2 orbital of carbon 2.
There are two C1–C2 bonds. One is a σ bond formed by overlap of an sp3 orbital of carbon 1 with an sp2 orbital of carbon 2. The other is a π bond formed by overlap of a p orbital of carbon 1 with a p orbital of carbon 2. All four atoms connected to the carbon–carbon double bond lie in the same plane, and all bond angles between these atoms are 120°. The bond angle between hydrogen and the sp3-hybridized carbon is 109°.
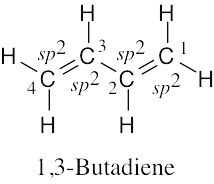
Draw a line-bond structure for 1,3-butadiene, H2C=CH–CH=CH2. Indicate the hybridization of the orbitals on each carbon, and predict the value of each bond angle.
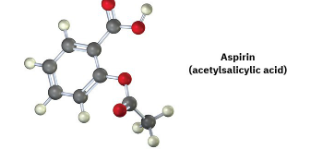
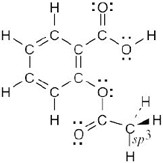
A molecular model of aspirin (acetylsalicylic acid) is shown. Identify the hybridization of the orbitals on each carbon atom in aspirin, and tell which atoms have lone pairs of electrons (black = C, red = O, gray = H).
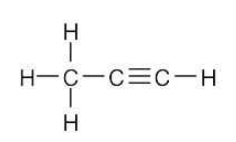
Draw a line-bond structure for propyne, CH3C≡CH. Indicate the hybridization of the orbitals on each carbon, and predict a value for each bond angle.
The C3-H bonds are σ bonds formed by overlap of an sp3 orbital of carbon 3 with an s orbital of hydrogen.
The C1-H bond is a σ bond formed by overlap of an sp orbital of carbon 1 with an s orbital of hydrogen.
The C2-C3 bond is a σ bond formed by overlap of an sp orbital of carbon 2 with an sp3 orbital of carbon 3.
There are three C1-C2 bonds. One is a σ bond formed by overlap of an sp orbital of carbon 1 with an sp orbital of carbon 2. The other two bonds are π bonds formed by overlap of two p orbitals of carbon 1 with two p orbitals of carbon 2.
The three carbon atoms of propyne lie in a straight line: the bond angle is 180°. The H–C1≡C2 bond angle is also 180°. The bond angle between hydrogen and the sp3-hybridized carbon is 109°.
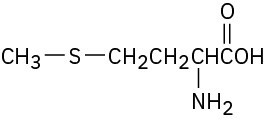
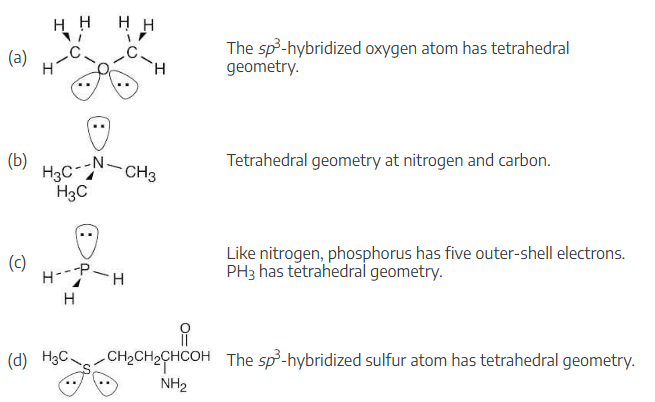
Identify all nonbonding lone pairs of electrons in the following molecules, and tell what geometry you expect for each of the indicated atoms.
(a) The oxygen atom in dimethyl ether, CH3–O–CH3
(b) The nitrogen atom in trimethylamine,
(c) The phosphorus atom in phosphine, PH3
(d) The sulfur atom in the amino acid methionine,
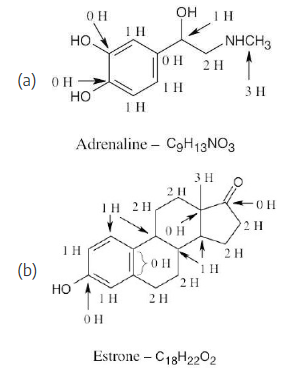
How many hydrogens are bonded to each carbon in the following compounds, and what is the molecular formula of each substance?
Remember that the end of a line represents a carbon atom with 3 hydrogens, a two-way intersection represents a carbon atom with 2 hydrogens, a three-way intersection represents a carbon with 1 hydrogen and a four-way intersection represents a carbon with no hydrogens.
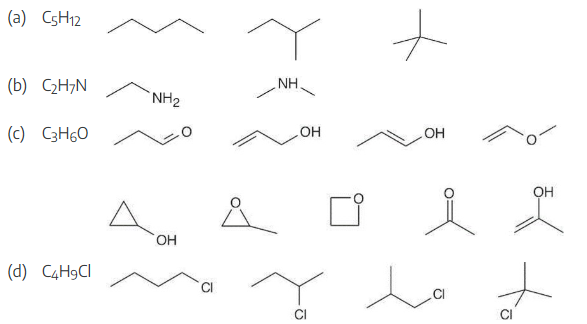
Propose skeletal structures for compounds that satisfy the following molecular formulas: There is more than one possibility in each case.
(a) C5H12
(b) C2H7N
(c) C3H6O
(d) C4H9Cl
Several possible skeletal structures can satisfy each molecular formula.
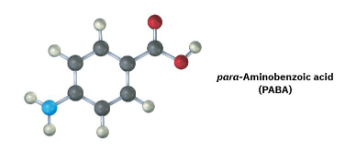
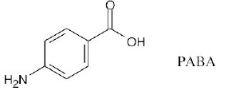
The following molecular model is a representation of para-aminobenzoic acid (PABA), the active ingredient in many sunscreens. Indicate the positions of the multiple bonds, and draw a skeletal structure (black = C, red = O, blue = N, gray = H).
Which element in each of the following pairs is more electronegative?
(a) Li or H
(b) B or Br
(c) Cl or I
(d) C or H
After solving this problem, use Figure 1.20 to check your answers. The larger the number, the more electronegative (EN) the element.
| ||||||||||||||||
Carbon is slightly more electronegative than hydrogen. |
Use the δ+/δ– convention to indicate the direction of expected polarity for each of the bonds indicated.
(a) H3C-Cl
(b) H3C-NH2
(c) H2N-H
(d) H3C-SH
(e) H3C-MgBr
(f) H3C-F
As in Problem 1.19, use Figure 1.20. The partial negative charge is placed on the more electronegative atom, and the partial positive charge is placed on the less electronegative atom.
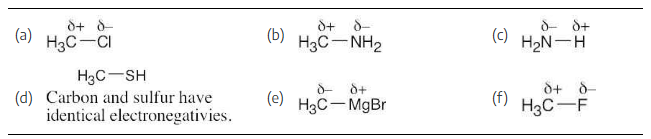
Use the electronegativity values shown in Figure 1.20 to rank the following bonds from least polar to most polar: H3C–Li, H3C–K, H3C–F, H3C–MgBr, H3C–OH
Use Figure 1.20 to find the electronegativities of each element. Calculate ΔEN and rank the answers in order of increasing ΔEN. | |||||||||||||||||||||||||||||||||||||||||||
The most polar bond has the largest ΔEN. Thus, in order of increasing bond polarity: H3C — OH < H3C — MgBr < H3C — Li, H3C — F < H3C — K |
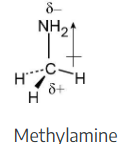
Look at the following electrostatic potential map of methylamine, a substance responsible for the odor of rotting fish, and tell the direction of polarization of the C–N bond:
In an electrostatic potential map, the color red indicates regions of a molecule that are electron-rich. The map shows that nitrogen is the most electronegative atom in chloromethane, and the direction of polarity of the C–N bond is:
Calculate formal charges for the nonhydrogen atoms in the following molecules:
To find the formal charge of an atom in a molecule, follow these two steps:
(1) Draw an electron-dot structure of the molecule.
(2) Use the formula in Section 1.14 (shown below) to determine formal charge for each atom. The periodic table shows the number of valence electrons of the element, and the electron-dot structure shows the number of bonding and nonbonding electrons.
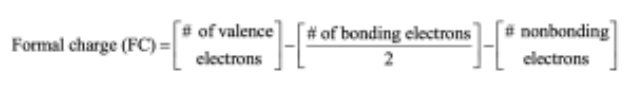
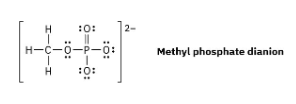
Organic phosphate groups occur commonly in biological molecules. Calculate formal charges on the four O atoms in the methyl phosphate dianion.
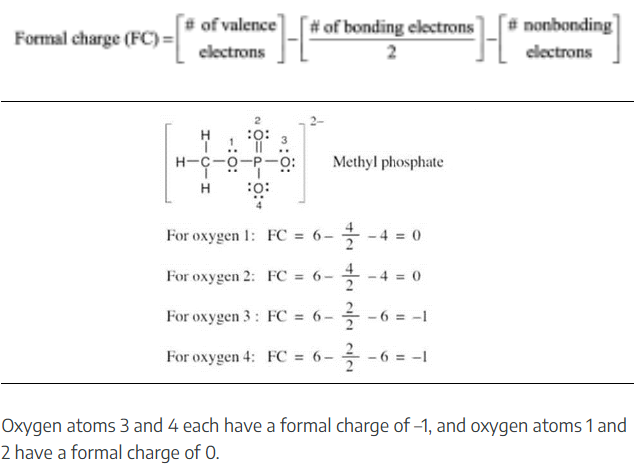
Nitric acid (HNO3) reacts with ammonia (NH3) to yield ammonium nitrate. Write the reaction, and identify the acid, the base, the conjugate acid product, and the conjugate base product.
When an acid loses a proton, the product is the conjugate base of the acid. When a base gains a proton, the product is the conjugate acid of the base.
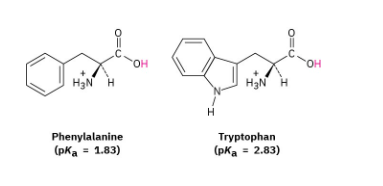
The amino acid phenylalanine has pKa = 1.83, and tryptophan has pKa = 2.83. Which is the stronger acid?
Recall from Section 1.18 that a stronger acid has a smaller pKa and a weaker acid has a larger pKa. Accordingly, phenylalanine (pKa = 1.83) is a stronger acid than tryptophan (pKa = 2.83).
Amide ion, H2N–, is a much stronger base than hydroxide ion, HO–. Which is the stronger acid, NH3 or H2O? Explain.
HO–H is a stronger acid than H2N–H. Since H2N– is a stronger base than HO–, the conjugate acid of H2N– (H2N–H) is a weaker acid than the conjugate acid of HO– (HO–H).

Will either of the following reactions take place to a significant extent as written, according to the data in Table 1.6?
Use Table 1.6 to find the strength of each acid. A reaction takes place as written if the stronger acid is the reactant.

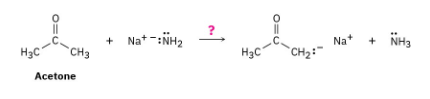
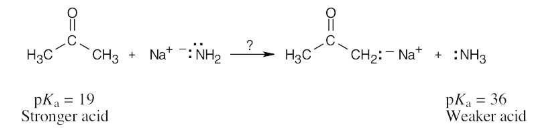
Ammonia, NH3, has pKa ≈ 36, and acetone has pKa ≈ 19. Will the following reaction take place to a significant extent?
As written, the above reaction will take place to virtual completion due to the large difference in pKa values.
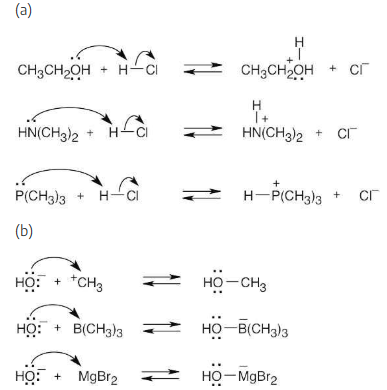
Using curved arrows, show how the species in part (a) can act as Lewis bases in their reactions with HCl, and show how the species in part (b) can act as Lewis acids in their reaction with OH–.
(a) CH3CH2OH, HN(CH3)2, P(CH3)3
(b) H3C+, B(CH3)3, MgBr2
Locate the electron pair(s) of the Lewis base and draw a curved arrow from the electron pair to the Lewis acid. The electron pair moves from the atom at the tail of the arrow (Lewis base) to the atom at the point of the arrow (Lewis acid). (Note: electron dots have been omitted from Cl– to reduce clutter.)